Abstract
Objectives:
A large retrospective database study was conducted to assess the incidence rate of treatment-emergent renal impairment/failure, seizure, and hemolytic anemia in doripenem and imipenem intravenous (IV)-exposed patients treated for complicated urinary tract infection (cUTI) and complicated intra-abdominal infection (cIAI) in US hospitals.
Methods:
Using the Premier Perspective™ Database (PPD), which maintains hospital discharge records for over 309 million patients, the incidence rate of treatment-emergent renal impairment/failure, seizure, and hemolytic anemia in the doripenem-treated compared with imipenem IV-treated population was examined.
Results:
The unadjusted doripenem rate ratio (RR) for renal impairment/failure relative to imipenem IV was 1.13 [95% confidence interval (CI) 1.07–1.21; p < 0.0001]. The unadjusted doripenem rate ratio for seizure relative to imipenem IV was 0.74 (95% CI 0.52–1.05; p = 0.07). In the comparative safety analysis, adjusted incidence rates of renal impairment/failure between doripenem-exposed patients and a propensity score-matched comparator cohort of imipenem IV-exposed patients showed no statistically significant difference in cUTI [RR = 1.02; 95% CI 0.93–1.12; p = 0.71] or cIAI (RR = 1.17; 95% CI 1.00–1.36; p = 0.05). Likewise, there was no statistically significant difference in adjusted incidence rates for seizures in doripenem-treated versus matched imipenem-treated patients for cUTI (RR = 0.69; 95% CI 0.41–1.14; p = 0.15) or cIAI (RR = 0.45; 95% CI 0.15–1.41; p = 0.17). No hemolytic anemia events were observed in this study.
Conclusions:
In this large retrospective cohort study of US hospitalized patients, no statistically significant differences in the adjusted relative rates of renal impairment/failure and seizure were observed between doripenem and a propensity score-matched comparator cohort of imipenem IV patients in the treatment of cUTI and cIAI.
Introduction
Carbapenems have the broadest spectrum of antibacterial potency among all antibiotics, with demonstrated effectiveness against gram-positive and gram-negative bacteria. As such, they are commonly used to treat severe and complicated bacterial infections [Hawkey and Livermore, 2012]. There are currently four marketed carbapenems in the United States (US): imipenem, meropenem, ertapenem, and doripenem.
Doripenem is approved in the US and in a number of European, Asian Pacific, and Latin American countries for various indications [Livermore, 2009]. Adverse reactions associated with doripenem included diarrhea, headache, increased hepatic enzymes, nausea, oral candidiasis, phlebitis, pruritus, rash, vulvomycotic infection, Clostridium difficile colitis, and hypersensitivity reactions. Further, anaphylaxis, neutropenia, thrombocytopenia, seizures, Stevens–Johnson syndrome, and toxic epidermal necrolysis have also been reported and identified as adverse drug reactions during post-approval use of doripenem. The most common adverse reactions (⩾5%) are headache, nausea, diarrhea, rash, and phlebitis [Ortho-McNeil-Janssen Pharmaceuticals, Inc., 2007].
While seizure risk is associated with carbapenems, doripenem may have a lower risk propensity based on animal and clinical studies [Horiuchi et al. 2006; Kaniga et al. 2010]. Horiuchi and colleagues investigated the convulsive liability of doripenem in comparison with various β-lactam antibiotics using several animal species and found little convulsive activity. In intravenous (IV) injection studies, doripenem caused no changes in the electroencephalogram (EEG) and behavior in rats at 400 mg/kg. In mouse intracerebroventricular (ICV) injection studies, doripenem did not induce convulsions at up to 100 μg/mouse. Similarly, in dog ICV injection studies, doripenem had no effects on the EEG and behavior in dogs at any doses. Convulsion induced by β-lactam antibiotics is considered to be related to the inhibition of gamma amino butyric acid (GABA) receptor binding. The lower epileptogenicity of doripenem found in animal studies may be due to its low affinity for the GABA receptor compared with that of other β-lactam antibiotics [Redman and File, 2009].
A recent meta-analysis of randomized clinical trials found that carbapenems had a significantly higher risk of seizures than non-carbapenem antibiotics [odds ratio (OR) 1.87, 95% confidence interval (CI) 1.35–2.59], with imipenem having the greatest risk (OR 3.5, 95% CI 2.23–5.49) and doripenem having the lowest risk (OR 0.44, 95% CI 0.13–1.53) [Cannon et al. 2014]. However, the analysis for doripenem was limited by the small number of studies that included this carbapenem (n = 2). At the time the current study was requested by the US Food and Drug Administration (FDA), seizure was an adverse event rarely associated with doripenem.
A large retrospective study of US hospitalized patients treated with doripenem and imipenem IV for complicated urinary tract infection (cUTI) and complicated intra-abdominal infection (cIAI), the two indications approved in the US, was conducted to assess the safety of doripenem. Imipenem IV was chosen specifically as a comparator for doripenem as it was the carbapenem with indications most similar to doripenem. This provided a comparator group of patients most similar to doripenem. The Premier Perspective™ Database (PPD) contains data from more than 309 million patients and over 40 million hospital discharge records. This database allowed us to examine the safety of doripenem and imipenem IV in a large number of hospital patients and hospital environments under real-world settings. The incidence rate of treatment-emergent renal impairment/failure, seizure, and hemolytic anemia events were quantified and compared in cUTI and cIAI hospital patients treated with doripenem or imipenem IV from 1 January 2010 to 31 December 2011.
