Abstract
Objective(s):
The aim of this study was to conduct a meta-analysis to assess the clinical safety of ceftolozane-tazobactam for the treatment of acute bacterial infections in adult patients.
Methods:
The PubMed, Embase, and Cochrane databases were searched from their inception until May 2020 for relevant randomized controlled trials (RCTs). Only RCTs evaluating the risk of adverse events (AEs) for ceftolozane-tazobactam and comparative treatments for acute bacterial infections in adult patients were included.
Results:
Overall, four RCTs including a total of 2924 patients (1475 in the ceftolozane-tazobactam group and 1449 in the control group) were included in the meta-analysis. The rate of treatment-emergent AEs was 51.3% (748/1458) in the ceftolozane-tazobactam group, which was comparable to the control group, 49.9% [714/1430; odd’s ratio (OR), 1.06; 95% confidence interval (CI), 0.91–1.25; I2 = 0%]. In addition, no difference was observed between the ceftolozane-tazobactam and control groups in terms of the risk of serious AEs (OR, 1.22; 95% CI, 0.93–1.61; I2 = 15.5%) and the risk of discontinuing the study drug due to AEs (OR, 0.85; 95% CI, 0.55–1.33; I2 = 0%). The rate of all-cause mortality did not significantly differ between the ceftolozane-tazobactam and control groups (OR, 1.11; 95% CI, 0.82–1.50; I2 = 0%). The only exception was the risk of Clostridiodes difficile (C. difficile) colitis, where ceftolozane-tazobactam treatment was associated with a significantly higher risk compared with the control group [0.72% (10/1376) versus 0.14% (2/1391), OR, 3.84; 95% CI, 1.23–11.97; I2 = 0%].
Conclusion:
Ceftolozane-tazobactam treatment is as tolerable as comparative treatment options for acute bacterial infections in adult patients, however it has an increased risk of C. difficile infection. As a novel broad-spectrum antibiotic, ceftolozane-tazobactam could be a safe therapeutic option for use in common clinical practice.
Plain language summary
Introduction
Ceftolozane-tazobactam is a combination of the broad-spectrum cephalosporin and a ß-lactamase inhibitor. 1 Ceftolozane, an oxyimino-aminothiazolyl cephalosporin, is structurally similar to ceftazidime, but the in vitro increases in the minimum inhibitory concentration (MIC) of ceftazidime due to porin loss, are not observed for ceftolozane. 2 Tazobactam, a β-lactam sulfone, is a potent β-lactamase inhibitor of most common class A and C β-lactamases. 1 Many in vitro studies3–7 have shown that this novel combination exhibits potent activity against most clinically important gram-negative bacteria, including multidrug-resistant (MDR) bacteria. Clinically, ceftolozane-tazobactam has also shown favorable efficacy for the treatment of complicated intra-abdominal infection (cIAI) and uncomplicated/complicated urinary tract infection (cUTI) in Japan.8,9 In addition, several randomized controlled trials (RCTs) have demonstrated that the clinical efficacy of ceftolozane-tazobactam is comparable with other alternative agents for the treatment of acute bacterial infections.10–12 In the ASPECT-cUTI trial, 10 ceftolozane-tazobactam was non-inferior to levofloxacin for composite cure (76.9% versus 68.4%, 95% CI 2.3–14.6) for the treatment of complicated lower-UTIs or pyelonephritis. In the ASPECT-cIAI trial, 11 the clinical response of ceftolozane-tazobactam plus metronidazole was comparable to meropenem in adult patients with cIAIs, including MDR pathogen-associated infections. In the ASPECT-NP trial, 12 high-dose ceftolozane-tazobactam was non-inferior to meropenem in terms of both the 28-day all-cause mortality and the clinical cure at test for Gram-negative nosocomial pneumonia in mechanically ventilated patients. 12 Even in the post hoc analysis of these RCTs,13–15 the clinical efficacy of ceftolozane-tazobactam was non-inferior to comparative treatments for infections caused by antibiotic-resistant pathogens, including ESBL-producing Escherichia coli and Klebsiella pneumoniae, MDR Pseudomonas aeruginosa, and levofloxacin-resistant Enterobacteriaceae. In addition, empiric use of ceftolozane/tazobactam for the treatment of cUTI could also be a cost-effective choice. 16 These findings suggest that ceftolozane-tazobactam could be an effective antibiotic for the treatment of acute bacterial infections. In addition to clinical efficacy, safety issues are another important concern when physicians use novel antibiotics in clinical practice. However, an updated meta-analysis comparing the safety of ceftolozane-tazobactam with comparative drugs for the treatment of acute bacterial infection is lacking. We conducted this meta-analysis to provide evidence on the safety of ceftolozane-tazobactam in adult patients with acute bacterial infection.
