Abstract
Background
Medication harm can lead to hospital admission, prolonged hospital stay and poor patient outcomes. Reducing medication harm is a priority for healthcare organisations worldwide. Recent Australian studies demonstrate cardiovascular (CV) medications are a leading cause of harm. However, they appear to receive less recognition as ‘high risk’ medications compared with those classified by the medication safety acronym, ‘APINCH’ (antimicrobials, potassium, insulin, narcotics, chemotherapeutics, heparin). Our aim was to determine the scale and type of medication harm caused by CV medications in healthcare.
Methods
A narrative review of adult (>16 years) medication harm literature identified from PubMed and CINAHL databases was undertaken. Studies with the primary outcome of measuring the incidence of medication harm were included. Harm caused by CV medications was described and ranked against other medication classes at four key stages of a patient’s healthcare journey. Where specified, the implicated medications and type of harm were investigated.
Results
A total of 75 studies were identified, including seven systematic reviews and three meta-analyses, with most focussing on harm causing hospital admission. CV medications were responsible for approximately 20% of medication harm; however, this proportion increased to 50% in older populations. CV medications were consistently ranked in the top five medication categories causing harm and were often listed as the leading cause.
Conclusion
CV medications are a leading cause of medication harm, particularly in older adults, and should be the focus of harm mitigation strategies. A practical approach to generate awareness among health professionals is to incorporate ‘C’ (for CV medications) into the ‘APINCH’ acronym.
Plain language summary
Patient harm from cardiovascular medications
Background
• Harm from medications can cause poor patient outcomes.
• Certain medications have been identified as ‘high risk’ and are known to cause high rates of harm.
• ‘High risk’ medications are included in medication guidelines used by health professionals.
• Cardiovascular medications (e.g. blood pressure and cholesterol medications) are important and have many benefits.
• Recent studies have found cardiovascular medications to cause high rates of harm.
• Cardiovascular medication harm is often under-recognised in clinical practice.
• Some guidelines do not consider cardiovascular medications to be ‘high risk’.
Method
• This review investigated the extent of harm caused by cardiovascular medications in adults across four healthcare settings:
(1) at the time of hospital admission;
(2) during hospital admission;
(3) after hospital; and
(4) readmission to hospital.
• Harm caused by cardiovascular medications was ranked against other medication classes.
• We investigated the type of cardiovascular medications to cause harm and the type of harm caused.
Results
• Seventy-five studies were reviewed across 41 countries.
• Cardiovascular medications were ranked within the top five medications to cause harm.
• Cardiovascular medications were a leading cause of harm in each healthcare setting investigated.
• Harm caused by cardiovascular medications was common in older adults (>65 years).
• Cardiovascular medications often caused preventable harm.
• Medications to treat high blood pressure and abnormal heart rhythms were the most common causes of harm.
• We reported kidney injury, electrolyte changes and low blood pressure as common types of harm.
Conclusion
• Increased focus on cardiovascular medications in clinical practice is needed.
• Health professionals need to carefully prescribe and frequently review cardiovascular medications, especially in older adults.
• Patient and health professional discussions should be based on both the benefits and harms of cardiovascular medications.
• Cardiovascular medications should be included in all ‘high risk’ medication guidelines.
Keywords
Key points
• Medication harm is a major priority area for healthcare organisations worldwide.
• Cardiovascular medications contribute to significant medication harm (~20–50%) across both ambulatory and inpatient clinical settings.
• Adapting a medication safety acronym is recommended to generate awareness about the optimisation and rationalisation of cardiovascular medications, particularly in older adults.
