Abstract
Proton pump inhibitors (PPIs) are among the most frequently prescribed
medications. Their use is likely even higher than estimated due to an increase
in the number of PPIs available without a prescription. Appropriate indications
for PPI use include
Introduction
The management of acid peptic disease was revolutionized by the introduction of
proton pump inhibitors (PPIs) into clinical practice almost three decades ago.
Today, PPIs remain among the most widely prescribed medications in the world. PPIs
as a class of medication also have a high prevalence of being prescribed for poorly
defined reasons or for conditions where PPIs have not been shown to be beneficial.
The current evidence suggests PPIs are often overused with 25–70% of
prescriptions having no appropriate indication.1,2 In the United States (US) alone,
PPIs account for >$10 billion in healthcare costs and the global costs
exceed $25 billion/year.
3
Clinicians can legally prescribe medications based on their
individual interpretation of the scientific evidence or clinical judgment regardless
of the narrow approval of the US Food and Drug Administration (FDA). The
‘off-label’ use of medications is prevalent in the intensive care
environment, and PPIs account for the highest off-label use (as high as 55%
prevalence) in intensive care units.
4
The widespread and often
open-ended use of medications, even those with a relatively safe profile, can have
negative, unintended consequences in the long term. Some of the main inappropriate
uses of PPIs are for the prevention of gastroduodenal ulcers in low-risk patients,
low-dose steroid therapy without additional risk factors, systemic anticoagulation
without additional risk factors for gastroduodenal injury, and the overtreatment of
functional dyspepsia. The US FDA and the National Institute for Clinical Excellence
in the United Kingdom published guidelines on the indications for prescribing PPIs
(especially in hospitalized patients). The major indications included erosive
esophagitis, nonsteroidal anti-inflammatory drug (NSAID)-induced dyspepsia and
sequelae, critically ill patients on mechanical ventilation, and the treatment of
A large number of mainly observational studies on a wide range of possible associations with the use of PPIs have been published in the past decade. Additionally, a number of meta-analyses on the same subject have almost doubled the number of publications reporting on the long-term effect of PPI use in varied patient populations in the last decade.
In this review, we sought to evaluate the major reported associations and gain some clarity on the long-term effects of PPIs.
The major associations reported in the literature about the potential adverse effects of the long-term use of PPIs are outlined below:
PPIs and
PPIs and dementia
PPIs and pneumonia
PPIs and antiplatelet agents
PPIs and kidney disease
PPIs and micronutrient deficiency
PPIs and bone mineral density
i. Long-term use of PPIs and the risk of C.
difficile infection
The introduction of PPIs into clinical practice revolutionized the management of
acid peptic disease and gastroesophageal reflux disease (GERD). The use of PPIs
has increased several-fold over the last two decades and one of the
inappropriate indications often attributed to this rise is the use of PPIs for
the prevention of gastroduodenal ulcers in low-risk patients. As the data
accumulated with years of usage, an epidemiologic association between the use of
hypochlorhydric agents and the increased risk of acquired enteric infections
such as
Studies evaluating the association between PPI use and the risk of
developing

ABX, Antibiotics; ASR, adjusted sequence ratio; CDI,
Studies evaluating the association between PPI use and the risk of
developing recurrent

CDI,
The mechanistic basis of this association is not clear at this time. Especially because the association was based on retrospective observational data. Thus not completing eliminating confounders in the analyses.
Postulated mechanisms linking CDI with PPI use
There are a number of potential pathophysiologic mechanisms that have been
described in the literature. Although
Summary
The various strategies in the prevention of CDI should begin with the cessation of the medications without strong indications and close reassessment of PPI use, especially in the intensive care patient population
ii. Long-term use of PPIs and the risk of dementia
Dementia is a silent and progressive disorder characterized by deterioration in cognitive ability that severely debilitates the individual and affects their ability to live independently. It is a disorder of age, with the incidence increasing as age advances and more importantly does not have a cure at this time. Besides the tremendous social, emotional and caregiver burden that dementia imposes, the associated worldwide financial costs of dementia patients were estimated at greater than 600 billion dollars in 2010. 13 With worldwide efforts directed at primary prevention, the focus on modifiable risk factors becomes key. One such factor is long-term medication use for other conditions. One of the early, large epidemiological studies based on the German ageing, cognition and dementia databases showed a significantly elevated risk of developing dementia in patients exposed to long-term PPI therapy. 13 A subsequent study conducted on a longitudinal sample of elderly patients from the largest German statutory health insurer also showed an increased risk of developing dementia compared with patients with no exposure to PPIs. 14
Postulated mechanism linking dementia and PPI use
The buildup of beta amyloid has been implicated in the progression and pathogenesis of dementia syndromes such as Alzheimer’s disease in humans. Central nervous system microglial cells use enzymes such as V-ATPase to degrade and scavenge beta amyloid. Murine models suggest that PPIs interfere with the activity of scavenger enzymes such as V-ATPase leading to the accumulation of beta amyloid. 15 Further studies are needed to elucidate the mechanism linking PPI usage with dementia in humans.
