Abstract
Background:
The aim of this study was to assess trends and variations in coprescribing of simvastatin or atorvastatin with interacting drugs in Thailand.
Methods:
Outpatient prescriptions between 2013 and 2015 in 26 tertiary care hospitals were analyzed for statin coprescribing. The proportion of patients exposed to coprescribing was estimated for semi-annual changes, using a time-series analysis and for hospital variations, using an interquartile range (IQR).
Results:
The coprescribing of simvastatin with all contraindicated drugs in 10 university and 16 general hospitals, respectively, was 3.6 and 3.1% in 2013, then decreased to 3.2 and 2.6% in 2014 and to 2.6 and 2.0% in 2015. The drug most frequently coprescribed with simvastatin, on a decreasing trend (by 0.19 percentage points) was gemfibrozil (in 2013, 2014 and 2015, respectively; 2.9, 2.3 and 2.0% in university hospitals, and 2.5, 2.0 and 1.6% in general hospitals). A similar trend was found in atorvastatin-gemfibrozil coprescribing. Patients coprescribed simvastatin with the rest of the contraindicated drugs were relatively stable at 0.6–0.8%. No protease inhibitors were coprescribed with simvastatin and atorvastatin. The IQR of simvastatin coprescribing in the university hospitals was smaller than that in the general hospitals and decreased over time.
Conclusions:
Coprescriptions potentially leading to drug interactions with simvastatin in Thailand were observed although the contraindicated drugs were acknowledged. Mutual awareness among health professionals and the implementation of electronic prescribing should be strengthened as zero drug interaction was possible as in the case of protease inhibitors in the present study.
Introduction
Cardiovascular diseases, the leading causes of serious complications and mortality have been expected to rise due to aging population, increasing patients with diabetes, and increasing risk factors of atherosclerotic cardiovascular disease (ASCVD). Statins are strongly recommended for primary and secondary prevention of cardiovascular diseases in patients with an ASCVD risk. Evidence from clinical trials shows that statins are well tolerated, however, statin-induced myopathy has been recognized. 1 It can present with a variety of findings such as myalgia, myositis, asymptomatically increased creatine kinase, and rhabdomyolysis. Rhabdomyolysis is a rare event (0.02–0.05 per 1000 person-years) compared with muscle symptoms (0.1 per 1000 person-years), but it is fatal.2–5 Many patients have known risk factors for statin-induced myopathy, including advanced age, renal impairment, small body frame, frailty, and statins in high doses or prescribed with drugs that increase the statin plasma level. Approximately 50% of spontaneous case reports of rhabdomyolysis submitted to the United States Food and Drug Administration (US FDA) adverse event reporting system (FAERs) have resulted from concomitant use of statins plus drugs known to increase the statin plasma levels. 6
Based on Thai Vigibase, a voluntary hospital-based adverse event reporting system in Thailand, from 1996 to 2009, there were 198 cases of muscle-related events involving statins 7 and more than half (55.6%) were associated with rhabdomyolysis, of which mortality was 6.36%. Simvastatin was the most commonly implicated drug (163 cases), of which 40.9% interacted with gemfibrozil, HIV protease inhibitors, azole antifungals, and macrolides.
Each statin has unique physiochemical properties leading to different levels of sensitivity to drug–drug interactions.8,9 Simvastatin, lovastatin and atorvastatin are lipophilic and metabolized by cytochrome P450 (CYP)3A4, the most important drug-metabolizing CYP isozyme. Pitavastatin, pravastatin and fluvastatin are hydrophilic and metabolized by other pathways. Simvastatin and lovastatin are most sensitive to CYP3A4-mediated drug interactions.
In Thailand, simvastatin and atorvastatin are most frequently prescribed because both of them are included in the National List of Essential Medicines (NLEM) 10 which specifies the drugs reimbursable by public health insurance schemes. Simvastatin is recommended as the first choice, unless clinically contraindicated. Atorvastatin, included by the NLEM since May 2012, is reserved for patients who are contraindicated to, or do not respond to simvastatin. The rest of the statins are not covered by the NLEM, hence they have been seldom prescribed.