Methods
Patients
The cohort included US hospitalized patients who were administered doripenem or imipenem IV therapy for at least 1 day. Data were collected retrospectively from the PPD from 12 October 2007 onwards. Due to the length of time required for this study, two drug utilization analyses were performed. The first was conducted during the first quarter of 2010 and considered patients discharged from inpatient services between January 2004 and December 2009 for inclusion. The analysis was updated in 2011 to include patients discharged in 2010. The safety analysis period (1 January 2010 through 31 December 2011) was chosen based on the availability of data indicating whether or not medical conditions were ‘present on admission’ in order to capture only adverse events that occurred after hospitalization. A total of 12,205 eligible cUTI and 4392 eligible cIAI patients, who were administered doripenem or imipenem IV therapy for at least 1 day, with discharge dates in 2010 or 2011 were included in the safety analysis set.
Based on data for the management of cIAI (for which we had information on a background seizure rate), the prevalence of seizure from a review of 1754 patients treated with imipenem/cilastatin in phase III dose-ranging studies in the US was assumed to be 3.0% and renal impairment/failure 3.5% [Calandra et al. 1988]. This suggests that the minimum number of patients required to study the incidence rate of seizures would be 1628 patients for each treatment who had comparable clinical experience, and 1384 patients for renal impairment/failure [assuming α = 0.05, 80% power, in order to detect a relative risk of at least 2.0]. Hemolytic anemia is a rare event and if the frequency of this event was assumed to be as high as 2/1000, using the ‘rule of three’, the sample size needed to observe at least one adverse event with 95% probability was projected to be approximately 1500.
Patient demographics, all diagnoses, medications, procedures, and pharmacy data were extracted from the PPD. The selection and exclusion codes for cUTI were based on the definitions in the draft guidance developed by the FDA and were used to develop an algorithm to define cUTI [Center for Drug Evaluation and Research, 2012]. An algorithm combining selected International Classification of Diseases, Ninth Revision (ICD-9) and procedure codes for cIAI was developed using guidelines issued by the Surgical Infection Society, the American Society for Microbiology, and the Society of Infectious Disease Pharmacists [Solomkin et al. 2010]. The algorithms used for defining cUTI and cIAI are provided in Appendix A of the online Supplemental Material.
Study design
Data source
Data for the study were derived from hospital discharge data (available from January 2000 through March 2012) from the PPD, which was periodically updated. The safety analysis included data from 1 January 2010 through 31 December 2011. Doripenem and imipenem IV utilization data were analyzed for two periods: 2007–2009 (spans the time from the introduction of doripenem in the US up to the start of the safety study period) and 2010 (covers part of the safety analysis period). Patients who received both doripenem and imipenem IV (<1%) were excluded from the safety analysis set.
The PPD contains data from standard hospital discharge files, including a patient’s demographic and disease state, and information on billed services, including medications, laboratory, diagnostics, and therapeutic services in de-identified patient daily service records.
Variables
The primary study endpoint was a diagnosis of incident renal impairment/failure, seizure, or hemolytic anemia. All patients from the study cohorts presenting with any of these three adverse events during follow up (hospital stay) were identified. Adverse events were identified in the database through prespecified ICD-9 diagnosis codes and procedure codes.
Information on the use of doripenem and imipenem IV was obtained from the daily pharmacy billing records during the hospital stay and the prescription codes for these drugs. The billing records did not include the dose administered or frequency. Initiation of doripenem or imipenem IV was defined as the first pharmacy-billing day for that drug during the hospital stay. The time of follow up of each patient, from the first day during the hospital stay when the drug was billed to follow up end date (hospital discharge date), was considered as person–time at risk for the adverse event.
Doripenem or imipenem IV was considered first-line therapy if no other prior antibiotic was administered (as determined by the date of dispensing); second-line therapy if any other antibiotic was administered on a date prior to the initial administration of doripenem or imipenem IV; and multiple treatment if doripenem or imipenem IV was first administered on the same day of service as the initial administration of another antibiotic.
The risk factors considered in the analyses were age, gender, geographic region, duration of the hospital stay before receiving the first dose, intensive care unit (ICU) admission, and use of other antibiotics including other carbapenems (prior to receiving each drug), valproic acid, or probenecid during the same hospital stay. Diagnoses that were known risk factors for seizure, hemolytic anemia, or renal impairment/failure were also included (see Appendix B of the online Supplemental Material). For example, abnormal renal function is common in patients with cUTI and cIAI, as are many of the other listed risk factors for renal disease including sepsis, diabetes, and proteinuria. The list of risk factors for seizure included an admitting diagnosis for seizures, head trauma, intracranial bleeding, or several other conditions that would predispose the patient to seizures.
Statistical analyses
Descriptive statistics were used to analyze baseline characteristics by treatment cohort and patterns of doripenem and imipenem IV usage. Additional descriptive analyses included estimating incidence rates of the three adverse events of interest for each drug (calculated by dividing the number of events by the total person–time of drug exposure to doripenem or imipenem IV and expressed as the number of events per person-year) and 95% CIs. Incidence rates for renal impairment/failure and seizures were also stratified by cUTI and cIAI. All p-values were calculated using a Pearson chi-square or Wald chi-square method.
Propensity score-matched Poisson regression models were used to adjust for the appropriate factors that influenced the risk of renal disease or seizure under the rare event assumption for the event rates. Modeling of safety endpoints (renal impairment/failure and seizure) was performed using SAS PROC GENMOD with a Poisson distribution, log link, and an offset equal to the number of days of exposure to either doripenem or imipenem IV as appropriate and included risk factors that could not be adjusted for with propensity score matching. Both outcome variables were assessed as positive if the relevant outcome ICD-9 codes were coded as ‘Not Present on Admission’.
A comparative safety analysis of the incidence rates of renal impairment/failure and seizure was conducted based on distribution of propensity scores for receipt of doripenem or imipenem IV. Complete univariate demographic comparisons of doripenem- and imipenem IV-exposed patients were performed. All eligible patients were entered into a logistic model assessing the probability of receiving doripenem, i.e. the ‘propensity score’. Variables entered into the model included: age category, gender, admission source, medical or surgical discharge, hospital bed size category, hospital urban/rural status, hospital teaching status, hospital geographic region, discharge year, presence of a central line, presence of a urinary catheter, presence of a nasogastric tube, presence of an abdominal drain, receipt of corticosteroid, as well as the Charlson Comorbidity categories of renal disease, any malignancy, and chronic obstructive pulmonary disease. The propensity model used SAS PROC LOGISTIC with Mahalanobis Metric Matching algorithm.
