Abstract
Cardiovascular diseases (CVDs) represent a huge health burden for older patients with diabetes. Acetylsalicylic acid (ASA) has long been used as a cardioprotective agent in primary and secondary prevention of CVD. However, there are important issues regarding the benefits and risks of ASA therapy in primary prevention of CVDs, for the older group in general and for individuals of all ages with diabetes. In this review, we summarize the benefits and risks related to ASA therapy by outlining the evidence for older patients and for patients with diabetes. There appear to be significant gaps in knowledge. The balance of benefits and risks is not well defined but ASA treatment seems to be unfavorable in many older patients.
Introduction
Cardiovascular diseases (CVDs) represent an important health burden for older people. The proportion of people aged 60–79 years who have CVD is estimated at around 72% in the USA, reaching at least 80% of those aged 80 years and over [Roger et al. 2011]. While CVD-associated death rate has decreased over the years, it remains the highest among those aged 65 years and over [Public Health Agency of Canada, 2009], with cardiac diseases being the leading cause of death in this age group [Roger et al. 2011]. Considering the ageing of the population and the increases in prevalence of obesity and diabetes, the number of deaths due to CVD in the future might increase [Public Health Agency of Canada, 2009]. Moreover, the consequences of cardiovascular events are often dramatic for older patients because they can reduce global functioning, impair daily activities and reduce quality of life [Yazdanyar and Newman, 2009]. Therefore, strategies to decrease cardiovascular risk are fundamental, especially for older people who are at higher CVD risk, such as those with diabetes.
Implementing pharmacological strategies to decrease cardiovascular risk remains challenging for older patients because they present many physiological changes that make them a distinct group. For example, reduced renal clearance and modification in body fat composition may alter the pharmacokinetics of drugs. Moreover, pathological changes associated with ageing may alter the pharmacodynamics of drugs. For example, undernutrition may reduce plasmatic albumin, which in turn may increase the effect of certain drugs. The effect of acetylsalicylic acid (ASA) could for example be increased among older patients with undernutrition.
ASA has been used for many decades as a cardioprotective agent, but recent evidence has questioned its veritable place in pharmacotherapy. ASA constitutes the most frequently used antiplatelet drug, in part because of its low cost and apparent safety. It has been used as a preventive agent for myocardial infarction (MI), ischemic stroke and vascular mortality for a wide range of patients, either in primary or secondary prevention [Patrono et al. 2005]. While it has been largely recommended for individuals with diabetes in the last decade [Canadian Diabetes Association, 2003; American Diabetes Association, 2007], the therapeutic role of ASA has since been reviewed and criticized in primary prevention of CVD [Canadian Diabetes Association, 2008; Bell et al. 2011]. Table 1 illustrates the variations in recommendations from different national guidelines. The place of ASA in secondary prevention is generally not questioned in patients with diabetes [Canadian Diabetes Association, 2008; Pignone et al. 2010], although the evidence in this particular context is still scarce [Sirois et al. 2008].
National guidelines recommendations for use of acetylsalicylic acid (ASA) in primary prevention of cardiovascular events for people with type 2 diabetes

CV, cardiovascular; CVD, cardiovascular disease; NICE, National Institute for Health and Clinical Excellence; NSAID, nonsteroidal anti-inflammatory drug.
Apart from its therapeutic effect, ASA also brings along adverse effects that may restrain its use, especially in older people. In fact, the risk of bleeding complications increases with age [Patrono et al. 2004]. Moreover, the presence of clinical conditions such as gastric ulcer, cancer, polymedication or drug–drug interactions that are common in the older population also raises bleeding risks [Mahé et al. 2003; Russolillo et al. 2012].
Considering its potential benefits in reducing cardiovascular risk and its potential serious side effects, it is imperative to review the risk/benefits balance in using ASA in older people with diabetes. The aim of this perspective paper is therefore to overview the evidence of ASA use in older people with diabetes for primary CVD prevention. We proceeded by evaluating the literature for older patients and for patients with diabetes. Since there is no consensus on the age cutoff for the definition of older patients, we did not attempt to set a definition prior to our review. However, we assumed that we would mostly retrieve data for patients aged >65 years who are usually arbitrarily considered as older patients.
Primary prevention of cardiovascular diseases in older patients
Randomized clinical trials
There is little evidence from the literature of the benefits of ASA in primary prevention of CVD for older patients with or without diabetes. To our knowledge, only two randomized trials have specifically targeted older people. First, the PACE trial (Prevention with low-dose Aspirin of Cardiovascular disease in the Elderly) was a randomized, double-blind, placebo-controlled trial involving people aged 70 years and older [Silagy et al. 1991, 1994]. ASA dose was 100 mg daily. Unfortunately, to our knowledge, only the results obtained from the pilot phase of the study were published. The number of events in this pilot phase was lower than expected: among the 400 people who were followed up for a 12-month period, there were two fatal cardiovascular events, three nonfatal coronary events and eight nonfatal cerebrovascular events. Therefore, it was not possible to draw conclusions about the potential of ASA to reduce CVD risk. Nonetheless, the occurrence of side effects dictated caution: participants receiving ASA experienced a statistically significant decrease in mean hemoglobin levels compared with those receiving placebo [Silagy et al. 1993]. Moreover, clinically evident gastrointestinal bleeding occurred in six patients taking ASA (3%) whereas no such side effect occurred in patients taking placebo [Silagy et al. 1993].