Materials and methods
Study search and selection
This study was written and reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guideline. 17 All clinical studies were identified through a systematic search of the PubMed, Embase, and Cochrane databases from their inception until May 2020. The following search terms were used: ‘ceftolozane-tazobactam,’ ‘ceftolozane,’ ‘tazobactam,’ and ‘randomized’. Only RCTs that compared the risk of adverse events (AEs) for ceftolozane-tazobactam and comparative treatments for adult patients with acute bacterial infections were included. Articles published in all languages were eligible for inclusion. However, we excluded articles if they reported in vitro studies or pharmacokinetic–pharmacodynamic assessments. Two reviewers searched and examined the publications independently to avoid bias. Any disagreement was resolved and decided by a third reviewer. The following data was extracted from the included studies: authorship, year of publication, study design, countries, antibiotic regimen for ceftolozane-tazobactam and the comparative treatment, and the risk of AEs.
Definitions and outcomes
The primary outcome was the risk of AEs, including treatment-emergent AEs (TEAE), treatment-related AEs, serious AEs, discontinuation of the study drug due to an AE, and all-cause mortality. TEAEs were defined as an AE that occurred in a participant that was administered the study drug but that does not necessarily have a causal association with the study drug. Serious AEs were defined as AEs that could result in death, persistent or significant disability or incapacity, be life threatening, require or prolong an existing hospitalization, or another important medical event deemed such by medical or scientific judgment.
Quality assessment
The quality of each included study was assessed using a risk-of-bias assessment tool. 18 Two reviewers subjectively reviewed all included studies and rated them either ‘low risk’, ‘high risk’, or ‘unclear’ according to the following items: randomization sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and inclusion of intention-to-treat analyses.
Data analysis
A DerSimonian–Laird random-effects model was performed to calculate the pooled estimates of odd’s ratios (ORs). 19 A two-sided p-value of < 0.05 was considered to indicate a statistically significant difference. Study heterogeneity was presented using a χ2-based Cochran’s Q statistic and I2. Cochran’s Q was defined by summing the square of the amount that each study estimate deviated from the overall estimate. For the Q statistic, p < 0.10 were considered statistically significant for heterogeneity. For the I2 statistic, heterogeneity was assessed as follows: no heterogeneity (I2 = 0–25%), moderate heterogeneity (I2 = 25–50%), large heterogeneity (I2 = 50–75%), and extreme heterogeneity (I2 = 75–100%). To evaluate the effect of individual studies, leave-one-out sensitivity analyses were performed for primary outcomes. In addition, publication bias was assessed by using Doi plot and the asymmetry of Doi plot was examined by LFK index. 20 The Doi plot is more sensitive than the funnel plot if less than 10 studies are included. However, the publication bias could not be assessed if there are less than three studies included in the meta-analysis. All statistical analyses were performed using Review Manager (RevMan) version 5.3 and MetaXL.