Introduction
Adverse events in healthcare are defined as ‘incidents in which harm resulted to a person receiving care’. 1 These events are associated with poor patient outcomes and are often preventable. 1 Medication harm is a major subset of adverse events affecting healthcare systems worldwide.2 –5 It can cause hospital admissions, longer hospital stays, increased patient morbidity and mortality and greater resource utilisation.2 –7 In Australia, the annual fiscal burden of medication harm has been estimated to be AUD$1.4 billion.7,8 The World Health Organization has identified medication harm as a global priority and the Australian government lists medication safety as the country’s tenth National Health Priority.9 –11
To help clinicians recognise and mitigate medication harm, ‘high alert’ or ‘high risk’ medication lists have been promoted in clinical settings.12 –14 The most commonly acknowledged list is published by the Institute of Safe Medication Practices (ISMP). 14 ‘High alert’ medications are those with a heightened risk of causing devastating harm if not used correctly. 14 In Australia, there is no standard list; however, the ‘APINCH’ acronym (Figure 1) is widely used to advertise medication safety, encourage harm prevention strategies and raise awareness to the potential for catastrophic harm caused by certain medication classes. 12 It is not intended to be an exhaustive list and does not incorporate every medication linked to harm.12,15

The APINCHa ‘high risk’ medication acronym.
Cardiovascular (CV) medications are among the most frequently prescribed, particularly in the older population.16,17 Currently, 90% of Australians aged >75 years take a CV medication. 16 A strong evidence base for treating CV disorders (e.g. acute coronary syndrome and heart failure) promotes the concurrent use of multiple CV medications.18,19 However, polypharmacy is an independent risk factor for medication harm and the older population are more susceptible to adverse drug events (ADEs).20,21
Local studies have identified CV medications as prominent causes of patient harm during hospital admission, and it is likely that this extends into ambulatory care.22,23 The purpose of this narrative review was to investigate the international literature to determine the scale and type of medication harm caused by CV medications. A contemporary review is pertinent due to the increased use of CV medications over the last three decades. 16 We sought to identify common themes, and if necessary, propose an approach to promote awareness about the safe use of these medications in clinical practice.
Methods
Data sources
Given the breadth and heterogeneity of medication harm research, a systematic review of one clinical intervention was deemed too restrictive. Instead, a narrative review exploring and evaluating the major medication harm studies was considered more appropriate to capture the extent of the issue across multiple healthcare settings. A structured literature search (see Appendix 1) based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines was undertaken using PubMed and Cumulated Index to Nursing and Allied Health Literature (CINAHL) databases. Search terms such as ‘adverse drug reaction’, ‘adverse drug event’, ‘adverse reaction’, ‘adverse event’, ‘medication error’ and ‘medication harm’ were used. Citations and bibliography lists of identified articles were scanned for additional studies.
Inclusion criteria
Systematic reviews and meta-analyses, narrative reviews, case-control studies and observational cohort studies of adverse events and medication harm within both hospital and ambulatory settings were included. Included studies quantified the incidence of medication harm as a primary outcome measure and included information about the medication classes causing harm. If systematic reviews did not specify rank of CV medications, relevant observational studies within the systematic reviews were also analysed.
Exclusion criteria
We excluded studies only investigating paediatric medication harm (<16 years of age) and those using definitions such as ‘potential drug-related problems’ or ‘potential adverse drug events’ which indicated the potential for harm, not actual patient harm. Studies investigating specific patient groups (e.g. mental health or diabetic populations) or specific medication classes (e.g. antimicrobials) were excluded. Randomised clinical trials containing adverse drug event (ADE) data about specific CV medications and conference proceedings, editorials and magazines were also excluded. To link medication harm to contemporary prescribing patterns, studies published before 1990 were excluded.
Definitions and terminology
Multiple terms are often used synonymously to describe medication harm, including ADEs and adverse drug reactions (ADRs; Box 1, see Appendix 2).
24
For the purposes of this review, medication harm was defined inclusively as ‘any negative patient outcome or injury, related to medication use’.
24
Classification of medications as ‘high alert’
Studies investigating medication harm have used multiple categorisations for CV medications. In this review, CV medications were classified as those that directly act on the CV system. 25 This included antihypertensives [e.g. angiotensin-converting enzyme inhibitors (ACE-Is) and calcium-channel blockers (CCBs)], diuretics (e.g. loop and thiazide diuretics), antiarrhythmics (e.g. digoxin, amiodarone), hypolipidaemics (e.g. statins, fenofibrate and ezetimibe) and antianginals (e.g. nitrates). 25 Anticoagulant and antiplatelet medications were excluded, as these medications are already widely acknowledged as high-risk medications and are a focus of pre-existing harm mitigation strategies within existing literature.12,14,26 –28 Including anticoagulants and antiplatelets in this review would detract from the focus on the harm caused by CV medications.
