Abstract
Dabigatran is a newer oral direct thrombin inhibitor approved by the United States Food and Drug Administration and the European Medicines Agency (EMA). The proper dosage of the drug, the potential for adverse drug reactions and the nature of bleeds with use of this drug as with other novel oral anticoagulants (NOACs), in the elderly population are still areas of uncertainty. Despite the existence of a specific antibody, idarucizumab which is an antidote to dabigatran toxicity, management of dabigatran-induced bleeds is an undefined area especially in resource constrained settings. We report severe haematuria with dabigatran in three elderly Indian patients at the lowest recommended therapeutic dose and explore these grey zones in dabigatran therapy.
Introduction
Dabigatran is a newer oral direct thrombin inhibitor approved by the United States Food and Drug Administration (US FDA) and the European Medicines Agency (EMA).1,2 Bleeding is its most serious adverse effect.1,2 Studies show variable risk of extracranial bleed and a low risk of intracranial bleed compared with warfarin.3–6 We report severe haematuria with dabigatran in three elderly Indian patients at the lowest recommended therapeutic dose.
Case presentation
Case-1
A 70-year-old male with coronary artery disease and advanced Alzheimer’s dementia presented with haematuria for 1 day. He had been predominantly bedridden, catheterized for 15 days and diagnosed with right lower limb deep venous thrombosis (DVT) 1 month previously. Dabigatran 150 mg once daily had been started 4 days previously as the patient had labile international normalized ratio (INR; time for which INR stayed in the therapeutic range was <60%, while on warfarin). Prothrombin time (PT)-INR, activated partial thromboplastin time (aPTT) and routine blood investigations including renal function tests done at this time were normal (Table 1). Dabigatran was stopped and patient was given tranexamic acid. Bleeding subsided within 24 h. A month later dabigatran was restarted with informed consent at 150 mg once daily for unresolved DVT. The patient was readmitted within 2 days with severe haematuria. PT-INR and aPTT were again normal. There were no changes in renal function parameters during this period (Table 1). Dabigatran was stopped and the patient was managed conservatively.
Medications being received by the patients at dabigatran initiation and salient observations in the three cases.
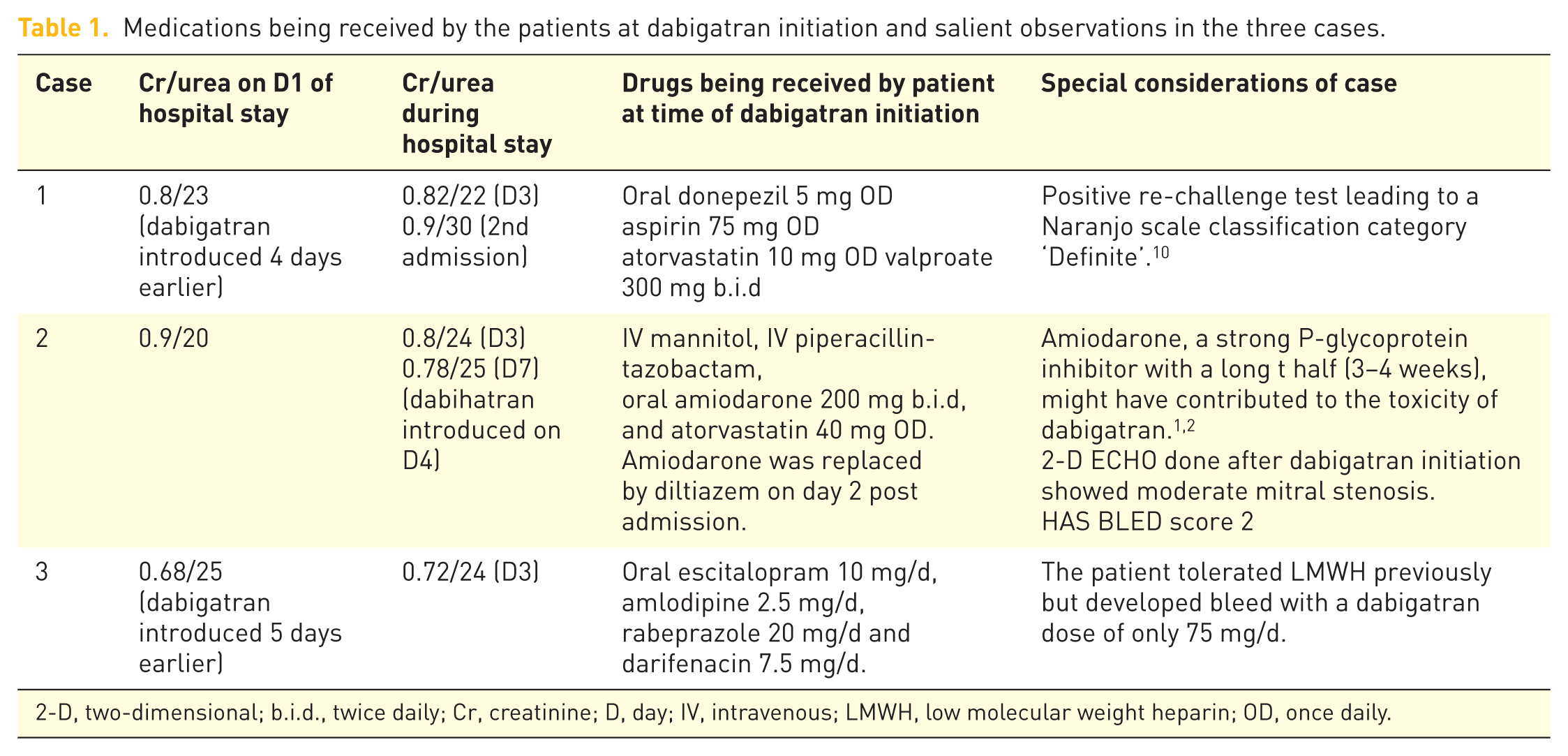
2-D, two-dimensional; b.i.d., twice daily; Cr, creatinine; D, day; IV, intravenous; LMWH, low molecular weight heparin; OD, once daily.
