Abstract
Inhaled anticholinergics (ipratropium bromide and tiotropium bromide) are widely used as maintenance treatment in chronic obstructive pulmonary disease. Previous studies have reported on their cardiovascular effects but relatively little is known about their effects on the bladder. Acute urinary retention is a medical emergency which can be associated with serious complications. Our objective was to evaluate the existing literature regarding the effects of inhaled anticholinergics on urinary retention among patients with chronic obstructive pulmonary disease. We searched PubMed and the United States Food and Drug Administration (FDA) adverse events database for case reports, observational studies, randomized controlled trials (or meta-analyses of such trials) that reported on the outcome of urinary retention with inhaled anticholinergics (ipratropium or tiotropium). We checked 27 published articles and identified relevant papers including two case reports, three pooled analyses, two observational studies and one randomized controlled trial. Two of the observational studies and a pooled analysis of randomized controlled trials reported a significant increase in the risk of acute urinary retention with inhaled anticholinergics. Older patients with benign prostatic hyperplasia seem to be at the highest risk of this adverse effect which tends to occur soon after treatment initiation. Although all the links in the chain have yet to be fully elucidated, the preponderance of evidence suggests the possibility of a causal relationship between inhaled anticholinergics and urinary retention. Clinicians should carefully balance these and other adverse effects of inhaled anticholinergics against their known symptomatic benefits on exacerbations, after eliciting patient preferences for various outcomes in a shared decision-making context.
Background
Acute urinary retention is a medical emergency which can be associated with serious complications. Previous studies have reported that the mortality rates in older men with precipitated acute urinary retention was double that of the general population [Armitage et al. 2007]. The mortality rates among men with acute urinary retention ranged from 9.5% in those aged between 45 and 54 years to 45.4% for men over 85 years of age [Armitage et al. 2007]. Hence, identification and avoidance of aetiological factors may have some impact in reducing risk of urinary retention and any associated medical complications.
Acute urinary retention is typically related to the presence of prostatic disease. However, some drugs have been known to be associated with urinary retention, including antipsychotics, tricylic antidepressants, calcium channel antagonists and anticholinergic drugs [Verhamme et al. 2008]. The bladder contains both M1 and M3 muscarinic receptors. The muscarinic M3 receptors play an important role in contracting detrusor smooth muscles [Verhamme et al. 2008]. Thus, acute urinary retention is associated with drugs that block muscarinic receptors.
The linkages between oral antimuscarinic agents and urinary retention are well accepted [Verhamme et al. 2008]. A database study in the UK demonstrated that the highest risk of acute urinary retention with oral antimuscarinic drugs was within the first month of treatment, with a significantly elevated relative risk (RR) of 8.3 [95% confidence interval (CI) 4.8–14.2], whereas longer term use over 30 days yielded a lower relative risk of 2.0 (95% CI 1.2–3.1) [Martín-Merino et al. 2009]. However, there remains some uncertainty whether the inhaled anticholinergic drugs share similar systemic adverse effects. A small pre–post study of 27 men with chronic obstructive pulmonary disease (COPD) and concomitant prostatic hyperplasia using inhaled tiotropium Handihaler (Boehringer Ingelheim, Germany) 18 μg failed to show any deleterious effects on prostatic symptoms or urinary flow rates [Miyazaki et al. 2008]. However, the small sample size, lack of suitable controls, exclusion of patients with history of urinary tract infections, and withdrawal of patients with systemic anticholinergic effect (such as dry mouth) limits definitive conclusions. Some have argued that the presence of a quaternary ammonium moiety may limit systemic absorption of inhaled tiotropium [Miyazaki et al. 2008]. Our objective was to evaluate the literature on the relationship between inhaled anticholinergic medications and acute urinary retention to ascertain the strength of the association in patients with COPD.
Methods
Search strategy and study selection
We carried out a search in June 2012 using PubMed. Our search strategy is shown in Box 1. We also identified additional studies reporting on urinary retention through our database of studies from previous systematic reviews of inhaled anticholinergics [Singh et al. 2008, 2011]. To identify spontaneous reports of urinary retention we also searched the US Food and Drug Administration (FDA) database of adverse events at www.adverseevents.com.