Results
Doripenem was introduced into the US marketplace in 2007 and by 2010 it accounted for approximately 50% of overall doripenem and imipenem IV utilization for all indications and 51.6% of cUTI and 46.7% of cIAI indications (Figure 1). The utilization of imipenem IV and doripenem for all indications increased from 2004 to 2010, likely due to the increased number of discharges and reporting hospitals in the PPD. In 2004, prior to the entry of doripenem into the market, 1.00% of all patients received imipenem IV whereas in 2010, 0.81% of all patients received either doripenem or imipenem IV.
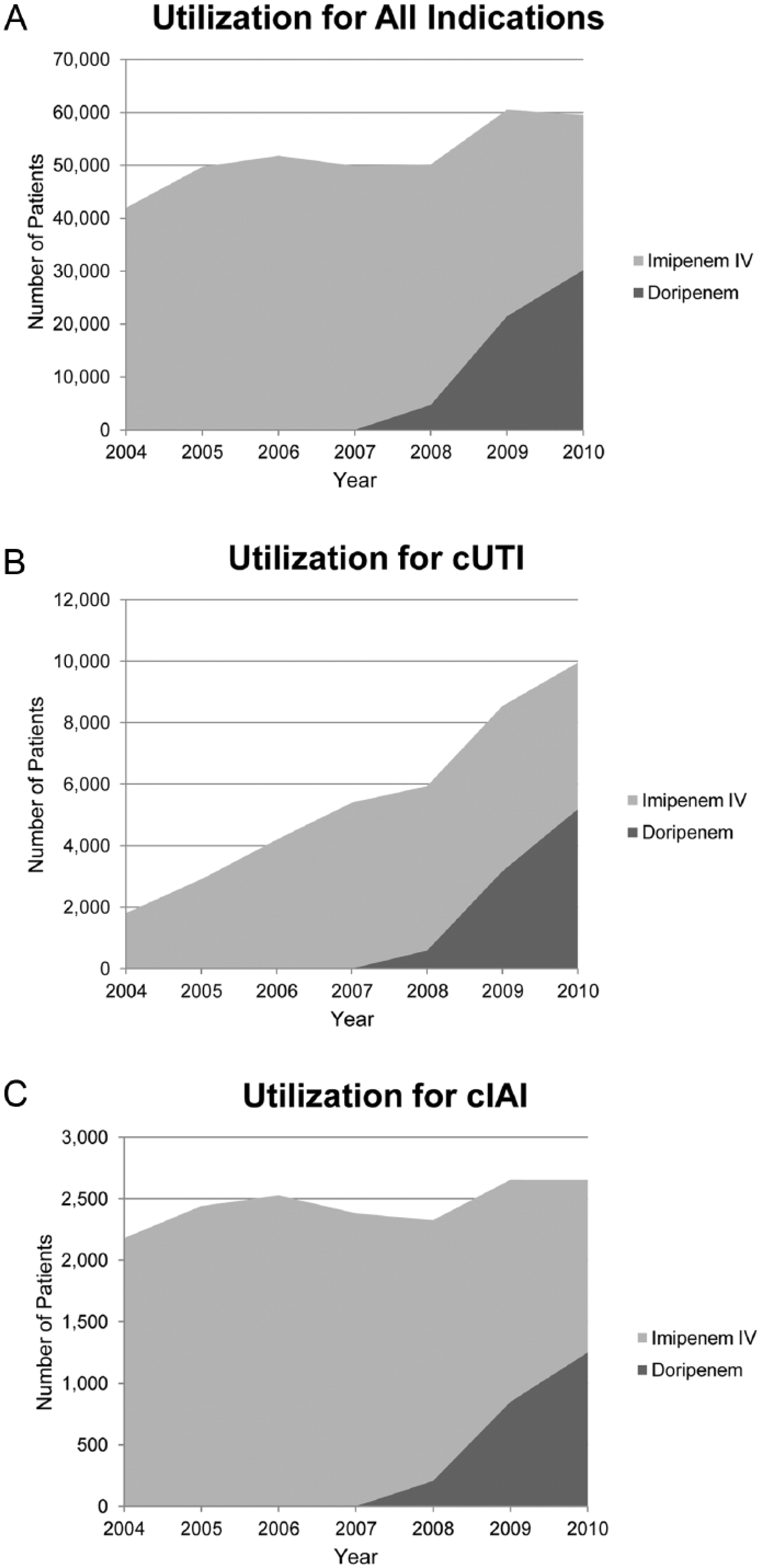
The utilization of doripenem or imipenem intravenous (IV) in US hospital settings from 2004–2010.
There were no remarkable differences in the doripenem and imipenem IV patient demographics of the 16,597 eligible patients in this study. However, doripenem-treated patients tended to have higher rates of extreme cases based on APR SOI (All Patient Refined Severity of Illness) and APR Risk of Mortality ratings as well as a higher percentage of patients receiving antibiotics before carbapenem administration (Table 1). There were significant differences in APR SOI between doripenem and imipenem-IV treated patients for both cUTI (p = 0.0011) and cIAI (p < 0.001) indications. The percentage of extreme cases based on APR SOI in doripenem compared with imipenem IV-treated patients was 45.3% versus 41.7%, respectively, for cUTI and 64.8% versus 57.5%, respectively, for cIAI. These differences may suggest that overall severity of illness before carbapenem exposure was somewhat greater in the doripenem cohort than in the imipenem IV cohort.
Patient demographics of the safety analysis set.
