Abstract
International guidelines recommend blood pressure-lowering therapy, statins and aspirin for people who have had a cardiovascular event but use of these medications is low, particularly for lower income countries. Clinical trials have demonstrated that combining these medications into a single pill or capsule (a ‘polypill’) improves adherence, systolic blood pressure and low density lipoprotein cholesterol compared with usual care in secondary prevention. Uptake of polypill-based care has been underwhelming, possibly due to safety concerns. Overall, results from the clinical trials of polypill use among people who have had a cardiovascular event show no immediate safety concerns. Increased use and adherence to medications will always be associated with side effects however use within a combination medication has not been shown to be any less safe than individual component medications. Research investigating the relative consequences of nonadherence to a polypill compared with individual components would be useful.
Introduction
Cardiovascular disease (CVD) is the major cause of global mortality, morbidity worldwide.1–4 An estimated 17.5 million people died from CVDs in 2012, representing 31% of all global deaths 5 and 37% of premature deaths resulting from all noncommunicable diseases. 6 Of these deaths, ischaemic heart disease was the most common and stroke the second most common cause of death in 20125 and this ranking is projected to remain the same in 2030. 7
CVD events and deaths are more common among people who have already had a CVD event than those who have not had a prior event.8,9 International guidelines are therefore focused on medical management for these people, in whom blood pressure (BP)-lowering therapy, statins and aspirin, as well as lifestyle advice, are recommended. However the use of these medications is low internationally, particularly for lower income countries. Only 44% of people with a prior CVD event in high-income countries (13% in upper-middle and 3% in lower-middle and low-income countries) reported taking at least three of four recommended preventive medications (aspirin, statin, angiotensin-converting enzyme inhibitor/angiotensin receptor blocker and another BP-lowering drug) in a survey published in 2011. 10
Use of fixed-dose combination medications or ‘polypills’ containing statins, BP-lowering and antiplatelet drugs is one strategy that has been shown to improve adherence, systolic BP and low density lipoprotein cholesterol (LDL) compared with usual care among patients with CVD or at high risk of their first event in an individual participant data meta-analysis of 3140 patients in six countries. 11 While the use of a polypill for CVD, along with the term itself, was popularized by Wald and Law who proposed giving a polypill to everyone above a certain age, 12 the most compelling application of polypill-based care is in the secondary prevention of CVD.12,13 In this context, a polypill is not a ‘magic bullet’; rather it is simply a mechanism for delivering medications with established indications in a high-risk group with known large and persistent treatment gaps.
Despite the strength of evidence supporting the use of polypill-based care in the secondary prevention of CVD, 11 the availability of polypills in more than 30 countries 14 and high acceptability to patients,15–17 uptake of this intervention has been underwhelming. 14 A number of safety concerns have been voiced about polypill-based care and these concerns may be limiting uptake. The purpose of this article is to consider whether or not these concerns are justified, in the light of best available evidence. Concerns have been broadly divided into those that can be addressed by clinical trials, and those that cannot.
Evidence from clinical trials
Relevant trials
BP-lowering therapy, statins and aspirin are all indicated for patients who have already have a CVD event, therefore only trials of polypills containing all three components and in which the comparator was active treatment are considered here. Given that all medications are indicated, a no treatment or placebo comparator would be unethical. Blinding of participants is inappropriate given that the intervention is a mechanism for delivering medication.
A 2017 Cochrane systematic review of polypill-based care for the prevention of CVD extensively searched the literature for trials of polypills containing at least one BP-lowering and one lipid-lowering component. 18 The systematic review identified four trials of relevance here: FOCUS (Fixed-Dose Combination Drug for Secondary Cardiovascular Prevention) 2014, 18 IMPACT (IMProving Adherence using Combination Therapy) 2014, 16 Kanyini-GAP (Guidelines Adherence with the Polypill) 2015 19 and UMPIRE (Use of a Multi-drug Pill in Reducing Cardiovascular Events) 2013 20 (Table 1). FOCUS compared a polypill with the three components separately for 9 months among 695 patients who had experienced a myocardial infarction within the preceding 2 years. 18 The other three trials (IMPACT 2014, 16 Kanyini-GAP 2015 19 and UMPIRE 201320), part of the SPACE (Single Pill to Avert Cardiovascular Events) collaboration, compared a polypill with usual care for a minimum of 12 months among 3140 patients with a prior CVD event (2385, 76%) or at high risk of their first event. 11 The three SPACE trials were based on the same trial design 21 and a prospective individual participant meta-analysis was conducted. 11 At the time of trial design in the SPACE collaboration, high-risk primary prevention patients were indicated for antiplatelet therapy according to the guidelines and hence were treated in the same way as secondary prevention patients in these trials. Since these trials were concluded the risk–benefit profile of aspirin has shifted for primary prevention patients and most guidelines would not recommend aspirin now in these patients.
