Abstract
Background:
Neuropsychiatric symptoms (NPS) in dementia impact profoundly on the quality of life of people living with dementia and their care givers. Evidence for the effectiveness and safety of current therapeutic options is varied. Cannabinoids have been proposed as an alternative therapy, mainly due to their activity on CB1 receptors in the central nervous system. However, little is known regarding the safety and effectiveness of cannabinoid therapy in people with dementia. A literature review was undertaken to identify, describe and critically appraise studies investigating cannabinoid use in treating NPS in dementia.
Methods:
We undertook a systematic review adhering to PRISMA guidelines. Twenty-seven online resources were searched, including Medline, PsycINFO and Embase. Studies assessing the safety and or effectiveness of cannabinoids in treating NPS in dementia in people aged ⩾ 65 years were included. Study quality was assessed using the Johanna Briggs Institute and Cochrane Collaboration critical appraisal tools.
Results:
Twelve studies met the inclusion criteria. There was considerable variability across the studies with respect to study design (50% randomized controlled trials), intervention [dronabinol (33%), nabilone (25%) or delta-9 tetrahydrocannabinol (THC; 42%)] and outcome measures. Dronabinol (three studies) and THC (one study) were associated with significant improvements in a range of neuropsychiatric scores. The most common adverse drug event (ADE) reported was sedation. A high risk of bias was found in eight studies. The highest-quality trial found no significant improvement in symptoms or difference in ADE rate between treatment arms. Included studies used low doses of oral cannabinoids and this may have contributed to the lack of demonstrated efficacy.
Conclusion:
While the efficacy of cannabinoids was not proven in a robust randomized control trial, observational studies showed promising results, especially for patients whose symptoms were refractory. In addition, the safety profile is favourable as most of the ADEs reported were mild. Future trials may want to consider dose escalation and formulations with improved bioavailability.
Introduction
Dementia is a group of diseases characterized by progressive and debilitating symptoms including cognitive decline, memory loss, changes in perception and personality. 1 In 2015, the World Alzheimer’s report estimated that there were 50 million people living with dementia worldwide, with projections of this population doubling every 20 years. 2
The most common types of dementia are Alzheimer’s disease (50–70%) and vascular dementia (20–30%) which have different aetiologies and disease course.1,2 Common to all dementia types is the suite of neuropsychiatric symptoms (NPS) including agitation, aggression, wandering, apathy, sleep disorders, depression, anxiety, psychosis and eating disorders.3–5 It is estimated that up to 90% of people living with dementia will experience at least one of these symptoms in their disease course. 6
The occurrence of NPS differs across the course of dementia, with anxiety and depression reported more common in the early stages and psychosis and aggression more common in the advanced stages of dementia. 3 Regardless of the stage of dementia, occurrence of NPS impacts profoundly on the morbidity and mortality of people living with dementia, often precipitating the use of additional medications, hospitalization and institutionalized care.3–5 It has been reported that behavioural symptoms of dementia have more significant consequences for both the patient and caregiver than cognitive decline, in part due to injury to either party through aggression and wandering. 7 Other reported impacts on carers include reduced quality of life, depression, distress and reduced employment income.8,9 Carers of individuals living with dementia and NPS report more distress and depression than carers of individuals living with dementia with no NPS. 9 First-line treatment for NPS in dementia involves a range of nonpharmacological interventions, despite a limited and disparate evidence base.6,7 These interventions are based upon identifying unmet physical and emotional needs that may be triggering the NPS and may include assessment for inadequately treated pain and unpleasant environmental factors.7,10 Complementary nonpharmacological interventions include carer education and carer support. 7