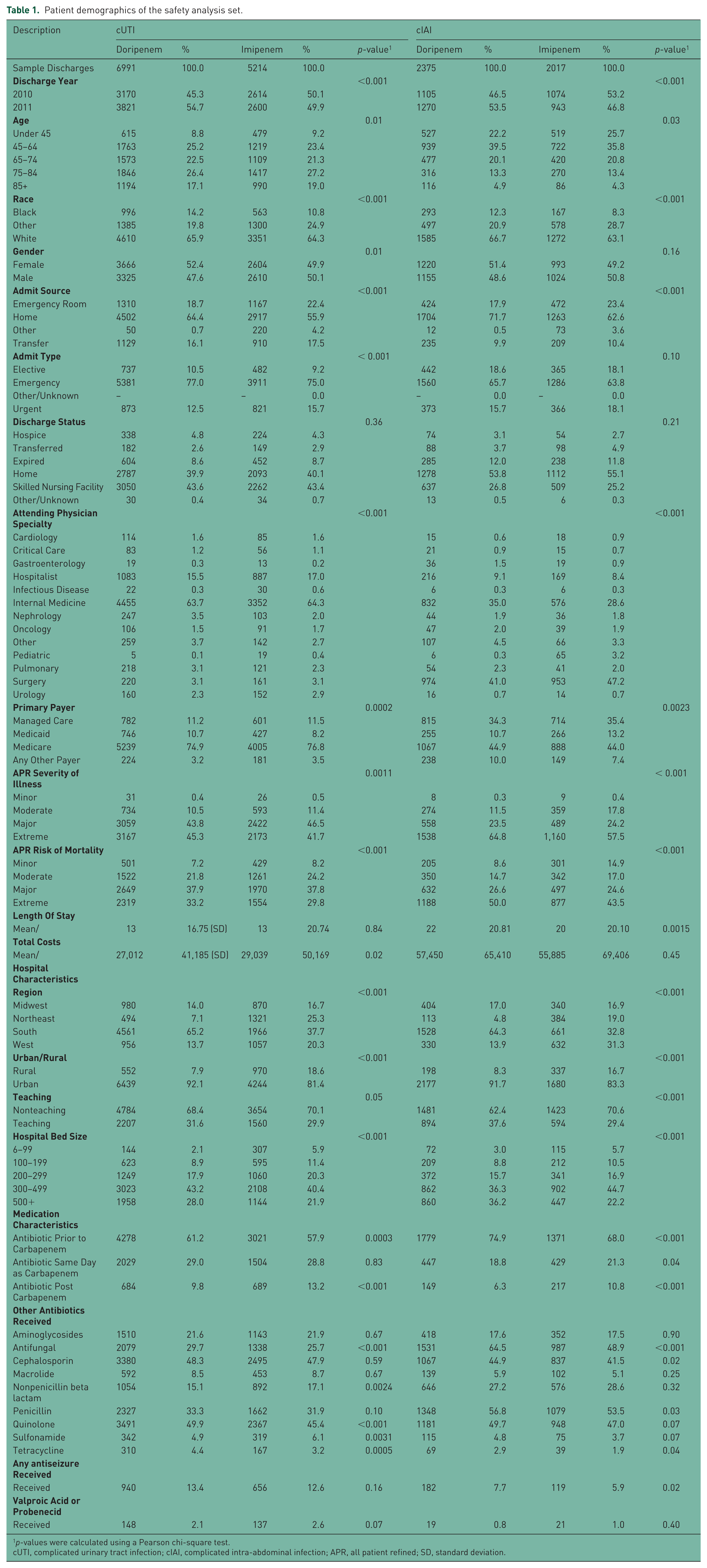
p-values were calculated using a Pearson chi-square test.
cUTI, complicated urinary tract infection; cIAI, complicated intra-abdominal infection; APR, all patient refined; SD, standard deviation.
For cIAI, patients treated with doripenem, average days of product administration was slightly greater than those treated with imipenem IV: 9.7 versus 9.3 days, respectively, in 2007–2009; and 9.8 versus 8.9 days, respectively, in 2010. Similarly, for cUTI, average days of product administration was slightly greater for doripenem compared with imipenem IV-treated patients: 6.5 versus 6.3 days, respectively, in 2007–2009; and 6.2 versus 5.8 days, respectively, in 2010.
There were no notable differences in rates of any other drug utilization or concomitant antibiotic use between doripenem and imipenem IV cohorts in the time periods studied (2007–2009 and 2010). Patients with cIAI generally received their first administration of doripenem or imipenem IV later than cUTI patients although there were no significant differences between the groups or time periods analyzed. Doripenem-treated patients had slightly greater number of days of administration than those treated with imipenem IV, whereas average initial, daily, and total doses were slightly higher among those treated with imipenem IV. Imipenem IV was also used at a somewhat higher rate than doripenem as a first line antibiotic. Patients treated with doripenem had a slightly higher rate of concomitant use of antifungals during both time periods compared with those treated with imipenem IV.
The safety analysis set includes all cUTI and cIAI hospital patients treated with doripenem (cUTI, n = 6991; cIAI, n = 2375) or imipenem IV (cUTI, n = 5214; cIAI, n = 2017) from 1 January 2010 to 31 December 2011. In the safety analysis set, the incidence rates per 100 treatment days for renal impairment/failure and seizure were estimated at 4.16 (95% CI 4.01–4.32) and 0.11 (95% CI 0.09–0.14), respectively, in doripenem-exposed patients (Table 2). The incidence rates per 100 treatment days for renal impairment/failure and seizure were estimated at 3.67 (95% CI 3.49–3.85) and 0.15 (95% CI 0.11–0.19), respectively, in imipenem IV-exposed patients (Table 2). Doripenem treatment had a significantly greater unadjusted rate of renal impairment/failures (1.13; 95% CI 1.07–3.85; p < 0.0001) compared with imipenem IV, but there was a nonsignificant trend toward a lower unadjusted rate of seizures for doripenem (0.74; 95% CI 0.52–1.05; p = 0.07).