Summary
These two large observational studies show a small effect size but have a low quality of evidence. The ongoing need for PPI therapy should be closely evaluated particularly in elderly patients. It is important to note that in these large population-based studies, the PPIs were not assigned at random but were related to patient characteristics (e.g. PPIs prescribed because of older age and NSAID-associated dyspepsia). This results in differences between PPI users and nonusers in factors that may impact study outcomes and confound results (residual bias).16,17
iii. Long-term PPI use and the risk of pneumonia
Healthcare-acquired pneumonia (HAP) and ventilator-associated pneumonia (VAP) are
a significant source of morbidity and mortality in critically ill patients. In
the late 1990s to early 2000s, several studies were published suggesting an
association between the use of PPIs in the outpatient setting and development of
community-acquired pneumonia. A meta-analysis of these studies found that the
risk of pneumonia was increased as early as the first month of
therapy.
18
This sparked an interest in the potential relationship
between PPI usage in the inpatient setting and the development of pneumonia. One
of the early studies examining this relationship evaluated the associated
between any type of acid suppression therapy and development of HAP. They found
the incidence of HAP was higher in patients receiving acid suppression therapy.
A stratified analysis according to subcategories of acid suppression therapy,
found this association was maintained in patients receiving PPIs but not in
patients receiving histamine-2 receptor antagonists (H2RAs).
19
Several
studies have compared the risk of pneumonia in patients receiving PPIs
There have also been several recent studies examining the association between
acid suppression therapy and pneumonia in specific patient populations. In
patients with nontraumatic intracranial hemorrhage, PPI prophylaxis was
associated with an increased risk of HAP
Postulated mechanism of HAP/VAP in patients on long-term PPI use
By suppressing gastric acid release, acid suppressants increase gastric pH, which may promote bacterial overgrowth leading to tracheal colonization and pneumonia.23,24 Evidence also indicates that PPIs may impair immune cell function, increasing the risk of infectious complications. 22
Summary
On the surface, based on the currently available literature, there appears to be an association between the long-term use of PPIs and development of HAP/VAP. Although the mechanism and association seems biologically plausible, the overall quality of evidence is low. The risk of developing VAP/HAP does not seem to be substantially increased and was observed in studies where residual confounding is highly likely. Thus, in reality, the clinical relevance of the observed association is probably less significant. For now, providers should aim to limit the use of acid suppression therapy to patients with a clear indication.
iv. Long-term use of PPIs and antiplatelet agents
One of the early concerns surrounding the use of PPIs was a potential interaction with the antiplatelet agent clopidogrel, a prodrug requiring activation by the CYP2C19 enzyme system. The active metabolite of clopidogrel is responsible for irreversibly binding to the ADP receptors on platelets, inhibiting their aggregation. 25 PPIs competitively inhibit CYP2C19 to varying degrees, with omeprazole likely the most significant inhibitor. Pantoprazole does not inhibit CYP2C19. A 2009 study, including 105 patients undergoing high-risk angioplasty, found that platelet reactivity was ~25% higher in patients receiving a PPI in addition to clopidogrel than in patients not receiving concomitant PPI therapy. 26 Several other studies suggesting a potential interaction between PPIs, primarily omeprazole, and clopidogrel prompted a 2009 US FDA label warning, recommending the avoidance of concomitant administration of clopidogrel and omeprazole due to a concern of a reduction in clopidogrel’s active metabolite levels and antiplatelet activity.
Since the US FDA warning, multiple studies have explored the safety of the
concomitant administration of PPIs and clopidogrel. A retrospective cohort study
of ~8200 patients assessed the risk of adverse outcomes associated with
concomitant use of PPIs and clopidogrel following acute coronary syndrome. It
found an increased risk of adverse outcomes in patients receiving clopidogrel
with a PPI
As previously mentioned, different PPIs inhibit CYP2C19 to varying degrees, with
omeprazole being the most potent inhibitor and pantoprazole being the least
potent. A cohort study including over 20,000 patients hospitalized with
myocardial infarction, coronary artery revascularization, or unstable angina did
not find a significant relationship between PPI use and an increased risk of
serious cardiovascular disease. It is noteworthy that the majority of patients
in this study were receiving pantoprazole and fewer than 10% were receiving
omeprazole.