A second study, the ASPREE trial (Aspirin in Reducing Events in the Elderly), is recruiting participants aged 70 years and above in Australia and the USA [Nelson et al. 2008]. Nineteen thousand participants are expected to be randomized. The study will examine the balance of benefits of low-dose (100 mg) ASA (in the prevention of CVD, cancer and dementia) and bleeding risks compared with placebo. Results are yet to be published. Therefore, to date, it is not possible to derive evidence-based conclusions from randomized controlled trials involving older patients only.
Subgroup analyses of randomized clinical trials
Other clinical trials that were performed to study the impact of ASA in primary prevention of CVD have involved, among others, participants aged 65 years and over. Table 2 lists the trials and the number of older people who were included, when specified. The information from the above trials regarding the effect of ASA in the older subgroups remains limited. In four [Peto et al. 1988; Medical Research Council’s General Practice Framework, 1998; Hansson et al. 1998; Collaborative Group of the Primary Prevention Project, 2001] out of nine studies, no analysis was performed to study the effect among older people.
Randomized clinical trials of acetylsalicylic acid (ASA) in primary prevention – evidence for the older subgroups.
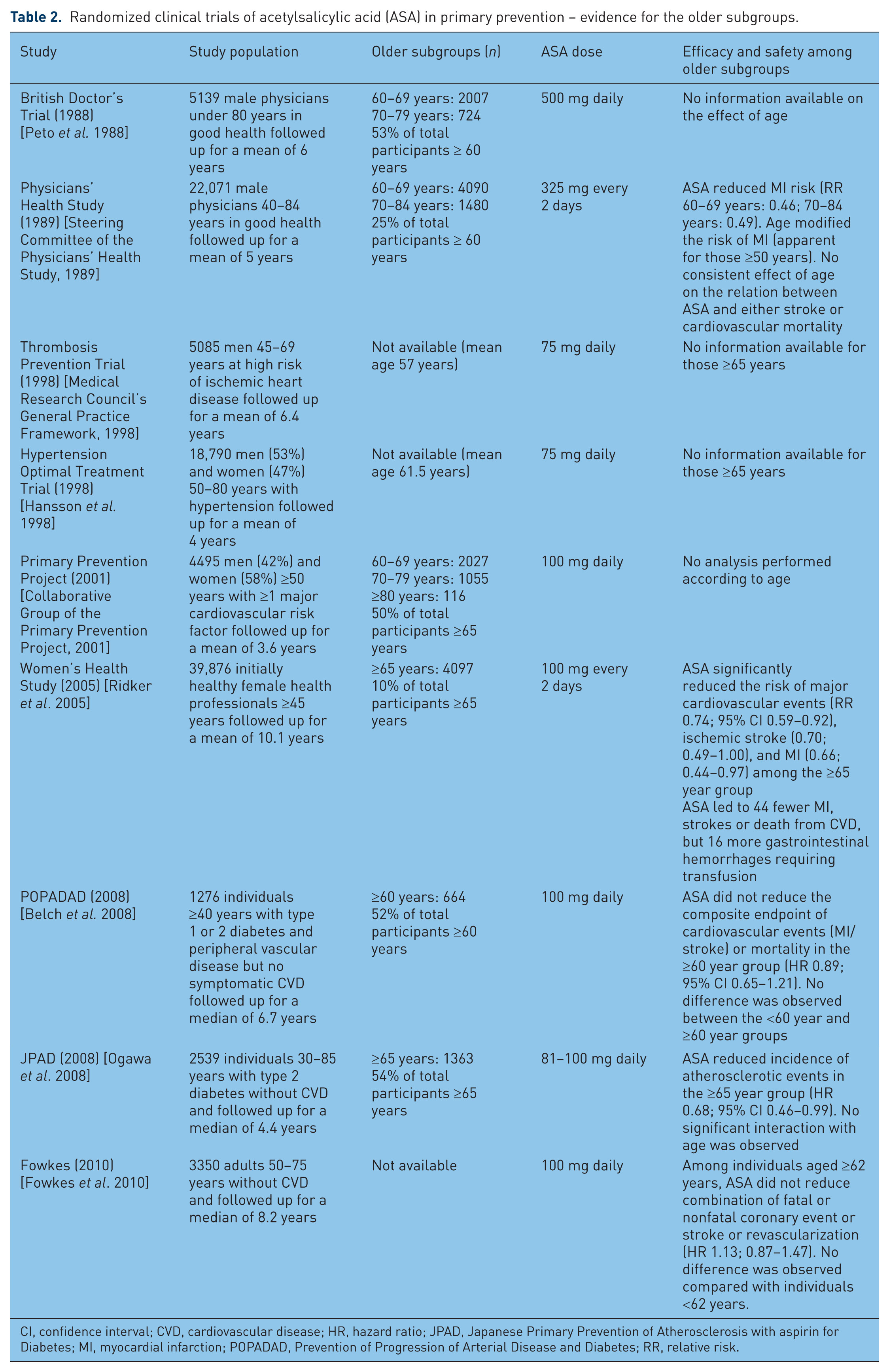
CI, confidence interval; CVD, cardiovascular disease; HR, hazard ratio; JPAD, Japanese Primary Prevention of Atherosclerosis with aspirin for Diabetes; MI, myocardial infarction; POPADAD, Prevention of Progression of Arterial Disease and Diabetes; RR, relative risk.