Unadjusted rates for renal failure, hemolytic anemia, and seizure diagnoses by indication and carbapenem exposure.
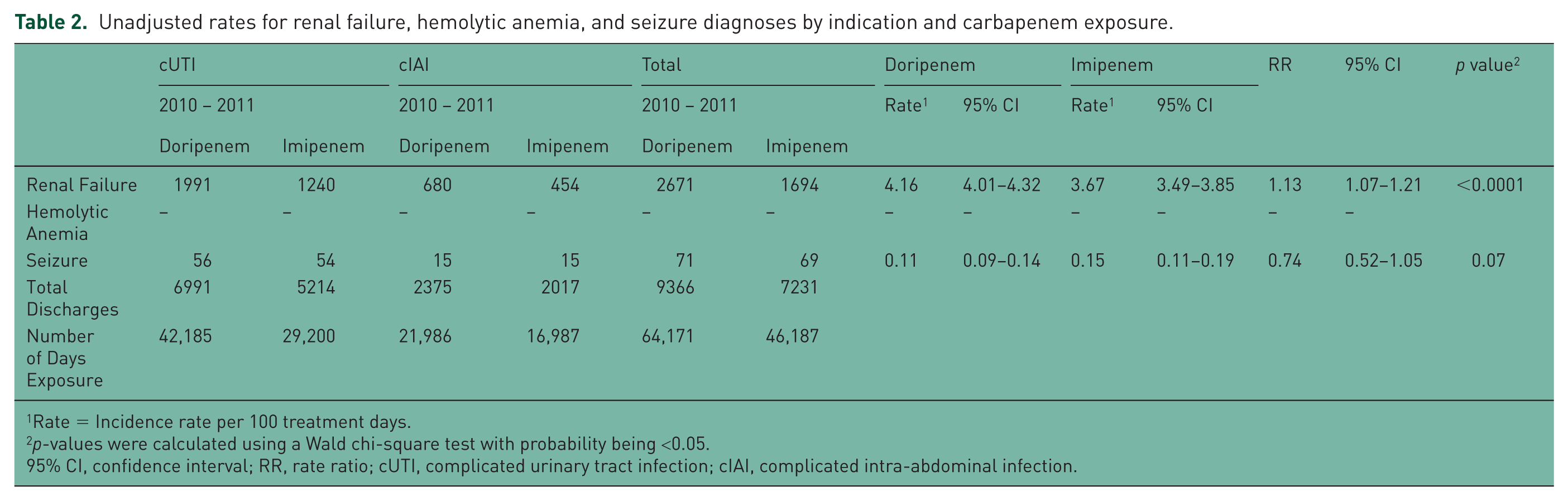
Rate = Incidence rate per 100 treatment days.
p-values were calculated using a Wald chi-square test with probability being <0.05.
95% CI, confidence interval; RR, rate ratio; cUTI, complicated urinary tract infection; cIAI, complicated intra-abdominal infection.
Within the cUTI and cIAI patient cohorts, doripenem was associated with a higher unadjusted rate per 100 treatment days of renal impairment/failures than imipenem IV in both cUTI (4.72 versus 4.24; p = 0.0035, respectively) and cIAI (3.09 versus 2.67; p = 0.02, respectively; Table 3). The unadjusted rates per 100 treatment days for cUTI patients indicated a nonsignificant trend toward a lower rate of seizures with doripenem compared with imipenem IV (0.13 versus 0.18;p = 0.08, respectively). The unadjusted rates per 100 treatment days for cIAI patients showed no difference in seizure rates between doripenem- and imipenem IV-treated patients (0.07 versus 0.09; p = 0.48, respectively; Table 3). No hemolytic anemia events were observed.
Incidence Rates of Renal Failure and Seizure Among All Patients Treated for cUTI or cIAI.
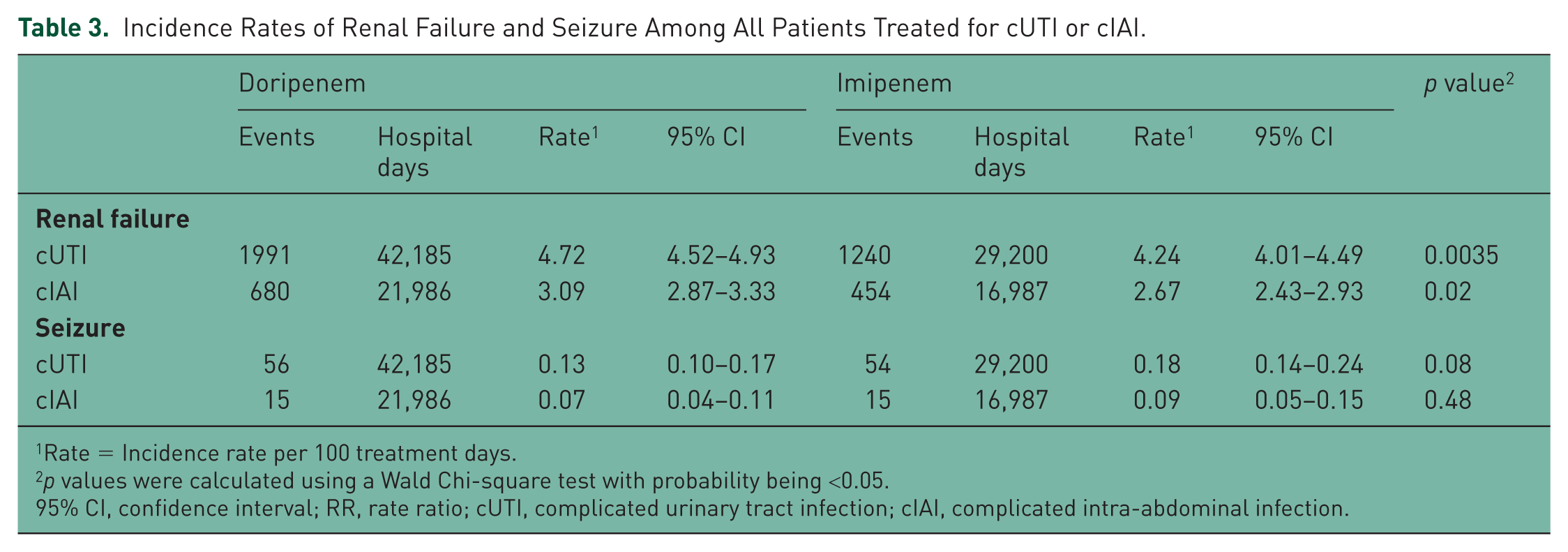
Rate = Incidence rate per 100 treatment days.