In recent years, regulatory agencies issued several warnings to healthcare professionals emphasizing the increased risk of muscle problems from the use of simvastatin in high doses or in combination with other drugs that increase the statin plasma level. Major changes in simvastatin labeling took place in June 2011 when the US FDA recommended limiting the maximum dosage of simvastatin to 80 mg in new patients and at certain doses when concomitantly used with other drugs, and revised the contraindication lists of drugs interacting with simvastatin. 11 In 2012, the Medicines and Healthcare Products Regulatory Agency (MHRA) in the United Kingdom (UK) updated a similar warning of simvastatin interactions. 12 In Thailand, the same recommendation has been issued in the NLEM guidance since 2013. Based on the US FDA, UK MHRA and Thai NLEM guidance, atorvastatin is classified as less sensitive to CYP3A4 metabolizing than simvastatin and has no contraindicated comedications.
The aim of this study is to assess trends after the NLEM guidance and hospital variations in the use of simvastatin that was coprescribed with contraindicated, interacting drugs in outpatient hospital settings in Thailand. Even though no interacting drugs have been listed to date as contraindicated to atorvastatin, it was examined additionally in the present study due to possibilities of drug interactions.
Method
Drugs interacting with simvastatin or atorvastatin
The risk of myopathy that is dependent on statin concentration is well recognized. An area under the curve (AUC) and maximum concentration (Cmax) are the two pharmacokinetic measures used by the US FDA to identify the presence of drug interactions. If the statin AUC increases 2–5 times and more than 5 times the normal level, an interacting drug is defined as a moderate and strong inhibitor, respectively. Drugs interacting with simvastatin or atorvastatin (Table 1) were obtained from the Thai NLEM 2013, which is in accordance with the US FDA and UK MHRA drug safety information. Drugs contraindicated to simvastatin include gemfibrozil, clarithromycin, erythromycin, itraconazole, ketoconazole, cyclosporine, danazol, nefazodone, and HIV protease inhibitors. All except gemfibrozil are CYP3A4 inhibitors. Unlike others, gemfibrozil increases the risk of myopathy due to an additive effect and inhibits hepatic glucuronidation of simvastatin. All drugs in Table 1 are contraindicated for simvastatin based on the evidence showing the increased AUC or Cmax and adverse events due to the interactions.
Lists of statin-interacting drugs and supporting evidence.
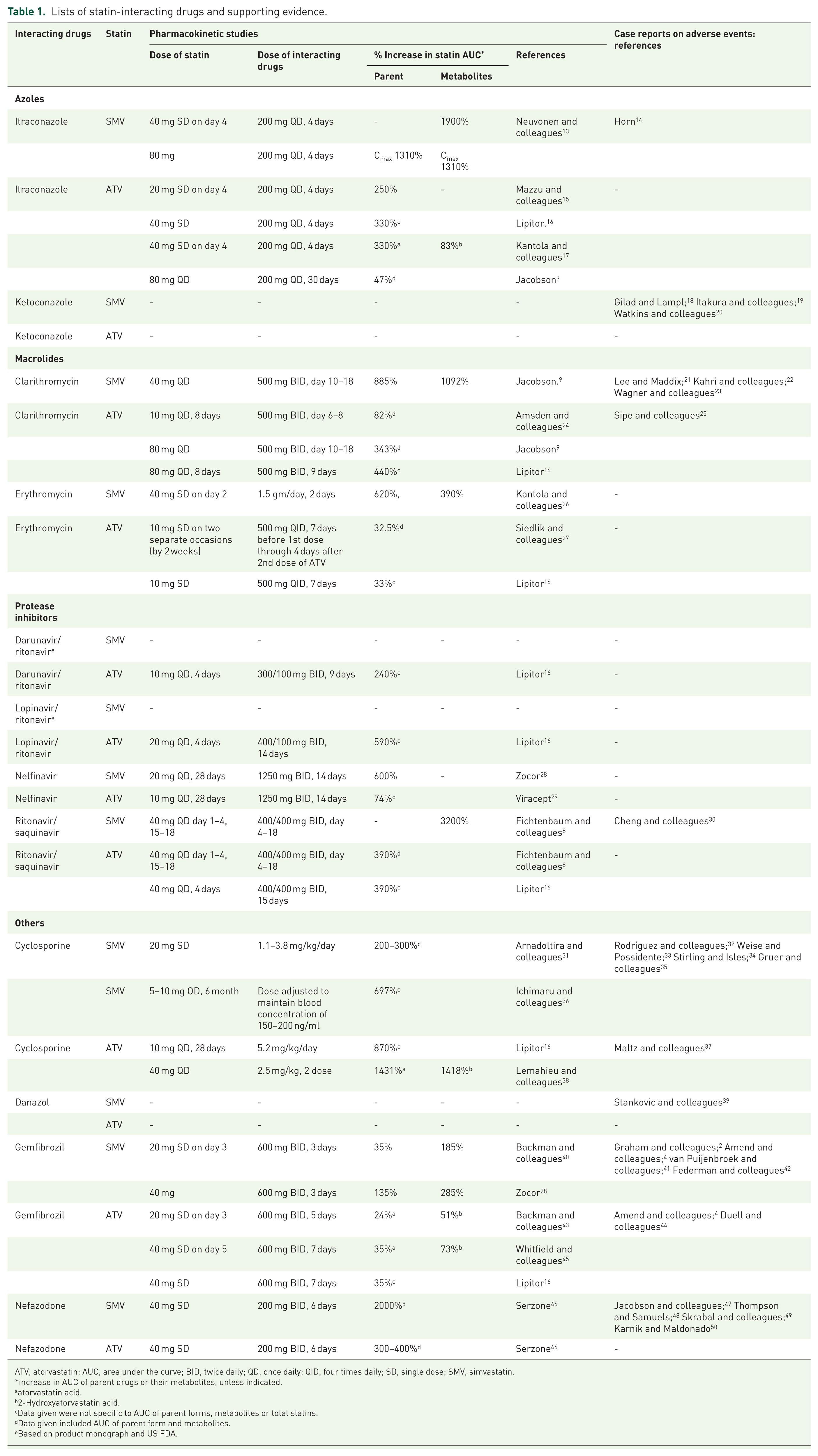
ATV, atorvastatin; AUC, area under the curve; BID, twice daily; QD, once daily; QID, four times daily; SD, single dose; SMV, simvastatin.
increase in AUC of parent drugs or their metabolites, unless indicated.
atorvastatin acid.
2-Hydroxyatorvastatin acid.
Data given were not specific to AUC of parent forms, metabolites or total statins.
Data given included AUC of parent form and metabolites.
Based on product monograph and US FDA.