Among the remaining five studies that addressed differences of effect according to age groups [Steering Committee of the Physicians’ Health Study, 1989; Ridker et al. 2005; Belch et al. 2008; Ogawa et al. 2008; Fowkes et al. 2010] three [Steering Committee of the Physicians’ Health Study, 1989; Ridker et al. 2005; Ogawa et al. 2008] demonstrated that older patients benefited from ASA therapy in primary prevention of CVD. The first to be published was the Physician Health Study (PHS) [Steering Committtee of the Physicians’ Health Study, 1989]. It was a randomized, double-blind, placebo-controlled trial performed among 22,071 healthy men to determine whether ASA (325 mg every other day) would decrease cardiovascular mortality, and whether β carotene would decrease cancer incidence. It involved physicians 40–84 years of age in the USA between 1982 and 1988. The ASA portion of the trial was terminated earlier than expected because of statistically significant reduction in the risk of MI [relative risk (RR) 0.56; 95% confidence interval (CI) 0.45–0.70] in the ASA group. Further analyses demonstrated that age modified the risk of MI, with ASA reducing the MI risk only for people aged 50 years and older. Among older patients, the RR of MI for people aged 60–69 years treated with ASA was 0.46, and 0.49 for those aged 70–84 years. There was no consistent effect of age on the relationship between ASA and either stroke or cardiovascular mortality.
While the PHS trial involved male physicians, the second trial that showed a positive effect of ASA on CVD prevention involved women: the Women’s Health Study [Ridker et al. 2005]. This randomized controlled trial was designed to evaluate the balance of risks and benefits of low-dose ASA (100 mg every other day) and vitamin E (600 IU every other day) in the primary prevention of CVD and cancer. A total of 39,687 initially healthy women aged 45 years and older were followed up for 10 years between 1992 and 2004. Globally, ASA reduced the risk of ischemic stroke in the ASA group (RR 0.76; 95% CI 0.63–0.93), but did not affect the risk of MI or death from cardiovascular causes. Nonetheless, there was evidence that age modified the effect of ASA on major cardiovascular events and MI (p value for interaction 0.05 and 0.03, respectively). Specifically, in the subgroup of women aged 65 years and older at study entry, the risk of major cardiovascular events was reduced among those who received ASA compared with those who received the placebo (RR 0.74; 95% CI 0.59–0.92). Similarly, the risk of ischemic stroke was lower in the group assigned to ASA treatment (RR 0.70; 95% CI 0.49–1.00), and so was the risk of MI (RR 0.66; 95% CI 0.44–0.97). Further analyses showed that menopausal status, the use or non-use of hormone replacement therapy after menopause, or global cardiovascular risk status did not modify the effect of ASA. Interestingly, the authors mentioned that in this group of 4097 women aged 65 years and older, the use of ASA led to 44 fewer MI, strokes or deaths from cardiovascular causes (p = 0.008), but it caused 16 more gastrointestinal hemorrhages requiring transfusion (p = 0.05). It thus appears that the balance of benefits and risks should be emphasized in treatment decision.
The last trial that showed a positive effect of ASA for an older subgroup in primary prevention of CVD is the JPAD study (Japanese Primary Prevention of Atherosclerosis with aspirin for Diabetes) [Ogawa et al. 2008]. This study aimed to examine the efficacy of low-dose ASA for the primary prevention of atherosclerotic events in patients with type 2 diabetes. It was a randomized, open-label trial of ASA 81–100 mg per day performed in Japan. A total of 2539 individuals aged 30–85 years were followed up between 2002 and 2008. The risk of CVD did not differ between the group receiving ASA and the one that did not [hazard ratio (HR) 0.80; 95% CI 0.58–1.10]. However, among individuals aged 65 years and older, the incidence of atherosclerotic events was significantly lower in the group of 719 patients who received ASA compared with the group of 644 patients who did not (HR 0.68; 95% CI 0.46–0.99). Such difference was not observed for individuals younger than 65 years (HR 1.0; 95% CI 0.57–1.70). Yet statistical testing did not show significant interaction with age. Finally, the composite of hemorrhagic stroke and major gastrointestinal bleeding was not significantly different between the two groups. However, the authors did not report the bleeding risk among older individuals.