Analysis of studies
Studies were separated according to healthcare setting to investigate both inpatient and ambulatory populations. These included medication harm causing hospital admission, occurring during hospital stay, after discharge or in ambulatory care and readmission. If studies investigated medication harm causing hospital presentation or admission, these were separated based on whether index admissions or readmissions were investigated.
All retrieved studies were reviewed to determine the type of medication harm investigated and the incidence rate of both ‘all cause’ medication harm and harm caused specifically by CV medications. CV medications were then ranked comparative with other medication classes to determine if CV medications were a leading cause of harm. If specified, the types of CV medications implicated were ascertained.
Results
Study inclusion, exclusion and rationale are shown in a flow diagram included in Online Resource 1. Overall, 75 studies were included, of which 10 were systematic reviews/meta-analyses. Most studies investigated medication harm as a cause of hospital admission (
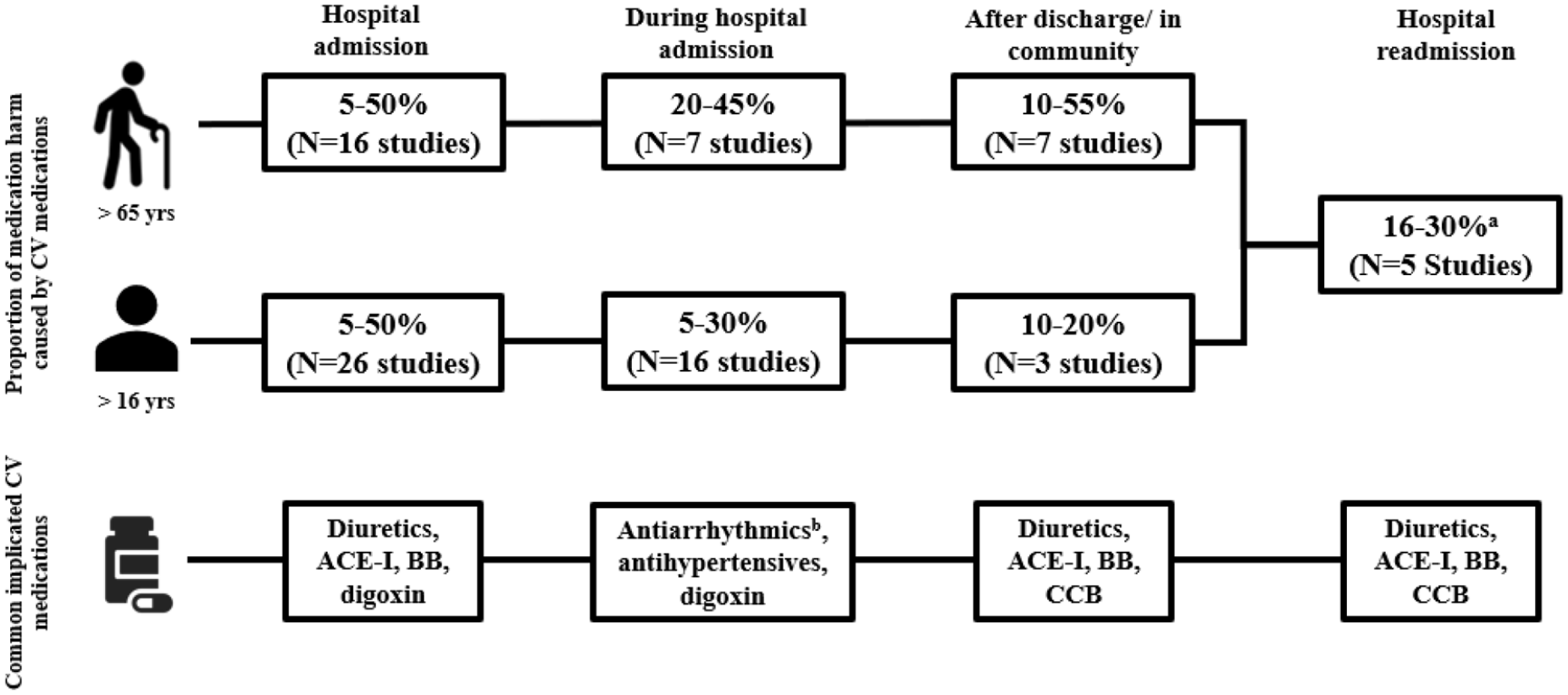
An overview of cardiovascular medication harm across four healthcare settings.