Relevant trials.
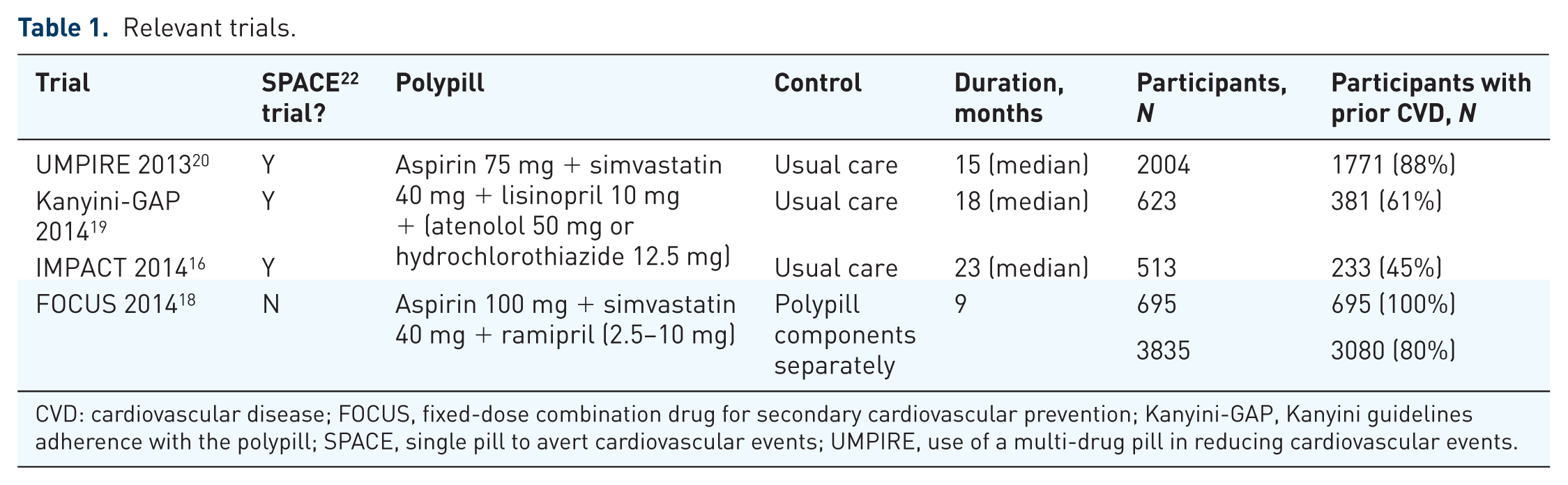
CVD: cardiovascular disease; FOCUS, fixed-dose combination drug for secondary cardiovascular prevention; Kanyini-GAP, Kanyini guidelines adherence with the polypill; SPACE, single pill to avert cardiovascular events; UMPIRE, use of a multi-drug pill in reducing cardiovascular events.
Serious adverse events
There was no statistically significant difference in the number of patients reporting one or more serious adverse events (SAEs) between groups in FOCUS (polypill 21/350, 6.0%, separate components 23/345, 6.6%, p value not reported). In the SPACE trials 360 patients (22.9%) in the polypill arm reported one or more SAEs compared with 316 (20.1%) in the usual care arm [relative risk (RR), 1.12, 95% confidence interval (CI) 0.99–1.27, p = 0.07]. Of these SAEs a numerical excess occurred at the 1 and 6 month visits in the polypill group with more SAEs reported as ‘medically important’ (145 versus 115, p = 0.04) but no significant excess of other SAEs (i.e. fatal, life-threatening, leading to hospitalization). When SAEs were categorized according to different MedDRA system organ classes there were no significant differences between arms except for miscellaneous vascular disorders (35 versus 17; of which hypotension accounted for 11 versus 0) and breast and reproductive disorders (6 versus 17 events, of which prostate issues including benign prostatic hyperplasia, accounted for 3 versus 10).