Case-2
A 65-year-old female patient of recurrent stroke with atrial fibrillation and suspected coronary artery disease was admitted with a Glasgow Coma Scale of E1V1M1 and a heart rate of 140/min. The only abnormal blood investigation was a slightly raised leucocyte count with neutrophilia. Non-contrast computed tomography of the head showed ischaemic changes in the right frontoparietal lobe. Dabigatran was added on day-4 for stroke prophylaxis at the dose of 150 mg per day and aspirin was discontinued. Baseline PT-INR, aPTT and renal function tests were normal (Table 1). A two-dimensional ECHO done on the night of day 5 of the hospital stay showed moderate mitral stenosis. The same night, the patient developed severe haematuria with a 3.4 g/dl fall in haemoglobin. Dabigatran was stopped immediately and tranexamic acid was started. On day 6, INR was 1.35 and aPTT was 32 (control 27.2). She was transfused with fresh frozen plasma and parenteral vitamin K was initiated for persistent bleeding on day 8. Intermittent haematuria continued till day 11 with eventual fatality on day 13.
Case-3
A 72-year-old hypertensive osteoporotic male with L1 and S3 vertebral fracture who was bedridden presented with haematuria of 3 days duration. Dabigatran 75 mg/d was started 5 days earlier for DVT prophylaxis replacing subcutaneous enoxaparin 40 mg/d. The patient had stable vitals with normal sensorium. Routine blood investigations including PT-INR, aPTT and renal function parameters were normal (Table 1). Dabigatran was discontinued and tranexamic acid was started. Bleeding ceased within 24 h. After a week, the patient was started on enoxaparin with no bleeds thereafter.
Discussion
The recommended dose of dabigatran is 150 mg twice daily for stroke prophylaxis and DVT treatment whereas for DVT prophylaxis, it is 110 mg twice daily.1,2 Dose modification is required in the elderly, renal compromised and when used with strong P-glycoprotein inhibitors (dronaderone and ketoconazole).1,2 A reduced need for monitoring makes dabigatran an attractive option. However, the monitoring of response of newer anticoagulants and management of bleeding remains a grey area. aPTT is prolonged with the use of dabigatran, but with no dose–response relationship. Diluted thrombin time and ecarin clotting time are shown to have a dose–response relationship with dabigatran, 8 but these are seldom available and were not done in the present cases. Prothrombin complex concentrates, rF-VIIa, activated charcoal and induced diuresis are being tried in dabigatran-induced bleeding but without compelling evidence. 8 The US FDA-approved monoclonal antibody, idarucizumab, an antidote to dabigatran is not commercially available in India and many other countries. 8 Tranexamic acid intravenous was given to manage bleeding in all the cases as a part of routine first line management of bleeding events in patients. The approach was successful in two of the patients while one patient (case number 2) continued to bleed despite fresh frozen plasma and tranexamic acid. 9
There are many reports of dabigatran-induced bleeding in the elderly, the majority of them being in the presence of renal insufficiency and at the dose of 150 mg twice daily. 7 Here we have reported three cases of severe haematuria with dabigatran at the lowest possible therapeutic dose and in the presence of a normal renal function. The lower doses were employed intentionally considering the age of the patients. All three patients were on indwelling urinary catheters. In the presence of this local factor, dabigatran may have induced bleeding. This may explain the variance from available data which states that gastrointestinal bleeding is the commonest with dabigatran. None of the patients’ urine routine microscopy examination revealed acanthocytes or red blood cell casts. We hypothesize that the urinary tract was responsible for the bleeds and not glomerular haemorrhage. Whereas the bleeding was manageable with dabigatran withdrawal and supportive measures in case 1 and case 3, the bleeding continued to occur in case 2 despite fresh frozen plasma transfusion. The Naranjo causality association scale classified the adverse drug reactions as ‘probable’ in cases 2 and 3 and as ‘definite’ in case 1. 10 The medications being received by the patients at the time of dabigatran initiation and salient observations are presented in Table 1.
The case series raises a doubt over the safety and proper dosage of NOACs in elderly patients. Whether warfarin and low molecular weight heparin can be preferred in resource-limited hospital settings remains unanswered. Interestingly, the cases were the first three patients to be prescribed dabigatran in the geriatric unit since the drug was approved by the Indian regulator. The bleeding events compelled the authors to regularly follow all subsequent elderly patients being administered dabigatran in a prospective study whose results may appear later.
Footnotes
Acknowledgements
Authors 1 and 2 should be treated as first authors as they contributed equally to the manuscript.
Data regarding all adverse drug reactions in elderly patients attending the Division of Geriatrics is covered as part of a larger ADR monitoring study approved by the Institutional Ethics Committee of the Institute of Medical Sciences, Banaras Hindu University, Varanasi, India, which is an independent ethics committee. The IEC approval number for this study is: Dean/2015-16/IEC/411 (EC Reg. No. ECR/526/Inst/UP/2014)
Written informed consent was taken from the legal guardians/primary caregivers of patient 1 (advanced Alzheimer’s dementia incapable of giving consent) and patient 2 (acute stroke in altered sensorium) and the patient himself for patient 3 for disclosure of their information
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
