Abstract
Background:
Programmed cell death protein (PD-1) inhibitors constitute the standard adjuvant therapy for cutaneous melanoma (CM), but well-established strategies for acral melanoma (AM) remain limited.
Objective:
To compare the efficacy of adjuvant anti-PD-1 immunotherapy versus high-dose interferon α-2b (HDI) in patients with stage IIB-IV AM and CM.
Design:
This multicenter, retrospective study enrolled 511 patients with resected stage IIB-IV AM and CM between January 2017 and December 2023.
Methods:
Patients were divided into four groups by subtype and treatment: patients with CM and treated with anti-PD-1 (CM-PD-1), patients with CM and treated with HDI (CM-HDI), patients with AM and treated with anti-PD-1 (AM-PD-1), and patients with AM and treated with HDI (AM-HDI). Recurrence-free survival (RFS), overall survival (OS), and patient safety were evaluated.
Results:
This study comprised 362 AM and 149 CM cases. AM cases presented with thicker primary lesions, higher ulceration rates, and fewer BRAF mutations. Median follow-up was 49 months. Among patients with stage IIB/C, median RFS was not reached in any groups. Among stage III/IV patients, median RFS was 14.6 (CM-PD-1), 13.7 (CM-HDI), 13.3 (AM-PD-1), and 11.7 months (AM-HDI), and median OS was 61.6, 40.7, 42.4, and 53.4 months, respectively, without significant intergroup differences. Anti-PD-1 significantly improved RFS in patients with stage III/IV AM with KIT mutations (9.1 vs 5.0 months, p = 0.048) and ⩾4 lymph node metastases (10.5 vs 6.6 months, p = 0.036). Anti-PD-1 had significantly fewer adverse effects than HDI (60.4% vs 88.6%, p < 0.001), including fewer grades 3–4 events (4.6% vs 30.7%, p < 0.001).
Conclusion:
Adjuvant anti-PD-1 therapy provides RFS comparable to that of HDI in AM and CM, with a superior safety profile. Patients with AM harboring KIT mutations derive greater benefits from adjuvant anti-PD-1 therapy.
Plain language summary
Acral melanoma (AM) is one type of melanoma that grows on palms, soles, or subungual regions. Cutaneous melanoma (CM) is another type that grows on other parts of the skin. For CM, doctors often recommend anti-PD-1 therapy after surgery to reduce the risk of recurrence and metastasis. However, there’s no clear advice on what treatment to use after surgery for AM. We analyzed 511 patients (362 AM, 149 CM) treated with adjuvant anti-PD-1 therapy or high-dose interferon α-2b (HDI) between 2017 and 2023. The patients were divided into four groups: CM patients treated with anti-PD-1 (CM-PD-1), CM patients treated with HDI (CM-HDI), AM patients treated with anti-PD-1 (AM-PD-1), AM patients treated with HDI (AM-HDI). We found that AM patients had thicker tumors, more ulcers, and fewer changes to a gene called BRAF than CM patients. For both AM and CM patients, anti-PD-1 therapy and HDI kept the cancer away for similar lengths of time, and didn’t affect how long people lived much differently. But there was an exception: AM patients who had changes to a gene called KIT, or whose cancer had spread to 4 or more lymph nodes, stayed cancer-free longer when they used anti-PD-1 therapy. Also, anti-PD-1 therapy caused fewer side effects: 60.4% of people had side effects, compared to 88.6% of those who used HDI. Severe side effects were also less common—only 4.6% with PD-1 inhibitors, versus 30.7% with HDI.
Keywords
Introduction
Acral melanoma (AM), originating on the palms, soles, and nail beds, is a rare subtype of melanoma in Caucasian populations. It is the predominant subtype in Asian, African, and Hispanic populations, accounting for approximately 50% of melanoma cases in these groups.1–4 Unlike cutaneous melanoma (CM), which demonstrates a high tumor mutational burden with ultraviolet signature, AM is characterized by lower mutational burdens, distinct oncogenic drivers, more structural chromosomal changes, and lower numbers of tumor-infiltrating lymphocytes.3,5–7 AM is often diagnosed at an advanced stage and has a worse prognosis than CM.4,5 Additionally, it is challenging to achieve wide negative margins in AM due to its anatomical locations, leading to a high postoperative recurrence rate.8–10 Robust evidence supporting the efficacy of adjuvant therapy in Asian patients with AM remains limited.