CV medication harm resulting in hospital presentation or admission
Forty-two studies were identified, including six systematic reviews and one meta-analysis that examined medication harm as a cause of admission to hospital. Thirty-five studies were analysed and tabulated (Table 1). The results of the remaining seven studies are included within the systematic reviews.29 –35 The majority of studies were undertaken in general medical or aged care populations. The rate of medication harm varied, with higher rates reported in older populations (range 0.16–41.3%).36,37
CV medication harm causing presentation or admission to hospital.

Includes medication errors.
Includes ADRs, overtreatment, under-treatment and adherence problems.
Definition for CV medications incorporated antithrombotics and anticoagulants.
Includes non-compliance, untreated indications, improper drug selection, sub/supratherapeutic dose, ADRs, drug interactions and drug use without indication.
Includes ADR, dosage too high, dosage too low, ineffective drug, needs additional drug therapy, unnecessary drug therapy and noncompliance.
Includes drug–drug interactions.
Includes ADR, ADEs and medication errors.
ACE-I, angiotensin-converting enzyme inhibitor; ADE, adverse drug event; ADR, adverse drug reaction; AE, adverse events; anti-HTN, antihypertensives; ARB, angiotensin receptor blocker; AU, Australia; AUD, Australian dollars; BB, beta-blockers; CCB, calcium channel blocker; CCS, case-control study; CSS, cross-sectional study/survey; CV, cardiovascular; DRA, drug-related admission; DRP, drug-related problem; DTF, drug therapeutic failure; ICU, intensive care unit; LOS, length of stay; LR, literature review; MA, meta-analysis; MM, medication misadventure; NR, not reported; OS, observational study; pADE, preventable adverse drug event; PCS, prospective cohort study; POS, prospective observational study (includes CCS/PCS); RCS, retrospective cohort study; ROS, retrospective observational study (includes CCS/RCS); SR, systematic review; USA, United States of America.
As per Table 1, 22 studies (63%) ranked CV medications in the top three causes of medication harm. Of the remaining 13 studies, a further five categorised CV medications within the top five causes of harm and another seven were unable to be ranked but described CV medications as a leading cause of harm. The majority of studies investigating older adults [73% (
In Australia, Runciman
Types of CV medications causing harm
Diuretics, antihypertensives and digoxin were most frequently identified as causes of harm.32,34,51 Diuretics have been implicated in up to 30% of admissions due to medication harm, including renal failure and serious electrolyte imbalances.34,39 Included within the systematic reviews of Howard
CV medication harm occurring during hospital stay
A total of 23 inpatient studies were found, including one systematic review, two meta-analyses, 16 observational studies and three literature reviews. Twenty-one studies were analysed and tabulated and the results of the remaining two studies are included within the systematic reviews (Table 2).2,69 There were wide variations in the reported rates of inpatient medication harm (2–46% of patients).51,70,71 This may be attributed to the different methodologies used to identify harm and to the different patient populations studied. Studies investigating older adults reported higher rates of harm compared with those including all age groups.22,51,71,72
CV medication harm within hospitals.

Includes medication errors.
n.b. leading drug type was ‘other’.
Includes ADEs causing admission; inpatient rate not separated in analysis.
terms used interchangeably.
Includes ADEs causing admission, readmission and inpatient ADEs.
ADE, adverse drug event; ADR, adverse drug reaction; AE, adverse events; antiHTN, antihypertensives, AU, Australia; CCS, case-control study; CV, cardiovascular; FADR, fatal ADR; ICU, intensive care unit; LOS, length of stay; LR, literature review; MA, meta-analysis; MRH, medication-related harm; NR, not reported; OS, observational study; pADE, preventable ADE; pADR, preventable ADR; PCS, prospective cohort study; POS, prospective observational study (includes PCS/CCS/cross-sectional study); RCS, retrospective cohort study; ROS, retrospective observational study (includes RCS/CCS/cross sectional study); SR, systematic review; USA, United States of America.
