Abstract
With recent increase in the use of direct oral anticoagulants (DOACs), several new cases of adverse drug reactions (ADRs) have been identified in pharmacovigilance surveys. These ADRs can result in significant mortality and morbidity if not identified and treated promptly. It is important for physicians to recognize that immunologically mediated delayed hypersensitivity reactions, although rare in occurrence, can have significant impact on patient’s quality of life. To the best of our knowledge, we report the first case of lichenoid eruption associated with apixaban. We further provide evidence of tolerance to rivaroxaban in the same patient.
Plain language summary
Well documented case reports, although providing evidence of probable causal relationship between a drug and specific adverse drug reactions (ADRs), can increase awareness amongst clinicians treating patients with direct oral anticoagulants (DOACs), especially with its rapid utilization. Rare ADRs are difficult to detect as clinical trials of DOACs lacked enough patient sample, making post-marketing reporting of such events important so both patients and clinicians can be vigilant to help with prompt recognition of such symptoms. We report the first case of lichenoid eruption hypersensitivity reaction associated with apixaban in patient with tolerance to rivaroxaban.
Keywords
Introduction
Direct oral anticoagulants (DOACs) have become the preferred choice of anticoagulant for treatment of atrial fibrillation (AF) and venous thromboembolism (VTE) due to less frequent monitoring, reduced drug–drug interactions, simplified dosing, and less intracranial bleeding as compared with warfarin.1–3 Adverse drug reactions (ADRs) associated with anticoagulation use have been estimated to range between 0.01% and 7.5%. With the rise in the utilization of DOACs, several hypersensitivity reactions have become increasingly apparent in post-marketing surveillance.4–6 Rivaroxaban and dabigatran have been the main culprits amongst the DOACs, as these were the first two in the drug class to become available on the market. There have been at least 16 hypersensitivity reaction cases related to rivaroxaban and 8 cases associated with dabigatran, as compared with only a few cases reported for apixaban in the literature. 7
According to the package insert for apixaban, the incidence of skin rash and anaphylactic reactions, such as angioedema, are reported in <1% of patients receiving treatment. 8 Availability of case reports describing apixaban-induced hypersensitivity reactions are limited, and very few, if any, describe experience on diagnostic or biopsy tests.9–11 In addition, the cross reactivity across DOACs as a class is also unknown. To the best of our knowledge, there is currently no reported case of lichenoid eruption induced by apixaban. We further provide evidence of tolerance to rivaroxaban in the same patient.
Case report
A 78-year-old male with past medical history significant for atrial fibrillation (AF) with CHA2DS2VASC score of 2, HAS-BLED score of 1, and rheumatoid arthritis well controlled on etanercept therapy since 1997, was referred to an anticoagulation clinic pharmacist for management of apixaban 3 months ago. The patient’s allergy history was pertinent for developing rash to fluoroquinolones and Stevens Johnson Syndrome to trimethoprim/sulfamethoxazole. The patient presented to the emergency department at Salem Veterans Affair Medical Center (SVAMC) with a chief complaint of rash on both his upper extremity (UE), primarily forearms, and torso that started about 1 month previously. The patient denies any changes in clothing, detergents, or contact with chemicals. No oral or genital lesions were found. No other new medications were added recently, and no other medication dosage changes were noted. Physical examination revealed the presence of erythematous