Second-line treatment of NPS involves the use of pharmacological interventions, usually in addition to the nonpharmacological strategies. Pharmacological intervention is usually warranted when a person becomes a risk to either themselves or others and nonpharmacological interventions have been unsuccessful. 6 The most common class of medications used to treat NPS of dementia is atypical antipsychotics (e.g. risperidone, olanzapine and quetiapine), although not all countries approve the use of these medications to treat NPS in dementia. 11 In Australia, risperidone is funded under the Pharmaceutical Benefits Scheme (PBS) for treating aggression and psychotic symptoms in patients with Alzheimer’s disease for up to 12 weeks. 12 A similar arrangement is available in the UK. 13 Use of antipsychotics ‘off label’ is widespread in the aged-care setting. A retrospective cohort study of over 300,000 nursing home residents in the US, reported that 23.5% of residents were prescribed at least one antipsychotic and 86% of this prescribing was ‘off label’. In addition, residents with dementia were 3.2 times more likely to use these medications ‘off label’ compared with residents without dementia. 14 The evidence for effectiveness of atypical antipsychotics to treat NPS of dementia is limited.15,16 Systematic reviews have found modest improvements in agitation and psychosis in dementia. 6 There have been many studies showing an association of these medications with harmful outcomes in patients with dementia including falls, cerebrovascular events and death.15,16 Antidepressants and antiepileptics are also used to manage NPS of dementia but are not approved for this indication. A Cochrane review undertaken in 2011, found the antidepressants sertraline and citalopram to be associated with a reduction in agitation and psychosis in dementia and were well tolerated compared with antipsychotics, however the studies were small, limiting the generalizability of these results. 17 A recent review of studies investigating the efficacy and safety of antiepileptic drugs for treating agitation and aggression in dementia found that while carbamazepine was effective at reducing symptoms, its clinical use is limited by poor tolerability and the potential for drug–drug interactions. 18 The use of valproate is not recommended due to questionable efficacy and poor tolerability and there is currently insufficient evidence to recommend the use of other antiepileptic medications in this setting. 18
Given the limited range, questionable effectiveness and side-effect profile of current therapeutic options for treating NPS of dementia, research into alternative therapies is a priority for this growing and vulnerable population. In recent years, research has focused on developing novel therapeutic agents to treat a range of NPS in dementia. Following the identification of two main cannabinoid receptors (CB1, predominantly in the central nervous system and CB2, predominantly in the immune system), cannabinoids have been investigated for their safety and efficacy in treating NPS of dementia.
19
While little is known regarding the mechanism by which cannabinoids exert their effects in NPS of dementia,
This systematic review was undertaken to identify, describe and critically appraise all studies investigating cannabinoid use in treating NPS of dementia. The objectives of this review were to identify safety and effectiveness criteria used in the trials with a focus on the risk–benefit profile of each cannabinoid and the criteria used to measure outcomes.
Methods
This study was registered in the PROSPERO database (the international prospective register of systematic reviews, registration number CRD42018086202). The study design complies with the PRISMA guidelines (Preferred Reporting Items for Systematic Reviews and Meta-Analysis protocols). 22
Study eligibility criteria
All original peer-reviewed studies assessing the safety and or effectiveness of any cannabinoid in treating NPS in patients with a diagnosis of any type of dementia were included, regardless of publication date. Where possible, all sources and healthcare databases were searched from inception to 1 January 2018.
The inclusion criteria included studies of any design, conducted in the older population (⩾65 years) and assessing the use of any cannabinoid in treating one or more of the known NPS of dementia. Included studies needed to assess one or both of the safety or effectiveness of cannabinoids in the study population and be written in English. Exclusion criteria included studies undertaken in younger populations (<65 years), studies assessing only the delay of onset or progression of any type of dementia and studies where the full text was not available.
Identification and selection of studies
In total, 27 online resources were systematically searched as shown in Table 1. This included healthcare databases, clinical trial databases, Alzheimer’s disease and dementia advocacy group websites, cannabinoid advocacy websites and Google Scholar. A reference checklist of identified studies deemed suitable for inclusion was also undertaken. The search terms used for Medline were:
Databases and websites searched (including search terms) from inception to 1 January 2018.

CBC, cannabichromene; NORML, National Organization for the Reform of Marijuana Laws; THC, delta-9 tetrahydrocannabinol; WHO, World Health Organization.
