Abstract
Immune checkpoint inhibitor therapy nowadays became a treatment for a wide range of cancers, and may be responsible for various dermatologic adverse effects, including bullous eruptions. In our report, we present a case of late-onset immunotherapy-induced eruption in a 62-year-old woman treated with anti-programmed cell death-L1 agent durvalumab for metastatic squamous cell carcinoma. Diagnosed as lichenoid dermatitis upon initial presentation, this eruption evolved into necrotic bullous dermatitis after several weeks of phototherapy, with histology and direct immunofluorescence study favoring lichen planus pemphigoides. Thus, this case may be regarded as durvalumab-induced lichenoid dermatitis with phototherapy-triggered progression to necrotic lichen planus pemphigoides-like eruption. The patient’s eruption responded to oral prednisone and immunotherapy interruption. Interestingly, durvalumab reintroduction in this patient led to recurrent lichenoid dermatitis without bullous component. This case of immunotherapy skin toxicity is rather distinctive by its clinical and histopathologic features, with phototherapy as an additional triggering factor.
Keywords
Introduction
Cutaneous side effects of immune checkpoint inhibitor therapy are multiple and may sometimes lead to cancer treatment discontinuation. Immunotherapy-induced bullous eruptions are part of this spectrum and include bullous pemphigoid, bullous lichenoid eruption and bullous exfoliative reactions. 1 They develop in about 1% of patients treated with anti-programmed cell death 1 (anti-PD1) or anti-programmed cell death ligand 1 (anti-PD-L1) therapy, with anti-PD-L1 agents alone associated with somewhat higher incidence, between 1.3% and 5%.1,2
Phototherapy is a useful treatment modality for lichenoid dermatoses, including immunotherapy-triggered cases. Short-term cutaneous side effects of narrow-band UVB include erythema, pruritus, herpes simplex reactivation and occasionally bullae. Immunobullous diseases, especially bullous pemphigoid, may be triggered or exacerbated by phototherapy. In this case report, we describe a patient presenting with a distinctive durvalumab-induced cutaneous eruption, initially lichenoid, and subsequently evolving into necrotic bullous eruption under phototherapy treatment, with histologic elements of lichen planus pemphigoides and prominent epidermal necrosis.
Case report
A 62-year-old woman presented to dermatology clinic with acute pruritic cutaneous eruption. She has been diagnosed with metastatic squamous cell carcinoma of unknown origin and was being treated with experimental anti-PD-L1 agent durvalumab given every 4 weeks for the last 2 years. The patient developed immunotherapy-induced hypothyroidism 1 year into treatment.
On her first dermatology visit, the patient complained of recent facial eruption which partially cleared with topical clindamycin lotion and new onset of extremely pruritic skin lesions and painful oral ulcers. She also noted fatigue and generalized muscle weakness. Physical examination revealed multiple erythematous, crusted and excoriated papules on the trunk (Figure 1) and extremities, several pink facial papules and superficial erosions of her buccal mucosa. Three skin biopsies showed lymphocytic interface dermatitis with central epidermal necrosis of traumatic origin, favoring lichenoid drug eruption. No immunofluorescence study was performed at this stage. The patient was treated with clobetasol cream but kept developing new symptomatic skin lesions. In this setting, durvalumab was suspended, and narrow-band UVB phototherapy three times a week was started, but did not halt skin disease progression. Two weeks later, the patient presented with extensive bullous lesions and necrotic crusted papules and plaques on her trunk and extremities (Figure 2), and phototherapy was interrupted. New skin biopsies demonstrated lichenoid interface change, mixed inflammatory infiltrate in the papillary dermis composed of neutrophils, eosinophils and lymphocytes, subepidermal bullae and moderate to severe epidermal necrosis. Direct immunofluorescence showed linear IgG and C3 deposits along the basement membrane with n-serrated pattern. The patient was started on oral prednisone 60 mg daily, continued clobetasol cream and applied fucidic acid cream on necrotic and eroded lesions. She stopped developing new lesions and the older ones were healing gradually.