29
A small, prospective trial of patients with acute
myocardial infarction found that platelet function, assessed using the Verify
Now system, was significant higher in patients who received omeprazole
Postulated mechanism of relationship between PPIs and antiplatelet agents
PPI have been shown to inhibit certain CYP enzymes; the degree of inhibition varies among the different agents within the class. Some antiplatelet agents rely on these CYP enzymes to be metabolized to their active form. In theory, the use of a PPI could prevent an antiplatelet agent from being activated and decrease the antiplatelet effects.
Summary
While the COGENT study remains the only large-scale prospective randomized controlled trial exploring the clopidogrel–omeprazole interaction, multiple other smaller or retrospective studies have been conducted with varying results. The two meta-analyses, each including over 80,000 patients, found a higher rate of adverse cardiovascular events in patients receiving concomitant PPI-clopidogrel therapy. 31 While it appears there is certainly a pharmacodynamic interaction between PPIs and clopidogrel, the clinical significance of this interaction for most patients is not well elucidated. Given the conflicting data currently available, further study is indicated.
v. Long-term PPI use and the risk of kidney disease
Chronic kidney disease (CKD) affects more than 10% of the US population, and is associated with significant morbidity and mortality, as well as a considerable economic burden to the healthcare system.32,33 Shortly after the introduction of PPIs, case reports suggesting an association between their use and the development of acute interstitial nephritis (AIN) emerged. 35 This association has been substantiated by further studies, but it is only in the last decade that the potential relationship between PPIs and the development or progression of CKD has been examined. The first large-scale study published examining the relationship between PPI use and CKD included two individual patient cohorts intended to represent the general population. In each group both the adjusted and unadjusted analysis found a significant positive relationship between PPI use and the development of CKD. Interestingly, H2RA use was not found to be associated with CKD in either cohort. 32 The results of a similarly designed study by Xie and colleagues paralleled those of the Lazarus study. They found that PPI use was associated with a higher incidence of CKD, kidney disease progression and end-stage renal disease (ESRD). 35 They also found that increased duration of exposure was associated with an increased risk of adverse renal outcomes up to 720 days of exposure; after 720 days, this association disappeared. A subsequent large, Swedish cohort study also found an association between cumulative PPI use and CKD progression. 36 The most recent evaluation of PPI exposure and CKD by Li and colleagues using complex pharmacoepidemiologic tools to estimate the effect of unmeasured/unknown confounders on the relationship of PPI use and risk of CKD suggests that confounding factors alone were unlikely to explain the reported association. Please refer to table 4 for summary of the studies.