p values were calculated using a Wald Chi-square test with probability being <0.05.
95% CI, confidence interval; RR, rate ratio; cUTI, complicated urinary tract infection; cIAI, complicated intra-abdominal infection.
In the comparative safety analysis, incidence rates of renal impairment/failure and seizure were compared between doripenem-exposed patients and a propensity score-matched comparator cohort of imipenem IV-exposed patients using multiple Poisson regression models. Both the cUTI and cIAI models had c-statistics over 0.80 (cUTI = 0.84, cIAI = 0.81). No statistically significant difference was observed in the adjusted rate ratio of renal impairment/failure for doripenem compared to imipenem IV among cUTI (1.02; 95% CI 0.93–1.12; p = 0.71) and cIAI (1.17; 95% CI 1.00–1.36; p = 0.05) patients, although the adjusted rate ratio did approach statistical significance in cIAI patients (Table 4). Likewise, the adjusted rate ratio for seizures in doripenem versus imipenem IV patients did not show any significant difference in cUTI (0.69; 95% CI 0.41–1.14; p = 0.15) and cIAI (0.45; 95% CI 0.15–1.41; p = 0.17; Table 5). Significant differences still remained within the doripenem and imipenem IV-treated cohorts that made up the comparative safety analysis set. The imbalances remaining in the model indicated that the doripenem population had a greater level of comorbidity before doripenem exposure that was not adjusted for by the matching exercise. Therefore, it is unclear whether the observed adjusted risks are due to carbapenem exposure or to residual confounding that could not be adjusted in the analysis. The rates of hemolytic anemia were not compared due to an inadequate number of events observed.
Comparative safety analysis of renal failure rates among propensity score-matched patients treated with doripenem or imipenem IV.
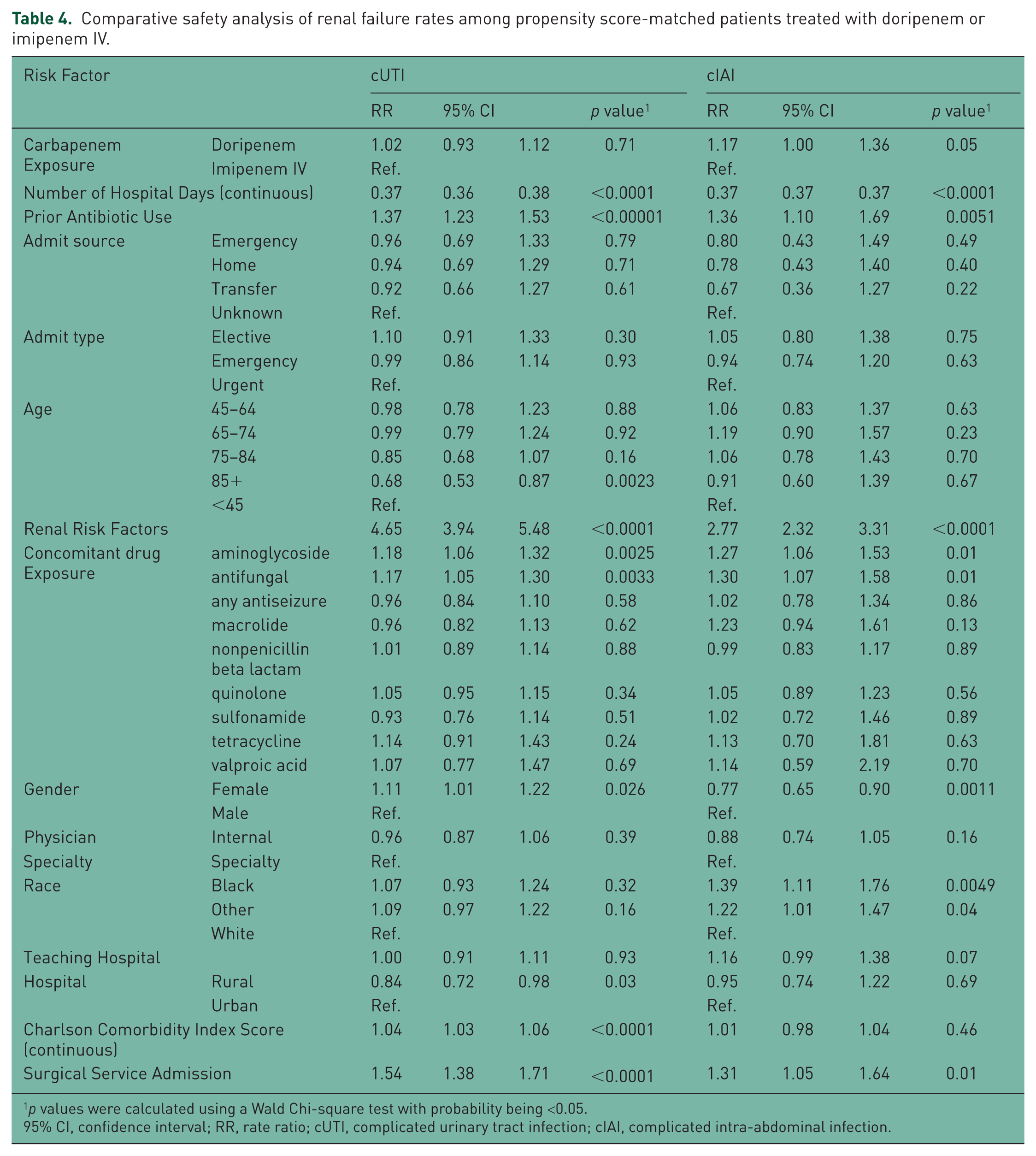
p values were calculated using a Wald Chi-square test with probability being <0.05.