CV medications were found to be one of the top five medications to cause harm during admission, and the prevalence of CV medication harm increased in older populations (Table 2).2,4,5,22,44,51,57,69 –72,74,76,83 A meta-analysis and systematic review found CV medications were the second and third most frequently implicated medications in inpatient ADEs and ADRs, respectively. 74 In addition, CV medications were the fourth most frequently involved drug class in fatal ADRs after antithrombotics, sedatives and antineoplastics. 74 Another literature review found CV medications to be implicated in causing 17.9% of preventable ADEs and recommended that they be a high-priority focus for harm prevention strategies. 70
In the Harvard Medical Practice Study, 3.7% of 30,195 patients experienced harm, with medications responsible for 19% of harm.
4
CV medications were the fourth highest cause (8.5%) of ADEs.
4
Antihypertensives, classified separately, were the seventh highest cause (5.0%).
4
In the Quality in Australian Health Care Study, CV medications were implicated in causing 20% (
Two studies investigated medication harm in critical care.75,82 A scoping review of 30 studies investigated medications as contributors to clinical deterioration or the need for critical care. 75 Sedatives, analgesics and CV medications were most commonly implicated, although the quality of evidence was low due to small sample sizes and few primary medication-related outcomes. 75 A Columbian study investigating ADRs as a cause of admission to the intensive care unit (ICU) found antiplatelet drugs (bleeding) and renin–angiotensin-receptor-blocking drugs (renal impairment) were most frequently responsible for harm. 82
Type of CV medication causing harm
Of the studies that delineated CV medications, antiarrhythmics, antihypertensives (e.g. beta-blockers, ACE-I) and diuretics were common causes of medication harm.22,44,74,76 Notably, digoxin was frequently implicated, likely a result of its narrow therapeutic range.44,76,84 Diuretics and antihypertensives have been reported to cause up to 33% and 17% of medication harm events, respectively.57,71 Renal impairment and electrolyte imbalances caused by these medications were frequent, and in severe cases, led to ICU admission and a prolonged length of hospital stay.22,44,51,71,82
CV medication harm after discharge from hospital
The transition of patients from hospital raises safety challenges due to the risk of medication harm.85 –88 Ten studies, one systematic review and nine observational studies, investigated medication harm after discharge or in ambulatory care. This included a systematic review and observational studies with a post discharge follow-up period of up to 365 days, and observational studies conducted in outpatient departments, multi-specialty clinics or a community setting (e.g. residential/continuing care facilities and general practitioners). Six studies were analysed and included in Table 3 as the remaining four observational studies were encompassed within the systematic review.87 –90 An examination of all adverse events in 400 general medical patients after discharge, found that ADEs accounted for the majority (66%) of events. 91 However, similar to medication harm on/during hospital admission, there was a wide variation in rates, with the highest rates reported in older populations (0.4–51.2%).88,92
CV medication harm after hospital discharge and in ambulatory care.

Includes medication errors.
Includes ADR, medication errors or harm caused by non-adherence.
ACE-I, angiotensin converting enzyme inhibitors; ADE, adverse drug event; ADR, adverse drug reaction; AE, adverse events; anti-HTN, antihypertensives; BB, beta-blockers; CV, cardiovascular; ME, medication error; MRH, medication-related harm; NR, not reported; OS, observational study; pADE, preventable adverse drug event; PCS, prospective cohort study; POS, prospective observational study; RCS, retrospective cohort study; ROS, retrospective observational study; SR, systematic review; USA, United States of America.