Results
Study selection and characteristics
Our search yielded 150 results from Pubmed (n = 23), Embase (n = 87) and the Cochrane library (n = 40). After excluding 60 duplicates, we screened the titles and abstracts of the remaining 90 studies and seven were retrieved for a full-text review. Finally, four studies10–12,21 fulfilled the inclusion criteria and were included in this meta-analysis (Figure 1). All studies10–12,21 had a multicenter design (Table 1). Three RCTs10–12 were phase III studies and one RCT 21 was a phase II study. Two studies focused on cIAI,11,21 while one focused on cUTI 10 and one focused on nosocomial pneumonia. 12 The study by Kollef et al. 12 used high dose ceftolozane-tazobactam at 3 g every 8 h, while the other three RCTs10,11,21 used 1.5 g ceftolozane-tazobactam every 8 h. Meropenem (three studies) and levofloxacin (one study) were used as the comparator treatments.
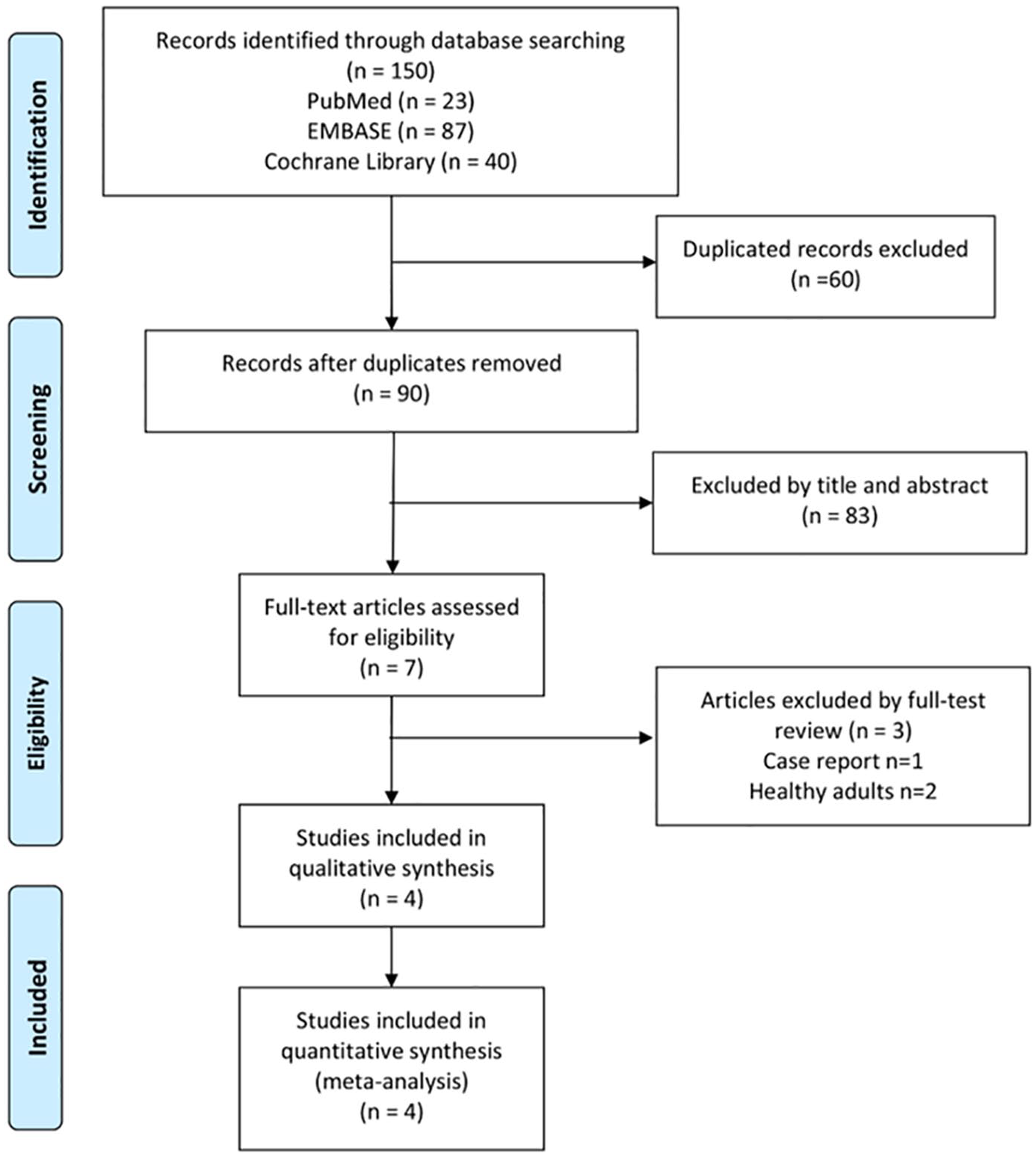
Algorithm for screening and identifying studies.
