Abstract
Introduction:
Little is known about the potential safety issues associated with apixaban in clinical practice and their reporting in spontaneous adverse event (SAE) databases.
Objective:
To describe SAE reports associated with the oral anticoagulant apixaban from Australia, Canada and USA and to examine associated concomitant medicine use.
Methods:
SAE report databases from Australia, Canada and the USA were examined for all reports of adverse events associated with apixaban and concomitant medicines from 1 January 2012 to 30 September 2014. Disproportionality analysis (proportional reporting ratio (PRR) and reporting odds ratio (ROR)) was conducted for the quantitative detection of signals using the USA database.
Results:
There were 97 SAE reports associated with apixaban from Australia, 77 from Canada and 2877 from the USA. Reporting of haemorrhage (any type) was common, ranging from 18% for USA to 31% for Australia. Gastrointestinal (GI) haemorrhage was the most commonly reported haemorrhage, accounting for approximately 10% of adverse event reports across all countries. Positive signals were confirmed in the USA data (haemorrhage (any type) PRR, 12.1; χ2, 5582.2 and ROR, 13.4; 95% CI: 12.13–14.6; GI haemorrhage PRR, 11.8; χ2, 2325.4 and ROR, 12.3; 95% CI, 10.8–14.0). Reporting of concomitant use of medicines with the potential to increase bleeding risk ranged from 47.6% in Canada to 65.5% in Australia.
Conclusion:
A large proportion of adverse event reports for apixaban were associated with use of concomitant medicines which may have increased the risk of haemorrhage.
Introduction
Atrial fibrillation (AF) is associated with increased risk of ischaemic stroke and prior to the development of novel oral anticoagulants (NOACs), warfarin was the mainstay of treatment. 1 Whilst highly efficacious in reducing the risk of stroke, the use of warfarin can be limited by the need for frequent monitoring, variable dose response and increased risk of bleeding. 2 Apixaban, a selective factor Xa inhibitor, is one of the most recently available NOACs for the prevention of ischaemic stroke in AF and venous thrombotic events (VTE). Clinical trial data namely from the ARISTOTLE trial in patients with AF and the AMPLIFY trial for treatment of VTE, have shown that apixaban has similar efficacy and safety as warfarin.3-5 The risk of intracranial bleeding, one of the most devastating complications of anticoagulation in patients with AF, was more than halved in patients treated with apixaban by comparison to warfarin (HR, 0.42; 95% CI, 0.30–0.58) in the ARISTOTLE study. 3 However, the median time in treatment range, a widely used measure of the quality of warfarin anticoagulation was 66% 3 in the ARISTOTLE trial. An increased risk of gastrointestinal (GI) bleeding has been reported for dabigatran and rivaroxaban in comparison with warfarin,6,7 but no increased risk has been observed for treatment with apixaban. 3
Despite this, concerns regarding the safety profile of the newer anticoagulants have been raised in part, due to differences in treatment populations between premarketing randomized controlled trials and clinical practice. 8 The introduction of the NOACs on the market has resulted in an increased overall utilisation of oral anticoagulants, possibly reflective of use in patients previously considered unsuitable for treatment with warfarin. 9 Real-life patients in whom NOACs are used are likely to be older, have impaired renal function, multiple comorbidities and concomitant medicine use, due to the increased prevalence of AF in the older population.1,8 Whilst currently under various stages of development and availability, the lack of an effective antidote to reverse the anticoagulant effect of the NOACs in the event of haemorrhage is an additional safety concern. 10 Further, concomitant use of medicines commonly used in the older population, such as antithrombotics and nonsteroidal anti-inflammatory drugs (NSAIDs), may increase the risk of haemorrhagic adverse events. 11 Post-marketing surveillance studies to date, examining spontaneous adverse event (SAE) reports for dabigatran and rivaroxaban have shown the most common types of adverse events reported were for bleeding specifically GI haemorrhage.12,13 However, little is known about the potential safety issues with use of apixaban in clinical practice and reporting in SAEs databases. The aim of this study was to analyse SAE reports for apixaban from Australia, Canada and United States with a specific focus on the risk of haemorrhagic events and to examine the use of concomitant medicine that may increase the risk of adverse events.
