Abstract
Background:
The most common cause of persistent hypoglycemia in infancy is hyperinsulinemic hypoglycemia. When conservative measures fail, providers often use medications to treat persistent hypoglycemia. Diazoxide is first-line therapy for neonatal hypoglycemia and works by inhibiting insulin secretion. Diazoxide is associated with fluid retention, and less commonly with respiratory decompensation and pulmonary hypertension. Case reports documenting these severe adverse events exist in the literature, although the overall incidence, risk factors, and timing for these effects in a newborn are not clearly defined.
Methods:
We performed a retrospective chart review of all infants admitted to the neonatal intensive care unit (NICU) at Regional One Health from 1 January 2013 until 15 August 2019, who received diazoxide as a treatment for persistent hypoglycemia secondary to hyperinsulinism. Patients were stratified as either having no adverse event or having an adverse outcome to the medication. A severe adverse outcome was defined as any known major side effect of the medication, which a patient developed within 2 weeks of medication initiation that led to medication discontinuation.
Results:
From our pharmacy database, we identified a total of 15 babies who received diazoxide for persistent hypoglycemia. Of these patients, eight (53%) were classified as having a complication requiring discontinuation of the medication. Six out of eight patients required intubation with mechanical ventilation and five out of eight patients developed pulmonary hypertension. All patients returned to their baseline respiratory support after drug discontinuation.
Conclusions:
A total of 53% of our study population had an adverse outcome to diazoxide. Previous studies suggest 5% of patients may have respiratory decompensation and require ventilatory support while on diazoxide; however, 40% of our patients deteriorated and then required mechanical ventilation. Based on our data, respiratory deterioration may be more likely to occur when diazoxide is used in preterm infants, those with lower birth weight and intrauterine growth restriction.
Plain language summary
Newborns could experience a transient period of low blood glucose levels soon after birth. However, some may progress to persistent low blood glucose levels that cannot be controlled with adequate glucose infusion and may require other ways of treatment. Diazoxide is the first-line drug approved by the US Food and Drug Administration (FDA) for this condition. However, certain cases have reported the development of respiratory deterioration, including increased blood pressure in lung circulation after its use. This prompted a black box warning in 2015 by the FDA. The incidence of neonatal low blood glucose levels seems to have increased and so has the use of this drug. Our study identifies 15 newborns who received diazoxide at Regional One Health neonatal intensive care unit in the past 6 years and reports a significantly higher rate of adverse events in our population leading to drug discontinuation in almost 53% of our cases.
Keywords
Introduction
Newborns experiencing a transient period of hypoglycemia soon after birth are not uncommon. It occurs within the process of independent glucose regulation during the transitional period from the intrauterine environment.1,2 Neonatal hypoglycemia that persists beyond the transitional period or occurs for the first time past the initial days of life is most often the result of underlying hyperinsulinism. Hyperinsulinism could pertain to genetic defects or can be transient secondary to perinatal stress. 3 This hyperinsulinemic hypoglycemia (HH), if left untreated, could seriously affect cognitive and functional neurological development in their early stages due to deprivation of glucose as the necessary brain fuel.4,5 In addition, the inhibitory effect of insulin on lipolysis and ketogenesis prevents the formation of alternative brain substrates. Thus, early diagnosis and timely intervention are crucial. 3
The immediate effective treatment strategy would be to achieve normal plasma glucose (PG) concentration through adequate feeding and/or intravenous dextrose infusion. Sometimes the suppression of insulin secretion may be required to achieve normoglycemia. Diazoxide is the US Food and Drug Administration (FDA) approved first-line drug therapy indicated in HH when PG is not stable on conservative feeds or fluids. It primarily targets the K+ ATP channels at β cells and blocks the secretion of insulin hormone. 6 Since it has been approved for HH, it is usually well tolerated except for some common side effects of hypertrichosis, edema, and electrolyte imbalance.7,8 However, in the past decade, some cases have been reported suggesting severe adverse events (SAEs) including pulmonary hypertension, respiratory decompensation, necrotizing enterocolitis9,10 and congestive heart failure related to the use of diazoxide specifically in neonates.11,12 In 2015, the FDA issued a drug safety black box warning. 13 They placed specific emphasis on the risk of pulmonary hypertension in patients with risk factors such as neonates with meconium aspiration syndrome, pneumonia, sepsis, and congenital heart disease. SAEs such as pulmonary hypertension and cardiopulmonary decompensation are reported in the range of 2.4–9% in the larger referral center studies.14–16 Due to the rarity of this adverse event, the data that describe the association of SAEs with diazoxide dose, duration, and patient demographics are very limited.11,17,18
Certain distinctive findings suggest a higher incidence of SAEs in neonates with perinatal stress hyperinsulinism than in those with genetic forms of hyperinsulinism (16.7 versus 3.6%; p = 0.01). 16 Comparably low birth weight (LBW) and small for gestational age (SGA) infants were found to be at higher risk. 15
Considering all this, diazoxide use has significantly increased over the past two decades and so has the rate of severe cardiorespiratory adverse events. 19 The purpose of our report was to review the use of diazoxide in neonates for persistent hypoglycemia in the past 6 years at our neonatal intensive care unit (NICU) and review the pulmonary, circulatory, and glycemic outcomes observed at our facility specifically in the very low birth weight population.