For atorvastatin that has no contraindicated, coprescribing drugs, the maximum recommended daily dose is up to 10 mg when prescribed with cyclosporine and up to 20 mg when prescribed with clarithromycin, itraconazole, and HIV protease inhibitors (product monograph of Lipitor™). Therefore, all interacting drugs were evaluated for both simvastatin and atorvastatin in the present study.
Data sources
In Thailand, outpatient departments of hospitals are the main source of drug prescriptions and a single prescription usually contains more than one drug item. Unlike in most of developed countries where separation between prescribing and dispensing is a norm, drugs prescribed in a hospital are dispensed by the hospital pharmacy department.
Our study obtained data on outpatient prescriptions during 2013–2015 from 10 university hospitals and 16 general hospitals nationwide. All studied hospitals are the tertiary care facilities with service load of more than 100 thousand prescriptions a year. The analytic dataset contained hospital codes, encrypted patient identification, and dates plus identification of the prescribed drugs, which were uniquely defined according to the anatomical, therapeutic and chemical (ATC) classification system. The study statins (ATC codes) were simvastatin (C10AA01) and atorvastatin (C10AA05). Variables on drug dosage regimens, patient demographics and clinical conditions were not available. The analysis framework focused on time trends and hospital variations in the number of patients exposed to statin coprescribing with interacting drugs listed in Table 1. Confidentiality was preserved because none of the individual patients were identifiable. This study was approved by the Ethics Committee for Human Research, Khon Kaen University (HE592234).
Data analysis
Statin coprescribing with an interacting drug was identified when statins appeared in the same prescriptions with the drugs summarized in Table 1. The number of patients exposed to the coprescribed interacting drugs was determined. For a patient exposed to more than one interacting drugs, each interacting drug was counted as a separate event unless the analysis was conducted as an overall. The magnitude of a potential problem of statin interaction was estimated semi-annually and annually in terms of the coprescribing rate, which was calculated as a percentage of all patients prescribed each statin. To examine whether the statin coprescription with interacting drugs decreased over time as a response to the NLEM guideline towards an awareness of the interaction, trends in the statin coprescribing patterns, one with gemfibrozil and the other with the rest of interacting drugs, were examined, using a time-series analysis. Changes in the magnitude of statin coprescription with interacting drugs during 2013–2015 were estimated in terms of an absolute difference in the percentages on average over a total of six half-year periods. To account for a serial correlation between observations in adjacent years, a generalized least squares model, using a Prais–Winsten transformation, was employed. 51 Variations across hospitals in the statin coprescription with interacting drugs over a year were described, using an interquartile range (IQR). All analyses were conducted separately between the university and general hospitals.
