Abstract
Background:
Medication errors are common in neonatal care and can lead to significant harm. We sought to explore utilization of various medication error prevention strategies across Australian and New Zealand neonatal units (NNUs) through a clinical practice survey.
Methods:
An electronic survey was distributed in October 2016 to relevant staff at each of the 29 level III NNUs identified as members of the Australian and New Zealand Neonatal Network (ANZNN). The survey contained questions relating to a range of medication error prevention strategies identified from a previous systematic review on the topic. The evaluated interventions targeted different aspects of the medication-use process including prescribing, evaluation/checking of orders by clinical pharmacists, transmission, preparation and dispensing of orders, storage of medications, and medication administration.
Results:
From the 20 respondents, the evidence-based strategies most commonly utilized were use of smart pumps (n = 18; 90%), and ward-based clinical pharmacists (n = 17; 85%). Interventions least commonly utilized included barcode scanning with medication administration (n = 0; 0%), electronic prescribing and clinical decision support (n = 1; 5%), and dedicated medication administration nurse (n = 2; 10%). The total number of evidence-based medication error prevention strategies utilized in each NNU ranged from 2 to 10 (median = 7), 10 of 16 strategies were utilized by less than 50% of NNUs.
Conclusion:
While evidence supports utilization of a number of medication error prevention strategies, these appear inconsistently utilized across current practice settings.
Introduction
Medication errors represent a significant cause of neonatal morbidity and mortality and in many cases are potentially preventable. Neonates have been described as being more prone to medication errors across each step of the medication use process due to the inherent increased need for calculations, dilutions, and manipulations of medications. 1 In many cases, medications are used off label, that is, they are not licensed for use in neonates, meaning that evidence to guide safe use in the neonatal setting is often limited and available dosing strengths and formulations are less suitable for neonatal use. 2
In light of the complexity of medication use in the neonatal setting, the high frequency with which medications are used and the potential for serious adverse events of even minor medication errors, intervention strategies to increase medication safety are of great importance. Previous systematic reviews have highlighted common causes of medication errors in the neonatal setting (e.g. dilution of medications, prescribing errors, medication selection errors),2,3 or have outlined a range of interventions which have been demonstrated to reduce the incidence of medication errors including electronic prescribing (e.g. electronic prescribing, standardized infusion concentrations, ward-based clinical pharmacy services, use of smart pumps for medication administration, smart pump medication administration library, and barcode medication administration system).3–5
While there has been interest in identification of factors contributing towards medication errors and the evaluation of interventions for improving medication safety in neonatal care, there is a paucity of comprehensive data evaluating the utilization of specific error prevention strategies in neonatal units (NNUs). Therefore, the objective of this study was to explore utilization of various medication error prevention strategies in routine clinical practice across Australian and New Zealand NNUs.
Methods
We undertook a systematic review of interventions for reducing medication errors in neonatal care. Based on these findings, we developed a survey designed to evaluate utilization of 16 evidence-based interventions across various clinical practice settings. The evaluated interventions targeted different aspects of the medication use process. These were broadly assigned to three categories including prescribing and evaluation of medication orders, obtaining and storing medications, and medication administration. The survey was expanded to include an additional six interventions that were considered to be of uncertain efficacy. These were included based on knowledge of interventions studied in adult settings, or those utilized in the study author’s own NNU, but where published evidence to support such strategies in the NNU setting was unavailable.
Pilot testing of the survey was undertaken at two sites, resulting in minor modifications to improve clarity. The electronic survey was distributed between October 2016 and January 2017 to relevant staff at each of the 29 level 3 neonatal units (NNU) identified as members of the Australian and New Zealand Neonatal Network (ANZNN). Surveys were emailed to pharmacists and neonatal unit managers at each site. Survey recipients at the same site were asked to assist each other with providing one consolidated response. Reminder emails were sent at 4 and 8 weeks if no response had been received following the original email. Recipients were advised that responses received would be de-identified and collated for analysis and publication of results. Individual participant consent was not required, as this clinical practice survey was considered exempt from ethical review by the Southern Adelaide Clinical Human Research Ethics Committee.