Methods
In this retrospective observational study, we reviewed the records of infants admitted to our NICU who received diazoxide for the treatment of persistent hypoglycemia, from 1 January 2013 to 15 August 2019. After obtaining institutional review board approval from the University of Tennessee Health Science Center (approval number 19-06822-XP), we identified the subjects, and data pertaining to the study were collected through an electronic medical record system, using the unique medical record number of those patients. The data included patients’ demographics, birth history, the reason for hypoglycemia, diazoxide dosage and duration, respiratory support before and after the diazoxide use, echocardiogram findings before and after the medication usage, development of SAEs, and mortality. A severe adverse outcome was defined as any known major side effect of the medication, which a patient developed within 2 weeks of medication initiation that led to medication discontinuation.
For the comparative analysis, we stratified the patients into two groups: one group of patients having no adverse events on the medication and the other group of patients having a SAE after medication. By using the chi-square test of independence and student’s t-test, we compared patient demographics, birth history, medication cumulative dose, and duration between both groups. Statistical significance was set at the p-value of 0.05, the analysis was done using SPSS version 26 for Windows.
Results
During the study period, we identified 15 newborn babies who received diazoxide during their birth hospitalization in the NICU for persistent hypoglycemia. The general and comparative demographics are described in Table 1. The average day of life the diazoxide was started was 33.7 ± 34. The average duration of therapy was 18.4 ± 16 days and ranged from 2 to 55 days. All patients received the starting dose of diazoxide within the dose range of 2.5–15 mg/kg/day.
Comparing the patient demographics and diazoxide dose-dependent variables between the groups of neonates with complications versus those without complications.

SD, standard deviation, bold indicate p<.05
Out of 15 neonates, eight babies (53%) developed SAEs related to diazoxide. The other seven (47%) babies had no adverse events on the medication, five of which tolerated the medication well and were discharged home on diazoxide. One out of the other two had refractory HH) not controlled by diazoxide and thus was transferred to a specialty center. All babies survived up to hospital discharge. The main adverse events during hospital stay were pulmonary hypertension (5/8), respiratory failure (6/8) and suspected necrotizing enterocolitis (NEC) with abdominal distension (2/8) which resolved after discontinuation of the medication and supportive management. Sixty per cent (9/15) of newborn babies developed a clinical mild to moderate level of edema within 7 days after the use of diazoxide. A total of four patients were treated with diuretics while on diazoxide. Three of the eight patients with adverse outcomes were on scheduled diuretics (chlorothiazide or furosemide) throughout the duration of their diazoxide therapy and still developed respiratory failure and signs of fluid overload.
Two patients required inhaled nitric oxide (iNO) within 2 weeks of diazoxide use for severe pulmonary hypertension. They were diagnosed with aneuploidies (trisomy 13 and 21). Figure 1 demonstrates the duration (7–13 days) within which the patients required a higher level of respiratory support from minimal to no support after diazoxide initiation. Of the six patients with respiratory failure who required intubation and mechanical ventilation due to respiratory decompensation, four patients were in room air before diazoxide was initiated. Diazoxide was discontinued in all these six patients soon after they developed respiratory deterioration, and five of them eventually returned to room air within 9 to 54 days after discontinuation of therapy. One patient with trisomy 21 did not improve with respiratory support right away and was transferred to a higher center for g-tube placement due to dysphagia. An echocardiogram was performed in only 10 of 15 patients before the start of diazoxide therapy. While nine out of 15 patients had echo within 2–3 weeks after the start of the diazoxide, seven of these patients were the ones who developed cardiorespiratory adverse events from diazoxide. Echo findings of moderate to large size of patent ductus arteriosus (PDA) (50% versus 29%), right ventricular hypertrophy (RVH) (63% versus 0%), atrial septal defect (ASD), and ventricular septal defect (VSD) (38% versus 14%) were more common in patients who developed cardiorespiratory adverse events from diazoxide than the group of babies with no SAEs on diazoxide. Echo performed after the beginning of diazoxide was also consistent with right ventricular dilation in 71% (5/7) and left ventricular dilation in 43% (3/7) in patients who developed respiratory and cardiac decompensation. All the patients who developed pulmonary hypertension clinically also had positive echo findings for the same.