excoriated patches on the upper chest area, and violaceous patches with lacy scales on the left UE, while right UE exam showed more atrophic patches with little scales. At this time, the patient was diagnosed to most likely have drug-induced lichen planus with the possibility of apixaban being the culprit agent, and was offered biopsy for further evaluation. The patient was prescribed steroid ointment (clobetasol propionate 0.05% ointment) to see if this helped alleviate his symptoms, and biopsy was scheduled in 1 month. The patient was instructed to discontinue steroid ointment 1 week prior to the biopsy. Physical examination at a follow up visit revealed that steroid ointment had cleared some of the rash, but did not completely resolve, and the patient reported no itching. The patient had new violaceous macules on the left side of the neck, while keratotic lesions had appeared on the right clavicular area. Several violaceous macules with lacy scales were noted on both UE. At this visit, two biopsies were obtained: shave biopsy of the right clavicular area and right forearm, and 3.5 mm punch biopsy from the left upper arm. Pathology report (Figure 1) revealed compact orthokeratosis, a thin atrophic epidermis with leukocytosis, and chronic interface dermatitis with superficial lymphohistiocytic inflammatory infiltrate with melanophages that extended focally to the mid-dermis level. Vacuolar alterations and several Civatte bodies in the epidermis and along the dermoepidermal interface was consistent with lichenoid eruption; however, atrophic lichen planus cannot be ruled out. Using further clinical correlation with patient-specific factors, it was determined that lichenoid drug eruption was the most probable diagnosis. The patient was advised to resume steroid ointment but avoid the biopsy sites, and to return in 6 weeks. The patient reported some relief with continual use of steroid ointment at this follow up. He still had rash on chest and forearms, with some hypopigmented patches where he had the inflammation before. However, the patient denied itching. He was advised to continue topical steroids at this time for an additional 2 weeks. A month after this encounter, the patient presented with resolution of previously developed rash; however, a new erythematous irregular and rough lesion had appeared on the right forearm. Per patient, the rash that had resolved with steroid use seemed to be coming back since he stopped using it 2 weeks ago. Around the same time, the patient also presented to the Ear, Nose and Throat (ENT) department for a left side cervical adenopathy, which was noted as a necrotic left submandibular gland mass with left level II and III adenopathy. He underwent submandibular gland excision, for which he had to stop apixaban for 5 days before the procedure, and reported almost complete resolution of rash during this time. The rash reappeared after apixaban was resumed post-surgery; the patient self-discontinued it after 2 weeks and called the primary care physician to inform of this. Several attempts were made to contact the patient to switch to another DOAC; however, the anticoagulation clinic pharmacist was unable to reach him. Almost 4 weeks after submandibular excision surgery, the patient was seen in the anticoagulation clinic, where he was then switched to rivaroxaban. At this visit, the patient reported complete resolution of the rash developed secondary to apixaban exposure post-surgery. At the subsequent anticoagulation clinic follow up at ~2 months, the patient reports continued tolerance to rivaroxaban and denies any rash or itching symptoms. For graphic representation, a timeline review of the patient case is shown in Figure 2.
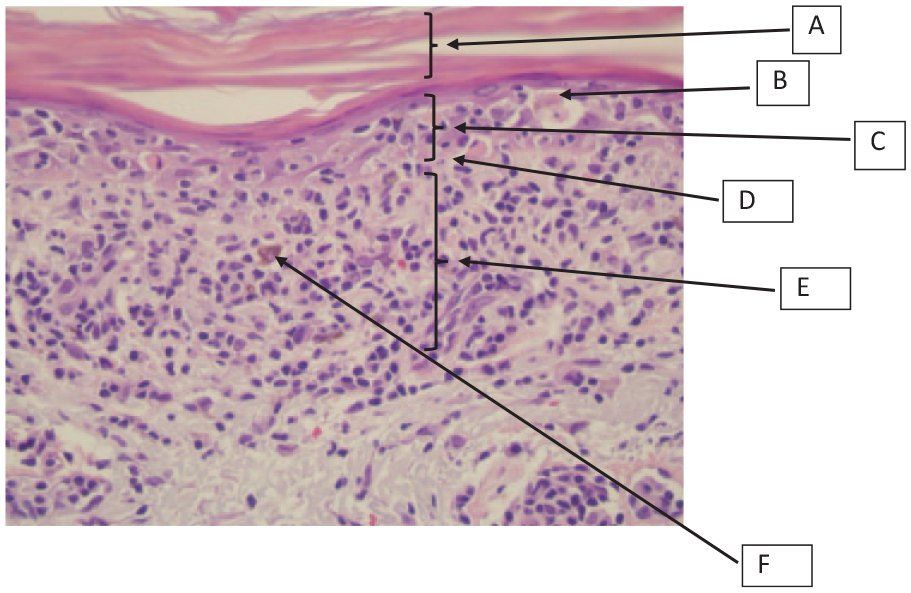
Skin biopsy histopathology revealing lichen planus type-drug reaction-atrophic variant.

Timeline review of patient case.