Methods
Data source and study design
SAE reports for apixaban were obtained from the Australian Therapeutic Goods Administration’s Database of Adverse Event Notifications for Medicines, 14 Health Canada’s Vigilance Adverse Reaction Online Database 15 and the USA Food and Drug Administration Adverse Events Reporting System (FAERS) database 16 between 1 January 2012 and 30 September 2014. The USA FAERS database includes international reports, but our analysis was limited to reports from the USA. To provide a quantification of apixaban usage by comparison to reporting of adverse events, monthly utilisation of apixaban was examined over a similar period between 1 January 2013 and 31 December 2014 in Australia only and data were obtained from Medicare Australia Statistics https://www.medicareaustralia.gov.au/statistics/pbs_item.shtml) for all apixaban prescriptions. The total number of prescriptions per month for apixaban was calculated per 100,000 Australian population using population statistics obtained from the Australian Bureau of Statistics. 17
Outcome definitions
We analysed the frequency of adverse event reports separately for all datasets, grouped by the Medical Dictionary for Regulatory Activities (MedDRA) System Organ Class (SOC). Where a single report included more than one adverse event classified under the same MedDRA SOC classification, these were treated as one adverse event. Analyses were performed at the SOC level (including GI disorders, nervous system disorders, vascular disorders and cardiac disorders) and at the MedDRA preferred term level. Within preferred terms any report of haemorrhage was aggregated within each of the SOC.
We analysed the prevalence of reporting of potentially inappropriate concomitant medicines in the adverse event reports. The Australian Therapeutic Guidelines 18 and Australian Medicines Handbook 19 were used to categorize concomitant medicines that may increase the risk of bleeding into three groups: concomitant antithrombotics (warfarin, heparins, platelet aggregation inhibitors, direct thrombin inhibitors and direct Factor Xa inhibitors; ATC Codes B01A, excluding B01AF02 – apixaban), concomitant other bleeding risk medicines (nonsteroidal anti-inflammatory drugs (M01A), selective serotonin reuptake inhibitors (N06AB) and oral (systemic) corticosteroids (H02A)) and concomitant medicines with potentially clinically significant interactions (P-glycoprotein or CYP3A4 inhibitors, including systemic azole antifungals (J02AB, J02AC), macrolide antibiotics (J01FA), HIV protease inhibitors (J05AE), cyclosporine (L04AD01), dronedarone (C01BD07), verapamil (C08DA01), amiodarone (C01BA01) and quinidine (C01BA01)).18,19
Statistical analysis
The frequency of adverse events by MedDRA term as a proportion of all adverse event reports for apixaban within each of the datasets was calculated. Recording of concomitant medicines is optional in SAE reports and the prevalence of potentially inappropriate concomitant therapy for each of the datasets was calculated using the total number of adverse event reports where concomitant medicines were reported as the denominator. The proportional reporting ratio (PRR) and the reporting odds ratio (ROR) were used to quantitate the strength of the association between reported adverse events and apixaban from SAE reports using the USA FAERS database.20,21 The total number of individual adverse events reported by MedDRA terms is required for these disproportionality analyses. and is only publicly available in this dataset. An adverse event signal is detected for a PRR ⩾2.0 with an associated χ2 value of ⩾4.0 20 and for the ROR if the lower bound of the 95% two-sided confidence interval exceeds 1. 21 Each adverse event report may contain multiple medicines and multiple adverse events with PRR and ROR calculated using each medicine/adverse event pairs reported. All analyses were performed using SAS 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
There were totals of 97, 77 and 2877 SAE reports associated with apixaban from Australia, Canada and USA respectively, between 1 January 2012 and 30 September 2014. Examination of utilisation of apixaban in Australia over a similar time period shows that from its introduction on the market use increased to approximately 80 apixaban prescriptions per 100,000 population per month (Figure 1). Table 1 shows the demographics and types of adverse events reported. The average age ranged from 72 to 74 years old and reports were approximately equally gender distributed. GI and nervous system adverse events (at the MedDRA SOC level) were the most commonly reported type of adverse event for all three countries (Table 1). At the preferred term level haemorrhage was common, particularly GI haemorrhage, which ranged from 8.0% of all adverse events reported with apixaban in the USA to 12.9% in Canada. At least a fifth of all adverse event reports associated with apixaban across all three countries reported some type of haemorrhage as the outcome, with GI haemorrhage accounting for 33.3% of all haemorrhage events in Australia to 50% in Canada. One in ten adverse events in Australia and Canada reported cerebrovascular thrombosis, but accounted for only 3% of reports in the USA (Table 1).