Meta-analyses
Meta-analyses offer the opportunity to get a global vision of the effect of ASA observed in randomized controlled trials. The Antithrombotic Trialists’ (ATT) Collaboration published their most recent meta-analysis on the role of ASA in the primary and secondary prevention of vascular disease in 2009 [ATT Collaboration et al. 2009]. It included six primary prevention trials [Peto et al. 1988; Medical Research Council’s General Practice Framework, 1998; Hansson et al. 1998; Collaborative Group of the Primary Prevention Project, 2001; Steering Committtee of the Physicians’ Health Study, 1989; Ridker et al. 2005] which totaled 95,000 people at low average risk (660,000 person-years) to study serious vascular events and major bleeds. According to the authors, the proportional reductions in serious vascular events did not depend significantly on age in their analysis. Moreover, sex, smoking history, blood pressure, total cholesterol, body mass index, history of diabetes and predicted risk of coronary heart disease did not influence the proportional reductions in serious vascular events. In fact, there was no significant trend in the proportional effects of ASA in individuals at all levels of estimated coronary heart disease risks. The authors concluded that, the proportional risk reductions being similar in different subgroups, the absolute risk reduction brought by ASA use would depend chiefly on the patient’s own basal CVD risk. Of note, the authors also mentioned that each decade of advancing age was associated with about a twofold increased risk of major coronary events (RR 1.84; 95% CI 1.74–1.95); and so was diabetes (2.43; 2.27–2.65). Moreover, these risks factors were also associated with hemorrhagic events [age (per decade): 2.15 (1.93–2.39); diabetes: 1.55 (1.13–2.14)].
In conclusion, all cardiovascular risks being equivalent it seems that ASA would not be more efficacious in the older patient than it would be in a younger patient. The corresponding bleeding risk is however not well defined.
Observational data/virtual modeling
We retrieved two observational studies in which the association between ASA use and CVD among older patients was assessed. In one study conducted in 1981, Paganini-Hill and colleagues used a postal questionnaire survey sent to a Californian retirement community to elicit details on ASA use [Paganini-Hill et al. 1989]. Among the 13,987 people (61% of a total of 22,781) who responded to the questionnaire, 2826 were excluded because they reported a history of CVD. The cohort of participants (median age 73 years) was then followed up for 6.5 years using hospital discharge summaries and death certificates. The incidence of ischemic heart disease was raised among daily users of ASA compared with nonusers for men (RR 1.9; 1.1–3.1) and women (1.7; 1.1–2.7). Likewise, the risk for all CVD was raised among daily users of ASA compared with nonusers (men: 1.5; 1.2–1.8; women: 1.4; 1.2–1.7). Nonetheless, there was a nonsignificant reduction in risk of MI of 36% and 27%, respectively, among those who took ASA daily and those who took it less often. There was no association between ASA use and gastrointestinal bleeding. Of note, the authors mentioned that people at high risk of gastrointestinal problems most probably avoided taking ASA, thereby reducing the observed risk of bleeding. Indeed, in a resurvey of a sample of their cohort who took analgesics daily, they found that a quarter of the 190 respondents avoided analgesics with ASA because of a history of gastrointestinal problems. Thus the results of this study could not confirm any benefits for ASA therapy in primary prevention of CVD in older patients living in the community.
In the second study, Kronmal and colleagues evaluated the risk of incident stroke in an observational cohort of 5011 older people (mean age 72 years) followed for a mean of 4.2 years [Kronmal et al. 1998]. A total of 3310 participants did not have CVD at study entry. Among those, 15% reported frequent ASA use whereas 12% used ASA infrequently at study entry. For women without CVD, frequent ASA use at study entry was associated with an increase in ischemic stroke risk compared with nonusers (2.11; 1.13–3.92), whereas infrequent use was associated with a nonsignificant increase (1.87; 0.92–3.83). For men without CVD, the estimates did not reach statistical significance for any type of ASA users (frequent users: 1.28; 0.65–2.52; infrequent users: 1.00; 0.44–2.27). ASA use was reassessed every year in the study. Most participants who were nonusers or frequent users in the prior year stayed in the same category in the following year. However, there was frequent switching for the infrequent users category. Using these data, time-dependent analyses were conducted to take into account the variation in ASA exposure in time. The results were slightly different. In fact, there remained no statistically significant association between ASA use and stroke for persistent frequent users among women (RR 1.67; 0.75–3.68) and men (RR 1.25; 0.51–3.06). Furthermore, the risk was not higher for sporadic and/or infrequent users compared with nonusers (women 1.04; 0.57–1.88; men: 1.32; 0.73–2.41). Hence, time-dependant assessment of ASA use resulted in a neutral effect of ASA on ischemic strokes. Bleeding risks were not evaluated in the study.
Finally, Nelson and colleagues conducted an epidemiological modeling in a hypothetical population of individuals aged 70–74 years to study the effect of low-dose (75–150 mg) ASA in the primary prevention of CVD [Nelson et al. 2005]. For 10,000 men receiving life-long treatment with ASA, 389 MIs (95% CI 213–581) would be prevented at the expense of 499 severe gastrointestinal hemorrhages (95% CI 266–740). Similarly, the 321 coronary heart diseases (95% CI 170–484) prevented in women would be offset by the 572 major gastrointestinal hemorrhages (95% CI 308–849). The authors specify that the findings are tempered by the wide confidence intervals: the final outcome could either be beneficial or adverse. Nonetheless, their results warrant caution for the use of routine ASA, which could cause more hemorrhages than MI or stroke preventions.