[Cannabinoid.mp. or exp Cannabinoids/cannabidiol.mp. or exp Cannabidiol/THC.mp. or exp Dronabinol/Cannabis.mp. or exp Cannabis/nabiximol.mp. or exp nabiximol/exp Dementia/ or exp Dementia, Multi-Infarct/or exp AIDS Dementia Complex/or exp Dementia, Vascular/or exp Frontotemporal Dementia/exp Alzheimer Disease/or alzheimers.mp./exp Depression/or exp Antipsychotic Agents/or BPSD.mp. or exp Behavioral Symptoms/Mental Disorders/Akathisia, Drug-Induced/or Antipsychotic Agents/or Psychomotor Agitation/or agitation.mp./aggression.mp. or exp Aggression/wandering.mp. or exp Attention/or exp Wandering Behavior/appetite.mp. or exp Appetite Depressants/or exp Appetite/or exp Appetite Stimulants/or exp Appetite Regulation/depression.mp. or Depression, Chemical/or exp Depression/or Long-Term Synaptic Depression/insomnia.mp. or exp ‘Sleep Initiation and Maintenance Disorders’/exp Anti-Anxiety Agents/or exp Anxiety Disorders/or anxiety.mp. or exp Anxiety/Caregivers/or exp ‘Quality of Life’/or carer burden.mp. or exp ‘Feeding and Eating Disorders’/or exp Stress, Psychological/adls.mp. or exp ‘Activities of Daily Living’/22. limit 21 to (English language and humans and ‘all aged (65 and over)’)]
One reviewer (JH) undertook the database and website searches and sifted through the titles and abstracts to identify suitable studies. Two reviewers (JH and GC) independently assessed the abstracts and full text of identified studies for inclusion. Disagreements were resolved with a third reviewer (NS). Data extraction and assessment of study quality was undertaken independently by two reviewers (JH and GC) and disagreements resolved with a third reviewer (NS).
Data collection and study appraisal
Four reviewers (JH, GC, CA and NS) developed and agreed upon the study protocol, search terms and data extraction tool. The data extraction tool included the following variables divided into the six categories of study description, intervention, study participant demographics, outcome measures, results and study quality.
(1) Study description: author(s), year of publication, country of origin, study design, assessment of safety, assessment of effectiveness, study setting, length of study (days) and analytic plan.
(2) Intervention: cannabinoid used, administration route, dose(s), exposure period (days) and if a comparator or placebo was used.
(3) Study participants: number of participants, mean age/median age, proportion of males, types of dementia included, criteria used to verify dementia diagnosis and history of NPS of participants and baseline cognition.
(4) Outcome measures: primary and secondary outcome measure(s) and the tools used to evaluate outcomes.
(5) Results: primary and secondary outcome measure(s) and effect size (if reported).
(6) Study quality: individual study quality was assessed using the relevant critical appraisal tool for each study design developed by The Johanna Briggs Institute (observation studies) and the Cochrane Collaboration (randomized controlled trials; RCTs).23,24
Synthesis of findings
For the included studies, data are presented for each study as described by the data extraction tool, including both quantitative and qualitative information. Where possible, aggregate data have been presented in proportions. Due to the expected paucity and heterogeneity of identified studies, a meta-analysis was not planned, nor undertaken.
Results
The search results are presented in the PRIMSA flowchart (Figure 1).

PRISMA flow diagram 22 of systematic review search results.
We identified a total of 4951 studies of which 12 met the inclusion criteria (Figure 1). Study descriptions and participant baseline characteristics are summarized in Table 2. Included studies were published over a 20-year period (1997–2017). Six studies were RCTs, two were cohort studies and four were case series or studies. Four studies were from the Netherlands, two from the UK and USA, respectively, and one each from Germany, Israel, Switzerland and Canada. Seven studies were undertaken in hospital (psychogeriatric units), two studies in the community, two were in both the community and hospital, and one study was undertaken in the community and nursing home settings. Five studies included participants with any type of dementia (Alzheimer’s disease, vascular dementia, frontotemporal dementia and mixed dementia), four studies included participants with Alzheimer’s disease only and three studies included one or two of a selection of dementia types. The cannabinoids used in the included studies were dronabinol (dose range = 2.5–7.03 mg/day), delta-9 tetrahydrocannabinol (THC; Namisol®; dose range = 1.5–15 mg/day) or nabilone (dose range = 0.5–2.0 mg/day) and were all administered orally.
Included studies: study description and participant baseline characteristics.