CV medications were a leading cause of harm post discharge. Most studies found approximately one in five medication harm events were caused by CV medications, increasing in some studies to over half of events among older patients.88 –90,92,95 –97 The aforementioned systematic review of patients greater than 65 years of age found that CV medications were the leading cause of ADRs and ADEs, implicated in 18.8–55.7% of events. 92 Within the review, a 1999 study found that the number of newly prescribed medications at the time of discharge was a significant risk factor for medication harm. 97 CV medications were the most commonly prescribed medications at the time of discharge, a potential reason for the high rates of harm in this group (18.8%). 97
Types of CV medications causing harm
Medication harm was most frequently attributed to antihypertensives which included diuretics, ACE-Is, beta-blockers and CCBs.87,88,93 Harm caused by these medications was deemed highly preventable, causing 40.5% of preventable ADEs. 88 They were also implicated in causing 42% of serious ADEs. 93 While the high prevalence of harm caused by CV medications may simply reflect their high prescribing rates, one study found that, after correcting for this factor, CV medications still contributed to excess harm. 93
Readmission caused by CV medication harm
While a number of studies investigated medication harm that caused readmission, most of these studies were summarised in one systematic review. 86 This review of 19 studies showed a wide variation in the reported incidence of medication harm causing readmission (3–64%). 86 The mean readmission rate caused by medication harm at 30 days and 12 months was found to be approximately 20% (range 7–61%). 86 CV medications were frequent causes of preventable readmissions in six studies within the systematic review, causing as many as 30% of ADR readmissions.20,86,98,99 Diuretics causing renal impairment were common and, in severe cases, were linked with death.86,98 Postural hypotension, arrhythmias and peripheral oedema caused by ACE-Is/diuretics, beta-blockers and CCBs, respectively, were also reported.98,100
Discussion
This review identified CV medications as a leading cause of medication harm, consistent with results reported from recent, local studies.22,23 Irrespective of clinical setting, approximately one in five medication harm events were found to be caused by CV medications and they were also ranked within the top five classes of medications to cause harm. Antihypertensives, diuretics and antiarrhythmics (e.g. digoxin) were most frequently implicated. The latter is consistent with a 2014 study that found that despite decreasing rates of digoxin prescribing, emergency presentations due to toxicity remained high, with >5000 visits estimated annually in the US. 101
CV medications and the ageing population
An emergent theme was that older persons are particularly susceptible to CV medication harm, as shown in Figure 2. 17 This class accounted for greater than 50% of all events reported in some studies, and studies specifically involving older patients are summarised in Online Resource 3.22,62,96 Similar to the findings for all adults, antihypertensives, such as diuretics and ACE-Is, and antiarrhythmics, were frequently implicated (Figure 2).
As the population ages and the prevalence of CV disease increases, harm caused by CV medications will likely increase. As a result of the ageing process, body systems undergo a progressive decline in physiological functioning, including the CV, pulmonary and renal systems. 102 This results in altered pharmacokinetics/pharmacodynamics of medications which clinicians must consider when managing CV medications.17,102
The importance of individualising and rationalising therapy in older adults is essential for patient care. A number of deprescribing tools have been developed and incorporate CV medications.103 –105 The application of decision support is particularly prudent in frail older persons in whom risks of medications often outweigh their benefits.106,107 Of particular interest is optimising the use of antihypertensives by using agreed treatment goals, absolute CV disease risk and appropriate blood-pressure target levels. 107 Knowing when to review or deprescribe CV medications in older patients is challenging in light of the strong evidence base for these medications in reducing CV disease risk. 108 However, it is imperative to note that most clinical trials have not included the frail and multimorbid older patients who are frequently encountered in routine clinical practice.109,110 It is important to recognise that deprescribing is indeed concordant with ethical principles when serving patient-centred interests (i.e. beneficence, non-maleficence, autonomy, and justice). 111
The under-representation of multimorbid, older adults, the focus of drug approval processes on efficacy and the lack of long-term efficacy and data on harm in clinical trials of CV medications has been described in the literature.110,112,113 These factors result in a discrepancy between the incidence, type and severity of harm reported in clinical trials and that reported in clinical practice (post-marketing). 112 While the benefits of CV medications are widely acknowledged, a recent push for active pharmacovigilance programmes and deprescribing in patients with CV diseases has emphasised the need to recognise the harms linked with CV medications.110,112,113 Patient–clinician interactions should allow for informed treatment decisions about the benefits and harms of CV medications.110,114 In addition, opportunities for rationalisation through withdrawal or dose reduction should be considered regularly with a focus on realistic treatment goals.110,114 The studies included in this review are representative of real-world treatment populations, and the findings support the need for emphasis to be placed on the rationalisation of antihypertensives and other CV medications, particularly within older populations.