95% CI, confidence interval; RR, rate ratio; cUTI, complicated urinary tract infection; cIAI, complicated intra-abdominal infection.
Comparative safety analysis of seizure rates among propensity score-matched patients treated with doripenem or imipenem IV.
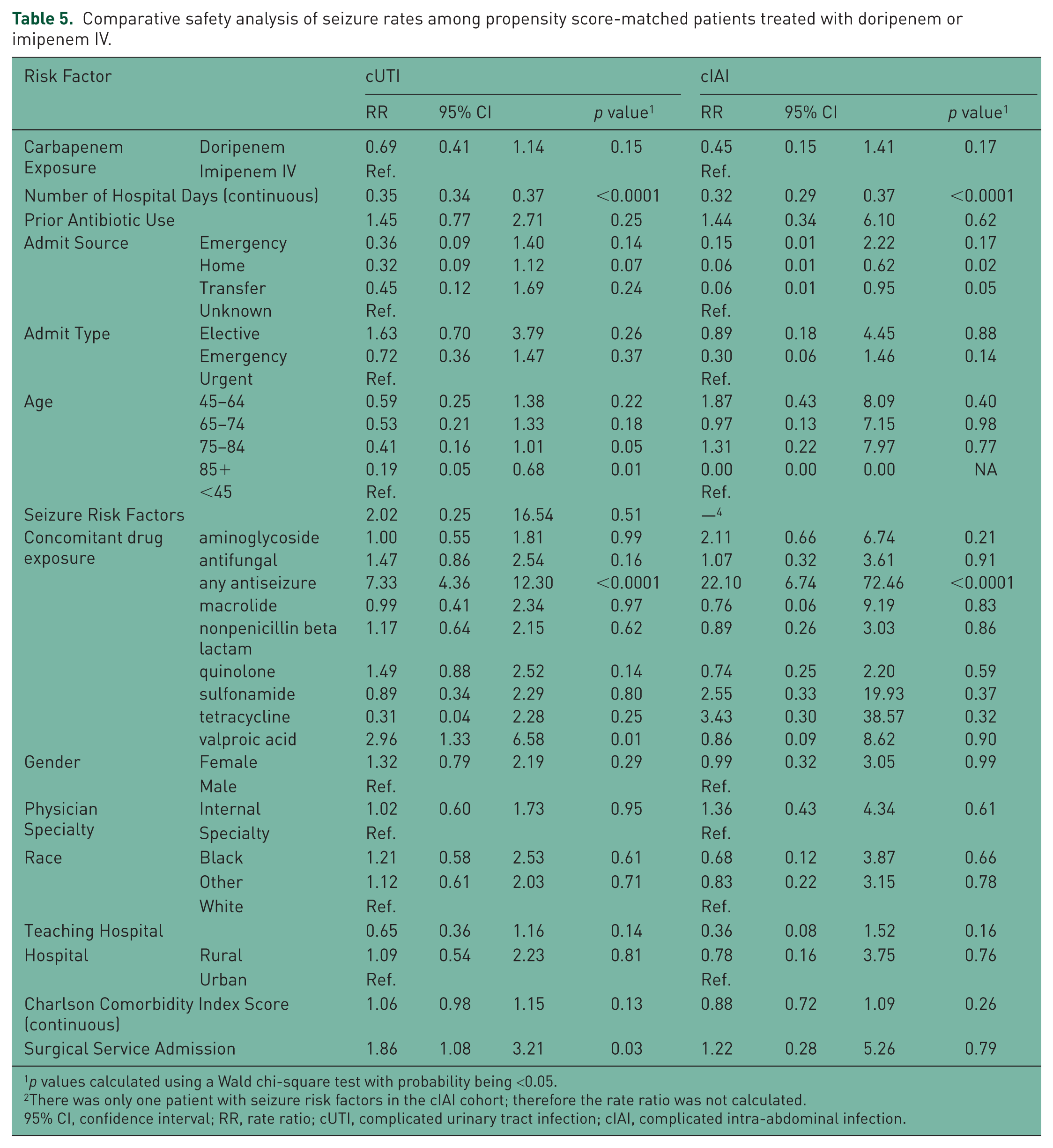
p values calculated using a Wald chi-square test with probability being <0.05.
There was only one patient with seizure risk factors in the cIAI cohort; therefore the rate ratio was not calculated.
95% CI, confidence interval; RR, rate ratio; cUTI, complicated urinary tract infection; cIAI, complicated intra-abdominal infection.
A number of factors were associated with decreased or increased risk of renal impairment/failures and seizures. For renal impairment/failure, decreased risk was associated with number of hospital days, patients over age 85, and rural hospital settings for cUTI patients and number of hospital days and females for cIAI patients (Table 4). Factors associated with an increase in renal impairment/failures risk for both cUTI and cIAI were similar (prior antibiotic use, presence of renal failure risk factors, use of aminoglycosides and antifungals, and surgical service admission) with additional risk factors being females and increase in Charlson Comorbidity Index Score for cUTI patients and a designation of black or other race for cIAI patients. For seizures, decreased risk was associated with number of hospital days and patients over age 85 for cUTI patients and number of hospital days and admission from home or transfer from another hospital for cIAI patients. Increased risk for seizures was associated with the use of antiseizure medication for both cUTI and cIAI patients, with valproic acid use and surgical service admission being additional risk factors for cUTI patients (Table 5).