Results
The number of patients receiving simvastatin or atorvastatin in university and general hospitals during 2013–2015 and the statin recipients who were coprescribed with gemfibrozil or other interacting drugs are presented in Table 2. The total number of recipients of simvastatin was approximately 3–4 times of that of atorvastatin. In the university hospitals, the use of atorvastatin increased noticeably over the 3-year period.
Statin recipients and exposure to coprescribed interacting drugs.

Number of patients were presented with omitted percentage.
The first and second most coprescribed interacting drugs in both hospital types were gemfibrozil and clarithromycin, respectively. Others among the top five interacting drugs by number of recipients in the university hospitals were cyclosporine, itraconazole and ketoconazole, and in the general hospitals were itraconazole, ketoconazole and cyclosporine. Fewer than 20 patients received simvastatin or atorvastatin concomitantly with erythromycin or danazol each year and none received the two statins with protease inhibitors or nefazodone during the study period.
In the university hospitals, patients who were prescribed simvastatin or atorvastatin concomitantly with any interacting drugs decreased from 3.6% in 2013 to 2.6% in 2015. From 2013 to 2015, the statin recipients who were exposed to potential drug interactions with gemfibrozil decreased from 2.9 to 2.0% and from 2.8 to 2.0% of patients receiving simvastatin and atorvastatin, respectively. In contrast, the statin recipients coprescribed with interacting drugs other than gemfibrozil were much lower and relatively stable at 0.6–0.8% over the same period.
In the general hospitals, the exposure to the overall coprescriptions potentially leading to drug interactions was lower than in the university hospitals. From 2013 to 2015, the recipients of simvastatin and atorvastatin who were exposed to any interacting drugs decreased from 3.1 to 2.0% and 2.6 to 1.9%, respectively. Patients exposed to statin-gemfibrozil coprescribing decreased from 2.5 to 1.6%, 1.9 to 1.4% for simvastatin and atorvastatin, respectively. For drugs other than gemfibrozil, exposure to the coprescribing of statin-interacting drugs in the general hospitals was similar to that in the university hospitals, 0.4–0.7% for simvastatin and atorvastatin.
A time-series analysis over six semi-annual periods (first half of 2013 to the second half of 2015) revealed a decreasing trend in the coprescribing of simvastatin or atorvastatin with gemfibrozil in both hospital types (Table 3). In the university hospitals, the percentage of simvastatin recipients exposed to the coprescribed gemfibrozil decreased significantly by 0.19 percentage points (%pt.) per half year, which was a little faster than that of atorvastatin-gemfibrozil (by 0.17%pt.). In the general hospitals, the significantly decreasing rate of coprescribing with gemfibrozil was similar for simvastatin (by 0.19%pt.) and a little slower for atorvastatin (by 0.16%pt.).
Absolute changes in statin recipients who were exposed to coprescribed interacting drugs.

CI, confidence interval.
For the interacting drugs other than gemfibrozil, a decreasing rate of coprescribing with atorvastatin in the university hospitals was statistically significant (−0.018%pt.). Even though the simvastatin coprescribing rate decreased by a greater magnitude (−0.024%pt.), it did not reach statistical significance level (
Regarding distribution of the statin coprescribing rates, there were variations in patient exposure to potential drug interactions across individual hospitals. In the university hospitals, patients prescribed simvastatin concomitantly with any interacting drugs accounted for 2.4, 3.2 and 4.0% of the statin recipients in the hospitals at the 25th, 50th and 75th percentiles, respectively in 2013 [Figure 1(a)]. The 25th, 50th and 75th percentiles of the hospitals by coprescribing rates decreased monotonically to 2.0, 2.8 and 3.2% in 2014, and to 1.7, 2.4 and 2.7% in 2015, respectively. Noticeably, the IQR of simvastatin coprescribing rates narrowed down over time, largely due to a rapid decline of the coprescribing rates in hospitals at the 75th percentile (by 0.8 and 0.5%pt. in 2014 and 2015, respectively). For atorvastatin, the 25th, 50th and 75th percentiles of the hospitals had coprescribing rates in 2013 of 2.1, 3.7 and 4.0%, respectively. A decrease in the coprescribing rates for atorvastatin in 2014 and 2015 was largely driven by those in the median and the 75th percentile hospitals (by 0.5 and 0.4–0.5%pt., respectively each year). At the 25th percentile, the coprescribing rate increased by 0.3%pt. in 2014, then declined by 0.8%pt. in 2015. The median coprescribing rates for atorvastatin was noticeably close to the 75th percentiles and were relatively higher than the median for simvastatin in every year.