Several randomized controlled trials in the last decade have demonstrated that immune checkpoint inhibitors, such as pembrolizumab and nivolumab, substantially enhance recurrence-free survival (RFS) in patients with completely resected stage IIB-IV CM.11–17 The SWOG S1404 study comparing adjuvant pembrolizumab with ipilimumab or high-dose interferon α-2b (HDI) for the treatment of patients with stage III/IV CM reported that pembrolizumab resulted in a statistically significant 23% improvement in RFS. 17 Consequently, anti-PD-1 monotherapy is now recommended as the standard adjuvant treatment for patients with CM, superseding the long-applied yet less efficacious interferon. 18 However, immunotherapy is less effective in patients with advanced AM, with a lower objective response rate of 14–26% and a shorter progression-free survival of 3.2–4.1 months.19–21 The efficacy of adjuvant anti-PD-1 therapy in AM remains uncertain, as only a small proportion of clinical trials included patients with AM, thus, well-established adjuvant treatment guidelines for AM are currently lacking. In Asia, the limited data on the efficacy of adjuvant anti-PD-1 immunotherapy for AM, along with the lack of a comprehensive comparison with other melanoma subtypes, prompted this investigation. We compared the efficacy of adjuvant anti-PD-1 therapy with that of HDI in Chinese patients with stage IIB-IV AM and CM.
Materials and methods
Patients
This was a multicenter, retrospective study of anti-PD-1 versus HDI as adjuvant therapy in patients with AM and CM treated at Peking University Cancer Hospital and Fujian Cancer Hospital. The eligibility criteria were (1) age ⩾ 18 years old, (2) diagnosis of completely resected stage IIB/IIC/III/IV AM or CM, (3) diagnosis between January 2017 to December 2023, and (4) receipt of pembrolizumab or toripalimab with recommended dosage, or HDI (18 × 106 IU/m2 per day interferon-α2b subcutaneously on days 1–5 for 4 weeks, followed by 9 × 106 IU/m2 per day interferon-α2b, thrice a week subcutaneously) within 3 months after surgery. The patients were divided into the following four groups: patients with CM and treated with anti-PD-1 (CM-PD-1), patients with CM and treated with HDI (CM-HDI), patients with AM and treated with anti-PD-1 (AM-PD-1), and patients with AM and treated with HDI (AM-HDI). The primary endpoint was RFS. In addition, distant metastasis-free survival (DMFS) and overall survival (OS) were evaluated. As the lead institution for the research, the Institutional Review Board of Peking University Cancer Hospital approved the study. This study adhered to the STROBE statement (Table S1). 22
Statistical analysis
All statistical analyses were performed using R statistical software version 4.2.2. Pearson’s χ2 statistic or Fisher’s exact test was used to identify differences between groups. The Kaplan–Meier method was used to plot survival curves, and differences in survival between the groups were identified using the log-rank test. Independent prognostic factors and hazard ratios (HR) were identified using univariate and multivariate Cox proportional hazard models. Statistical significance was defined as a two-sided p-value of <0.05.
Results
Patient characteristics
A total of 511 patients (362 with AM and 149 with CM) were enrolled between January 2017, and December 2023. Compared to patients with CM, patients with AM were more likely to present with thicker primary lesions and ulcerations. BRAF mutations were more prevalent in patients with CM than in those with AM (30.2% vs 8.8%), whereas KIT (2.7% vs 6.6%) and NRAS (10.7% vs 15.7%) mutations were less common. Among patients with AM (Table 1), 182 patients were treated with adjuvant anti-PD-1 therapy and 180 were treated with HDI. Among patients with AM, the proportions of females, stage II, thicker primary lesions, absence of lymph node metastasis, and wild-type tumors were significantly higher in the HDI group than in the anti-PD-1 group. Among patients with CM, 101 received anti-PD-1 and 48 received HDI; the anti-PD-1 group had significantly fewer patients younger than 60 years and fewer with stage II disease than did the HDI group.