Lichenoid dermatitis with excoriated lesions at initial presentation. The patient has been receiving durvalumab for 2 years.
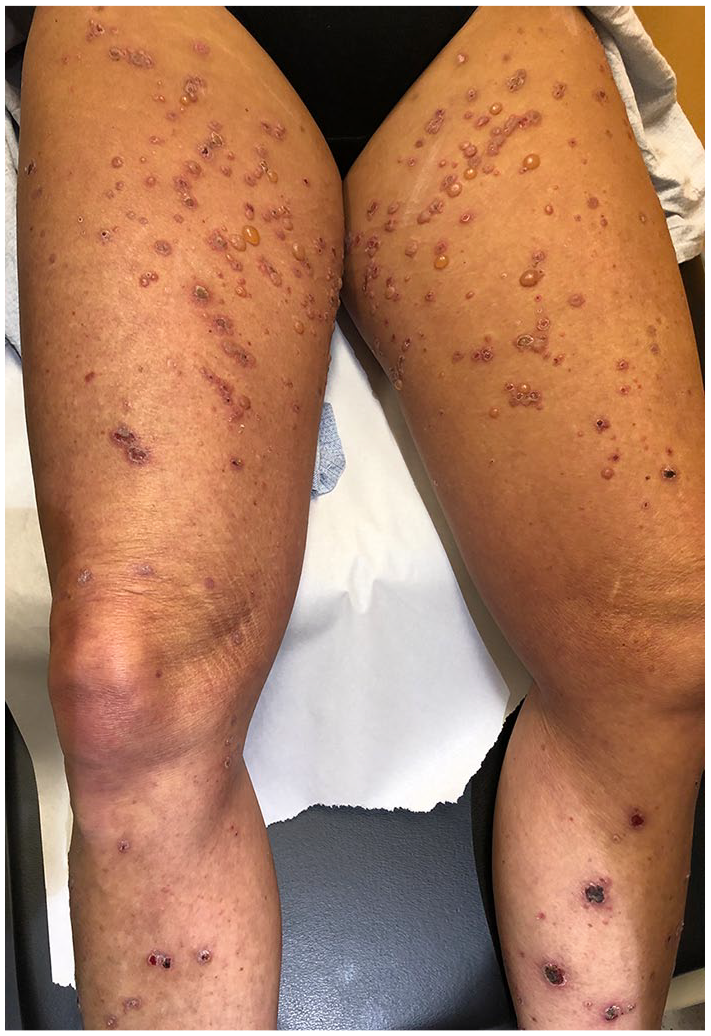
Tense bullae arising 2 weeks following the initiation of narrow-band UVB phototherapy, shortly after durvalumab discontinuation.
Durvalumab treatments were resumed 2 months following bullous eruption given a clinical progression of nodal metastasis. Despite oral prednisone maintained at 10 mg daily, cutaneous pruritic eruption similar to the initial presentation reappeared on day 15 following durvalumab treatment, but no bullae were seen (Figure 3). The eruption quickly resolved with ultrapotent corticosteroid cream (see Table 1 for the timeline of the patient’s treatment history).

Recurrent lichenoid eruption upon rechallenge with durvalumab.
Timeline of the patient’s treatment history and treatment-related complications.

NB-UVB: narrow-band UVB.
Discussion
This case is an interesting late-onset grade 3 cutaneous adverse reaction to an anti-PD-L1 therapy agent durvalumab. The constellation of clinical and histologic observations described herein may be regarded as immunotherapy-induced, phototherapy-triggered lichen planus pemphigoides with prominent necrotic component. Recurrent lichenoid eruption in this patient upon drug rechallenge was luckily self-limited and did not evolve into life-threatening drug eruption, a complication much feared by the patient’s oncologist and the dermatology team.
Lichen planus pemphigoides differs from bullous lichen planus by its positive direct immunofluorescence and onset of bullae in non-lesional skin. 3 Reports of phototherapy-triggered lichen planus pemphigoides exist in the literature, as do reports of this condition induced by anti-PD1 or anti-PD-L1 agents, but not both.3,4 None of the reports mention this specific reaction to durvalumab. Bullous reactions in the setting of immunotherapy frequently lead to its discontinuation, in addition to systemic steroids treatment and, in some cases, steroid-sparing immunosuppressive agents. Many immunotherapy-induced cutaneous reactions are currently being evaluated as potential prognostic markers for tumor response. 1 One recent retrospective study suggests that immunotherapy-induced bullous pemphigoid may represent positive prognostic value for cancer patients. 5
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Informed consent
Informed consent has been obtained from the patient by the authors.