Hospital variations in statin recipients who were exposed to coprescribed interacting drugs.
In the general hospitals, variations in the coprescribing rates for both statins were wider than those in the university hospitals, as the 25th percentiles and the medians were lower than in the university hospitals but the 75th percentiles were higher in every year [Figure 1(b)]. For simvastatin, the IQR was 1.4–4.3% in 2013, then decreased to 0.9–3.6% in 2014 and to 0.6–3.1% in 2015. For atorvastatin, the IQR was 1.0–4.2% in 2013, then decreased to 1.0–3.4% in 2014 and to 0.5–4.3% in 2015. Another distinction is that the median coprescribing rates, especially for atorvastatin in the general hospitals were towards the 25th percentiles, while those in the university hospitals were to the 75th percentiles. This resulted in the median coprescribing rate in the general hospitals of atorvastatin being lower than that of simvastatin.
Discussion
The coprescribing rate of simvastatin with all contraindicated, interacting drugs in university and general hospitals, respectively was 3.6 and 3.1% in 2013, then decreased to 3.2 and 2.6% in 2014 and to 2.6 and 2.0% in 2015. The most common interacting drug coprescribed with simvastatin was gemfibrozil (2.9, 2.3 and 2.0% in the university hospitals, and 2.5, 2.0 and 1.6% in the general hospitals in 2013, 2014 and 2015, respectively), which had a decreasing trend (per half year by 0.19%pt. equally in both hospital types). All other interacting drugs contraindicated to simvastatin coprescribing were found in less than 1% of the simvastatin recipients. Among the common ones were clarithromycin, cyclosporine, itraconazole and ketoconazole. Variation in the coprescribing rates of simvastatin with contraindicated, interacting drugs was higher in the general hospitals than that in the university hospitals. Given that the simvastatin-interacting drugs were not contraindicated to atorvastatin, the overall coprescribing rates of atorvastatin should be higher than that of simvastatin. In the university hospitals, the coprescribing rates for the two statins were similar. In the general hospitals, the overall coprescribing rate for atorvastatin was a little lower than that for simvastatin. In addition, a decrease in the coprescribing rate for atorvastatin was a little slower than that for simvastatin.
Coprescribing between simvastatin and the contraindicated, interacting drugs in Thailand was lower than that in other countries. However, reports on this issue were mainly based on drug utilization data before the US FDA and UK MHRA warnings related to the safety of simvastatin and recent reports are lacking. The coprescribing rate of simvastatin in the present study with gemfibrozil (2.0–3.6%) was lower than that in US during 2003 and 2009 (2.9–6.8%) 52 and with clarithromycin (0.2–0.3%) was lower than that in Korea (2.6%). 53 In Thailand, fenofibrate, an alternative to gemfibrozil, and azithromycin, an alternative to clarithromycin, are covered by the NLEM. Risk of rhabdomyolysis from the coprescribing of simvastatin and gemfibrozil was higher than that from the use of simvastatin alone and in combination with fenofibrate 12 times and 3–4 times, respectively. 4 A large cohort study reported an increased risk of rhabdomyolysis [relative risk, 2.17; 95% confidence interval (CI), 1.03–4.52] in elderly patients who received simvastatin concomitantly with clarithromycin or erythromycin, compared with that with azithromycin. 54 It is recommended that simvastatin should be avoided when taking clarithromycin, or azithromycin should be used instead if the coprescribing of simvastatin and macrolides was unavoidable. Our study also found coprescribing between simvastatin and ketoconazole or itraconazole (0.04–0.09% in the university hospitals and 0.17–0.24% in the general hospitals), which were less frequent than that in previous studies (ranging from 0.1 to 1.6%).53,55,56 Itraconazole is a potent inhibitor of CYP3A4 that could increase the AUC of simvastatin acid 19-fold. 21 Fluconazole, the less potent CYP3A4 inhibitor, is the best alternative for patients taking simvastatin. For cyclosporine, an immunosuppressant drug, the coprescribing in the present study with simvastatin in the university hospitals (0.2%) was more frequent than in the general hospitals (0.02%). A study in Finland reported prevalence of concomitant use between simvastatin and cyclosporine was 1.1%. 56 Cyclosporine increases the AUC of simvastatin 6–8-fold and fluvastatin 2–4-fold. 57 Atorvastatin may be considered as the substitute but the daily dose should not exceed 10 mg. 57 Rosuvastatin which is mainly metabolized by CYP2C9, is the optimal choice for patients who have to use cyclosporine in long term, such as the transplant recipients. The relatively low coprescribing rates with the decreasing trends during 2013–2015 found in our study reflect an increasing awareness of recommendations by regulatory agencies on the coprescribing of statins that may lead to drug interactions.