Clinical characteristics of patients with AM and CM treated with adjuvant anti-PD-1 or HDI.

AM, acral melanoma; CM, cutaneous melanoma; HDI, high-dose interferon α-2b.
Efficacy
The median follow-up time was 49 months for all patients, based on the cutoff date of December 10, 2024. The median follow-up time was 51.7 months (95% CI: 44.6–60.4) in the CM-PD-1 group, 62.9 months (95% CI: 61.2–78.5) in the CM-HDI group, 39.9 months (95% CI: 35.0–45.9) in the AM-PD-1 group, and 51.8 months (95% CI: 45.2–61.6) in the AM-HDI group. As tumor stage is a major prognostic factor and stage distribution differed significantly between anti-PD-1 and HDI groups, subsequent survival analyses were performed using stage-stratified subgroups.
In patients with stage IIB/C melanoma, the median RFS, DMFS, and OS were not reached in the four groups, and no significant differences were observed among the groups (Figure 1(a)–(c)). The estimated 36-month RFS rate was 75.5% in the CM-PD-1 group, 64.3% in the CM-HDI group, 72.2% in the AM-PD-1 group, and 54.9% in the AM-HDI group.

Kaplan–Meier curves of RFS, DMFS, and OS in patients with stage IIB/C (a–c) AM and CM treated with anti-PD-1 or HDI.
Among patients with stage III/IV melanoma, 56 (63.6%) patients with CM treated with anti-PD-1 and 26 (76.5%) patients with CM treated with HDI relapsed. Furthermore, 114 (67.1% patients with AM treated with anti-PD-1 experienced relapse, whereas 75 (73.5%) patients with AM treated with HDI experienced disease recurrence. The median RFS was 14.6 months (95% CI: 12.3–26.8), 13.7 months (95% CI: 8.6–33.5), 13.3 months (95% CI: 10.3–17.1), 11.7 months (95% CI: 8.2–15.1) in the CM-PD-1, CM-HDI, AM-PD-1, AM-HDI groups, respectively. No significant difference was noted in median RFS among the four groups (Figure 2). The RFS rates at 1, 3, and 5 years were 60.2%, 33.3%, and 31.8%, respectively, for patients in the CM-PD-1 group, compared to 50.4%, 23.5%, and 20.2%, respectively, for patients in the CM-HDI group. The RFS rates at 1, 3, and 5 years were similar between the AM-PD-1 and AM-HDI groups (53.4% vs 49.0%, 23.7% vs 25.3%, and 16.5% vs 19.5%, respectively). No notable differences were observed in the DMFS 19.5 months vs 21.6 months vs 20.5 months vs 19.7 months ((b)) across the four groups. Patients with CM in the anti-PD-1 group reported a longer OS than those in the HDI group 61.6 months vs 40.7 months, p = 0.373 ((c)), whereas those with AM in the anti-PD-1 group reported a shorter OS than those in the HDI group (42.4 months vs 53.4 months, p = 0.829). Subgroup analyses demonstrated that adjuvant anti-PD-1 therapy improved RFS in patients with KIT mutations (Figure S1(a)). The RFS of the anti-PD-1 group was significantly longer than that of the HDI group in patients with AM (9.1 months vs 5.0 months, p = 0.048). A beneficial trend of anti-PD-1 therapy was noted in patients with AM with BRAF mutation (10.6 months vs 7.3 months, p = 0.318) and NRAS mutation (10.8 months vs 4.8 months, p = 0.120). However, adjuvant anti-PD-1 and HDI displayed similar RFS in BRAF-mutant patients with CM (9.9 months vs 8.6 months, p = 0.340), whereas anti-PD-1 therapy had a shorter RFS than HDI in NRAS-mutant patients (6.0 months vs 40.1 months, p = 0.053). In addition, adjuvant anti-PD-1 therapy yielded a longer RFS than HDI in patients with AM and patients with CM with at least four lymph node metastases (AM: 10.5 months vs 6.6 months, p = 0.036, CM: 17.2 months vs 3.5 months, p = 0.088, Figure S1(b)).