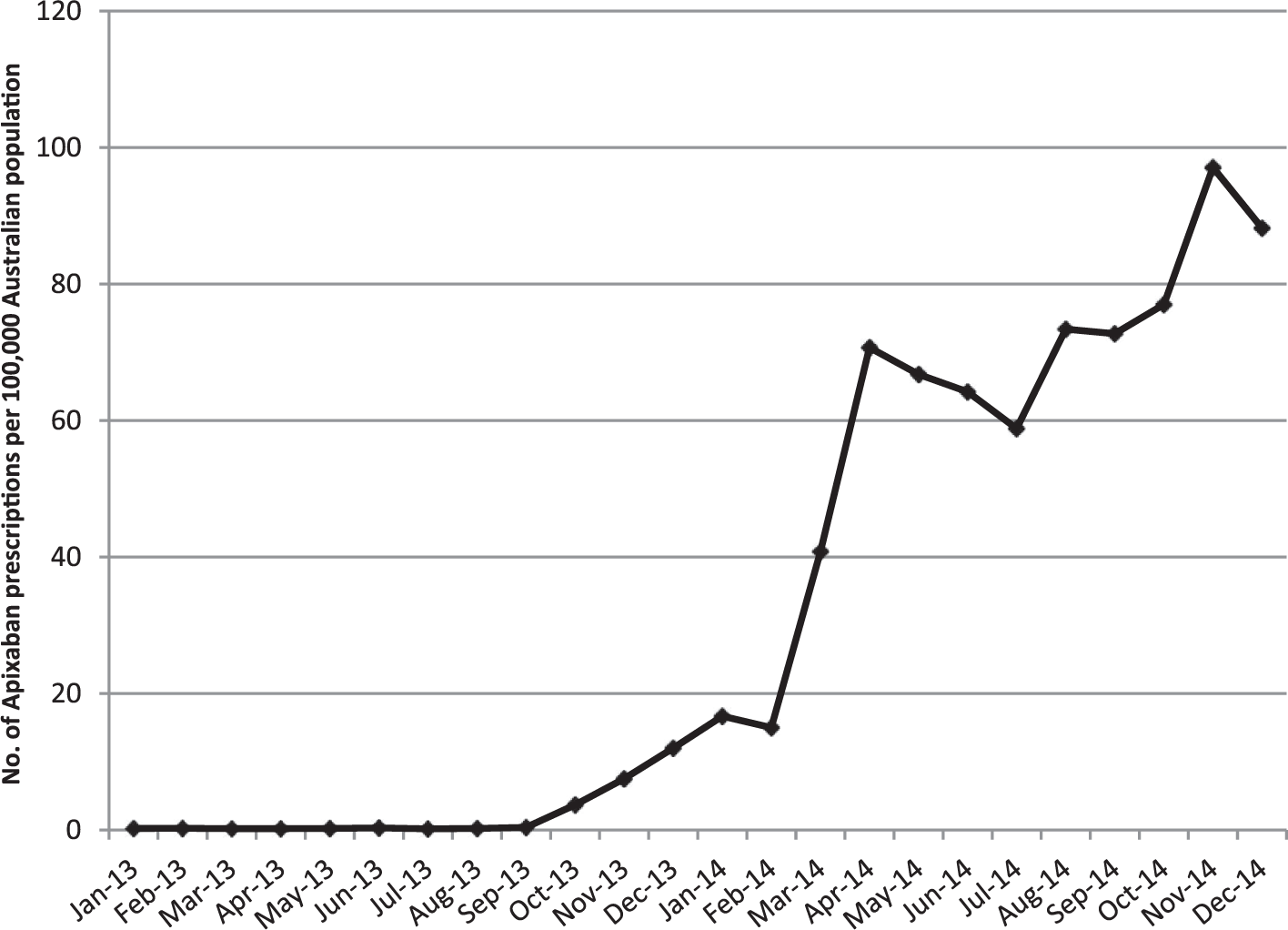
Utilisation of apixaban in Australia between January 2012 and December 2014.