Box 1. Search strategy
We searched PubMed in June 2012 based on the search terms:
((“tiotropium” [Supplementary Concept] OR “tiotropium” [All Fields]) OR (“ipratropium” [MeSH Terms] OR “ipratropium” [All Fields]) OR inhaled-anticholinergic [All Fields]) AND ((“urinary tract” [MeSH Terms] OR (“urinary” [All Fields] AND “tract” [All Fields]) OR “urinary tract” [All Fields] OR “urinary” [All Fields]) OR (“urinary retention” [MeSH Terms] OR (“urinary” [All Fields] AND “retention” [All Fields]) OR “urinary retention” [All Fields]))
There were no language restrictions.
Eligible studies were case reports, observational studies, randomized controlled trials (or meta-analyses of such trials) that reported on the outcome of urinary retention with inhaled anticholinergics (ipratropium or tiotropium). Both authors (SS and YKL) screened titles and abstracts independently, and identified potentially relevant papers for further detailed checking prior to deciding on study inclusion. For the observational studies and randomized trials, YKL extracted data on study design and setting, population, sample size, limitations and outcomes. The data extraction was checked by SS. Owing to the diverse range of included study designs, it was not possible to summarize overall risk of bias with any single tool or to pool the data in a meta-analysis.
Results
The process of study selection is shown in Figure 1. We included eight articles comprising two case reports, two observational studies, one randomized controlled trial and three pooled analyses of trial data.
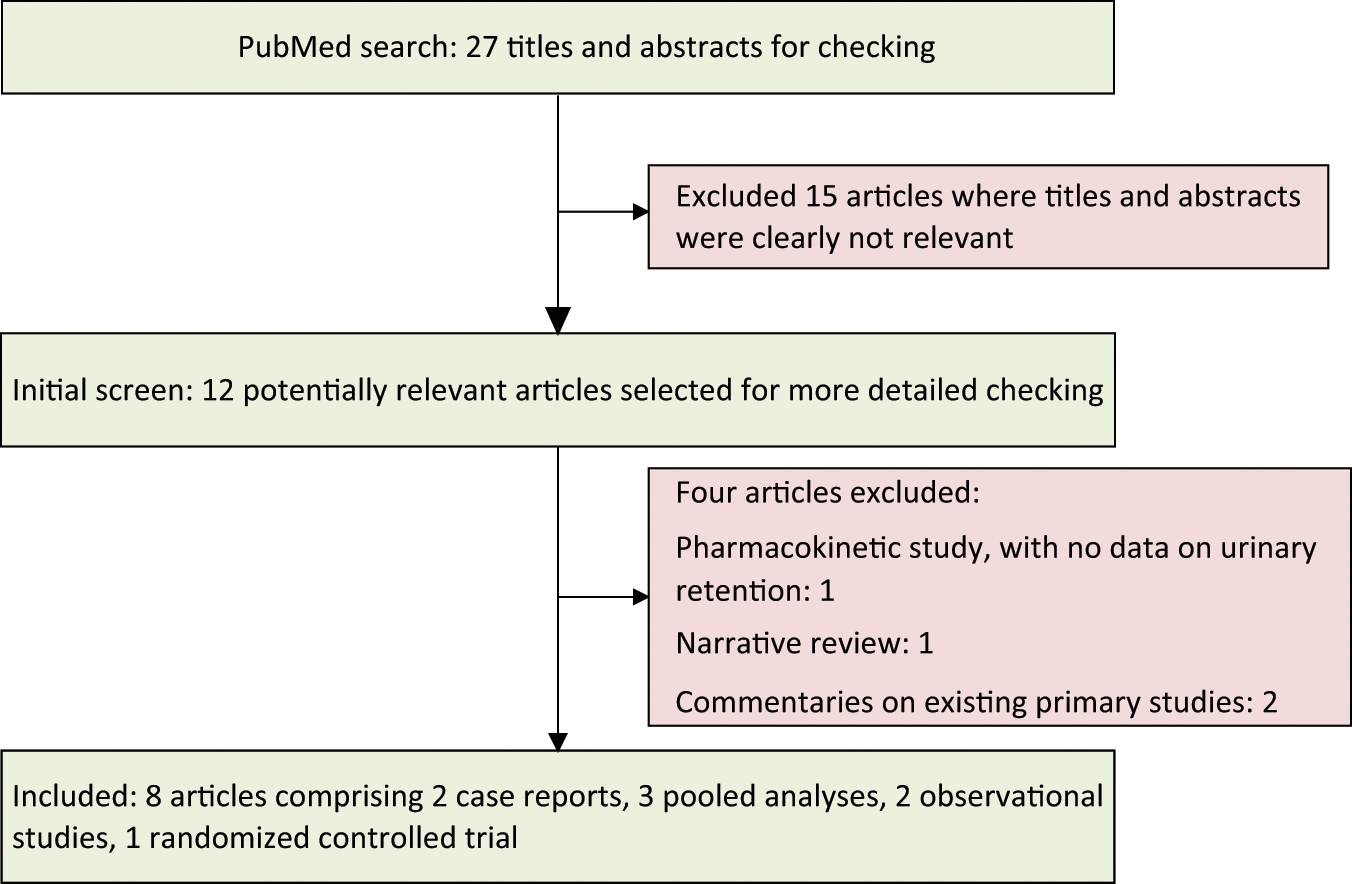
Flow diagram of the process of article selection.