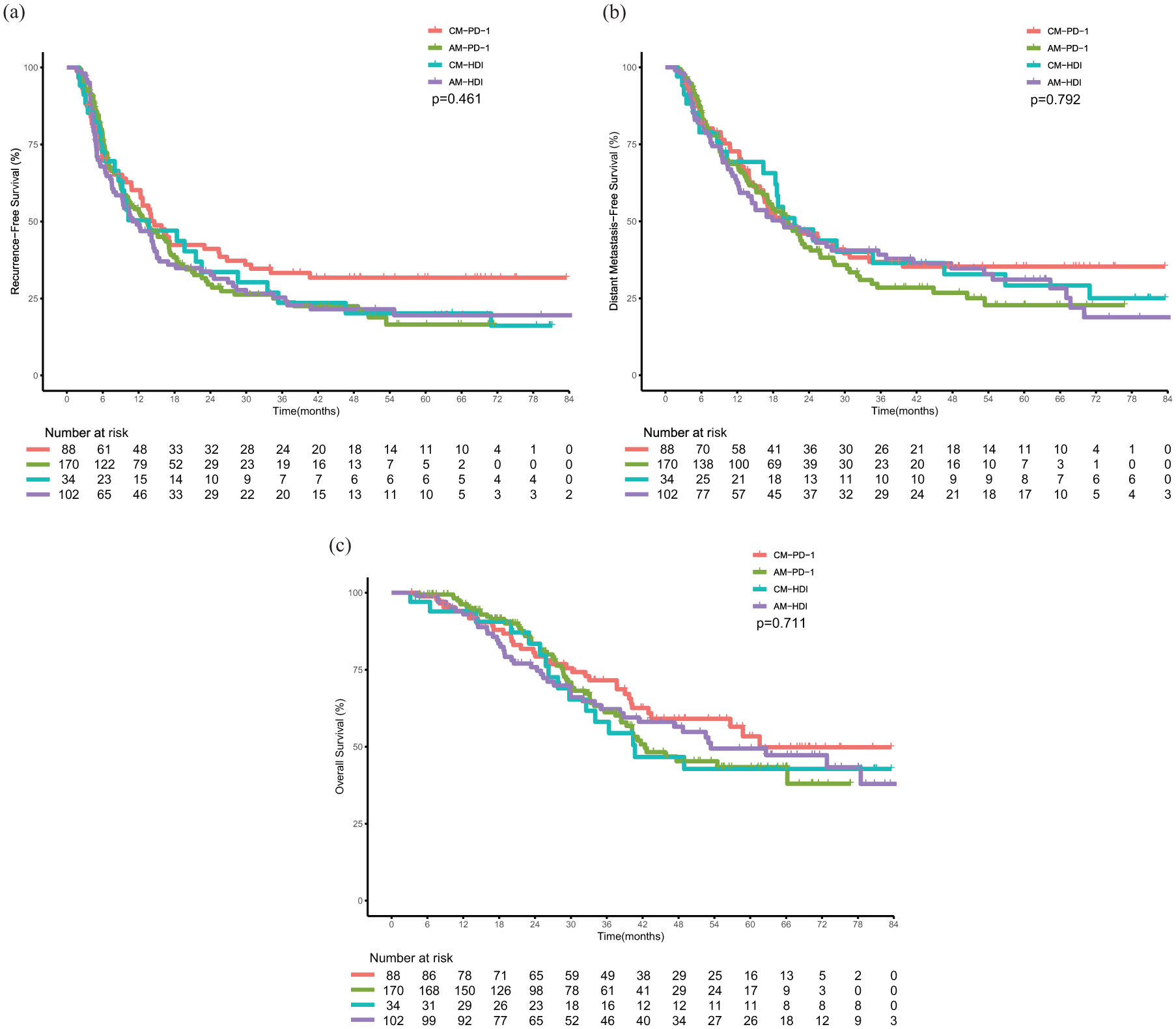
Kaplan–Meier curves of RFS, DMFS, and OS in patients with stage III/IV (a–c) AM and CM treated with anti-PD-1 or HDI.