Discussion
In this large, US hospital-based retrospective study, we evaluated the safety of doripenem in the treatment of cUTI and cIAI. To our knowledge, this is the first large epidemiologic study that has evaluated the safety of doripenem in the treatment of cUTI and cIAI with respect to the rates of renal impairment/failure, seizure, and hemolytic anemia. The study evaluated patients across multiple hospitals with different geographic and administrative characteristics where doripenem is most likely to be administered, thus our findings may be generalizable to real-world hospital settings.
We found that doripenem-treated patients had a significantly higher unadjusted rate ratio for renal impairment/failure compared with imipenem IV-treated patients, and the unadjusted doripenem rate ratio for seizure relative to imipenem IV was lower, although it did not reach statistical significance. The unadjusted rate ratios should be viewed with caution as they do not account for channeling or confounding factors that may bias the results. In addition, the overall severity of illness before drug exposure may have been greater in the doripenem-treated cohort than in imipenem IV-treated patients. The rates of hemolytic anemia were not compared due to an inadequate number of events observed.
The propensity score-matched comparative analysis did not show a statistically significant difference in the rates of renal impairment/failures or seizures in doripenem-treated patients compared with imipenem IV-treated patients. The relative rate for renal impairment/failure in cIAI approached statistical significance; however, the increase may be due to a higher level of morbidity before receipt of doripenem that could not be adjusted for in the regression models.
Carbapenems have an important role in the treatment of serious nosocomial infections; however, the potential for seizures is a primary safety concern. The highest rates of seizures have been reported for imipenem (up to 5% in some studies) [Sutter et al. 2015]. Animal studies suggest that doripenem has less potential to induce seizures than imipenem and meropenem due to lower affinity for binding to GABA receptors. Data from clinical trials and postmarketing experience support the low seizure-inducing potential of doripenem [Redman and File, 2009; Zhanel et al. 2009]. Although rare, acute renal failure/renal insufficiency events have been reported with the use of other carbapenems [Merck & Co., Inc., 2008, 2009]. Cases of renal failure and renal impairment with the use of doripenem have also been reported in clinical trials and the post-marketing setting; however, a causal relationship has not been established [data not published]. Our study provides additional data on the safety profile of doripenem in a broad hospital-based population of doripenem-treated patients. Further epidemiologic studies are needed on the utilization of doripenem in the real-world clinical setting, particularly in high-risk patients.
Study limitations include the use of an administrative database, which is subject to the biases of analyzing secondary databases. All variables were defined by ICD-9 codes and lack the full clinical details available in medical charts. In addition, there were no ICD-9 codes for cUTI or cIAI; therefore, we relied on algorithms to define these indications as well as risk for renal impairment/failure and seizure. These algorithms may have resulted in misclassification of some variables. Misclassification may have also resulted from loss of data due to patients who were lost to follow up or essential data that was not collected as routine fields. Another limitation was that exposure to doripenem and imipenem IV was determined by billing records. As such, the analysis did not consider dosing administration, frequency, or dose adjustment. However, the validity of the algorithms was very good when compared with source medical records. In addition, the PPD differs from other observational databases in how ICD-9 and Current Procedural Terminology (CPT) codes are captured. The PPD captures all discharge diagnosis codes recorded by the hospital without any restriction on number of diagnoses captured. This feature would suggest that comorbidities would be more accurately assessed using the PPD compared with other observational sources.
Our data were also subject to real-world prescribing practices that may have biased the results in the comparative safety analysis. The decision to prescribe doripenem or imipenem IV was likely influenced by patient characteristics, prescriber assessment of the health status and risk profile, and local policies and formularies. Patient populations were comparable across most key factors; however, doripenem patients tended to have higher rates of extreme cases based on APR SOI than imipenem IV-treated patients. A propensity score-matched cohort analysis was conducted to adjust for confounding that might have been related to the safety outcomes of interest, but differences in the two cohorts were observed. Patients treated with doripenem may have had a higher level of morbidity compared with imipenem IV-treated patients; therefore, residual confounding may still have influenced the results.
Overall, there was a nonsignificant trend toward lower unadjusted seizure rates between the doripenem and imipenem IV cohorts; whereas unadjusted renal impairment/failure rates were significantly higher in doripenem compared with imipenem IV patients. There was evidence that residual confounding due to higher levels of morbidity among patients prior to exposure to doripenem may have caused bias in this assessment. In a propensity score-matched comparative analysis, there were no statistically significant differences in adjusted seizure rates or renal impairment/failure rates between the doripenem and imipenem IV cohorts.
Footnotes
Acknowledgements
We thank Dr. Tracy T. Cao (Source One Technical Solutions) for providing writing assistance and Dr. Bradford Challis (Janssen Research & Development) for providing additional editorial support for this manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Janssen Research & Development, LLC (formerly known as Johnson & Johnson Pharmaceutical Research & Development, LLC), Raritan, NJ, USA.
Conflict of interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Mrs. Pai and Dr. Chavers are employees of Janssen Research & Development, LLC. Mr. Magee and Ms. Baumer are employees of Premier Inc., which was paid by Janssen to conduct the study. All authors declare that, except for income received from their primary employer, no financial support or compensation has been received from any individual or corporate entity over the past 3 years for research or professional service that could be perceived as constituting a potential conflict of interest. Authors may be shareholders of Johnson & Johnson, the parent company of the Janssen companies. All authors meet ICMJE criteria and all those who fulfilled those criteria are listed as authors. All authors had access to the study data, contributed to the analysis and interpretation of the data, were involved in the manuscript from its conception, critically reviewed each draft, provided comments and guidance on its direction, made the final decision about where to publish these data, and take responsibility for the work.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