Demographics and type of spontaneous adverse events (SAEs) reported by MedDRA systems organ classification (SOC) for apixaban. a
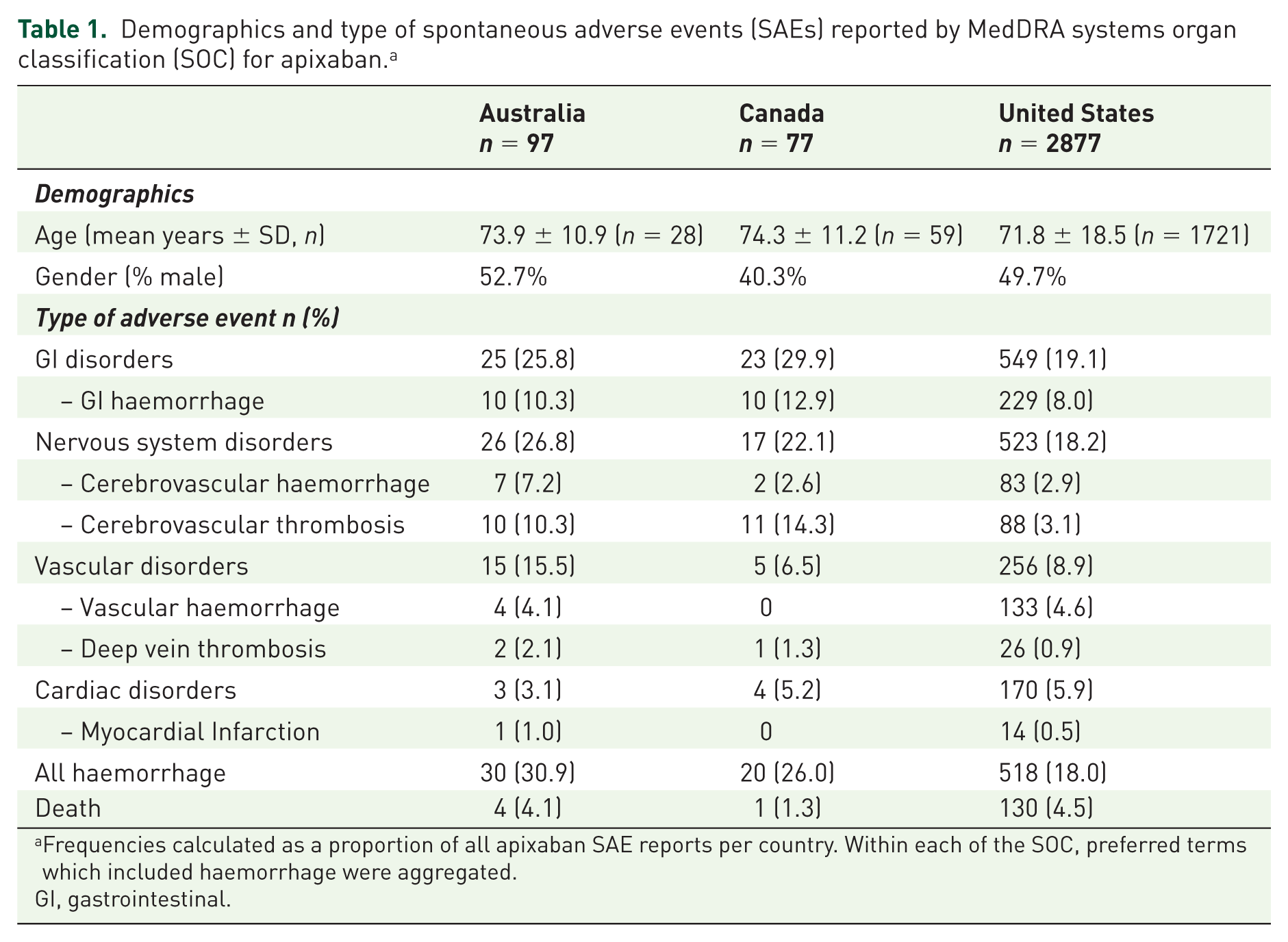
Frequencies calculated as a proportion of all apixaban SAE reports per country. Within each of the SOC, preferred terms which included haemorrhage were aggregated.