We conducted univariate and multivariate Cox proportional hazards regression analyses to further elucidate the prognostic factors for RFS. Adjuvant anti-PD1 or HDI did not affect the RFS of patients with AM (Table S1) or CM (Table S2). Age <60 years, advanced disease stage, and the presence of NRAS mutations or KIT mutations were associated with poorer RFS in patients with AM. Univariate regression analysis revealed that tumor stage and number of lymph node metastases influenced the RFS of patients with CM, whereas multivariate analysis indicated that they were not independent prognostic factors.
Safety
Most adverse events were grade 1 or 2 (Table 2). The incidence of adverse events in the anti-PD-1 group was significantly lower than that in the HDI group (60.4% vs 88.6%, p < 0.001). The most common adverse events in the anti-PD-1 group were hypothyroidism, elevated transaminases, rashes, and vitiligo. Leukopenia, pyrexia, fatigue, nausea, and elevated transaminases were the most frequent adverse events in the HDI group. Furthermore, the HDI group had a higher proportion of grades 3–4 adverse events, with a significantly higher incidence of leukopenia and elevated transaminases than the anti-PD-1 group. None of the adverse events led to mortality in either group.
Adverse effects in patients with AM and CM treated with adjuvant anti-PD-1 or HDI.

AM, acral melanoma; CM, cutaneous melanoma; HDI, high-dose interferon α-2b.
Discussion
We conducted a multicenter retrospective study wherein we compared anti-PD-1 with HDI as an adjuvant therapy in patients with stage IIB-IV AM and CM. The results demonstrated that adjuvant anti-PD-1 therapy and HDI had comparable efficacy in both stage IIB/C AM and CM, with median RFS, DMFS, and OS all not reached. Similarly, no significant differences in RFS, DMFS, or OS were found among the four groups of patients with stage III/IV melanoma, suggesting that anti-PD-1 immunotherapy may provide clinical benefits comparable to those of classic HDI across different melanoma subtypes, with a superior safety profile. Compared to other studies, this study included a relatively large sample size and preliminarily explored the efficacy of adjuvant anti-PD-1 versus HDI in stage IIB/C AM.
Interferon was the first approved adjuvant therapy for resected melanomas. A meta-analysis of 15 randomized trials in more than 7500 patients with melanoma reported that adjuvant interferon therapy significantly reduced the risk of relapse and improved OS compared to observation. 23 Mao et al. conducted a randomized phase II clinical trial involving 158 patients with stages IIB-IIIC AM treated with 1 month or 1 year of adjuvant HDI. 24 No significant difference in RFS was noted between 1 month HDI and 1 year HDI groups (17.9 months vs 22.5 months, p = 0.72); however, patients with stages IIIB-IIIC melanoma were more likely to derive benefit from 1 year HDI treatment. The RFS in our study was 11.7 months for patients with stages III-IV AM in the HDI group, which was consistent with the findings of previous studies involving Chinese patients with melanoma.24–27 However, HDI was no longer the preferred standard adjuvant therapy for melanoma due to its high toxicity and limited antitumor activity.