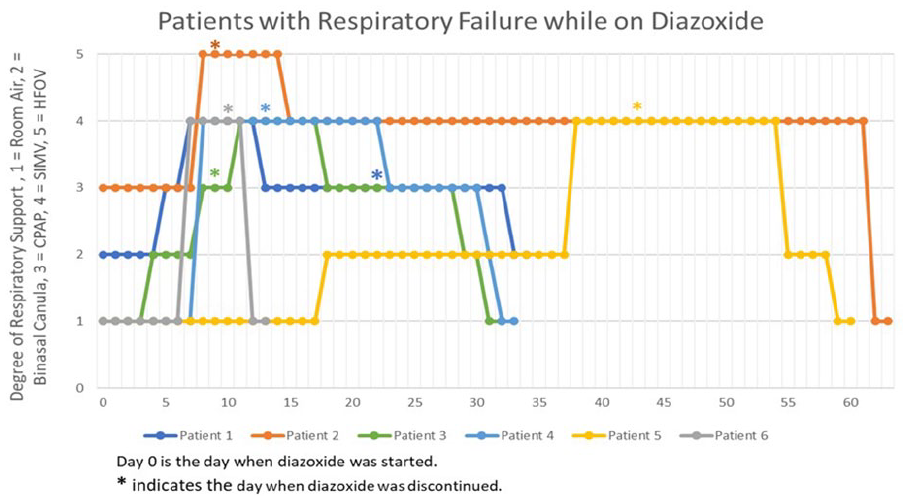
The timeline shows the change in respiratory support in infants after the initiation and discontinuation of diazoxide.
Adverse outcome versus benign course
For comparative analysis, we divided our study population into two groups. One group included patients who developed diazoxide induced complications and the other group included patients whose condition remained uneventful on diazoxide. Table 1 compares the patient demographics, birth history, and diazoxide-related variables between both groups. The neonates who developed severe adverse outcomes after diazoxide therapy had significantly lower birth weight and gestational age and a higher rate of intrauterine growth restriction (IUGR). There was no difference in the cumulative dose, maximum dose, or duration of diazoxide therapy between both groups.
Discussion
HH is a major cause of persistent and recurrent hypoglycemia of the neonatal period, and often requires prompt management to prevent long-term brain injury. 3 This dysregulation of insulin secretion could either be due to a primary congenital genetic defect or secondary to risk factors such as intrauterine growth retardation, perinatal stress, maternal diabetes, etc. Transient hyperinsulinism, the term used to describe HH developing secondary to perinatal or maternal risk factors, often persists longer than expected and requires medical treatment with diazoxide.20–22 In our study sample, no patient was found to have any known genetic defects for HH and most of them had idiopathic congenital hyperinsulinism (6/15) or persistent transient hyperinsulinism (9/15).
Regardless of the type of HH, diazoxide is the first-line FDA approved therapy for the treatment of hypoglycemia. Historically, the drug was used for the treatment of severe hypertension, and due to its side effect of hyperglycemia, was later discontinued from the line of antihypertensive agents. In neonates, the common side effects of diazoxide are hypertrichosis, fluid overload/edema, and electrolyte imbalance which usually resolves after discontinuation of the drug. Thus, the concurrent use of diuretics, mainly chlorothiazide, is advocated with the use of diazoxide for fluid restriction. Experts recommend the use of a diuretic in every patient who receives diazoxide to prevent the effects of cardiorespiratory fluid overload; 15 however, the use of diuretics was selective involving only four of our patients. In the group of patients who developed SAEs, three of eight patients received diuretics throughout the period of time that they developed respiratory deterioration and fluid overload. However, they did not show any signs of improvement until the diazoxide was discontinued. In addition, similar to the findings from the existing literature, the development of edema was not dose dependent. 15
Pulmonary hypertension (PH) is a major serious adverse event recently identified with diazoxide use in infants. After the reported cases illustrating the severity of drug-dependent PH, the FDA issued a black box warning for diazoxide use and advised close monitoring in infants at risk of PH; mainly neonates with meconium aspiration syndrome, respiratory distress syndrome, transient tachypnea, pneumonia, sepsis, congenital diaphragmatic hernia or congenital cardiac disease.13,18 Since then, evidence in the literature describing severe pulmonary and cardiac adverse effects of diazoxide has been growing, indicating the incidence of PH is more common than previously thought. However, the supportive published studies are mainly institutional case series and case reports. 11 In our study, there were more infants with bronchopulmonary dysplasia with adverse outcomes. Underlying lung disease may increase the risk for PH with diazoxide use. This group of infants should be monitored carefully.