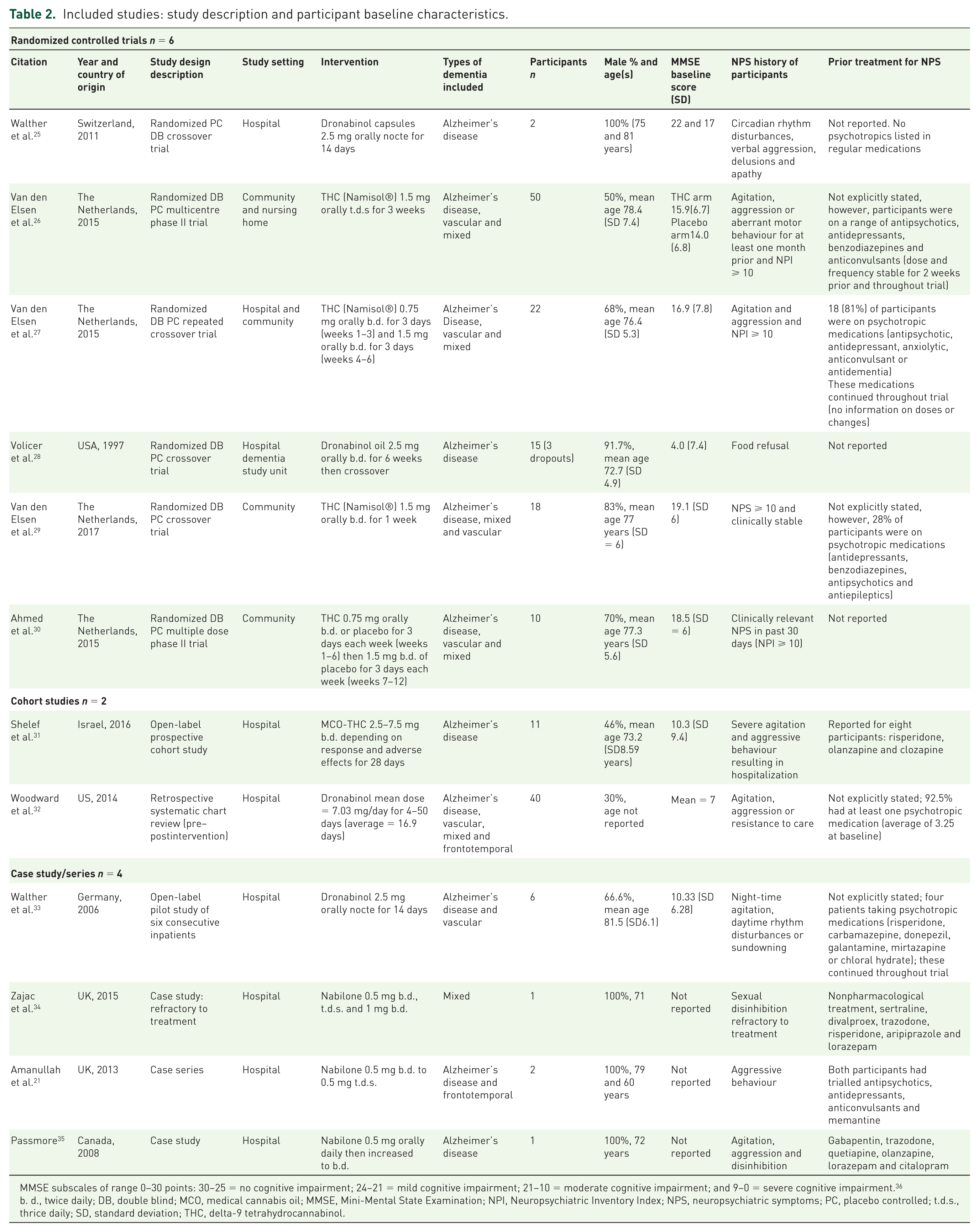
MMSE subscales of range 0–30 points: 30–25 = no cognitive impairment; 24–21 = mild cognitive impairment; 21–10 = moderate cognitive impairment; and 9–0 = severe cognitive impairment. 36
d., twice daily; DB, double blind; MCO, medical cannabis oil; MMSE, Mini-Mental State Examination; NPI, Neuropsychiatric Inventory Index; NPS, neuropsychiatric symptoms; PC, placebo controlled; t.d.s., thrice daily; SD, standard deviation; THC, delta-9 tetrahydrocannabinol.
The number of participants in the studies ranged from 2 to 50. The mean age ranged from 72.7 to 81.5 years, with the proportion of males included ranging from 30 to 100%. Baseline cognition (assessed using the Mini-Mental State Examination or MMSE) was reported in nine studies and scores ranged from 4 (severe cognitive impairment) to 22 (mild cognitive impairment). In four studies (all RCTs), participants had to score ⩾ 10 on the Neuropsychiatric Inventory Index (NPI) to be included. Reporting of prior treatment for NPS varied across the included studies but overall, was limited. Studies reporting concomitant medication use at the time of the trial recorded multiple medication use, including antipsychotics, antidepressants and neuromodulators; however, it was often unclear which medications were indicated for treating NPS of dementia.