GI, gastrointestinal.
Disproportionality analyses within the USA FAERS database found positive signals for GI, cerebrovascular and vascular haemorrhage, as well as all types of haemorrhage (Table 2). A positive signal was also detected for apixaban reports and cerebrovascular thrombosis. Signal strengths were similar for both types of disproportionality analyses (Table 2).
Signal detection for apixaban-associated adverse events in the USA FAERS database.
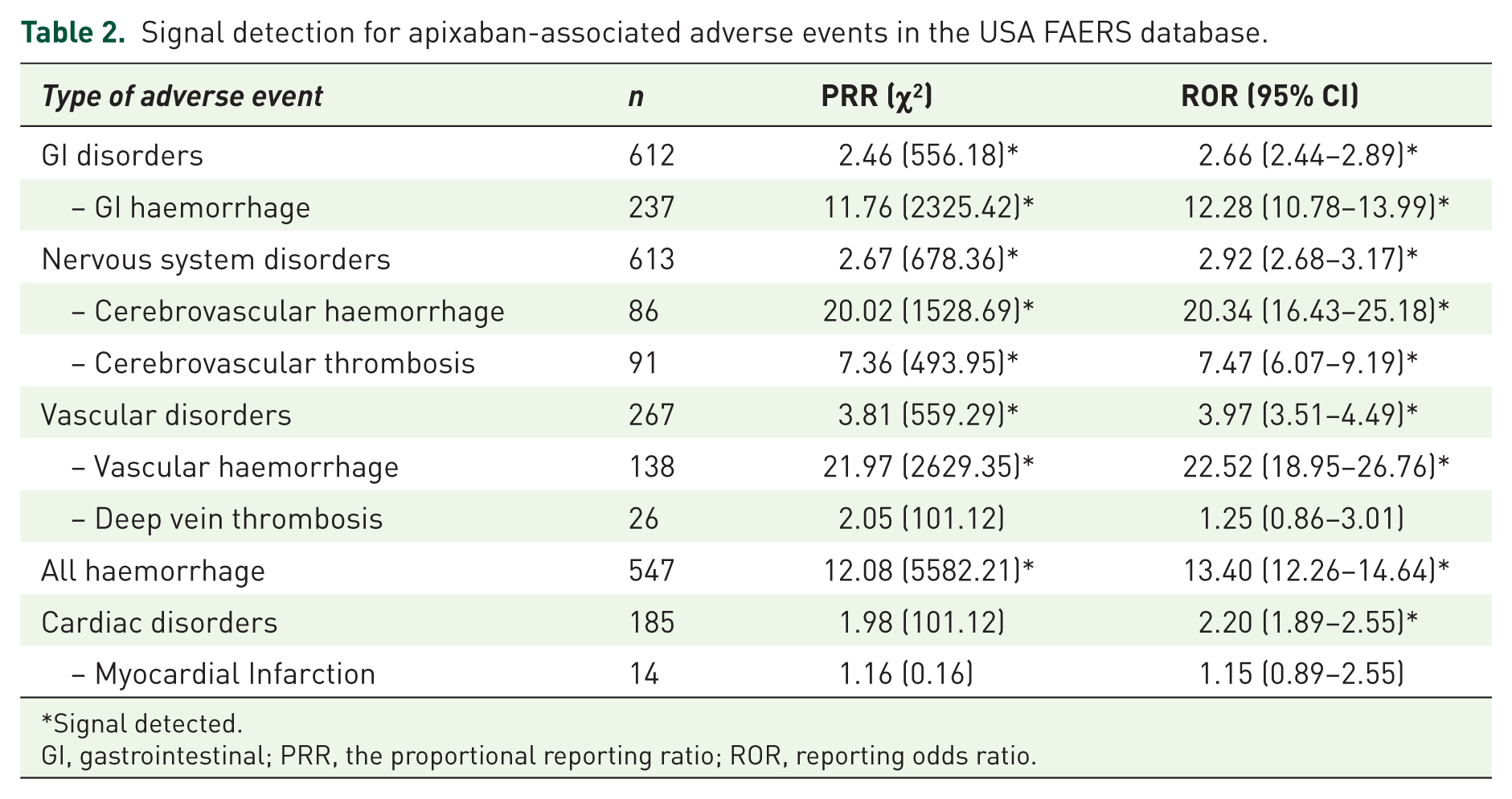
Signal detected.