Characteristics of included studies.

cIAI, complicated intra-abdominal infection; cUTI, complicated urinary tract infection; HAP, hospital-acquired pneumonia; VAP, ventilator-associated pneumonia.
Overall, a total of 2924 patients (1475 in the ceftolozane-tazobactam group and 1449 in the control group) were included in this meta-analysis. Each study was classified as having a low risk of bias for all domains considered for analysis (Figure 2).
Risk of bias summary.
Risk of AEs
Overall, the rate of TEAEs was 51.3% (748/1458) in the ceftolozane-tazobactam group, which was comparable with the control group at 49.9% [714/1430; OR, 1.06; 95% confidence interval (CI), 0.91–1.25; I2 = 0%; Figure 3]. In addition, no significant difference was observed between ceftolozane-tazobactam and the control group for the risk of serious AEs (OR, 1.21; 95% CI, 0.91–1.60; I2 = 16%; Figure 3) and the risk of discontinuation of the study drug due to AEs (OR, 0.85; 95% CI, 0.55–1.33; I2 = 0%; Figure 3). The rate of all-cause mortality did not differ significantly between the ceftolozane-tazobactam and the control groups (OR, 1.11; 95% CI, 0.82–1.50; I2 = 0%; Figure 3). Only two RCTs reported the rate of treatment-related AEs, and the pooled analysis for these two RCTs showed that the risk of treatment-related AEs was similar between the ceftolozane-tazobactam and control groups (OR, 055; 95% CI, 0.07–4.05; I2 = 92%; Figure 3).

Forest plot of risks of adverse events (AEs).
In the subgroup analysis, which compared ceftolozane-tazobactam and meropenem, no significant difference was observed for the risk of TEAEs (OR, 1.10; 95% CI, 0.89–1.35; I2 = 0%), serious AEs (OR, 1.29; 95% CI, 0.97–1.71; I2 = 10%), discontinuation of the study drug due to TEAEs (OR, 0.85; 95% CI, 0.55–1.33; I2 = 0%) or all-cause mortality (OR, 1.10; 95% CI, 0.81–1.49; I2 = 0%.). In addition, the pooled analysis of the three phase III RCTs revealed that a similar trend was observed between ceftolozane-tazobactam and alternative antibiotics in terms of the risk of TEAEs (OR, 1.06; 95% CI, 0.90–1.25; I2 = 0%), serious AEs (OR, 1.19; 95% CI, 0.94–1.51; I2 = 0%), discontinuation of the study drug due to TEAEs (OR, 0.85; 95% CI, 0.55–1.33; I2 = 0%), and all-cause mortality (OR, 1.09; 95% CI, 0.81–1.48; I2 = 0%).
Regarding specific AEs, nausea was the most common AE (5.3%), followed by diarrhea (4.6%), headache (4.2%), anemia (4.0%), and pyrexia (3.9%). However, no significant difference was observed between ceftolozane-tazobactam and the control groups for the risk of elevated aspartate aminotransferase (AST), alanine aminotransferase (ALT), γ-glutamyl transferase (GGT), anemia, atrial fibrillation, diarrhea, dizziness, headache, hypokalemia, hypertension, ileus, insomnia, nausea, urinary tract infection, vomiting, or wound dehiscence (Table 2). The only exception was the risk of Clostridiodes difficile colitis, where ceftolozane-tazobactam was associated with a significantly higher risk compared with the control group [0.72% (10/1376) versus 0.14% (2/1391), OR, 3.84; 95% CI, 1.23–11.97; I2 = 0%] (Table 2).