Outcomes reported in the included trials varied substantially with heterogeneity of cannabinoid type used and evaluation of effectiveness criteria (Table 3). Half the identified studies used more than one set of criteria to evaluate effectiveness. The most common tool for assessing effectiveness of cannabinoid therapy for treating NPS of dementia was the NPI, which was used in five studies. Of these five studies, two RCTs reported no significant improvement in NPI,26,27 one case series reported significant improvement in overall NPI score and three subscales (aberrant motor behaviour, agitation and night-time behaviours), 33 one cohort study reported significant improvement in 6 of the 13 NPI subscales (agitation, disinhibition, irritability, aberrant motor behaviour, night-time behaviour and caregiver burden). 31 The fifth study, an RCT with two participants, reported improvement in overall NPI score. 25 The Cohen–Mansfield Agitation Inventory (CMAI) was used in three RCTs with no significant changes reported.26–28
Summary of study outcomes and quality.

ADEs, adverse drug events; ADL, activities of daily living; b.d., twice daily; BMI, body mass index; BP, blood pressure; CGI, Clinical Global Impression (-S, severity); CCGIC, Caregiver Clinical Global Impression of Change; CI, confidence interval; CIBRS, CONSORT, Consolidated Standards of Reporting Trials; Cumulative Illness Burden Rating Scale; CMAI, Cohen–Mansfield Agitation Inventory; ECG, electrocardiogram; GAF, Global Assessment of Functioning; GP, general practitioner; HR, heart rate; ITT, intention to treat; KSBA, Kensington Standardized Behaviour Assessment tool; MMSE, Mini-Mental State Exam; n/a, not applicable; NPI, Neuropsychiatric Inventory; NH, National Health; NPS, neuropsychiatric symptoms; PAL-WMSR, Paired Associate Learning Wechsler Memory Scale Revised; PAS, Pittsburg Agitation Scale; Tinetti–POMA, p.r.n., as required; RCT, randomized controlled trial; t.d.s., thrice daily; Tiinetti–Performance-Oriented Mobility Assessment; TUG, Timed Up and Go; UTI, urinary tract infection; VAS, Visual Analogue Scale; ZBI, Zarit Burden Index.
Reporting of adverse drug events (ADEs) was the most frequently used method for assessing safety in the included trials, used in 10 studies (one case series and one case study did not report ADEs; Table 3). Methods for ascertaining ADEs included participant and carer reports, predetermined lists of ADEs to aid in identification, medical notes or clinical observation or a combination of these. Four RCTs reported no significant difference in ADEs between treatment arms26,27,29,30 and two studies (one RCT and one case series) reported no ADEs during the trial.25,33 One RCT used a blinded independent physician to rate causality of an ADE to treatment with a cannabinoid. In this study, dizziness, fatigue and agitation were all considered related to Namisol® administration and were dose related (Table 3). 30 Serious ADEs were reported in three trials. One RCT reported three serious ADEs (one seizure and two serious infections). 28 Another RCT reported four serious ADEs (gastroenteritis, worsening of NPS, exacerbation of vestibular disorder and malignancy). 27 One cohort reported three serious ADEs (dysphagia, fall and confusion). 31 Overall, the most common ADE reported was sedation.
In addition to reporting ADEs, a variety of physical parameters related to safety were monitored in seven studies and included blood pressure, heart rate, electrocardiogram and weight. One RCT and two cohort studies reported no significant change in parameters.26,31,32 One RCT reported significant increases in body sway, stride and change in internal perception. 27 Another RCT reported a significant increase in weight. 28 One RCT reported significant decrease in balance and increase in stride. 29 Lastly, an RCT reported significant changes in perception, increase in body sway, increase and decrease in systolic blood pressure (dose dependent) and increase in heart rate. 30 Two RCTs formally assessed caregiver burden using the Zarit Burden Interview 27 and Caregiver Clinical Global Impression of Change (CCGIC) 26 with no significant differences reported between treatment and placebo arms, respectively. A case series reported families felt the treatment to be effective and with a reduction in their emotional burden. 21
Overall, the quality of included trials was low with a high risk of bias assessed for 8 of the 12 studies (66.7%; Table 3). Half (3/6) of the RCTs were assessed as low quality mainly due to small sample size, unclear methods of randomization and blinding, and selective reporting of results. The two cohort studies were of moderate and low quality, with both inadequately identifying confounding factors. In addition to the inadequacies identified above, the cohort study rated lowest quality had time-varying exposure to dronabinol and incomplete reporting of results. 32 The four included case series/studies were all assessed as low quality, as there was insufficient clinical and demographic information reported. The highest-quality trial was a randomized placebo-controlled crossover trial, which had a low risk of bias and was sufficiently powered to find a clinically significant change in NPI. 26 This study found no significant improvement in NPI compared with baseline between placebo and THC 1.5 mg three times daily for 21 days. Similarly, this study found no significant differences between placebo and treatment arm for ADEs or other physical parameters.