The incidence of PH after the use of diazoxide is reported to be from 2.4% to 7% by studies from larger institutions or subspecialty institutes where HH infants are more commonly managed.14,15 However, in our study population, 33% (5/15) of patients developed PH while 40% (6/15) of total patients developed respiratory failure. Our population is at high risk, including two babies with aneuploidy. Patients with trisomy 13 tend to show higher insulin levels at hypoglycemia.23,24 In our small cohort, this may lead to an increased rate of SAEs compared to other studies. A recently published single largest cohort study, describing the use of diazoxide in newborn infants in the NICU setting, has demonstrated a significantly increasing trend in the use of the drug for hypoglycemia from 1997 to 2016. 19 Serious concerns of respiratory adverse events from diazoxide have been raised but are limited, with minimal evidence about the patients’ risk factors. Thus, it becomes crucial to use caution for diazoxide use in the NICU where HH is not frequently treated. Neonates who developed SAEs from diazoxide use at our center were found to have significantly lower birth weight and a higher prevalence of IUGR and SGA compared to the group of neonates with an uneventful hospital course. Respiratory adverse events related to diazoxide also have a close association with structural heart disease. Echocardiography before the start of the diazoxide treatment is recommended, but it was done in 10/15 (67%) of cases in our study. Another study has documented the use of echo in about 88% of cases. 15 The development of pulmonary hypertension has been closely correlated with the presence of congenital cardiac abnormalities supported by the echo findings in our study population as well. The literature suggests that patients who develop pulmonary hypertension frequently had potential risk factors, including respiratory failure and structural heart disease. 15
The American Pediatric Endocrine Society recommends early evaluation of plasma glucose within 48 h of age so that the period of transitional glucose regulation has passed and persistent hypoglycemia could be excluded before the discharge. 25 As hyperinsulinism is the most common form of hypoglycemia in neonates, diazoxide is being used more frequently in the setting. 19 However, regarding preterm neonates and especially IUGR, careful consideration should be given before the initiation of diazoxide. Diazoxide toxicity leading to cardiac and pulmonary decompensation remains a concern. As we found in our study, other studies also support the higher risk of the development of respiratory and cardiac adverse events in newborns with SGA, IUGR, and LBW.14,15,17 One of our study babies developed NEC while on diazoxide, which demonstrates questionable significance. A recently published study indicated a possible association between exposure to diazoxide and the development of NEC. Interestingly, the risk of NEC associated with diazoxide usage was increased in IUGR babies and developed within the first week of drug use as in our patient.9,10
Our high-risk NICU cohort included two babies with aneuploidy. Patients with trisomy 13 tend to show higher insulin levels at hypoglycemia.23,24 In a small cohort this may lead to an increased rate of SAEs compared to other studies. Also, diazoxide is highly protein bound in the body and low serum protein may increase free diazoxide. It has been speculated that low serum protein combined with poor renal function may lead to increased side effects of diazoxide.14,26 Although in our small cohort the serum protein and creatinine levels were not statistically different at the time of initiation of therapy.
Apart from the important findings and concerns that our study reports, it also has some limitations. It is a retrospective study, so data gathering was limited to the chart review. Our study sample size was small with a higher rate of SAEs (53%) reported, which could potentially misinterpret the actual population. A study published from Philadelphia Children’s Hospital data described a nearly higher incidence rate (16.7%) of SAEs in neonates with perinatal stress hyperinsulinism in those with risk factors of IUGR, LBW, and SGA. 16 Our study center, Regional One Health NICU, infrequently treat such patients with diazoxide. A larger randomized controlled trial or prospective study and research on conservative management of HH especially in perinatal stress-related hyperinsulinism would be important evidence for future consideration.
Conclusions
Diazoxide use has been increasing especially in neonates with persistent hypoglycemia secondary to hyperinsulinism. 19 Cases related to severe adverse effects on pulmonary hypertension and respiratory deterioration have increased in general. A total of 53% of babies developed SAEs from diazoxide in our study population and interestingly they were more prevalent in those with SGA and IUGR. Special consideration and close monitoring must be followed whenever diazoxide is used in these patients.