The advent of immune checkpoint inhibitors has revolutionized the treatment landscape of CM, delivering substantial survival benefits across both the advanced and adjuvant treatment phases. Large clinical trials, including CheckMate 238, 11 IMMUNED, 12 KEYNOTE-054, 13 and SWOG S1404, 17 have demonstrated that nivolumab and pembrolizumab reduced the risk of recurrence or death by 23%–43% and significantly increased RFS and DMFS in patients with resected stage III/IV CM. Considering their preferred antitumor activity and safety profile, current guidelines recommend PD-1 inhibitors as standard adjuvant treatments for stage III/IV CM. However, whether adjuvant anti-PD-1 therapy confers comparable survival benefits in patients with stage IIB/C CM to those with stage III/IV CM remains uncertain. The phase III KEYNOTE-716 study demonstrated that pembrolizumab as adjuvant therapy significantly reduced the risk of recurrence or death in patients with resected stage IIB/C CM versus placebo, with 12-month RFS rates of 90% and 83% and 18-month RFS rates of 86% and 77%, respectively. 16 The CheckMate 76 K study demonstrated that nivolumab yielded significant improvements in both RFS and DMFS versus placebo in patients with stage IIB/C CM. 28 With OS data still immature, the associated risks and benefits of adjuvant anti-PD-1 therapy for stage IIB/C CM warrant careful consideration. In our study, adjuvant anti-PD-1 therapy for Chinese patients with CM did not exhibit the prominent efficacy as reported in previous studies, which might be attributed to ethnic differences. Bai et al. reported that East Asian, Hispanic, and African patients with cutaneous and unknown primary melanomas treated with anti-PD-1 had poorer clinical outcomes than White patients. 29
Compared with CM, advanced AM lacks effective therapeutic options, with dismal response rates to conventional immune checkpoint inhibitors. Moreover, far fewer adjuvant treatment modalities have been validated for high-risk resectable AM, and related large-scale clinical trials are scarce. Jacques et al. conducted a multicenter retrospective study of 139 patients with AM and found that adjuvant anti-PD-1 was associated with improved RFS (HR = 0.69, p = 0.0127), DMFS (HR = 0.58, p = 0.0134), and OS (HR = 0.59, p = 0.0196), relative to the historical control cohort who did not receive adjuvant anti-PD-1. 30 A multicenter retrospective study from Turkey involving 114 patients with stage III/IV AM reported that 31 patients received adjuvant anti-PD-1 and 19 patients received interferon. 31 Median RFS was not reached in the anti-PD-1 group and 22 months in the interferon group, with no statistically significant difference detected between the two groups. Li et al. evaluated adjuvant anti-PD-1 versus conventional therapy for Chinese patients with stage III melanoma in a real-world retrospective study. 26 The RFS was 23 months in the anti-PD-1 group (126 patients), 15 months in the interferon group (31 patients), and 11 months in the observation group (42 patients). A notable difference in RFS was observed only between the anti-PD-1 and observation groups. No significant differences in RFS or DMFS were observed between the adjuvant anti-PD-1 and HDI groups, although the OS was significantly longer in the anti-PD-1 group. A small cohort study enrolling 46 Chinese patients with stage III CM and AM demonstrated that pembrolizumab had no advantage over HDI in RFS in either the CM or the AM subgroup. 25 Another retrospective study demonstrated no significant difference between adjuvant anti-PD-1 and HDI in 36 patients with AM, whereas adjuvant anti-PD-1 therapy yielded a significantly better RFS and DMFS than HDI in Chinese patients with CM. 32 However, a real-world study involving 174 patients with melanoma (118 with AM and 45 with CM) demonstrated that the anti-PD-1 group had a better RFS than the interferon and observation group (22 months vs 11 months). 27 In contrast to previous results, Maeda et al. observed that nivolumab (n = 5) was associated with an inferior disease-free survival to other treatments (n = 22), although without statistical significance. 33 Discrepancies in sample size, follow-up time, race, stage distribution, and gene mutation profiles may contribute to the divergent outcomes across these studies. However, our study included a relatively larger cohort with a longer follow-up period and exclusively enrolled Chinese patients with stages IIB-IV melanoma, demonstrating that adjuvant anti-PD-1 provided clinical benefits comparable to those of HDI in both AM and CM. Thus, considering the relatively high toxicity of HDI, adjuvant anti-PD-1 therapy remains an effective treatment choice for Chinese patients with stages IIB-IV AM and CM.