The implications of CV medication harm
While it is acknowledged that CV medications have a fundamental role in the reduction of major CV endpoints, such as morbidity and mortality, the harm caused by these medications should not be overlooked. Although medication harm, such as electrolyte imbalance and renal impairment caused by antihypertensives, can be potentially reversible and may not be considered severe, the findings of this review highlighted that these events are linked with poor outcomes. The included studies reported life-threatening events, admissions to the ICU, prolonged hospital stays and in some cases, fatal events, caused by antihypertensives and diuretics.22,34,39,44,51,71,82,88,98 Although underexplored, this medication harm has the potential to cause both unwanted physical and psychological ramifications for patients due to the distress associated with hospitalisation and intensive care stays. 115 Many studies also found a high proportion of preventable medication harm events to be caused by CV medications.5,62,70,77,88,98 The resultant financial impact and consumption of healthcare resources due to potentially avoidable hospital presentations and prolonged length of hospital stays should also be considered when measuring the magnitude of the harm. For patients where withdrawal of CV medications is not indicated (see above), prudent dose selection and stringent monitoring following guideline driven dose adjustments is warranted to reduce the risk of medication harm.
In this review, we wanted to investigate CV medication harm from an international perspective. The studies included in this review (including those within the systematic and literature reviews) were conducted across six continents including: North America, Europe, Australia/Oceania, South America, Asia and Africa, from 41 countries. This highlights that CV medication harm is a global challenge and does not discriminate across healthcare settings or healthcare systems worldwide. The World Health Organization’s third global patient safety challenge ‘Medication without harm’ has paved the way to improve medication harm from a global perspective. 9 The findings of this review suggest that improving CV medication harm should be prioritised along with other ‘high risk’ medications.
High risk medications
In responding to the high prevalence of medication harm, lists of ‘high risk/alert’ medications have been formulated. These include medications associated with an increased risk of patient harm, particularly if prescribed, dispensed or administered erroneously. 14 The lists are commonly promoted in hospitals to raise awareness about medication safety.
The ISMP list of ‘high alert’ medications is the most comprehensive and frequently used worldwide. 14 It is updated regularly based on medication error reports, medication harm literature and consensus from practitioners and safety experts. 14 The latest list consists of 21 different medication classes, with CV medications accounting for 19.0% of the listed medications. 14
A standardised list is important to remind clinicians of the risk of medication harm. In Australia, the current ‘high risk’ medications are within the acronym, ‘APINCH’ (Figure 1), which has been widely adopted nationwide. 12 It differs from the ISMP list in that it does not incorporate CV medications, such as antiarrhythmics (e.g. amiodarone), inotropes (e.g. digoxin) and adrenergic antagonists (e.g. beta-blockers).12 –14
The omitted ‘C’ in APINCH
In addition to the evidence for contributing to harm, there are other clinical reasons CV medications should be considered ‘high risk’. First, many CV medications require well-defined protocols to guide administration and often can only be prescribed by skilled staff. Second, ‘high risk’ medicines are defined as ‘medicines that have an increased risk of causing significant patient harm or death if they are misused or used in error’. 13 This criteria would apply to intravenously administered antiarrhythmics, such as digoxin, metoprolol and amiodarone. 116 Third, CV medication harm largely affects older populations who account for most hospital admissions and are particularly vulnerable to medication harm.36,50,117 Additionally, harm is prevalent across all healthcare settings and our findings suggest patients newly prescribed CV medications during hospital admission are at risk of medication harm in ambulatory care.92,97 This emphasises the need for clinicians to consider harm mitigation strategies at the time of discharge such as home medication reviews, additional follow-up general practitioner visits and ‘high-risk’ discharge clinics.
Given the results of our review, we propose that CV medications are brought to the forefront by incorporating ‘C’ into the ‘APINCH’ acronym. ‘CAPINCH’ or ‘APINCH-C’ would serve as a prompt to optimise the use of CV medications throughout hospitalisation, including at the time of discharge. We acknowledge that this may be a different application of the ‘APINCH’ acronym to what was originally intended. However, the addition of ‘C’ would provide a practical and timely initiative to generate awareness about the harm caused by CV medications.