A copy of the survey is included as an appendix (Appendix 1). All questions were positively worded, with a combination of closed and open-ended questions utilized. Responses consisted of either simple yes/no answers, or a four-item scale of ‘Always’, ‘Frequently’, ‘Sometimes’, or ‘Never’. The four-item scale was introduced following pilot testing of the survey, as respondents felt uncomfortable answering all questions as either yes or no where certain interventions were not utilized all of the time. Responses of ‘Always’ or ‘Frequently’ were combined (indicating ⩾50% use) to determine utilization of medication error prevention strategies.
De-identified survey responses were entered into a Microsoft Excel spreadsheet and reported using descriptive statistics. Comparisons in the median medication safety score according to country and hospital type were undertaken using the Wilcoxon rank-sum test. Statistical significance was defined as a two-sided p value of <0.05. All statistical analyses were undertaken using STATA IC 14 (Stata, College Station, Texas).
Results
Responses were received from 20 of 29 ANZNN NNUs (response rate 69%), with the response rate similar across Australia (n = 16/23; 70%) and New Zealand (n = 4/6; 67%). Responses were received from hospitals located in all states and territories within Australia, including the Australian Capital Territory (n = 1), New South Wales (n = 4), Northern Territory (n = 1), Queensland (n = 3), South Australia (n = 2), Tasmania (n = 1), Victoria (n = 3) and Western Australia (n = 1). Hospital types included a mixture of general hospitals (n = 14; 70%) and specialist maternity or paediatric hospitals (n = 6; 30%). The majority of surveys were returned electronically by pharmacists (n = 18).
A summary of 16 aspects of clinical care considered important for medication safety according to previous systematic reviews is provided in Figure 1. Interventions most commonly utilized across NNUs included use of smart pumps for medication infusions (n = 18; 90%), availability of a ward-based clinical pharmacist (n = 17; 85%) and requirement for nursing staff to complete a medication administration calculation test to be able to administer medications (n = 17; 85%). Interventions less commonly utilized included barcode scanning with medication administration (n = 0; 0%), electronic prescribing and clinical decision support (n = 1; 5%), and availability of dedicated medication administration nurse (n = 2; 10%). Of the 18 sites reporting a ward-based clinical pharmacy service, none reported having a 7-day service. Of the 17 sites reporting use of a medication administration calculation test, this was specific to the neonatal unit in 15 sites, and 6 sites reported that this was only required to be completed once, rather than repeated. Eight sites reported using an on-site sterile pharmacy service to prepare high-risk medications. The most commonly provided examples included morphine (n = 4), inotropes such as dopamine (n = 4), amphotericin B (n = 3), alprostadil (n = 3), indometacin (n = 3), amikacin (n = 2), ganciclovir (n = 2), fentanyl (n = 2) and midazolam (n = 2). Five sites reported having prediluted medications available on the ward. The most commonly provided examples included amikacin (n = 2), caffeine citrate (n = 2), gentamicin (n = 2), vancomycin (n = 2) and alprostadil (n = 1).