Data for older patients: conclusions
According to the primary prevention trials, there are very limited data that support the benefits of ASA for CVD prevention among older patients, and none pertain to individuals aged 80 years and over. Although the risk of MI was reduced in the PHS study for men, this study was performed three decades ago, and therefore it may be difficult to conclude ASA would have similar effects considering today’s use of other evidence-based pharmacotherapy. For women, the Women’s Health Study provided interesting results for prevention of different outcomes. Nonetheless, the balance of 44 events prevented with 16 hemorrhages requiring transfusions warrants caution. Finally, the recent JPAD study involved a rather small number of patients and provided limited data for efficacy, but had the advantage of including older patients with diabetes. While this study alone cannot resolve the dilemma of ASA benefit, it suggests that, if ASA does reduce CVD risk, its benefits remain relatively small. In fact, all those positive findings must be balanced with the results of five other trials in which benefits were not observed. It is also difficult to interpret the above findings without considering the bleeding risks associated with treatment. Unfortunately, there are very few details on the bleeding risks according to the age group, although bleeding risk was almost uniformly augmented in studies on the general population. Hence, without proper comparison, it is difficult to establish the balance between benefits and risks.
Data from observational studies are also very limited and cannot help establish whether ASA would constitute an acceptable therapy in a population-based perspective. The difficulty of defining ASA exposure in such studies poses a serious threat on the ability of evaluating ASA effect. The observed increased risk in the mentioned studies may also be explained by an indication bias: if people at higher risk use ASA in a greater proportion than those at lower risk, there would be a spurious association between ASA use and CVD. Once again, the evidence from observational studies is not high enough to either recommend or reject ASA use. Moreover, the balance of risk/benefits is most probably very thin, as suggested by the modeling performed by Nelson et al. [Nelson et al. 2005].
Along with age, diabetes represents a strong risk factor for CVD. Since patients with diabetes are at high absolute risk of CVD, any benefit of primary prevention with ASA might be most easily demonstrated, especially in older patients. The next section explores the evidence among people with diabetes.
Acetylsalicylic acid for people with diabetes
Recent years have witnessed much turmoil regarding the role played by ASA in primary prevention of people with diabetes. At first, the 1998 landmark study by Haffner and colleagues revolutionized the concept of diabetes treatment, suggesting diabetes could be considered as a CVD equivalent [Haffner et al. 1998]. As a consequence, the use of ASA was wholeheartedly recommended for cardiovascular prevention. In 2007 for example, the US Diabetes Association guidelines stated that, in primary prevention, all individuals who are at increased cardiovascular risk, including those over 40 years of age or who have additional risk factors, should receive ASA therapy in primary prevention (grade A level of evidence) [American Diabetes Association, 2007]. Yet considering diabetes as a CVD was questioned [Bulugahapitiya et al. 2009]. In 2006, Howard and colleagues suggested that only people with diabetes who also have numerous cardiovascular risk factors share the same cardiovascular risk as those who have already had a cardiovascular event [Howard et al. 2006]. The new perspective may result in different treatment strategy in primary prevention of CVD. Therefore, we present and discuss recent trials and literature reviews that have revisited ASA therapy for cardiovascular prevention for individuals with diabetes. Observational data are also evaluated to help construct a thorough picture of the effect of ASA in this population.
Randomized controlled trials
There is very little evidence regarding the benefits of ASA for individuals with diabetes in the primary prevention setting. Table 3 lists seven studies that have included individuals with diabetes [Peto et al. 1988; Hansson et al. 1998; Steering Committee of the Physicians’ Health Study, 1989; Ridker et al. 2005; Belch et al. 2008; Ogawa et al. 2008; Sacco et al. 2003]. Apart from the two recent studies conducted among individuals with diabetes only [Prevention of Progression of Arterial Disease and Diabetes (POPADAD) [Belch et al. 2008] and JPAD [Ogawa, 2008]), the number of individuals with diabetes varied from 2% [Peto et al. 1988] to 22% [Sacco et al. 2003]. Only two studies reported statistically significant results: PHS [Steering Committee of the Physicians’ Health Study, 1989] and WHS [Ridker et al. 2005]. In PHS, it seems that people with and without diabetes benefited from ASA use in primary prevention (diabetes did not modify ASA effect, p = 0.22). ASA use was associated with a 60% reduction in MI. In the WHS, ASA reduced the risk of stroke and ischemic stroke, but had no impact on major cardiovascular events and MI. In the Hypertension Optimal Treatment (HOT) study [Hansson et al. 1998], the authors stated that ASA would prevent 2.5 MIs per 1000 patient-years in patients with diabetes, but did not reveal whether or not this result was statistically significant. No significant results were obtained for the Primary Prevention Project [Sacco et al. 2003], POPADAD [Belch et al. 2008] and JPAD [Ogawa et al. 2008]. The available information does not provide clear evidence of the benefits of ASA therapy among people with diabetes. New studies are underway (A Study of Cardiovascular Events in Diabetes [ASCEND], Aspirin and simvastatin Combination for Cardiovascular Events Prevention Trial in Diabetes [ACCEPT-D]) and will help position the usefulness of ASA use in the context of the use of other evidence-based recommended treatments, such as statins. For the time being, meta-analyses might provide an additional look at the evidence.