Risk of specific AEs.
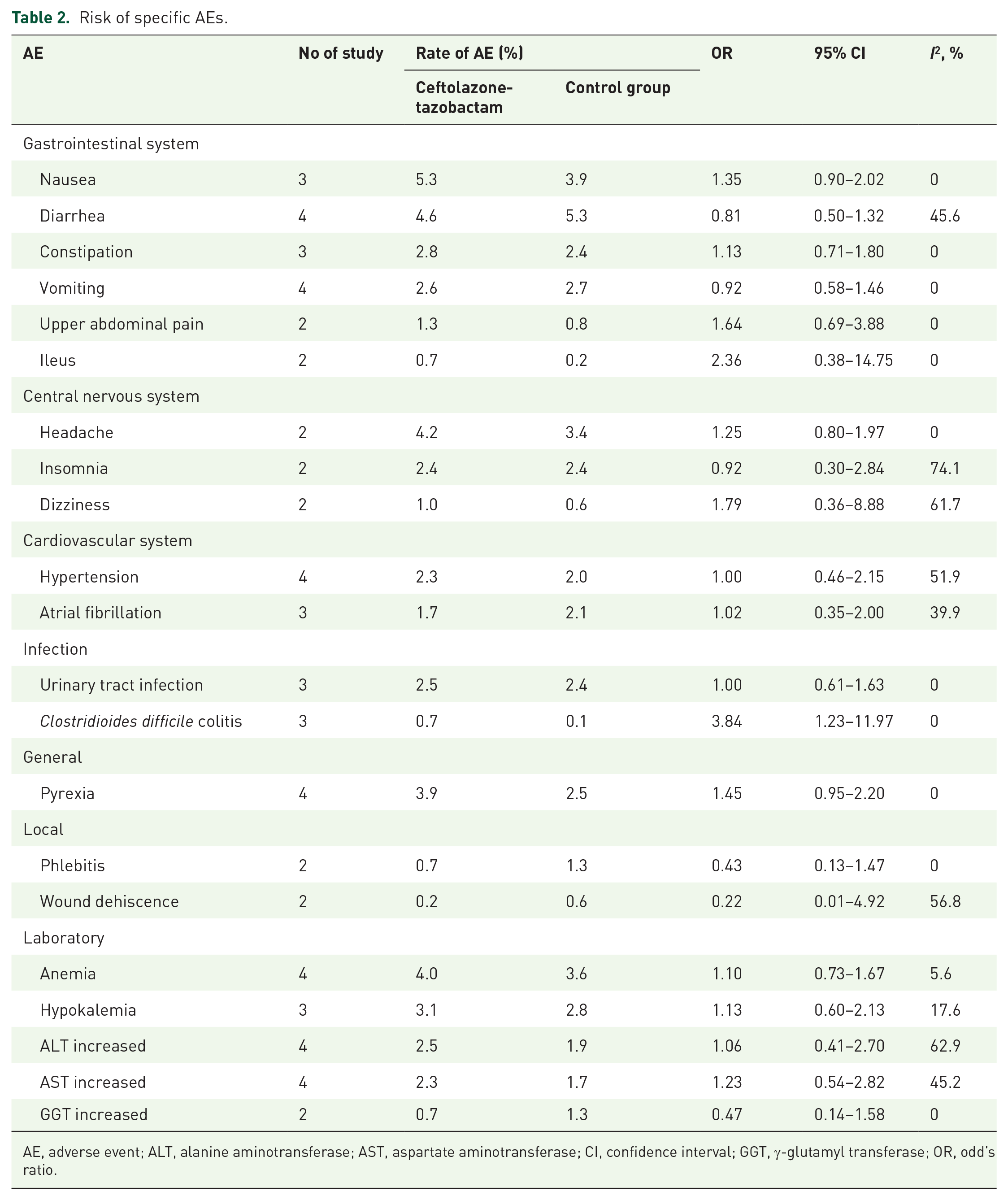
AE, adverse event; ALT, alanine aminotransferase; AST, aspartate aminotransferase; CI, confidence interval; GGT, γ-glutamyl transferase; OR, odd’s ratio.