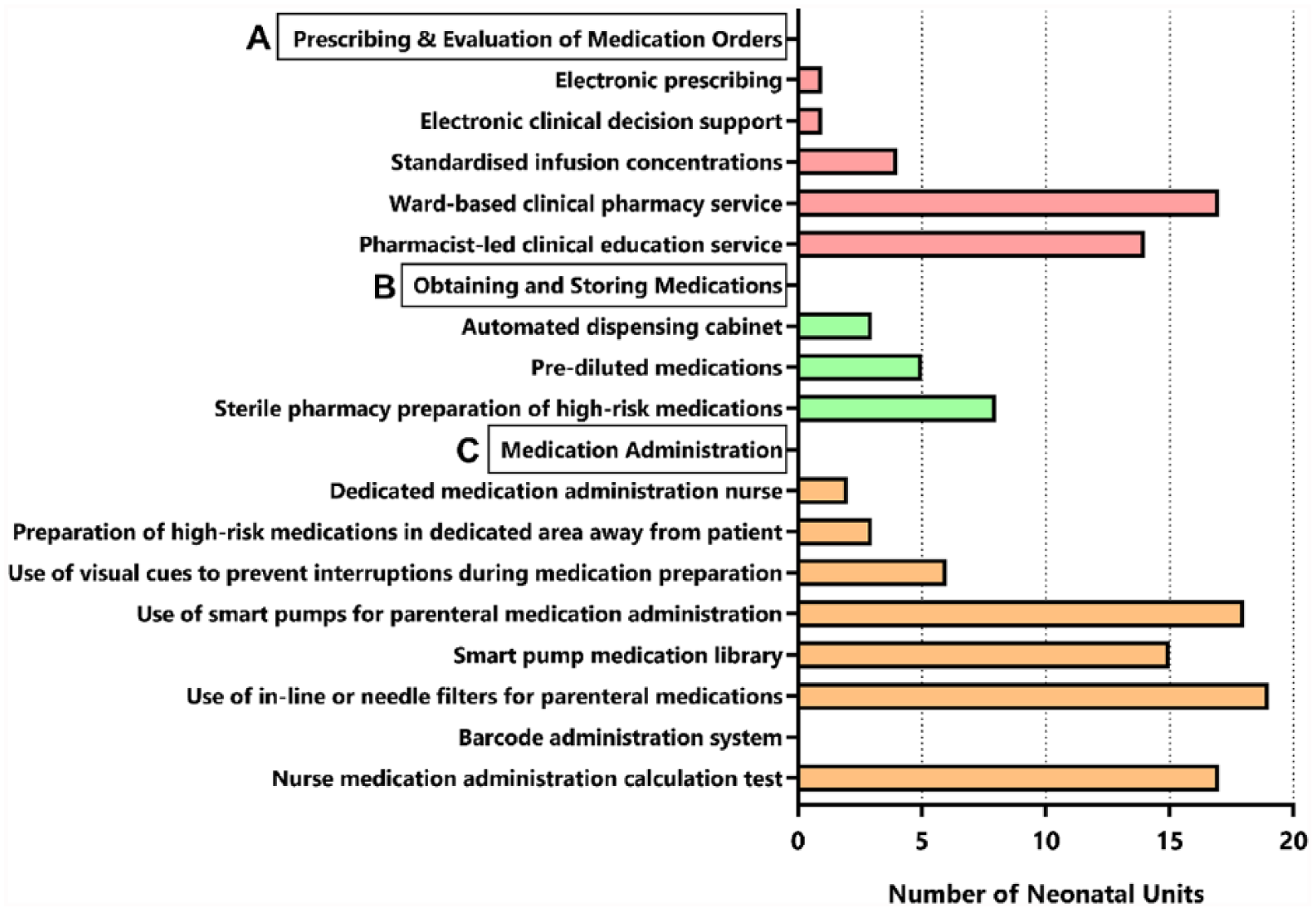
Utilization of medication error prevention strategies by Australian and New Zealand neonatal intensive care units.
The total number of evidence-based medication error prevention strategies utilized in each NNU ranged from 2 to 10 (median = 7; Figure 2). There was no difference in the total number of medication error prevention strategies utilized between Australian (7, range 2–10) and New Zealand (7, range 6–8; p = 0.847) NNUs, or between general hospitals (7, 2–10) and specialist maternity or paediatric hospitals (6.5, 5–9; p = 0.583).