Table 4 provides a summary of the overall risk–benefit profile of each type of cannabinoid used in the included studies. Dronabinol was used in four studies with a daily dose range of 2.5–7.03 mg. The use of dronabinol was associated with significant improvements in several NPS scores [Pittsburg Agitation Scale, negative affect, Clinical Global Impression (CGI) and NPI], increase in weight, reduction in nocturnal activity and increase in percentage of food consumed. Dronabinol was not significantly associated with any ADEs; however, at a dose of 2.5 mg twice daily, dronabinol was associated with three serious ADEs (one seizure and two serious infections). Nabilone was used in three studies with a daily dose range of 0.5–2.0 mg. Nabilone was not significantly associated with an improvement in NPS of dementia or any ADEs; however, at a dose of 1.5 mg twice daily, nabilone caused severe sedation which required withholding of the medication. THC was used in six studies with a daily dose range of 1.5 to 15 mg. THC was associated with significant improvement in NPS of dementia in one trial (improvement in CGI and NPI). 31 THC was the only cannabinoid to be significantly associated with ADEs, including increase in body sway, increase and decrease in systolic blood pressure and increase in heart rate and in one trial, there were two dropouts due to pneumonia and nausea.
Summary of effectiveness and safety outcomes by cannabinoid.

Indicates statistical significance.
ADEs, adverse drug events; BP, blood pressure; CGI, Clinical Global Impression; CCGIC, Caregiver Clinical Global Impression of Change; CMAI, Cohen–Mansfield Agitation Inventory; GAF, Global Assessment of Functioning; HR, heart rate; KSBA, Kensington Standardized Behaviour Assessment tool; NPI, Neuropsychiatric Inventory; NPS, neuropsychiatric symptoms; QuOL-AD, quality of life in Alzheimer’s disease; PAS, Pittsburg Agitation Scale; RCT, randomized controlled trial; ZBI, Zarit Burden Index.
Discussion
To our knowledge, this systematic review is the most comprehensive presentation of the effectiveness and safety of cannabinoids in treating NPS of dementia. Overall, it was difficult to make generalizations about the safety and effectiveness of cannabinoids in treating NPS of dementia due to the heterogeneity of the included studies (even within study design), the range of assessment tools used and the poor quality of identified studies. While the efficacy of cannabinoids was not proven in a robust RCT, observational studies showed promising responses, especially for refractory patients. In addition, the safety profile presented was favourable, as the majority of ADEs reported were mild.
Within the RCT group of included trials, inclusion criteria differed substantially with respect to baseline MMSE, prior treatment for, and severity of, NPS. In addition, these trials used a variety of tools to measure change in NPS of dementia. Evidence of effectiveness and safety was limited by the power of the RCTs to detect a statistically significant change: only one trial predetermined a clinically relevant change and powered the study to detect such a change. 26 Five of the six RCTs were therefore underpowered to detect true effectiveness of cannabinoids in treating NPS of dementia.
The quality of the observational studies was limited by inadequately described comorbidities and medication use making it difficult to determine if cannabinoids were the only contributing factor to improvement in NPS of dementia. More robust cohort studies adjusting for concomitant medications and comorbidities may give insight into the true effectiveness of cannabinoids in refractory patients.
The NPI was the most frequently used tool to assess NPS in dementia response to cannabinoid treatment. This tool uses an informant to report on the severity and frequency of a broad range of symptoms (depression, anxiety, apathy, hallucinations, delirium, agitation, sleep, irritability and elation). 37 The CMAI was the second most frequently used tool to measure effectiveness of cannabinoids in treating NPS of dementia. This tool is specific to agitation with information gained through observation and informant reporting. 38 Two RCTs used both the NPI and CMAI and both did not find significant differences in NPS between treatment arms on either scale. Improvement in NPS in dementia as measured by the NPI was reported for one RCT (two patients), one cohort and one case series. This may be due to observational study design characteristics such as closer and prolonged observation of patients, more in-depth knowledge of patients and nonblinding of the assessor. Several studies used multiple tools to assess response to treatment which covered a range of NPS and global functioning. There was minimal discussion in the included studies regarding the selection criteria for assessment tool(s) and who administered the test(s). Clear description of why each tool was chosen, how it was administered and how the tools relate to one another would assist with interpreting the results.