Spontaneous reports
The possibility of urinary retention with ipratropium therapy was first described in a case report [Pras et al. 1991]. The adverse event was recorded in a 69-year-old man who was treated with nebulized ipratropium for exacerbation of COPD. The problem was resolved after cessation of ipratropium therapy. Similarly, a 5-year old girl was found to have poor urinary flow and voiding difficulties after therapy with ipratropium and salbutamol for asthma. These symptoms improved after cessation of therapy but emerged again when the patient was rechallenged with both bronchodilators [Hooimeijer et al. 2007].
According to the FDA’s adverse events reporting system, there were 350 reports in which tiotropium was the primary drug suspected to cause urinary retention among 20,856 reports of serious adverse events with tiotropium from April 2004 to September 2011. Here, 72 of the patients were hospitalized and there were 10 fatalities. However, the lack of information on confounders or denominators precludes definitive assessment of the incidence of this complication.
Observational studies
The risk of acute urinary retention with inhaled anticholinergics was assessed in two separate nested case–control studies, one based in Holland and the other in Canada [Afonso et al. 2011; Stephenson et al. 2011]. Both studies consistently found that the risk of acute urinary retention was most prominent in patients who had recently started on inhaled anticholinergics, thus providing strong evidence of a temporal link between initiation of therapy and development of acute urinary retention. Both studies reported that men with benign prostatic hyperplasia appeared to be particularly susceptible. There were no major differences in risk between ipratropium and tiotropium. The characteristics of these database studies are summarized in Table 1.
Studies reporting on urinary adverse events associated with inhaled anticholinergics.
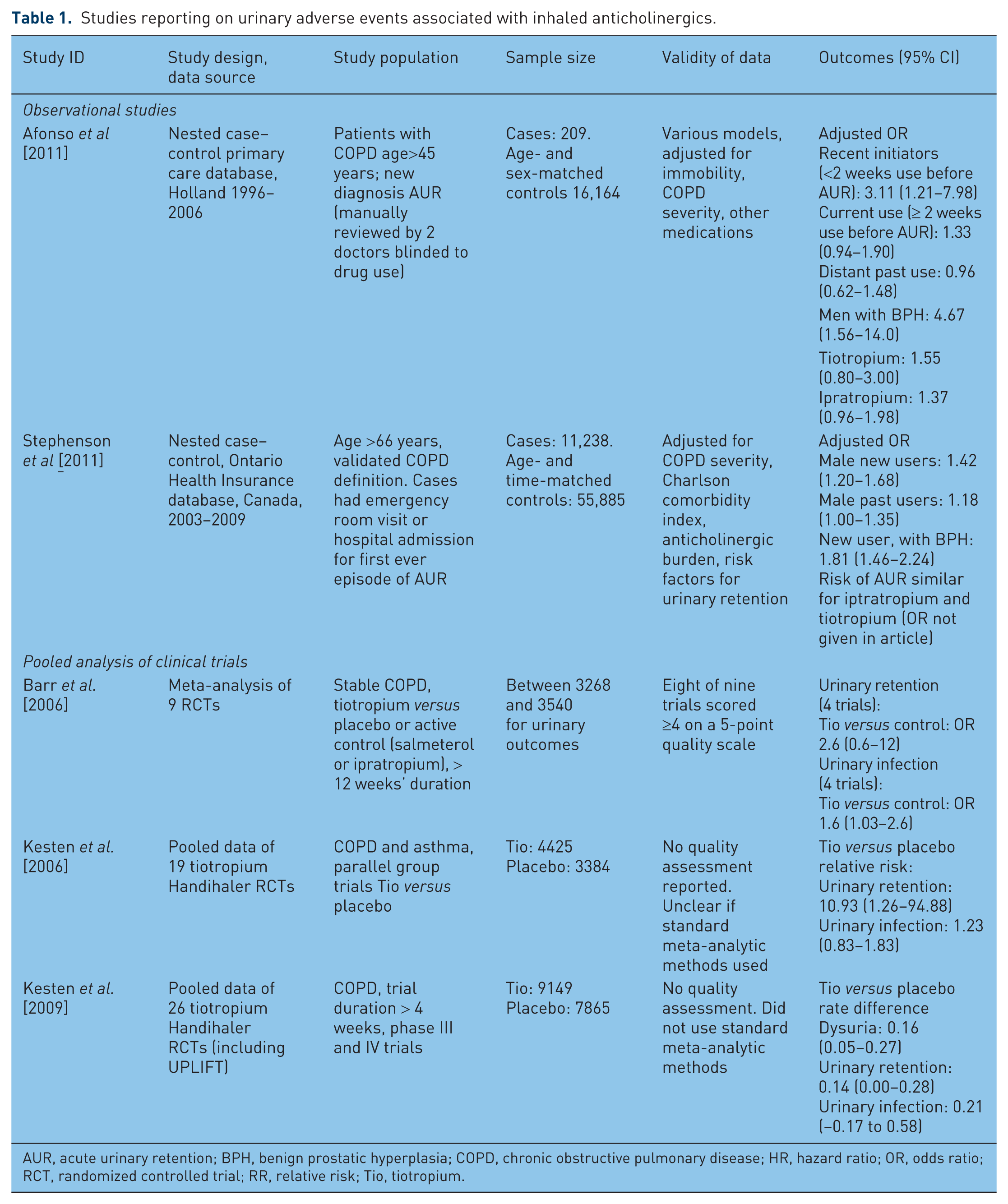
AUR, acute urinary retention; BPH, benign prostatic hyperplasia; COPD, chronic obstructive pulmonary disease; HR, hazard ratio; OR, odds ratio; RCT, randomized controlled trial; RR, relative risk; Tio, tiotropium.