Studies evaluating the association between PPI use and the risk of developing nosocomial pneumonia.
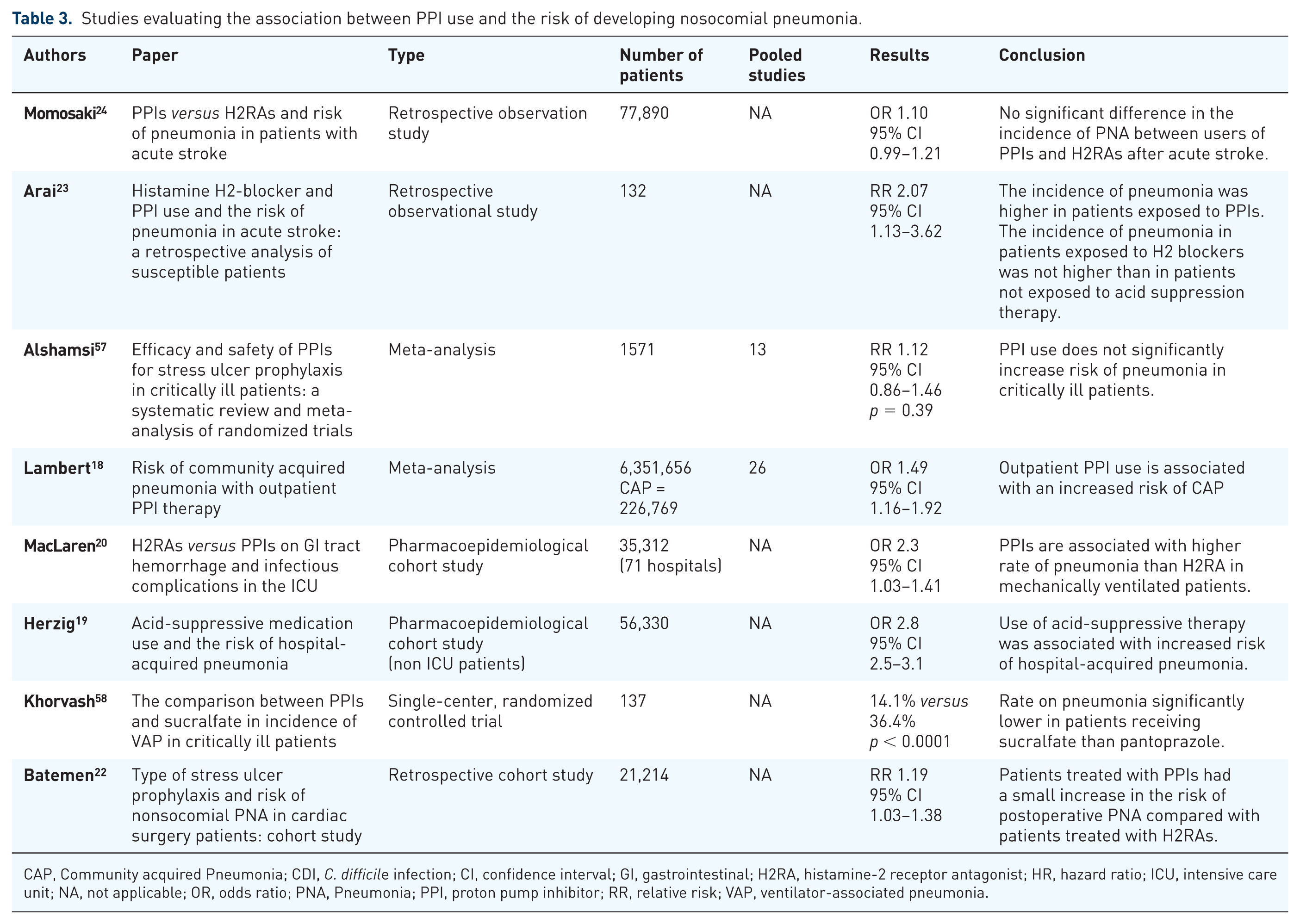
CAP, Community acquired Pneumonia; CDI,
Studies evaluating the association between PPI use and chronic kidney disease.

AKI, acute kidney injury; CI, confidence interval; CKD, Chronic kidney disease; ESRD, End stage renal disease; GFR, glomerular filtration rate; HR; Hazard ratio; PPI, proton pump inhibitor; RR, relative risk; Scr, Serum Creatinine.
Postulated mechanism of CKD in patients on long-term PPI use
The mechanism responsible for the association between PPI use and CKD is not well understood. While the risk of AIN-induced acute kidney injury with PPI use is well established, whether or not this is the sole mechanism by which PPIs are associated with CKD remains unanswered. Other proposed mechanisms by which PPIs may cause renal dysfunction include abnormalities in lysosomal acidification hydrogen/potassium adenosine triphosphatase enzyme system, decreased regeneration of renal tubular cells, increased oxidative stress, and altered gene expression. 37 Further study is needed to gain a better understanding of this relationship.
Summary
Given the current evidence, providers should be prudent in evaluating the need for initiation and continuation of PPIs, particularly in patients with known CKD or risk factors for development of CKD.
vi. Long-term PPI use and the risk hypomagnesemia
Hypomagnesemia occurs in up to 65% of critically ill patients and may increase the risk of both short and long-term complications.38,39 Acutely, magnesium has been shown to decrease inflammation, reduce platelet aggregation, and prevent arrhythmias. 38 Chronic hypomagnesemia may increase the risk of cardiovascular disease, diabetes, and osteoporosis. 39 While hypomagnesemia in critically ill patients is often multifactorial, the use of PPIs is a proposed contributor. The pathophysiology behind the relationship of PPI use and hypomagnesemia is not well understood. One hypothesis is that the pH change induced by PPI use alters the affinity of magnesium transport receptors for magnesium, decreasing the active transport of magnesium across the intestinal lumen.40,41
In 2011, the US FDA issued a drug safety communication regarding the potential
association between PPI use and hypomagnesemia.
42
A 2014 meta-analysis by
Park and colleagues including nine studies examining the relationship between
PPI use and hypomagnesemia found a higher incidence of hypomagnesemia in PPI
users than nonusers.
40
In an effort to further explore the relationship
between PPI use and hypomagnesemia, Kieboom and colleagues performed a
prospective cohort study including approximately 9000 patients. They found that
PPI use increased the risk of hypomagnesemia.