It is interesting that coprescribing between statins and protease inhibitors was not found in this study. In Thailand, the delivery of antiretrovirals has been strictly controlled in hospital settings and managed through a national program that requires multidisciplinary approaches. An intensive monitoring of the antiretroviral prescribing in the hospital setting is likely the key to avoidance of coprescribing leading to potential drug interactions.
Although our study found a decreasing trend of the coprescribing of simvastatin with contraindicated, interacting drugs, there was a wide variation across hospitals. A higher overall rate of simvastatin coprescribing with potentially interacting drugs in the university hospitals was mainly driven by cyclosporine that had a limited use in the general hospitals. However, the general hospitals had a higher coprescribing variation. The variation raised concerns on patient safety management in certain hospitals. It should be noted that contraindications and dose restrictions are different across individual statins and the details of these issues are recall burdens when prescribing the drugs. Electronic prescribing along with computerized alert systems could reduce prescribing prone to drug interactions. In the real-world practice however, overriding medication-related alerts occurred.58–60 Therefore, motivating an awareness of health professionals and use of electronic prescribing systems should be implemented in tandem.
Major strengths of this study included the use of a large prescribing database from several hospitals over multiple years. The claims database is a valid source to monitor events on drug exposure. The studied hospitals are all public and their services are the tertiary care level with a large volume of outpatient visits, hence representing the mainstream health services in Thailand.
Certain limitations remained in our study. First the study included only the large hospitals. Coprescribing of simvastatin with the interacting drugs can occur in small hospitals that have no medical specialists. The coprescribing rates among the excluded small hospitals are more likely to be higher than that reported at present due to a high turnover of general practice physicians, lack of computerized, updated decision support systems, and limited availability of alternative drugs. Second, the present study focused on coprescriptions of simvastatin with drugs that were contraindicated. Other well known, interacting drugs which have been classified by the US FDA as the caution lists, such as amiodarone, amlodipine, verapamil were not studied. Third, information on diagnoses, drug dosage regimens, patient demographics and clinical conditions, laboratory results, and health outcomes were not available in the studied database. Therefore, dose-dependent drug–drug interactions were beyond the scope of this study and clinical consequences of the potential drug interactions were not investigated.
Our study reported a decreasing trend and the modest prevalence of potential drug interactions with statins in tertiary care hospitals after public warning on statins coprescribing. In 2013, the prevalence of 6.8% was reported in simvastatin users in a district hospital in Thailand. 61 Further studies should update the situation in small hospitals.
Conclusion
The rate of coprescribing of simvastatin with the contraindicated, interacting drugs was small and decreased over time. Because of the wide use of simvastatin, patients who were exposed to the risk of drug interactions were not insignificant in number. Therefore, further efforts to increase the awareness of health professionals along with the effective use of electronic prescribing systems are warranted.
Key points
(1) During 2011–2013, regulatory agencies issued recommendations about contraindication lists of drugs interacting with simvastatin that increase risk of myopathy.
(2) In Thailand in the outpatient setting, the coprescribing rates of simvastatin with all contraindicated, interacting drugs in university and general hospitals, respectively were 3.6 and 3.1% in 2013, decreased to 3.2 and 2.6% in 2014 and to 2.6 and 2.0% in 2015.
(3) The most common interacting drug coprescribed with simvastatin was gemfibrozil.
(4) In university hospitals, the IQR of simvastatin coprescribing rates narrowed down over time, and was narrower than that in general hospitals.
(5) Further efforts to increase awareness of health professionals along with effective use of electronic prescribing systems are warranted to decrease an exposure to risk of simvastatin-contraindicated drug interactions.
Footnotes
Acknowledgements
The authors thank Dr Cynthia R. Gross for providing editing assistance.
Author contributions
T.R.: study concept and design, analysis and interpretation of the data, and drafting of the paper. C.L.: study concept and design, analysis and interpretation of the data, drafting of the paper, and revising it critically for intellectual content. O.W.: analysis and interpretation of the data. All authors are involved in the final approval of the version to be published and agree to be accountable for all aspects of the work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Faculty of Pharmaceutical Sciences, Khon Kaen University, Thailand.
Conflict of interest statement
The authors declare that there is no conflict of interest.