Afonso and colleagues evaluated a cohort of patients with COPD in the Dutch primary care database with at least 12 months of follow-up data. They identified 209 newly diagnosed cases of acute urinary retention. Controls were matched on age, gender and index dates, and statistical analysis was performed using multivariate conditional logistic regression. This demonstrated a significant association with recent (within past 2 weeks) current users of inhaled anticholinergics, odds ratio (OR) 3.11 (95% CI 1.21–7.98) [Afonso et al. 2011]. The magnitude of association appeared to diminish when considering current users with more than 2 weeks of inhaled anticholinergic use prior to the episode of acute urinary retention. Analysis of distant past users did not show any evidence of associated risk. The ORs for tiotropium and ipratropium were similarly elevated, although neither reached statistical significance, possibly due to smaller sample size The use of nebulized anticholinergics (OR 2.92; 95% CI 1.17–7.31), or the presence of benign prostatic hyperplasia (OR 4.67; 95% CI 1.56–14.0) were associated with elevated risk of acute urinary retention [Afonso et al. 2011].
Afonso’s study has limitations [Afonso et al. 2011]. Residual confounding and influence of unmeasured confounders is always possible in a database study. There was a relative paucity of tiotropium users, with ipratropium users representing almost 80% of this dataset. Tiotropium only became available for the latter half of the study period from 1996 to 2006. Exposure to inhaled anticholinergics was based upon computer records of general practice prescriptions. It is unclear whether these prescriptions were actually dispensed and how regularly the inhalers were used by patients. Any misclassification of exposure was likely nondifferential and unlikely to influence the significant association between inhaled anticholinergics and urinary retention. Outcome misclassification is also possible. However, case ascertainment was based on diagnostic codes, and although records of all potential cases were checked manually by two medical doctors, there were inconclusive reports that had to be classified as ‘possible’. Sensitivity analysis involving both ‘definite’ and ‘possible’ cases confirmed a significant association between inhaled anticholinergics and acute urinary retention among new users and men with benign prostatic hypertrophy. Such analyses among new users are considered more reliable than analyses among prevalent users [Ray, 2003].