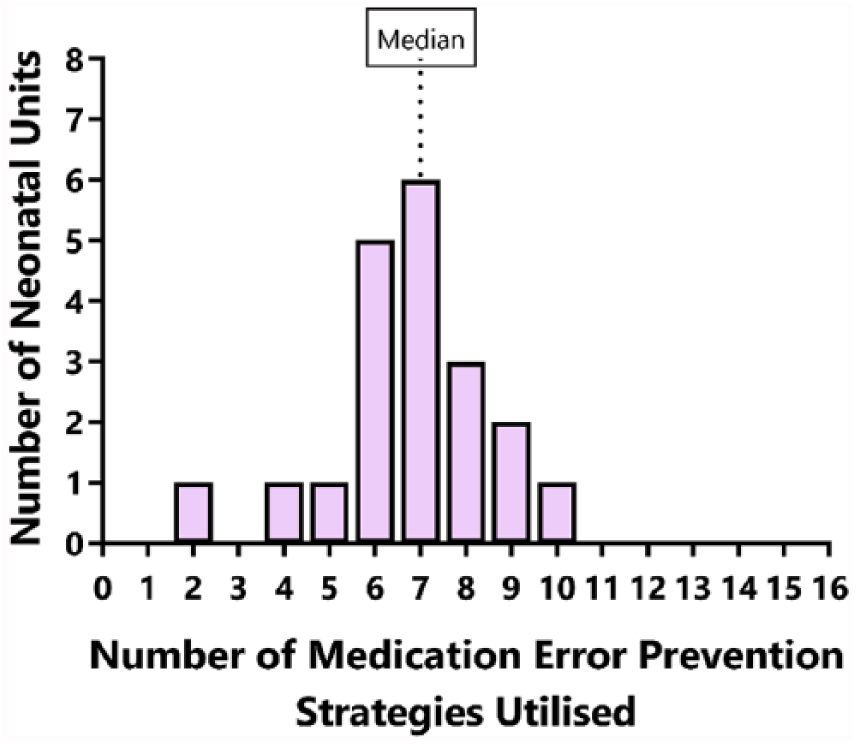
Histogram of number of medication error prevention strategies utilized by Australian and New Zealand neonatal intensive care units.
A summary of the additional six aspects of clinical care that were considered to be of uncertain benefit is provided in Supplemental Figure 1. All of these interventions were utilized by less than 50% of NNUs. These included pharmacist prescribing (n = 1; 5%), nursing documentation of dilution and administration calculations when administering medications (n = 4; 20%), standardized medication administration times for routine medications (n = 7; 35%), preprinted prescribing stickers (n = 8; 40%), routine use of electronic rather than paper-based medication guidelines (n = 8; 40%), and pharmacist attendance at ward rounds (n = 9; 45%). The one site utilizing pharmacist prescribing reported the ability of the pharmacist to prescribe cholecalciferol and nystatin. Among the eight sites reporting use of preprinted prescribing stickers, these were most commonly used for prescribing eye drops for eye examinations (n = 4) or sucrose for analgesia (n = 4). Individual sites reported use of preprinted stickers for prescribing other medications such as iron, vitamins, nystatin, probiotics and vancomycin.
Discussion
While evidence supports the role of various medication error prevention strategies in improving medication safety in neonatal care,3–5 our clinical practice survey of 20 NNUs across Australia and New Zealand identified that such strategies are poorly utilized. Underutilization of medication error prevention strategies may place vulnerable newborns at risk of potentially preventable medication-related harms.
To the best of our knowledge, we are not aware of previous surveys providing a comprehensive overview of the utilization of such error prevention strategies within current clinical practice in the Australasian setting. Our results, however, can be compared with the recent publication of a national survey of medication safety practices of NNUs in the United States. 6 Utilizing the same methodology, Greenberg and colleagues asked about utilization of a similar range of medication safety intervention strategies. 6 They obtained responses from 55% of surveyed NNUs, with more than 85% reporting adherence to practices including use of electronic health records, computerized physician order entry, and clinical decision support. 6 This is in stark contrast to our survey where such interventions were utilized by just one (5%) Australian NNU. A slightly lower percentage of NNUs in the United States reported routine use of barcoding of medication administration (72%), compared with no site using this technology in our survey. These findings demonstrate significant variability in the use of technology-based interventions across US, and Australian or New Zealand NNUs. In contrast, the presence of pharmacists on ward rounds (36%) was similar to that reported in our survey (45%).