Randomized clinical trials of acetylsalicylic acid (ASA) in primary prevention – evidence for individuals with diabetes.
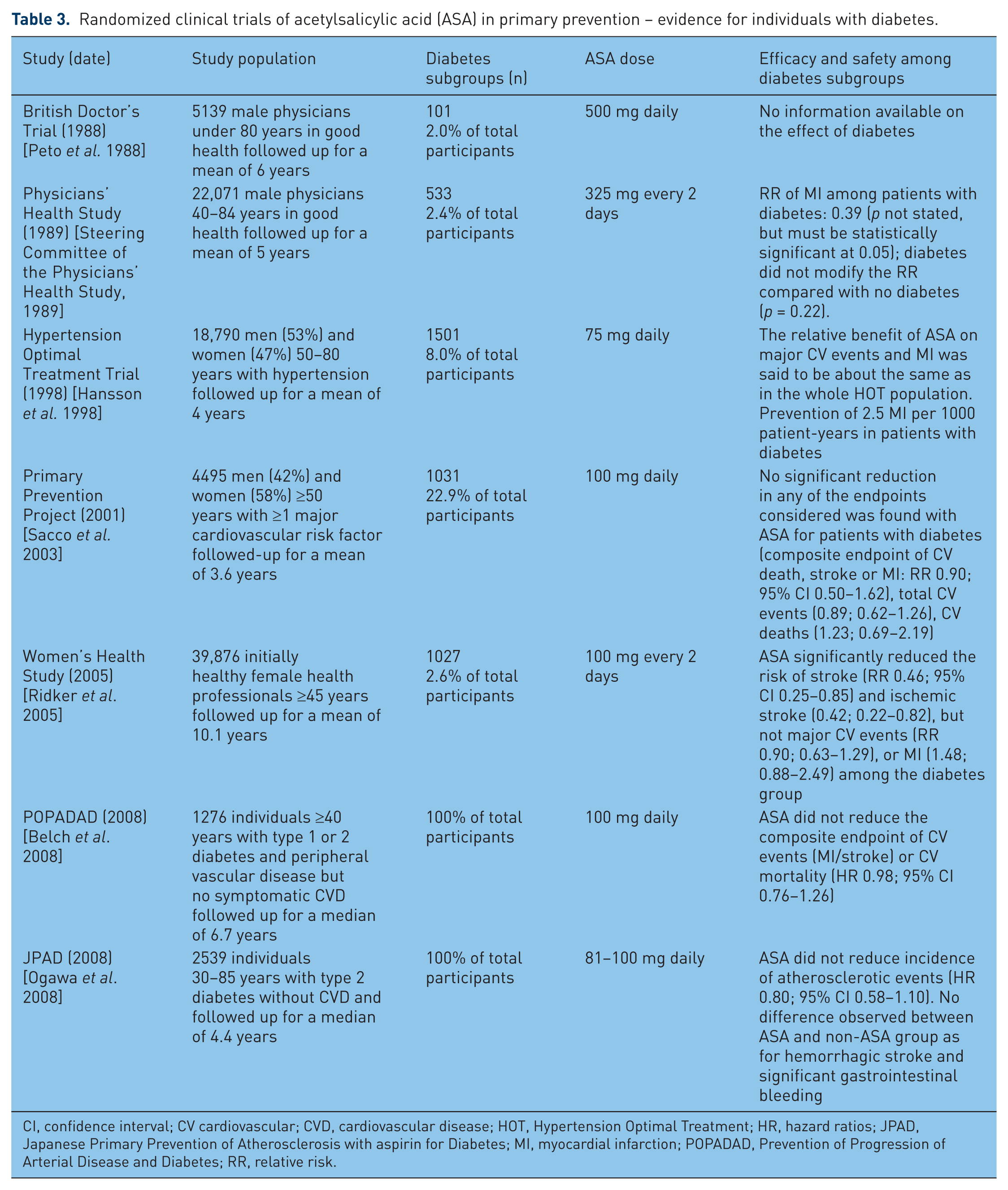
CI, confidence interval; CV cardiovascular; CVD, cardiovascular disease; HOT, Hypertension Optimal Treatment; HR, hazard ratios; JPAD, Japanese Primary Prevention of Atherosclerosis with aspirin for Diabetes; MI, myocardial infarction; POPADAD, Prevention of Progression of Arterial Disease and Diabetes; RR, relative risk.
