Abstract

Introduction
Beneficence and non-maleficence: these classic ethical principles 1 often contradict when atypical antipsychotics are prescribed to children. Expedient effects on development should be weighed carefully against harmful side effects, especially metabolic complications. Prescribing antipsychotic medication requires thorough monitoring of effects and side effects. 2
During this process, health professionals are often tempted to assign every inter-current symptom to the drug prescribed. However, sometimes this reasoning is false and thereby the decision to stop the medication. Effective treatment may be withheld based on erroneous reasoning, and this situation is sometimes avoidable. In this context, we present the following case:
Case
History
The patient was a 9-year-old healthy boy, with normal intelligence but severe difficulties in social interactions due to his autism spectrum disorder (ASD). He had a history of constitutional eczema, and a soy allergy, presenting as a generalized rash, which responded quickly to an oral antihistaminic. He had no bronchial hyper-reactivity. The patient was selectively vaccinated and presumably had had measles; other rashes were not documented.
After an initial ambulatory observational period, risperidone 0.5 mg was prescribed and the patient slowly became a little more responsive to human contact. Initial complaints of fatigue, malaise and feeling warm all disappeared after a few days. The dose was slowly increased over 8 months from 0.75 mg to 1.0 mg.
One week after this last increase, he was brought in with a rash. Accompanying complaints of nausea, headache and fatigue started after attending a birthday party a few days earlier, following the dose adjustment of the risperidone. At the birthday party the patient had not eaten anything novel, and had swum in the same pool where he had his regular lessons.
Within a few days a rash also developed, starting on the dorsal side of both feet, and subsequently appearing on the hands, elbows and perioral area. The spots were transient, and appeared and disappeared in hours. Most pronounced were the spots on the hands, feet and perioral area. There were no complaints of itch, pain, dyspnoea or cough. The mother was also feeling uncomfortable, with malaise without skin manifestations.
The boy’s mother feared an allergy for risperidone, especially because the pharmacy supplied a different brand when the dose was increased. On her way to the clinic, the worried mother visited the general practitioner, who administered 10 mg cetirizine because of probable allergy. This has not yet changed the symptoms. In accordance with ethical standards of the 1964 Declaration of Helsinki and its later amendments, written informed consent to publish this case study was given by the parents of the patients.
Physical examination
On examination the patient appeared tired and not ill, he occasionally made eye contact and executed simple commands. His mother reported that his behaviour was more subdued since the start of cetirizine. Results of the physical examination were: length 1.38 m, weight 27 kg, pulse 68/min regular and equal, blood pressure 92.68 mmHg and respiration 16/min. Examination showed no stridor, audible respiration or retractions, nor oedema of the head and neck region. Some skin-coloured papules, which we described clinically as weals with pale interiors and well-defined reddish margins, were seen periorally. Small, non-pitting, urticaria papules were visible with slight swelling on the dorsum of the feet and hands, the left elbow and right knee. There were no petechiae or itching marks. During the examination, new urticaria papules developed while others diminished. Muscle tension was normal. Skin and tendon reflexes were undisturbed, and pain and tactile senses were intact.
Differential diagnosis
We had the following diagnostic considerations for this clinical presentation dominated by urticaria.
We first considered an allergic response to risperidone following the increased dose, then we considered an excipient of the tablet, because the complaints had started after the use of the same drug but from a different brand. In addition, we considered allergic responses to an unknown agent, but thought these unlikely as he had not been exposed to new agents. Lastly, despite the history of incomplete immunizations we rejected an infectious viral or bacterial disease with urticaria because of the disparate clinical presentation.
After consulting the pharmacist, we considered an allergic response to an excipient as the most plausible. Earlier dose increases of risperidone had not caused urticaria and there had been ample time for sensitization. The urticaria and the course over time fitted a clinical picture of an immunoglobulin E-mediated type I allergic reaction. As a result of the favourable effect of risperidone on the patient’s behaviour, the dose of 1.0 mg was continued, while switching back to the original brand. The patient was frequently checked and within a few days the skin symptoms diminished. After a week, all weals had completely disappeared.
Discussion
An allergic response of urticaria concomitantly with use of risperidone in a child has not been reported before. Allergy 3 and urticaria 4 associated with risperidone have previously been reported in adults only. Our PubMed search with combined entries ‘risperidone, urticaria, children’ or equivalents showed no results.
Such adverse drug reactions can be an allergic response to the active agent, but can also be to an excipient. Generally, children are at increased risk for toxicity of excipients by their age-related pharmacokinetics. 5 Articles found with entries ‘risperidone, excipients’ only dealt with practical and pharmaceutical considerations of excipients, not with possible allergic responses.
Moreover, it is very plausible that the allergic response was caused by an excipient and not by the active component. This excipient allergy hypothesis scored 5 points on the Naranjo scale of adverse drug reactions (see internet supplement, Table 1), which is classified as probable, the second highest category of probability. 6 The composition of the new brand differed from the well-tolerated one in four excipients that might be causative: stearic acid, iron oxide red [E172] and titanium dioxide [E171] in the film coating, and croscarmellose in the core.