The investigators report that ‘this result in the SPACE trials could partly be due to a lower threshold for reporting events in the polypill group, as there was a numerical excess in 18 of 24 diverse SAE categories; and hypotension was reported for 11 patients in the polypill group but none in the usual care group, whereas hypotension is typically reported in the placebo group of placebo-controlled trials.’ 11
Although caution is advised when interpreting these data, when switching people onto a polypill from stable treatment regimens, or adding new medications a real increase in adverse events might be expected. The data from the SPACE collaboration analyses suggest that any increase is modest in size and occurs within 6 months of the switch. Differing polypill formulations for example with an angiotensin receptor blocker rather than an angiotensin converting enzyme (ACE) inhibitor, or a range of dose combinations, may reduce some of this potential increase in side effects.
Across trials there was considerable variation in the proportion of control participants experiencing an SAE, from 7% in FOCUS to 20% in the SPACE trials (UMPIRE 10%, Kanyini-GAP 41% and IMPACT 36%). These differences in absolute rates of the reporting of SAEs could be a reflection of study setting and trial duration; the lowest rates were observed in FOCUS and UMPIRE, which were based largely in secondary care settings, whereas Kanyini-GAP and IMPACT were based in primary care and were of longer duration than the other two trials.
Adverse events
Data on all adverse effects (AEs, not just SAEs) were only routinely collected by the FOCUS trial. FOCUS found no statistically significant difference between polypill and control groups in the proportion of participants experiencing any AE (124/350, 35.4% versus 112/345, 32.5%), a hospitalization (6.0 versus 6.7), a haematological AE (1.4% versus 1.7%), musculoskeletal adverse event (1.4% versus 3.8%), cough (1.4% versus 1.7%), dizziness (0.6% versus 0.6%), or hypotension (0.0% versus 0.2%) (no p values reported). 18
Treatment discontinuation
Of the four trials considered in this review, only the FOCUS trial was able to validly compare treatment discontinuation rates between polypill and control groups because the comparator was the separate components of the polypill being used, as opposed to heterogeneous usual care like the other trials. 18 FOCUS reported that treatment was discontinued due to AEs in 4.0% of polypill (14/350) and 3.7% (13/345) of control participants.
Polypill discontinuation (whether or not there was a suitable comparator) was reported by all four trials. As noted above, in FOCUS, the polypill was discontinued due to AEs in 4% (14/350) of participants randomized to this treatment. The SPACE trials collected data on all reasons for polypill discontinuation. A total of 28% (446/1569) of participants discontinued the polypill in the SPACE trials. The most commonly reported reason for discontinuation was an AE (10%, 160/1569), followed by advice of doctor (7%, 105/1569) and patient choice (5%, 85/1569).
The difference in polypill discontinuation due to AEs between the FOCUS trial and the SPACE trials could be a reflection of the difference in trial duration (9 months versus median 15 months).
Lifestyle risk factors
Concerns have been raised that if a polypill is portrayed or perceived as a preventive panacea, the use of a polypill may deflect attention from lifestyle risk factors. 23 The SPACE trial collected data on anthropometric measures (body mass index, waist circumference, weight) and lifestyle behaviours (smoking status, number of cigarettes smoked/day, alcohol use status, units of alcohol/week, minutes of physical activity/week). 24 An individual participant data meta-analysis of these measures and behaviours found no significant difference between polypill and usual care patients, combined or in a range of subgroups, and confidence intervals were narrow, excluding any major effect. 24
Other issues
Polypill components, titration and flexibility
The suitability of some polypill components in the light of current guidelines have been questioned along with perceived adverse impact of inflexibility of prescribing due to limited titration options among polypill components. Firstly, it must be noted that the availability of a broad range of individual medications with a wide range of titration options has not proven effective thus far in achieving adequate medication prescription and adherence even in high-income countries. Therefore, postulating that lack of such options will adversely impact clinicians’ capacity to adequately individualize patient treatment and achieve worse outcomes than are currently seen seems unlikely.
Secondly, much of this discussion is specifically related to the polypills offered within the clinical trials. Options for drug components and doses were limited due to the developmental nature of the trial drugs, and patent protection on more potent statins such as atorvastatin and rosuvastatin at the time of drug development. Patent protection of atorvastatin and rosuvastatin are no longer an issue and atorvastatin is now being routinely incorporated into ‘next generation’ commercial polypills 14 and rosuvastatin into polypills utilized in clinical trials. 25 A wider variety of BP-lowering drugs is now available in different polypills with two widely available polypills, Trinomia™ and Triveram™, also offering multiple doses of BP-lowering drugs within their compositions, allowing for some flexibility in titration. Ultimately, market availability of components and dose options will be driven by consumer demand. As further manufacturers come on board with marketed polypills there will be likely be substantially more variety available.