Discussion
DOACs constitute >60% of currently prescribed anticoagulants, and have replaced warfarin for the treatment of AF and VTE.12,13 Hypersensitivity drug reactions associated with anticoagulants are being increasingly reported in the literature. 14 Symmetric eruption of violaceous papules on the trunk and extremities with possible lacelike patterns are highly characteristic of drug-induced lichen planus resembling our patient case and is a relatively uncommon cutaneous drug reaction. The time to onset of this type of drug reaction is reported as ranging from a few months to a year or more depending on the offending drug type. 15 Whereas patch testing may have some role in determining the culprit agent, it is not performed routinely in patients’ experiencing lichenoid drug eruption. Only about 30–50% of patients with cutaneous drug reactions in a small observational study had a positive patch test, confirming that false negative results are frequent.16–18 A case of lichenoid eruption related to ticlopidine has been described in a 67-year-old male. In this case, histopathological findings from skin biopsy showed hypergranulosis and band-like lymphocyte infiltration along the dermal–epidermal junctions indicative of lichen planus resembling the biopsy finding in our case. In addition, the onset of drug reaction was ~2 months in the patient receiving ticlopidine, like our patient case. 19 This is the typical minimum period reported in the literature required to induce lichenoid eruption. 15
A few case reports describing apixaban-induced hypersensitivity reactions have been reported in peer-reviewed literature. Albalbissi and colleagues described a case of cutaneous drug reaction to apixaban in 78-year-old female that appeared 9 days after taking the drug that manifested mainly as a pruritic rash starting on the right upper arm and then progressing to her face 2 days later. No biopsy was performed and patch testing with apixaban, rivaroxaban, and edoxaban was negative; however, challenge was declined, and the patient was treated successfully with warfarin instead. 20 A case of apixaban-induced leukocytoclastic vasculitis manifesting as erythematous rash on lower limbs quickly progressing to purpuric itchy and burning rash, developed 10 days after drug exposure. Histopathology of skin biopsy revealed infiltration of neutrophils around and inside the superficial vascular plexus together with focal fibrinoid vessel wall necrosis in association with erythrocyte extravasation. The patient showed complete resolution with apixaban discontinuation and use of oral steroids. The patient was switched to rivaroxaban, and tolerated it well. 9 In a case study, palmoplantar psoriasiform eruption that started 3 days after drug initiation was linked to apixaban. A biopsy was performed and pathological findings such as keratosis, acanthosis, focal parakeratosis, and necrotic keratinocytes confirmed the finding. The patient showed gradual improvements over 3 weeks after drug withdrawal and use of topical corticosteroids. 10 In one of the cases of apixaban, a 60-year-old woman treated for AF developed rapid worsening of neurologic symptoms of imbalance, vertigo, headache, diplopia, and confusion. These symptoms ceased after the drug was discontinued and the patient returned to her baseline neurological state within 72 h. In this case patient self re-challenged herself to apixaban unknown to her medical team, producing a similar neurologic response, which was again resolved within 24 h of drug discontinuation. This patient was then transitioned successfully to rivaroxaban and tolerated it well. 21
A case study by Cortellini described widespread erythematous lesion and prurigo in a 70-year-old woman secondary to edoxaban with evidence of cross reactivity to apixaban. The patient in this case underwent patch testing prior to switching to apixaban and was found to test positive at 5 days to dabigatran and edoxaban. Although this patient had a negative patch test for apixaban initially, she only tolerated it for 7 days with graded challenge before developing widespread eczematous dermatitis, and was eventually transitioned to warfarin with no further side effects. In support of patch testing, this study recommends crushing whole tablets in a mortar and mixing it with Vaseline at 30%. This concentration appeared to be nonirritant and can be used for diagnosis. However, it failed to identify cross reactivity to apixaban, indicating that a delayed reading of test at more than 5 days may be indicated. 11 Another recently published case report described rivaroxaban-induced urticaria and angioedema, with possible cross reactivity to dabigatran, manifested as rash in a 63-year-old male. 22 There is limited availability of information on the cross reactivity across different DOACs in the literature.