GI, gastrointestinal; PRR, the proportional reporting ratio; ROR, reporting odds ratio.
Half of all reports across the three countries examined reported the use of at least one concomitant medicine that may have increased the likelihood of haemorrhage. The mean number of bleeding risk medicines reported ranged from 1.0 (SD ± 0.93) for Australia to 1.40 (SD ± 0.81) for USA (Table 3). Use of concomitant antithrombotic medicines were the most commonly reported, ranging from 23.8% in Canada to 38.2% of reports from the USA, with aspirin the most commonly reported antithrombotic for all countries (Table 3). Approximately 10% of the reports across all three countries reported use of a concomitant other bleeding risk medicine or one with potential for drug to drug interactions. Those medicines of most concern are NSAIDs, corticosteroids and amiodarone.
Prevalence of reporting of concomitant bleeding risk medicines in apixaban spontaneous adverse events (SAEs) reports. a
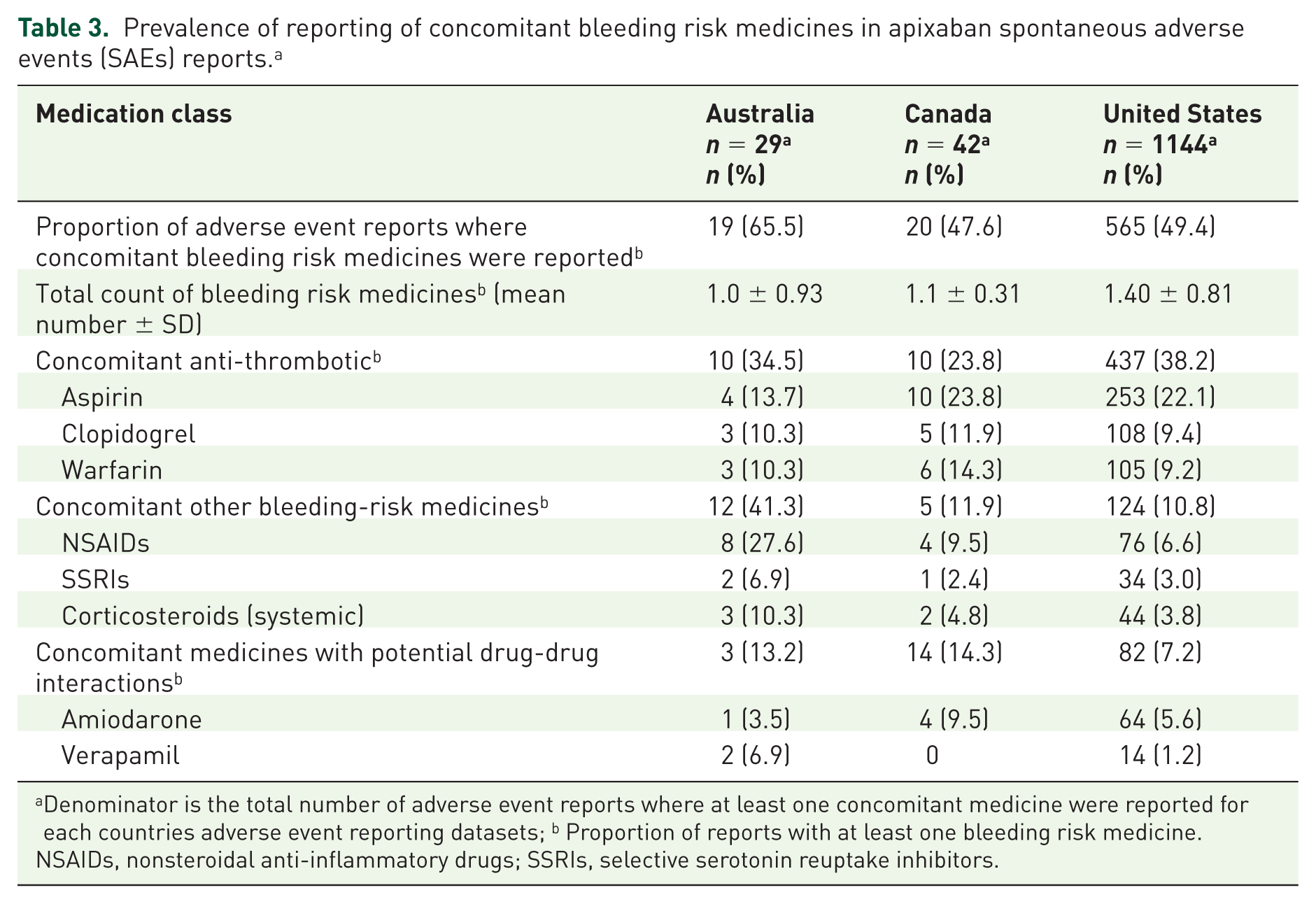
Denominator is the total number of adverse event reports where at least one concomitant medicine were reported for each countries adverse event reporting datasets; b Proportion of reports with at least one bleeding risk medicine.
NSAIDs, nonsteroidal anti-inflammatory drugs; SSRIs, selective serotonin reuptake inhibitors.
Discussion
The reporting of haemorrhagic adverse events with use of apixaban in the ‘real-world’ setting was common in this cross-country study. GI haemorrhage was the most commonly reported haemorrhage, accounting for approximately 10% of adverse event reports across all countries. Further, similar to the findings of the ARISTOTLE trial, we found at least a third of all reported haemorrhages were for a GI haemorrhage. 3 The balance between the benefits and harms associated with oral anticoagulants is a challenge for clinicians, and is complicated by the fact that the risk factors for stroke are similar to the risk factors for bleeding, particularly in the older population. 