Sensitivity analyses
Leave-one-out sensitivity analyses were performed to examine whether a single study may have any effect on the pooled results when included studies were removed one at a time. The results showed that no study had significant influences on all primary outcomes, except for TRAEs, where only two studies were included with opposite direction of association (Table 3).
Leave-one-out sensitivity analyses for AEs.

AE, adverse event; OR, odd’s ratio
Publication bias
There was minor asymmetry for treatment-emergent AEs (LFK index = 1.34) and serious AEs (LFK index = 1.03), and major asymmetry for all-cause mortality (LFK index = 7.42) (Figure 4), which indicated moderate or substantial publication bias for primary outcomes. Publication bias was not assessed for the outcomes – the risk of discontinuation of the study drug due to an adverse event and TRAEs because only two studies were included.

Publication of bias according to the outcome of (a) treatment-emergent adverse events (AEs), (b) serious AE, and (c) major asymmetry for all-cause mortality.
Discussion
This meta-analysis of four RCTs10–12,21 demonstrated that the safety of ceftolozane-tazobactam is comparable to that of alternative treatment options for patients with acute bacterial infections; this is supported by the following evidence. Firstly, in the pooled analysis of four RCTs,10–12,21 the overall risk of TEAEs, serious AEs, discontinuation of the study drug due to TEAEs, treatment-related AEs, and all-cause mortality for ceftolozane-tazobactam were similar to that of the comparison drug. This finding was consistent with previous meta-analysis with only three RCTs investigating the clinical efficacy and safety of ceftolozane-tazobactam.22,23 Secondly, no significant differences were observed in the subgroup analysis of only phase III trials10–12 that compared ceftolozane-tazobactam with alternative antibiotics and only RCTs11,12,21 that compared ceftolozane-tazobactam with meropenem. Thirdly, the risks for almost all specific AEs, except for the risk of C. difficile colitis, were similar between ceftolozane-tazobactam and the alternative treatment options. Finally, the overall risk of TEAEs, serious AEs, and discontinuation of the study drug due to TEAEs were 15.7%, 2.7%, and 8.2%, respectively. Although the overall all-cause mortality rate was 8.2% (n = 120) for ceftolozane-tazobactam, most of the deaths were from the study by Kollef et al., 12 which included patients with nosocomial pneumonia, in which the mortality rate was 29.1% (n = 105). 12 In contrast, only 15 deaths were reported in the three other studies10,11,21 for patients with cIAI and cUTI, and the pooled mortality was only 1.4%. These findings are consistent with those of previous retrospective studies,8,9 in which ceftolozane-tazobactam treatment was well tolerated for the treatment of acute bacterial infection. A multicenter, open-label, non-comparative study 8 investigated the usefulness of ceftolozane-tazobactam plus metronidazole in 100 Japanese patients with cIAI. It revealed that the rate of TEAEs, serious AEs, and drug-related AEs were 62%, 10%, and 19%, respectively; however, no drug related serious AEs were observed and no patients discontinued the drug due to an AE. Another nonrandomized, multicenter, open-label study 9 assessed the usefulness of ceftolozane-tazobactam in 114 Japanese patients with cUTI. It reported that the rate of TEAEs and drug-related AEs were 57.8% and 17.5%, respectively. AEs leading to ceftolozane-tazobactam discontinuation were observed in two patients, one patient with a moderate headache and the other with mild hepatic function abnormality, but no drug related serious AEs or death were reported. In summary, these findings suggest that ceftolozane-tazobactam is a tolerable therapeutic option for the treatment of acute bacterial infections.