The Canadian study looked at 565,073 patients with COPD enrolled in the Ontario healthcare database, and identified cases with diagnostic codes signifying a first ever healthcare visit for acute urinary retention [Stephenson et al. 2011]. The authors found (using multivariable conditional logistic regression) that new users of inhaled anticholinergics were significantly more likely to have sought medical care for acute urinary retention (OR 1.42; 95% CI 1.20–1.68) [Stephenson et al. 2011]. However, as with Afonso’s study above [Afonso et al. 2011], the association of inhaled anticholinergics was less apparent when considering past users. The associated risk was further elevated in patients with benign prostatic hyperplasia, but the authors also reported that cases had higher prevalence or risk factors such as neurological disease and urinary incontinence. The authors used their estimated OR and baseline event rate in unexposed men to estimate a number needed to harm of 263 for acute urinary retention with inhaled anticholinergics over 180 days [Stephenson et al. 2011]. However, the authors did not find any evidence of increased risk of urinary retention in women exposed to inhaled anticholinergics.
This study also has some limitations. Drug exposure was assessed based on dispensing records and they could not determine how frequently patients were actually using their inhalers. It was not always possible to determine whether the episode of urinary retention was acute or whether there was some background history of chronic retention. The database does not capture inhaled anticholinergics prescribed in hospital and it does not record urinary retention that may have been treated in an ambulatory outpatient setting [Stephenson et al. 2011].
Randomized controlled trials and their meta-analyses
Barr and colleagues conducted a Cochrane review involving randomized controlled trials of tiotropium and reported a nonsignificant trend towards increased risk of urinary retention (OR 2.6; 95% CI 0.6–12), and a significant increase in the risk of urinary infections (pooled OR 1.6; 95% CI 1.03–2.6) [Barr et al. 2006]. Another pooled analysis reported a significant OR of 10.9 (95% CI 1.26–94.9) for urinary retention with tiotropium [Kesten et al. 2006]. These reviews were based predominantly on trials with relatively short durations of follow up.
A further pooled analysis was carried out by Kesten in 2009 with additional data available from a large 4-year study entitled ‘Understanding Long term Impacts on Function with Tiotropium’ (UPLIFT). This pooled analysis showed only a borderline statistically significant excess of urinary retention for tiotropium versus control with a risk difference for incidence of urinary retention of 0.14 (0.00–0.28) [Kesten et al. 2009]. In itself, the UPLIFT trial reported that urinary retention occurred more often in the tiotropium group than in the placebo group (0.34 versus 0.21 case per 100 patient years at risk; rate ratio 1.65; 95% CI 0.92–2.93), although this difference was not statistically significant [Tashkin et al. 2008].
Discussion
Although all of the links in the causal chain have not been clearly elucidated, converging lines of evidence from a variety of data sources including clinical trials and observational studies suggest a significantly increased risk of acute urinary retention associated with inhaled anticholinergics in patients with COPD. While the precise magnitude of the effect is uncertain, the risk appears to be most prominent in men with benign prostatic hyperplasia. Nevertheless, there are some inconsistencies within the dataset that merit further exploration.
First, there was some variation in the number of participants who developed urinary retention in various UPLIFT trial reports, raising doubts about the adequacy of ascertainment and completeness of reporting of such systemic adverse effects [Tashkin et al. 2008; Tashkin, 2010a, 2010b]. Initially, only one serious adverse event of urinary retention was reported in each arm in UPLIFT [Tashkin et al. 2008]. Subsequently in 2010, 15 cases of urinary retention as serious adverse events among tiotropium-treated patients compared with eight cases among placebo-treated controls were reported in UPLIFT (0.5% versus 0.27% for tiotropium versus placebo) [Tashkin, 2010a).] Further reporting of data from UPLIFT could not rule out as much as a fourfold increased risk of urinary retention, although this difference was not statistically significant (rate ratio 1.73; 95% CI 0.73–4.09) [Tashkin, 2010b].
The significantly elevated risk of urinary retention in the earlier pooled analysis [Barr et al. 2006] was negated following inclusion of data from UPLIFT. This may have stemmed from differences in the participants recruited, the interventions delivered and incomplete ascertainment of adverse events in UPLIFT. The exclusion of participants with symptomatic prostatic hyperplasia or bladder neck obstruction and moderate to severe renal impairment, the recruitment of a high proportion of short-acting anticholinergic users at baseline (approximately 50%) and during the trial (approximately 14% in both arms), and the high rate of loss to follow up of participants of approximately 40% in UPLIFT could have resulted in an inability to reliably ascertain systemic anticholinergic adverse effects. ‘Depletion of susceptibles’ is another possible explanation of the discrepant findings of adverse effects between earlier short-term trials and longer-term studies [Hammad et al. 2011]. Since the risk of urinary retention is most prominent soon after starting on anticholinergic drugs [Afonso et al. 2011; Stephenson et al. 2011; Martín-Merino et al. 2009], patients most susceptible to drug-related bladder symptoms likely dropped out early on in UPLIFT, with only the healthier patients remaining in the intervention arm for long-term follow up. A database study of oral anticholinergic drugs yielded similar findings of greatest risk of urinary retention within the first 30 days of use, with a gradual decline in risk with time [Martín-Merino et al. 2009].