22 Disproportionality analysis of the USA FAERS data confirmed positive signals for increased reporting of haemorrhage associated with apixaban. In half of the SAE reports concomitant use of a bleeding risk medicine was reported, potentially contributing to the high number of reports of haemorrhage with apixaban use. The potent deleterious effects of antithrombotic medicines and NSAIDs urge caution with concomitant use and vigilance by prescribers regarding appropriateness and duration of therapy.
A consequence of having a bleeding event is the likelihood of cessation of anticoagulant treatment, which may increase the risk of subsequent thrombotic events. 23 Discontinuation of warfarin following a GI bleed was recently shown in a US study to increase the risk of death and thrombotic events 23 and in a study of over 68,000 outpatients with prior ischaemic vascular disease those with a serious bleed had a three-fold increase risk (HR, 3.25; 95% CI, 2.58–4.10; p < 0.0001) of a significant vascular outcomes (myocardial infarction, stroke, vascular death) compared with patients without a serious bleed. 24 Examination of clinical outcomes associated with major bleeding in patients with AF treated with either apixaban or warfarin from the ARISTOTLE trial, reported a 12-fold elevation in the risk of death or thrombotic events (ischaemic stroke or myocardial infarction) during the 30 days post a major bleeding event, with no significant difference between apixaban and warfarin. 25 Following a major bleed, 70.8% of the study cohort had an interruption of anticoagulant use with just over half resuming therapy within 2 weeks. The interruption of anticoagulant therapy post-bleed will likely result in these patients being less protected from thrombotic events, contributing to the increased risk of death and thrombotic events. 25 Together with the results of the current study, this highlights the need for identification of those most at risk of adverse events and reduction of modifiable risk factors where appropriate, to prevent risk of bleeding in patients receiving anticoagulation but also the potential for harm from thrombotic events due to cessation of anticoagulant therapy.