In contrast to previous meta-anlaysis,22,23 the present meta-analysis did a comprehensive investigation about the risk of each specific AE. In this meta-analysis, the gastrointestinal system was the most common system affected by AEs. The most common AE was nausea (5.3%) while the frequencies of all other AEs were <5%. Most importantly, most AEs in the included studies were mild to moderate.10–12,21 In the study by Solomkin et al., 11 the most common laboratory AEs were increased ALT and AST, which occurred in 2.5% and 1.6% of all patients, respectively. In the study by Wagenlehner et al. 10 the most common AEs were mild to moderate, and the incidence of treatment-limiting adverse events was <2% in each treatment group, while no laboratory abnormality resulted in an AE that led to premature discontinuation of the study drug. In the study by Kollef et al., 12 the most commonly reported treatment-related AEs in the ceftolozane–tazobactam group were abnormal liver function tests (3.3%, n = 12), C. difficile colitis (1.1%, n = 4), and diarrhea (1.1%, n = 4). In the single arm study by Arakawa et al., 9 the most common drug-related AEs were diarrhea and ALT increase (5.3% each), while AST increased in 3.5% of patients. In another single arm study by Mikamo et al., 8 the most common drug-related AEs were AST increase (11.0%), ALT increase (9.0%), diarrhea (3.0%), and nausea (2.0%). All cases of ALT or AST increase were resolved by the end of the treatment or during the study. Based on the findings of observational studies, Maraolo et al. 24 conducted a systematic review of the efficacy and safety of off-label use of ceftolozane-tazobactam in the treatment of a total of 130 difficult-to-treat Pseudomonas aeruginosa infections and found that only five AEs occurred among 48 available cases, including hypokalemia (n = 2), and each one of C. difficile infection, rash, and elevation of liver enzyme. In summary, gastrointestinal AEs and abnormal liver function were the most common AEs associated with ceftolozane-tazobactam, but most of them were mild to moderate.
In this meta-analysis, we found that C. difficile infection could be a concern when ceftolozane-tazobactam is administered. In the study by Solomkin et al., 11 ceftolozane-tazobactam-related serious AEs due to C. difficile infection were reported in one patient (0.21%). In the study by Kollef et al., 12 ceftolozane-tazobactam related C. difficile colitis was reported in four patients (1.1%), while in Wagenlehner et al.’s study 10 two serious AEs (0.38%) due to C. difficile infection were reported in the ceftolozane-tazobactam group, and these were deemed to be associated with the study treatment. These two patients had recovered by the follow-up. In Maraolo et al.’s 24 systematic review, the risk of C. difficile infection was 2.1% (2/48). 24 Although none of the C. difficile infections were reported to cause death, the pooled analysis of these three RCTs showed that the overall risk of C. difficile colitis was 0.7%, which was significantly higher than in the comparative treatments. As a result, clinicians should keep alert to the risk of C. difficile colitis when treating patients with ceftolozane-tazobactam.
A limitation of the current study was that only four RCTs were considered within the meta-analysis, and only three types of acute bacterial infections (pneumonia, cUTI, and cIAI) were included. In addition, four RCTs included different patients’ population and control groups are not all the same. Further large-scale research is warranted to assess the usefulness of ceftolozane-tazobactam as a treatment.
In conclusion, the safety profile of ceftolozane-tazobactam does not significantly differ from that of comparator drugs for the treatment of acute bacterial infections in adult patients. However, it has an increased risk of C. difficile infections. As a novel broad-spectrum antibiotic, ceftolozane-tazobactam could be a safe therapeutic option for use in common clinical practice.
Footnotes
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Cardinal Tien Hospital (CTH106A-2A18, CTH108A-2A28, CTH109A-2209, and CTH109A-2210).