Inconsistent findings have also been noted between the earlier short-term trials and the long-term trial UPLIFT for the cardiovascular adverse effects of inhaled tiotropium. Earlier randomized controlled trials have demonstrated an increased risk of cardiovascular events with inhaled anticholinergics [Singh et al. 2008]. However, the UPLIFT trial did not show a statistically significant increased risk of myocardial infarction or composite stroke with inhaled tiotropium Handihaler but only noted an excess of angina and tachyarrhythmias [Tashkin et al. 2008]. Another recent observational study also noted excess cardiovascular events with tiotropium [Jara et al. 2012], but the data from other observational studies have been inconsistent [Loke and Singh, 2012]. Inhaled tiotropium delivered via Respimat (Boehringer Ingelheim, Germany) has been shown to increase mortality in clinical trials, possibly due to an increase in cardiovascular deaths [Singh et al. 2011].
There are no recommendations on how prescribers of inhaled anticholinergics can reduce the risk of such systemic effects such as acute urinary retention in their patients. The influence of risk factors such as immobility, concomitant use of other anticholinergics, severity of prostatic disease and use of α2 blockers should be further evaluated so that prescribers can more accurately identify men who are at highest risk of acute urinary retention and perhaps consider alternative therapies for COPD. There should be additional precautions put in place for such high-risk patients that may include urological referral prior to initiating inhaled anticholinergics, or closer follow up and enquiry regarding any deterioration in urinary symptoms even before acute urinary retention occurs. The link between inhaled anticholinergics and systemic adverse effects such as urinary retention is not widely appreciated by the medical community. Educating prescribers about these potential risks and the need to consider cessation of inhaled anticholinergic therapy may be warranted.
Future research
Clinicians need reliable, accurate and comprehensive safety data for their patients with COPD. An intensive postmarketing cohort study may yield further information on the contribution of other risk factors (such as immobility or neurological disease) that predispose certain patients to acute urinary retention with inhaled anticholinergics. Such database studies should pay particular attention to vulnerable subgroups at the highest risk of systemic anticholinergic effects, such as older men with preexisting renal failure who have been excluded from randomized controlled trials [Singh and Loke, 2012]. Such studies should determine the prognostic significance of an episode of acute urinary retention with inhaled anticholinergics, including its impact on mortality. The newer long-acting muscarinic agents such as aclidinium bromide appear to have a better urinary safety profile than tiotropium in small studies [Gavaldà et al. 2012]. However, these findings need confirmation in larger trials of adequate statistical power. Such trials should also collect data on adverse effects from participants who withdraw from trials.
Conclusion
Despite the absence of definitive evidence, certain pragmatic recommendations can be made. Clinicians should inform patients with COPD about the risk of acute urinary retention associated with inhaled anticholinergics and be alert for any symptoms that emerge within the first 30 days of initiating inhaled anticholinergic use. The systemic risks of inhaled anticholinergics are particularly concerning in vulnerable subgroups at the highest risk of systemic anticholinergic effects, such as older men with benign prostatic hyperplasia. The specific risk of acute urinary retention should be seen in the context of the overall benefit:harm balance, and the efficacy and risks of alternative agents, including pneumonia [Singh et al. 2009; Singh and Loke, 2010] and fractures [Loke et al. 2011] with inhaled corticosteroids. Although no anticholinergic inhaler has been shown to affect the progressive decline in lung function or reduce mortality in COPD, there is evidence that inhalers reduce exacerbations, hospitalizations and improve quality of life, which are all important outcomes. To determine the optimal choice of therapy, clinicians and patients should carefully weigh the possibility of urinary retention and other adverse effects against their symptomatic benefits after eliciting patient preferences for various patient-oriented outcomes in a shared decision-making context.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
There are no potential conflicts of interest relevant to the article.