There has been a rapid adoption of the NOACs into clinical practice,9,26 and we observed increasing utilisation of apixaban in Australia since its availability. In a 3 years study of over 6000 AF patients in the USA, NOACs accounted for 62% of new prescriptions and 98% of anticoagulant-related medicine costs, with those at lower risk of adverse events more likely to receive a newer oral anticoagulant. 26 Apixaban has been found to be efficacious in preventing stroke in patients with AF and in the prevention of VTE and may be a viable option for patients who have specific contraindications to warfarin or where INR monitoring cannot be performed appropriately. 7 The prevalence of cerebrovascular thrombosis adverse event reports ranged from 3% in the USA to 14% in Canada databases, with positive signals detected from the disproportionality analysis. Compliance may be a concern given the requirement for twice daily dosage regimen for apixaban and the relatively short duration of anticoagulant activity. However, a recent study from Sweden showed at 12 months persistence was 85.9% (95% CI: 81.8–90.1%) with apixaban, greater than that observed for either dabigatran or rivaroxaban. 27
The characteristics of the patient populations between those in the clinical trials and those who receive apixaban in clinical practice are likely to be different. Generally, patients in clinical trials are younger and consequently less likely to have comorbid conditions, including renal or hepatic impairment and use fewer medicines, limiting the overall generalisability of safety and efficacy results obtained from randomized clinical trials.8,28 The average age of patients from SAEs reports associated with apixaban was 72 years and older. Only a third of patients included in the ARISTOTLE trial were aged 75 years and older, with 20% having a prior stroke or transient ischaemic attack (TIA), 16% having a prior clinically relevant bleed. 3 This is particularly pertinent for clinically relevant subgroups of patients with AF, most likely to use apixaban and where the risk of both stroke and bleeding are increased, including those aged 75 years or older, or with prior stroke/TIA or renal dysfunction. However, in a post-hoc analyses of clinical outcomes and management of major bleeding from the ARISTOTLE study, the median age of those with a major bleed was 74 years old (IQR 67-79) significantly different from those people who did not have a major bleed whose median age was 70 years old (IQR 63-76). 25
There are limitations of this study, including the use of SAE reporting data, which can be associated with underreporting, reporting biases and varying report quality.29,30 The numbers of adverse event reports for apixaban for Australia and Canada were low, possibly reflective of the short time since apixaban was introduced on the market. The specific types of adverse events reported in the current study are similar to those observed in clinical trial results, whether these reports are a true reflection of all adverse events that are occurring in clinical practice or are an underrepresentation of those which are less severe (e.g. minor bleeds) with apixaban remains unclear. We were unable to determine if reporting of concomitant medicines were truly co-administered with apixaban. The disproportionality analysis was only able to be conducted in the USA FAERS database and other signals may have been observed with the inclusion of other spontaneous report datasets if available.
Conclusion
The results of this study highlight the need for continued post-marketing surveillance of apixaban and the conduct of robust population-based observational studies using national linked datasets. It also demonstrates the need for increased awareness of potential for bleeding events, particularly in people with comorbid conditions where the use of concomitant medicines that may increase the risk of bleeding is common. Accurate assessment of individual clinical characteristics, balancing the risk of stroke and bleeding in the older population will facilitate the appropriate and safe use of apixaban in clinical practice.
Footnotes
Funding
This work was supported by an Australian Government National Health and Medical Research Council Centre of Research Excellence in Post-Marketing Surveillance of Medicines and Medical Devices grant (APP1040938).
Conflict of interest statement
Gillian E. Caughey, Lisa M. Kalisch Ellett, John D. Barratt and Sepehr Shakib have no conflicts of interest to declare.