Limitations
There are some limitations to this review. Medication harm literature is vast and heterogeneous, and studies differ in terminology and methodology which makes it difficult to compare studies. 24 Additionally, the breadth and exploratory nature of the research across four healthcare settings did not meet the explicit criterion for a systematic approach, such as PRISMA. Therefore, a narrative review was undertaken and consequently, some relevant studies may not have been included. To account for this, we employed a structured search strategy using elements of PRISMA (e.g. medical subject headings, abstract/title screening) with a focus on the major studies incorporating definitions that matched our medication harm definition. To aid in transparency, we distinguished between study design and definitions used throughout this review. Similarly, the classification of drug classes differed between studies. For example, some studies included antithrombotics (e.g. aspirin) as a CV medication. 36 As our focus was on medications directly acting on the CV system, antithrombotics were excluded; however, some studies did not specify what was included within the CV medication class. It should be noted that antiplatelets are not incorporated within the ‘APINCH’ acronym. 12 Due to the important role these medications play in practice, antiplatelets could be a potential focus for future reviews of medication harm. Finally, it is acknowledged that medication harm can be precipitated by drug–drug and drug–disease interactions and from medication omission (e.g. non-compliance). Unless it was specified by the authors of the study, we were unable to discern whether these underlying factors were major contributors to medication harm.
Conclusion
CV medications are frequently implicated in causing harm across all healthcare settings. A common theme was the high prevalence of harm in older adults, which leads to morbidity and hospital utilisation. A practical method for socialising the risk would be to adapt a well-accepted safety acronym.
Supplemental Material
sj-pdf-1-taw-10.1177_20420986211027451 – Supplemental material for Patient harm from cardiovascular medications
Supplemental material, sj-pdf-1-taw-10.1177_20420986211027451 for Patient harm from cardiovascular medications by Chariclia Paradissis, Neil Cottrell, Ian Coombes, Ian Scott, William Wang and Michael Barras in Therapeutic Advances in Drug Safety
Supplemental Material
sj-pdf-2-taw-10.1177_20420986211027451 – Supplemental material for Patient harm from cardiovascular medications
Supplemental material, sj-pdf-2-taw-10.1177_20420986211027451 for Patient harm from cardiovascular medications by Chariclia Paradissis, Neil Cottrell, Ian Coombes, Ian Scott, William Wang and Michael Barras in Therapeutic Advances in Drug Safety
Supplemental Material
sj-pdf-3-taw-10.1177_20420986211027451 – Supplemental material for Patient harm from cardiovascular medications
Supplemental material, sj-pdf-3-taw-10.1177_20420986211027451 for Patient harm from cardiovascular medications by Chariclia Paradissis, Neil Cottrell, Ian Coombes, Ian Scott, William Wang and Michael Barras in Therapeutic Advances in Drug Safety
Footnotes
Appendix 1
Appendix 2
Medication harm definitions and terminology. a .

| Acronym | Definition |
|---|---|
| ADE | Any untoward medical occurrence that may present during treatment with a pharmaceutical product but that does not necessarily have a causal relation to the treatment |
| ADR | A response to a medicine which is noxious and unintended, and which occurs at doses normally used in man |
| ME | Any preventable event that may cause or lead to inappropriate medication use or patient harm while the medication is in the control of the health care professional, patient, or consumer |
Based on definitions from the World Health Organization.
ADE, adverse drug event; ADR, adverse drug reaction; ME, medication error.
Acknowledgements
The authors thank Ms Christine Dalais, Liaison Librarian at The University of Queensland, who helped to develop, test and optimise the search strategy for this research.
Author contributions
CP, MB, IC and NC were involved in the conception and planning of the manuscript. CP researched, analysed and wrote the manuscript under the guidance of MB, IC and NC. MB, IC, NC, IS and WW reviewed and edited the manuscript. IS and WW provided medical expertise in the analysis and presentation of results and discussion points.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Code availability
Not applicable
Availability of data and material
All data included and analysed for this research are incorporated within the manuscript and supplementary files provided. These published works were obtained from PubMed and CINAHL databases.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