Meta-analyses
Seven meta-analyses have evaluated the benefits of ASA in primary prevention of CVD among people with diabetes [Pignone et al. 2010; ATT Collaboration et al. 2009; Calvin et al. 2009; De Berardis et al. 2009; Zhang et al. 2010; Butalia et al. 2011; Stavrakis et al. 2011] (Table 4). Risk reductions associated with ASA use were 10–15% for coronary events and stroke, but none of the results of these publication reached statistical significance. Pignone and Williams suggest that low-dose ASA (75–162 mg) would be reasonable for people with diabetes for whom the 10-year cardiovascular risk would be over 10%, in the absence of increased bleeding risk [Pignone and Williams, 2010]. Bleeding risk assessment should be based on a history of previous gastrointestinal bleeding or peptic ulcer disease, or concomitant use of other drugs that increase bleeding risk, such as nonsteroidal anti-inflammatory drugs or anticoagulants [Pignone et al. 2010]. No statistically significant increase in the risk of bleeding was observed in the meta-analyses. However, the risk of bleeding might be higher among older people living in community [Pignone et al. 2010].
Summary of meta-analyses performed on primary prevention of cardiovascular disease (CVD) among patients with diabetes.
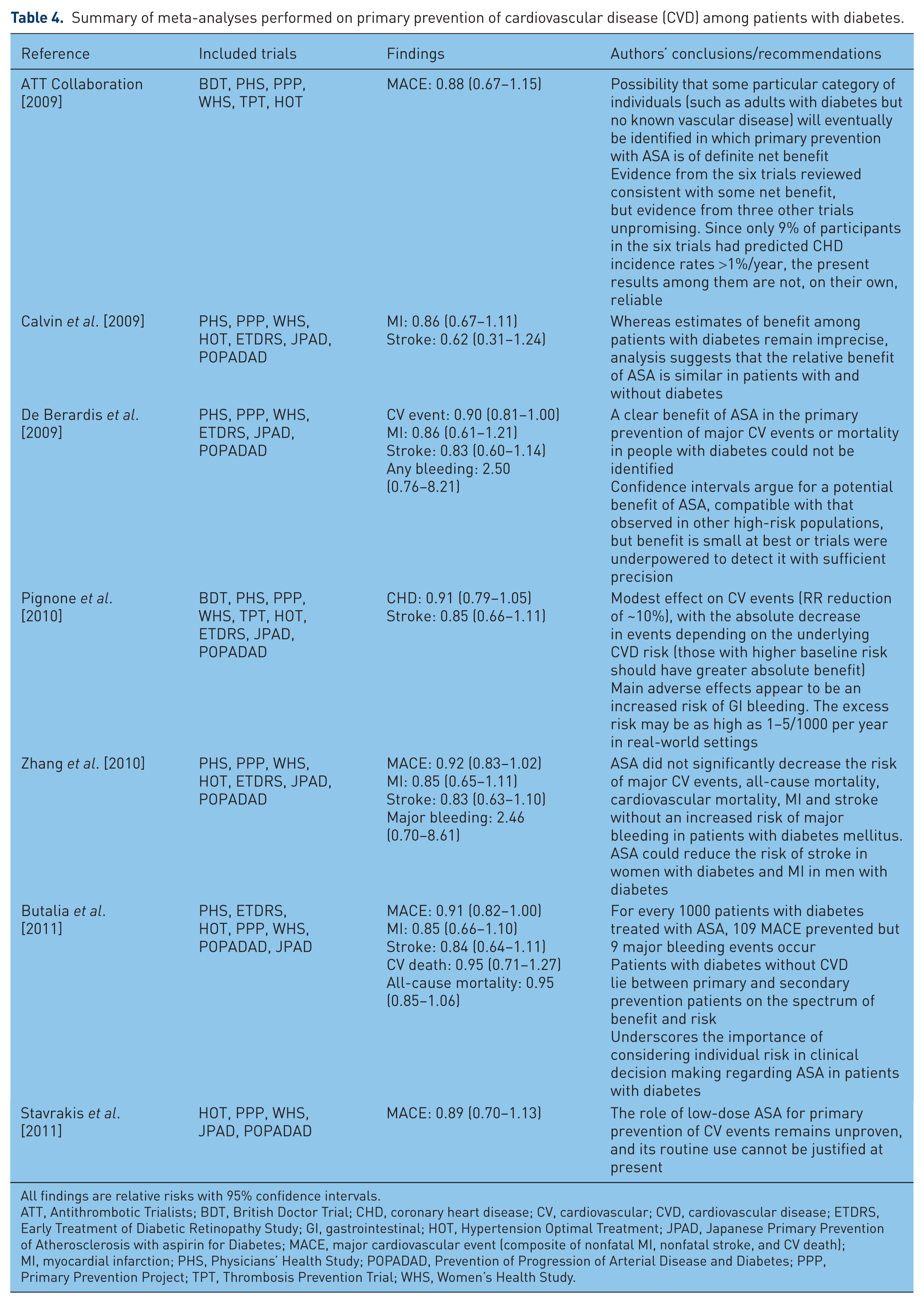
All findings are relative risks with 95% confidence intervals.