Findings from clinical practice surveys such as this provide an important snapshot of current clinical practice and highlight many opportunities for neonatal units to implement additional practices to reduce the risk of medication errors. Results can be utilized as part of benchmarking for NNUs to identify areas for further improvement in line with their own knowledge about what the incidence of medication errors are within their NNU and what the causes of these errors might be. Reporting of such findings also provides opportunity for shared learnings between NNUs with respect to the process of implementing, evaluating, and sustaining various practices.
While previous systematic reviews clearly demonstrate that a range of interventions are associated with reductions in medication errors, no single intervention has been demonstrated as clearly superior to others.3–5 Further, interventions differ in their abilities to reduce medication errors related to prescribing, administration or both.3–5 Given medication errors can occur across different stages of the medication use process, the findings of these reviews suggest that multifaceted approaches that target each aspect of the medication use process are essential for promoting medication safety in clinical practice.3–5 A notable limitation of the existing medication safety literature in the neonatal setting is the heavy reliance on observational study designs that are subject to a range of biases rendering interpretation of the true effect difficult. 5 A further limitation is the lack of data relating to cost effectiveness, particularly for technology-based interventions. The financial burden associated with some strategies may represent a significant barrier to their uptake and explain their relatively low utilization in this survey.
Our survey identified, albeit infrequent, utilization of a range of intervention strategies that were considered to be of uncertain effectiveness in reducing risk of medication errors. This highlights a range of interventions that should be subject to formal evaluation, such as pharmacy prescribing, pharmacist attendance at ward rounds, or requiring nursing staff to document dilution and administration calculations prior to administering medications. The concept of pharmacist prescribing is new to Australia, but is common in places like the United Kingdom. A recent survey of neonatal unit pharmacists in the UK identified that 47% were able to prescribe medications. 7 While there were many perceived benefits of pharmacist prescribing, including the potential for reducing prescribing errors, the practice is still lacking formal evaluation in this setting. Similarly, while pharmacist involvement in undertaking clinical review of patients in the neonatal unit is an important medication safety intervention, the benefit of attendance on consultant ward rounds is uncertain. Evidence supporting involvement can be taken from a recent systematic review of interventions to reduce medication errors in adult intensive care which identified a number of studies demonstrating reductions in medication errors following inclusion of a pharmacist in consultant ward round. 8 Whether their findings are generalizable to the neonatal intensive care unit setting is uncertain. Lastly, the rights of medication administration (e.g. right patient, drug, dose, route and time) is a familiar concept incorporated within hospital policies and guidelines and nursing education practices to reduce the risk of medication errors. 9 The requirement for nursing staff to document these rights, including dilution and administration calculations, prior to administering medications is seen by some as improving accountability, increasing the likelihood of nursing staff identifying prescribing errors, and reducing risk of administration errors. While this documentation process may assist in reducing unintended dosing errors due to incorrect dosing calculations, medication errors can still occur where nursing staff fail to identify that an incorrect dose was prescribed in the first place. Given the potential for dosing errors in the neonatal setting, formal evaluation of strategies for reducing administration errors are required.
A strength of this survey is the high response rate, with good representation of hospitals located across Australia and New Zealand. The response rate obtained in this study (69%) was higher than that of a similar recent medication safety intervention survey undertaken in the United States (55%). A limitation was that medication safety scores or utilization of various interventions were not able to be correlated with the incidence of medication errors within each NNU. This would be of interest for subsequent studies, noting, however, the difficulties of accurately determining the frequency and severity of medication errors when relying on medication incident reports, rather than detailed clinical audit. 10 A further limitation is that there may be additional aspects of clinical practice that may contribute towards medication safety that have not been captured by the survey. Also, the survey evaluated whether or not medication safety intervention strategies were utilized at each site but could not determine adherence to such interventions. For example, while some sites had a policy for high-risk medications to be prepared in a dedicated area away from the patient, we could not determine if staff were 100% adherent to this policy. Such adherence measures could only be evaluated through detailed observational studies at each site.