Etanercept has been associated with drug-induced lichen planus.23–25 Given that the patient in our case had been taking it since 1997 without any such side-effects, makes the causal relationship with the observed ADR highly unlikely, and it was ruled out as possible causative agent by the physician. An extensive literature search using PubMed and Google scholar did not yield any lichenoid eruption cases due to apixaban.
The United States Food and Drug Administration (FDA) adverse drug reporting system public dashboard (accessed 14 January 2020) reported 4528 cases of skin and subcutaneous tissue disorder since 2008, classified as rash, pruritus, urticaria, face swelling, angioedema, drug eruption, and other hypersensitivity reactions, with most of those cases reported in the age group of 65–85 years of age. However, the chances of this information being incomplete, inaccurate, and unverified, as stated on this official website, makes a causal relationship between apixaban and ADRs uncertain and difficult to interpret. 26 According to the eHealthme database of 11,904 people who reported side-effects to apixaban, 27 about 160 patients (1.34%) report experiencing rash. Patients who are older than 60 years of age, been taking the drug for 1–6 months, and on concomitant amlodipine seemed more prone to experiencing rash. According to this database, the risk of experiencing rash was ~50% in the first 1–6 months and is reduced to 0% after 1 year of initiating apixaban. In addition, other concomitant symptoms reported by patients who experienced rash include itching (30%), peripheral swelling (14.37%), malaise (13.75%), and diarrhea (13.75%).
To our knowledge, this is the first case of lichenoid eruption associated with apixaban in the literature. This case further includes biopsy to prove the type of hypersensitivity reaction as well as re-challenge to apixaban, further strengthening the causal relationship with the drug. Based on the type of reaction, and the time to its development, we conclude that this was most likely a type IV delayed type cellular hypersensitivity reaction according to the Gell and Coombs classification. 28 Well-documented case reports are an essential component of post-marketing safety data, but they typically suggest a probable reaction based on the Naranjo score and are inadequate for making causal inferences. 29 Using the Naranjo Adverse Drug Reaction Probability Scale (Supplemental appendix A), we calculated a score of 8 points out of 13 in our patient case, placing it in the “probable” causality category. 30
Conclusion
In this report, we described a case of apixaban-induced lichenoid eruption. We further provide evidence of tolerance to rivaroxaban in the same patient case. The exact pharmacologic mechanism by which apixaban causes this type of ADR is unclear; however, as DOACs become increasingly utilized and favored over warfarin because of their superior safety and convenience, post-marketing observations describing such drug hypersensitivity reactions are critical. We believe that our case report highlights a rare ADR associated with the apixaban and can help extend pharmacovigilance by further raising clinicians’ awareness of atypical ADR patterns, avoiding delays in their diagnosis and treatment.
Supplemental Material
Supplement_1_Naranjo_Adverse_Drug_Reaction_Probability_Scale – Supplemental material for A rare case report of apixaban-induced lichenoid eruption
Supplemental material, Supplement_1_Naranjo_Adverse_Drug_Reaction_Probability_Scale for A rare case report of apixaban-induced lichenoid eruption by Tanvi Patil, Suzanne Hanna and Wayne Torre in Therapeutic Advances in Drug Safety
Footnotes
Author contributions
TP (primary author) drafted the initial and final version of the manuscript and was involved in the care of this patient as anticoagulation clinic pharmacist. SH provided intellectual review of the manuscript as well as was involved in the care of this patient as family practice physician with expertise in primary dermatology. WT was involved in the patient care as pathologist and contributed towards the intellectual review of the manuscript, as well as assisting with procuring quality images of the skin biopsy histopathology and interpretation of the same. This case study does not represent the views of federal government or Veterans administration.
Conflict of interest statement
None of the authors have any conflict to disclose.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of ethical approval
The Institutional Review Board (IRB) of Salem Veterans Affair Medical Center determined that this case report does not meet the definition of human-subject research or exempt research; therefore, review and approval by the IRB or R&D Committee was not required for this activity. The case report was reviewed and waived for IRB submission.
Statement on informed consent
Verbal consent to use this case for publication was obtained from the patient during routine clinical care encounter. which was then documented in the medical records. This case report was deemed as being appropriate by the IRB for publication purposes as no patient specific identifiable health information was disclosed.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