Even without such component variability and titration options, additional drugs can also be prescribed on top of the polypill if additional medications are required. The Kanyini-GAP trial looked at additional prescribing on top of the polypill at the end of follow up (median 18 months). 26 Of those still taking the polypill, 44% were prescribed additional medications of which the majority (85%) were BP-lowering medications. Although this appears counterintuitive to the premise of the polypill in simplifying medication regimes, there was still an overall mean reduction in pill burden of 2 and the overall outcome for all participants in the study was a 49% improvement in adherence (RR 1.49, 95% CI 1.30–1.72).
Equivalent therapeutic benefit
In order to obtain regulatory approval for a cardiovascular polypill, complex pharmacodynamics and pharmacokinetic testing is required to demonstrate bioequivalence of the combination to its constituent components, and the product needs to undergo testing for stability and safety.27,28 Concerns therefore about whether or not combination medications confer the equivalent benefits to the individual medications are not an issue as long as the regulatory requirements are fulfilled. Demonstration of an effect of polypills on hard cardiovascular outcomes such as heart attack and stroke are not a requirement for regulatory approval among people who have had a CVD event because of the strength of the evidence and consistency in international guideline recommendations supporting the use of component medications in this group of patients.27,28
Stop taking one, stop taking all
One safety concern that has not been addressed in the clinical trials thus far has been the concern that, for patients who are not adherent to their medications, nonadherence to a polypill will necessarily mean nonadherence to all risk-reducing medications rather than perhaps only one or two. Generating evidence to address this issue is challenging. Randomization to nonadherence in a trial is obviously unethical. Observational data from patients are known to be prone to bias and confounding. Data from the trials confirm that patients taking the polypill are also prone to nonadherence albeit less than the individual medications. The effect of this on clinical outcomes and whether or not this confers more clinical risk than nonadherence to individual medications is however unclear. What is known is that in the SPACE collaboration group of trials, although patients in both arms over the course of follow up showed deterioration in adherence, at the end of follow up there was still a clear adherence advantage in the polypill arm compared with the usual care. 11
Safety in primary prevention
The SPACE collaboration trials also included a large number of patients who, although not yet having been diagnosed with CVD, were at high calculated risk of having a first event (>15% risk over 5 years). Although the risk–benefit ratio of taking antiplatelet medication in this group is more uncertain in 2017, guidelines still routinely recommend treatment with BP-lowering and statin medication in this group of patients. Nonaspirin polypills containing solely BP-lowering medication and statins are available in many countries and the safety issues discussed in this paper also pertain to high-risk primary prevention patients when thinking about statin and BP-lowering combination polypills.
Primary prevention patients who are at moderate risk require more careful consideration. Trials such as HOPE-3 29 have shown that concurrent use of statins and BP-lowering medication results in lower risk due to the statin component; however, benefit from BP-lowering was only seen in those with a higher baseline BP. Until guidelines routinely recommend the use of statins and BP-lowering agents in those at moderate risk, discussion of the use of a polypill, and thereafter safety issues, is premature in the light of few trials and limited evidence.
Widespread use of polypills in patients categorized by single risk factors, for example age, as per the original Wald and Law concept 12 is generally not commonly accepted and requires further evidence.
Conclusion
Overall, results from the clinical trials of polypill use in secondary prevention show no immediate safety concerns. Increased use and adherence to medications will always be associated with side effects; however, use within a combination medication has not been shown to be any less well tolerated than individual component medication. This field would benefit from additional research into any AEs of nonadherence to polypills compared with usual care.
Footnotes
Acknowledgements
RW is supported by an NHMRC Early Career Fellowship (APP1125044).
Funding
Funding was received from Dr Reddy’s Ltd (Hyderabad, India) for coordination of the SPACE program (![]() ). RW was the international coordinator of the SPACE collaboration and VS was the study director for the IMPACT trial. The polypills used in the SPACE trials were manufactured and supplied by Dr Reddy’s Ltd free of charge.
). RW was the international coordinator of the SPACE collaboration and VS was the study director for the IMPACT trial. The polypills used in the SPACE trials were manufactured and supplied by Dr Reddy’s Ltd free of charge.
Conflict of interest statement
The authors declare that there is no conflict of interest.