In conclusion, our clinical practice survey provides an important snapshot of current practice across Australian and New Zealand NNUs and demonstrates relatively poor uptake across a range of medication error prevention strategies. Further research should focus on exploring the key barriers to uptake of such strategies, while also expanding the strength of evidence pertaining to the effectiveness of various strategies (particularly their cost effectiveness). Finally, greater emphasis should be placed on facilitating shared learnings from the implementation of such strategies where they are being utilized and evaluating the impact of such strategies on medication errors and patient safety.
Supplemental Material
Supplemental_Figure_1 – Supplemental material for Utilization of neonatal medication error prevention strategies: a clinical practice survey of Australian and New Zealand neonatal units
Supplemental material, Supplemental_Figure_1 for Utilization of neonatal medication error prevention strategies: a clinical practice survey of Australian and New Zealand neonatal units by Nadine Matti, Minh-Nha R. Nguyen, Cassandra Mosel and Luke E. Grzeskowiak in Therapeutic Advances in Drug Safety
Footnotes
Appendix 1

| Survey on neonatal medication error prevention strategies | ||
|---|---|---|
| This clinical practice survey is considered exempt from ethical review according to the NHMRC National Statement on Ethical Conduct in Human Research 2007. | ||
|
|
|
|
| All answers provided below will be de-identified prior to analysis and presentation of results. | ||
|
|
□ Australia □ New Zealand | |
|
|
□ General hospital (adult and children) □ Specialist women’s/pediatric hospital |
|
Always = 85–100% Frequently = 50–84% Sometimes = 20–49% Never = 0–19% |
||
|
|
||
□ Yes → □ No (go to next question below) |
□ Yes □ No |
|
| □ Always □ Frequently □ Sometimes □ Never | ||
e.g. All Penta-vite® at 12 p.m. |
□ Always □ Frequently □ Sometimes □ Never | |
|
|
□ Electronically only □ Paper-based folder/s □ Either |
|
□ Yes → □ No (go to next question below) |
|
|
|
|
||
□ Yes → □ No (go to next question below) |
□ 5 days a week (Mon to Fri) □ 7 days a week □ Other, please describe: □ Always □ Frequently □ Sometimes □ Never |
|
□ Yes → □ No (go to next question below) |
|
|
□ Yes → □ No (go to next question below) |
□ Nursing staff □ Medical staff Both □ 2–3 times a week □ Once weekly □ Monthly □ Other, please describe: |
|
|
|
||
□ Yes → □ No (go to next question below) |
□ All medications □ Most medications □ Some medications □ Few medications □ Yes □ No |
|
e.g. vancomycin prediluted to 5 mg/ml rather than 500 mg stock vial □ Yes → □ No (go to next question below) |
|
|
Yes → No (go to next question below) |
□ On site □ Off site □ Both (i.e. may use a combination of both) □ Other, please describe: |
|
□ Yes → □ No (go to next question below) |
|
|
□ Yes □ No |
|
|
|
|
□ Any nurse who happens to be close by (as long as they are appropriately credentialled) □ The shift coordinator or senior nurse supervising the relevant clinical area □ A dedicated medication administration nurse who is not designated as the shift coordinator or senior nurse |
|
|
|
□ Bedside bench □ Portable medication trolley □ Separate medication preparation area |
|
□ Yes → □ No (go to next question below) |
|
|
□ Yes → □ No (go to next question below) |
□ Yes □ No |
|
|
|
□ Always □ Frequently □ Sometimes □ Never |
|
|
|
□ Always □ Frequently □ Sometimes □ Never □ Electronically □ Pre-printed calculation sheets □ Medication chart □ Other, please describe |
|
□ Yes □ No |
||
|
|
||
|
|
||
Acknowledgements
LEG acknowledges salary support provided through an Australian National Health and Medical Research Council (NHMRC) Early Career Fellowship (APP1070421).
Author contribution
MN, CM, and LEG conceived the review and contributed towards study design. MN and LEG acquired the data. NM and LEG analysed the data. All authors were involved in the interpretation of data. NM and LEG drafted the manuscript. MN and CM were involved in critical revision of the manuscript, with all study authors approving the final version for submission.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Supplemental Material
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