Naranjo scale.
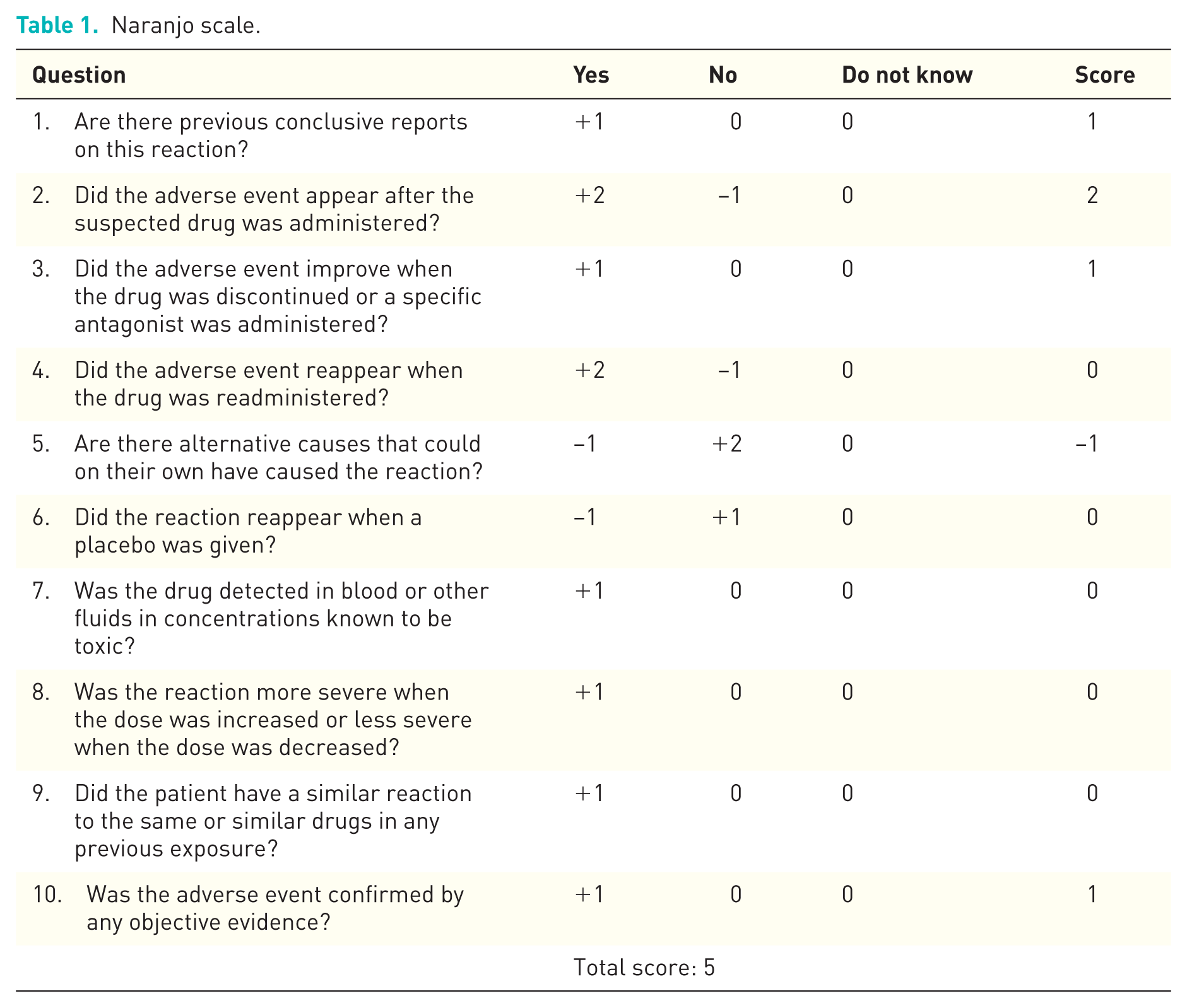
Naranjo algorithm – adverse drug reaction probability scale.

As the urticaria occurred following a dose increase, with no previous allergic symptoms for risperidone, we felt we could safely continue with the dose of the original brand in accordance with our excipient allergy hypothesis. This had a favourable effect on behaviour and the disappearance of the urticaria.
Two recent trends underscore the significance of describing our case. The first is a worldwide increase in the prescription of antipsychotic drugs to children and adolescents with ASD. 7 The second trend is the intensification of government-regulated switches from prescription drugs to generic drugs occurring in The Netherlands and elsewhere. As a consequence, pharmacies supply generic drugs, 8 often unbeknownst to the prescriber. These combined trends increase the likelihood of an excipient allergy in children with ASD using risperidone remaining unnoticed and unreported because their physicians will always first suspect an allergy to the active ingredient risperidone. However, physicians should be aware of the possibility of an allergic reaction to an excipient and switch to another brand if this is applicable and safe.
Footnotes
Ethical note
In accordance with ethical standards of the 1964 Declaration of Helsinki and its later amendments, written informed consent to publish these case details was given by the parents.
Funding
This research received no specific grant from anyfunding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