39
Another study examined
this relationship in over 400 hemodialysis patients. Serum magnesium levels were
significantly lower in PPI users than nonusers (0.94
Hypomagnesemia often goes undetected in the general population; thus, the true incidence of PPI-related hypomagnesemia is difficult to determine and is likely under reported. 41 Most evidence suggests that hypomagnesemia is a problem for chronic PPI users, rather than patients who receive them for short-term gastrointestinal prophylaxis in the intensive care unit. Patients with CKD and those receiving medications known to lower magnesium concentration, such as diuretics, are also thought to be at higher risk.
Other micronutrient deficiencies
Summary
PPI use may be associated with various micronutrient deficiencies. The development of these deficiencies is likely also highly correlated with additional patient risk factors, rather than being singly attributed to PPI use. Further study into the long-term effects and clinical implications of PPI-related micronutrient deficiency is warranted.
vii. Bone density, fracture risk and PPIs
In 2010, the US FDA revised the labeling of PPIs to include a warning about the possible increased risk of bone fractures with their use. This warning was retracted from the OTC PPI in 2011 due to insufficient evidence that short-term, low-dose OTC PPI use was associated with bone density changes. These negative effects on bone health may be related to nutritional deficiencies. As discussed previously, the increase in gastric pH with the use of PPIs may impede calcium absorption. 15 There is currently insufficient evidence to support routine monitoring of bone mineral density in patients on PPIs without other indications for monitoring.
viii. Spontaneous bacterial peritonitis
The data suggesting an association come primarily from observational studies, where causality cannot be established. This spontaneous bacterial peritonitis concern was highlighted especially in the liver disease and cirrhosis patient cohort. 15 The biologic plausibility of secondary bacterial infections in patients with long-term PPI exposure stems from the role of hypochlorhydria and subsequent pH changes in the colon (downstream effect) allowing for bacterial translocation and colonic transmigration leading to gram negative peritonitis. The current level of evidence does not allow for broad recommendations and certainly does not support withholding PPIs, if indicated, in patients with liver disease.
Long-term PPI use and overall risk of death from all causes
Some investigators have evaluated the role of PPIs and all-cause mortality following
prolonged exposure rather than individual organ system dysfunction. Xie and
colleagues evaluated patient data from the US Department of Veterans Affairs as a
longitudinal observational cohort study.
44
This study used complex
statistical analysis including time-dependent propensity score-matched cohorts and
high-dimensional propensity score-adjusted models to reduce the potential
confounding bias. In the primary cohort of new users of acid suppression therapy
followed for a median of 5.71 years, they showed an association between PPI
use and an increased risk of all-cause mortality
A word of caution about false alarms and broad conclusions
Despite a large number of studies, the overall quality of evidence for adverse effects of long-term use of PPIs is low to very low. Overzealous conclusions based on weak associations can be problematic and maybe partly responsible to the growing alarm about the prescription of PPIs today. It is worth noting that the ‘guilty by association’ problem facing PPIs is not unique; in fact, Hill eloquently addressed issues with observational studies in as early as 1965. 45 He proposed a set of nine ‘aspects of association’ (strength of association, consistency, specificity, temporality, biological gradient, plausibility, coherence, experiment, and analogy) to evaluate countless hypothesized relationships between exposures and disease outcomes.
Observational studies, no matter how well performed, may be inherently incapable of accurately discerning weak associations from null effects due to their susceptibility to systematic errors of bias/confounding and other methodological weaknesses. 46 Also, statistical significance only takes random errors related to sample size into consideration; it ignores systematic errors.
Laine and colleagues recently reported on the difficulty in eliminating residual bias in observational studies even with statistical adjustment, because all confounding factors are not recorded or even known. 16 This may be especially important when effect sizes are small (odds/hazard ratio < 2), and so it may not be possible to determine whether the association is valid or the result of residual bias.
Conclusion
We must be cautious about drawing broad conclusions on the current level of evidence with the long-term use of PPIs. This is especially important because the conclusions are overwhelmingly based on observational studies and meta-analyses, which frequently include the same observational studies.
PPIs have had a profound impact on the outcomes of patients with acid peptic disease since their introduction into clinical practice in the late 1990s. They continue to have a strong positive impact when used appropriately for the recognized indications. The optimal strategy for PPI prescription at this time is for patients with clear indications, avoiding broad off-label use and to have a prudent time-limited endpoint of prescription.
Footnotes
Funding
The study was supported by the Department of Anesthesiology ( Division of Critical Care) only.
Conflict of interest statement
The authors declare that there is no conflict of interest.