Previous studies have demonstrated that advanced AM is less responsive to immunotherapy than CM; however, whether immune responses differ between the two subtypes in the adjuvant setting remains unclear. Bloem et al. evaluated the efficacy of adjuvant anti-PD-1 in 1958 patients with stage III/IV melanoma (86 with AM and 1872 with CM) from the prospective of nationwide Dutch Melanoma Treatment Registry. 34 The RFS of patients with AM was 14.8 months, significantly shorter than the 37.4 months observed in patients with CM. Similarly, Muto et al. found that the 1-year RFS rate of 26 patients with stage III AM receiving adjuvant anti-PD-1 was significantly lower than that of 26 patients with stage III CM (26% vs 58%, p = 0.029). 35 Another retrospective study involving 37 patients with AM receiving adjuvant anti-PD-1 reported that the cutaneous subtype had significantly better RFS than the acral subtype (HR = 0.49 (95% CI: 0.27–0.92), p = 0.023). 36 Notably, patients with stage III/IV AM receiving adjuvant anti-PD-1 therapy had a median RFS of 13.3 months in our study, shorter than the 14.6 months in patients with CM; however, the difference was insignificant. The differential efficacy of adjuvant anti-PD-1 between the two subtypes may stem from a lower tumor mutational burden and fewer tumor-infiltrating lymphocytes in AM than in CM.6,7
The association between the efficacy of anti-PD-1 antibodies and mutation status has rarely been explored in previous studies. Certain clinical trials, such as KEYNOTE-054, CheckMate-238, and IMMUNED, reported that the BRAF mutation status might not affect the efficacy of PD-1 blockade.11–14 The CheckMate-037 trial demonstrated that nivolumab provided a better objective response rate than chemotherapy in the BRAF-wild-type group, whereas no statistical difference was observed in BRAF-mutant group. 37 A Chinese retrospective study detected a RFS benefit trend of adjuvant anti-PD-1 therapy compared to interferon in BRAF-mutant patients (RFS: 17 months vs 10 months, p = 0.302), which were consistent with our study; however, this trend was not observed in patients with NRAS or KIT mutation. 27 In our study, patients with AM harboring KIT mutation were more likely to benefit from adjuvant anti-PD-1 therapy rather than HDI. Adjuvant anti-PD-1 demonstrated numerically superior RFS compared to HDI for NRAS- or BRAF-mutated AM patients, although the difference was not statistically significant. However, these findings need to be interpreted with caution, given the limited sample size of the gene mutation subgroups.
Furthermore, we identified potential prognostic factors using Cox regression analysis. Age, Breslow thickness, ulceration status, regional lymph node metastasis, and stage correlated with the prognosis of melanoma in previous studies.38–45 In our study, Breslow thickness and ulceration status were not prognostic factors for the RFS of patients with AM; whereas age, stage, NRAS mutation, and KIT mutation were associated with RFS. In addition, adjuvant anti-PD-1 therapy demonstrated an RFS benefit in patients with AM and patients with CM with four or more lymph node metastases.
This study had some limitations. First, certain potential selection and information biases may have been present owing to the retrospective nature of the study. Second, the survival data may be influenced by disease progression and subsequent treatments. Third, the performance of diverse subgroup analyses led to the dispersion of patient numbers, which may have caused bias.
Conclusion
This study demonstrated that adjuvant anti-PD-1 therapy provided an efficacy comparable to that of HDI in prolonging RFS for patients with stage IIB-IV AM and CM. Nevertheless, the exploration of alternative adjuvant strategies to improve AM outcomes is warranted.
Supplemental Material
sj-docx-1-tam-10.1177_17588359261436189 – Supplemental material for Anti-PD-1 matches interferon as adjuvant therapy for acral melanoma: a retrospective study
Supplemental material, sj-docx-1-tam-10.1177_17588359261436189 for Anti-PD-1 matches interferon as adjuvant therapy for acral melanoma: a retrospective study by Yu Du, Yu Chen, Jiaxiang Wang, Jing Lin, Xiaoting Wei, Junjie Gu, Jun Guo, Lu Si and Lili Mao in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359261436189 – Supplemental material for Anti-PD-1 matches interferon as adjuvant therapy for acral melanoma: a retrospective study
Supplemental material, sj-docx-2-tam-10.1177_17588359261436189 for Anti-PD-1 matches interferon as adjuvant therapy for acral melanoma: a retrospective study by Yu Du, Yu Chen, Jiaxiang Wang, Jing Lin, Xiaoting Wei, Junjie Gu, Jun Guo, Lu Si and Lili Mao in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