The use of caregiver reporting of response to therapy provides additional insights. Although highly subjective in nature, these reports can represent a change in physical and emotional burden for caregivers. In one case series, caregivers reported improvement in NPS and carer burden post-treatment. However, this was not supported by the formal assessment of caregiver burden in two RCTs.
The safety profile of the cannabinoids used in the identified studies appears to be reasonable. Studies reported mostly mild adverse effects such as sedation, somnolence and fatigue. Changes in blood pressure, balance and infections were reported from several studies and are worth a more in-depth surveillance in future studies, as these effects may have serious outcomes in an older, frailer population. Long-term safety was not established in the studies due to short exposure times.
Conducting clinical trials for treating NPS in dementia has many obstacles. As demonstrated by the included studies, this cohort is frail with multiple comorbidities and multiple chronic medications making it difficult to conduct trials where there is only one variable of change. In addition, severity of a person’s NPS in dementia can vary over time. Several of the included RCTs used a crossover design which can assist with adjusting for confounders, however, will not adjust for within-person changes over time. The main deficits in the identified studies were incomplete reporting of study design, patient clinical characteristics and outcomes. Future studies should focus on improving the quality of study design and reporting as outlined in Table 5, which includes appropriate study design, accurate assessment and reporting of baseline cognition, medication use and rescue medications, calculation of effect size and study power
Clinical trial criteria for assessing NPS of dementia response to cannabinoids.
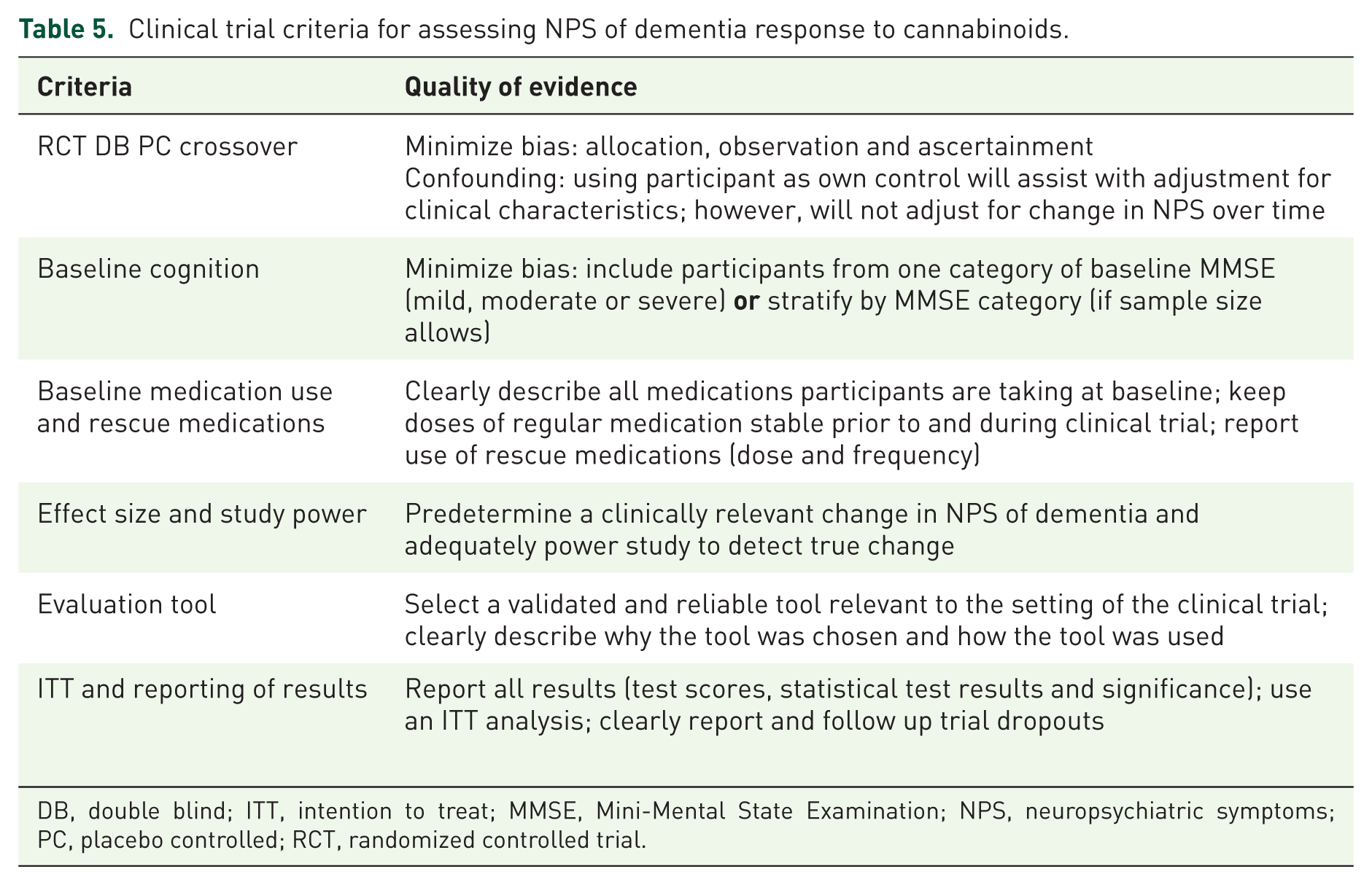
DB, double blind; ITT, intention to treat; MMSE, Mini-Mental State Examination; NPS, neuropsychiatric symptoms; PC, placebo controlled; RCT, randomized controlled trial.