ATT, Antithrombotic Trialists; BDT, British Doctor Trial; CHD, coronary heart disease; CV, cardiovascular; CVD, cardiovascular disease; ETDRS, Early Treatment of Diabetic Retinopathy Study; GI, gastrointestinal; HOT, Hypertension Optimal Treatment; JPAD, Japanese Primary Prevention of Atherosclerosis with aspirin for Diabetes; MACE, major cardiovascular event (composite of nonfatal MI, nonfatal stroke, and CV death); MI, myocardial infarction; PHS, Physicians’ Health Study; POPADAD, Prevention of Progression of Arterial Disease and Diabetes; PPP, Primary Prevention Project; TPT, Thrombosis Prevention Trial; WHS, Women’s Health Study.
Observational data
There are three observational trials that have evaluated the impact of ASA use on CVD in patients with diabetes [Ong et al. 2010; Welin et al. 2009; Leung et al. 2009], with contradictory results. From the Fremantle Diabetes Study, 651 patients with diabetes but free of CVD at entry in the study were followed from inclusion (1993–1996) until death or June 2007 [Ong et al. 2010]. ASA use was assessed at cohort entry. In this study, regular ASA use was associated with reduced CVD mortality (HR 0.30; 95% CI 0.09–0.95). However, the impressive reduction in mortality risk is puzzling, and could be attributable to several biases that may have affected the results (e.g. misclassification due to changes in therapy during follow up which were not accounted for, differences in protective drug exposures between those who were treated with ASA and those who were not).
In the second study, Welin and colleagues evaluated the effect of ASA on mortality and on serious bleeding (hospitalization with a diagnosis of bleeding event) in a group of 58,465 patients with type 1 and type 2 diabetes [Welin et al. 2009]. These authors used data from administrative databases between 2005 and 2006. Among patients for whom there was no formal indication for ASA other than primary prevention of CVD, ASA use was associated with a 17% increase in mortality (95% CI 1–36) at 50 years of age which increased to 29% (16–43) at 85 years. The authors estimated that in the group of patients taking ASA without a specific indication, there were 107 more deaths than among those not taking ASA. Moreover, patients receiving ASA without indication were at a higher risk of serious bleeding compared with those not receiving ASA, with a RR of 1.46 (95% CI 1.22–1.75). These results contrast with those observed in the group of patients for whom ASA was indicated, since ASA use was associated with a decrease in mortality (164 deaths prevented) and serious bleeding (RR 0.82; 95% CI 0.69–0.98). However, this study was limited by a very short follow up (1 year) and by the fact that, once ASA was dispensed, it was assumed that the patient continued on ASA until the end of the follow-up period or until death.
Finally, Leung and colleagues conducted an observational study involving 6454 Chinese patients with type 2 diabetes [Leung et al. 2009]. A total of 5731 patients constituted the primary prevention cohort. ASA use was defined dichotomously as use or nonuse. In the primary prevention cohort, ASA use was associated with an increased incidence of the composite endpoint of nonfatal MI, stroke or vascular death (HR 2.07; 95% CI 1.66–2.59).
These surprising results raise concerns that indication bias or other confounding bias may be present. The issue of ASA benefits in patients with diabetes has not been resolved in these studies, and there is still a need to study the benefits of ASA in an observational setting. Indeed, clinical trials are poorly representative of real-life settings and often do not include older patients. Appropriate assessment of ASA use is nonetheless paramount in observational studies, and future studies should focus on this particular issue to ensure reliable evaluations.
Acetylsalicylic acid for primary prevention in older patients with diabetes: yes or no?
Nonfatal cardiovascular events may result in devastating consequences for older patients, and they are most likely to produce greater disability than nonfatal bleeding. However, the small absolute reductions of cardiovascular events that were observed in randomized clinical trials of primary prevention have to be balanced with the small, but rather uniformly reported increased risk of ASA-induced serious bleeding. Such serious bleeding may have major consequences for older patients. Thus, one should not consider a priori ASA use as safe since, as with any other drug, it could potentially produce severe side effects. The risk of bleeding with ASA, for example, was comparable to that for warfarin among patients with atrial fibrillation [Hansen et al. 2010].
While ASA is not costly, is widely available and well accepted by patients, there is no evidence to show that adding ASA to other evidence-based pharmacotherapy will translate into additional primary CVD protection. Hence, according to the available evidence, ASA cannot be recommended as a population-based prevention strategy for older patients with diabetes.
Conclusion
Despite the higher absolute risk of CVD in older patients with diabetes, there is no robust evidence that the use of ASA leads to a favorable benefits-to-risk balance. The clinical benefits of ASA are yet to be proven in the older population, especially in relation to the higher probability of serious bleeding and anemia. As the aging population will increase the burden of CVD, future studies will have an important role in clarifying this issue. ASA is an affordable, well known drug, but its efficiency must be proven before population-based recommendations can be made for its use in primary prevention of CVD in older patients, including those with diabetes.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. CS is a recipient of the Fonds de la recherche en santé du Québec scholarship.
Conflict of interest statement
The authors declare that there is no conflict of interest.