Despite the paucity of evidence for safety and effectiveness of cannabinoids in treating NPS of dementia, it is important to recognize the identified trials’ contribution to knowledge in this field. Identified observational studies have shown potential for cannabinoid use in treating refractory NPS of dementia with minimal side effects. While larger RCTs did not show effectiveness of cannabinoids, the reported safety profile was acceptable. However, doses used in these studies may be a key issue in terms of safety and effectiveness of cannabinoids.
The included studies used low doses of oral cannabinoids relative to studies treating other indications, and this may have contributed to the lack of demonstrated efficacy. A systematic review of RCTs investigating the use of cannabinoids for treating chemotherapy-induced nausea and vomiting found that doses of oral nabilone 2 mg twice daily and oral dronabinol 10–15 mg/m2 up to six times daily were effective in reducing nausea and vomiting by up to 50%; however, adverse effects were commonly reported with fatigue significantly associated with treatment. 39 Therefore, dose escalation may be warranted in future studies investigating the efficacy and safety of cannabinoids in treating NPS in dementia.
Furthermore, orally administered cannabinoids have poor oral bioavailability due to high first-pass hepatic metabolism. 40 For example, a 10 mg oral dose of dronabinol has a bioavailability of approximately 6–7%. 40 Namisol® is a THC formulation with reported enhanced bioavailability (up to 30%) due to Alitra®, a lipophilic delivery technology. 41 Future studies investigating the efficacy and safety of cannabinoids in treating NPS of dementia should trial innovative administration modalities to improve bioavailability.
This study was a systematic review registered with PROSPERO and followed the PRISMA guidelines. However, we cannot confirm that we have correctly identified all relevant studies. The inclusion criteria of studies in the older population may have reduced the likelihood of including executive and frontal variants of dementia, which are more common in the younger dementia population. However, the symptoms of NPS are more likely in the older population. Two reviewers independently selected studies for inclusion and extracted the relevant information. The studies identified in our study are similar to those reported in the Cochrane systematic review of RCTs 42 and a recently published review. 43 Our systematic review identified two more RCTs than a review published in 2017 which assessed effectiveness of cannabinoids. 44 The 2017 systematic review assessed the quality of the included RCTs as ‘unclear’. In addition, we have graded the quality of each study and identified areas of bias in each study.
Conclusion
This systematic review has found that the quality of studies examining the use of cannabinoids to treat NPS of dementia is poor. While the efficacy of cannabinoids was not proven in a robust RCT, observational studies showed promising responses, especially for refractory patients. In addition, the safety profile appears favourable, as most ADEs reported were mild. However, formulations and doses of the cannabinoids used in the identified studies may have limited the ability to demonstrate cannabinoid efficacy and safety for this indication. A large, well-controlled trial is warranted given the current limited treatment options available for NPS in dementia patients.
Footnotes
Acknowledgements
We would like to thank Dr Rocco Iannello, Head of Research and Development (WardMM) and Dr Ric De Garis, Head of Clinical Trials (WardMM).
Funding
WardMM received support for this study from LeafCann Pty Ltd. LeafCann and its employees were not involved in the undertaking of this systematic review, nor the interpretation of the results.
Conflict of interest statement
The authors declare that there is no conflict of interest.
