Abstract
Objective
Rheumatoid arthritis (RA) is a chronic systemic autoimmune disease characterized by inflammatory arthritis and extra-articular involvement. Patients with RA are at increased risk of developing cardiovascular diseases. This retrospective cohort study aimed to determine whether acetylsalicylic acid (aspirin [ASA]) protects against major ischemic vascular events in patients diagnosed with RA.
Methods
Patients with RA were recruited from a population of 118, 500 participants who were enrolled into the Kailuan Cohort Study between 2014 and 2016. Information regarding demographic characteristics, anthropometric measures, and medical histories were documented. Patients with RA who underwent ASA therapy were propensity score matched at a ratio of 1:4 with patients with RA who did not receive ASA. The risk of ischemic vascular events (myocardial infarction and ischemic stroke) was examined. The relationship between ASA therapy and major ischemic vascular events was analyzed by Cox proportional hazards regression.
Results
A total of 939 patients with RA were included (189 who received ASA and 750 who did not). During the 3.3-year follow-up, six and 35 major ischemic vascular events occurred in the ASA and non-ASA groups, respectively, with no significant between-group difference in the cumulative incidence of such events. Cox proportional hazards regression analysis showed that ASA therapy was not associated with major vascular ischemic events and its subtypes.
Conclusions
ASA did not confer protection against major ischemic vascular events in patients diagnosed with RA.
Introduction
Rheumatoid arthritis (RA) is associated with an increased risk of morbidity and mortality, largely due to enhancement of atherosclerotic disease. 1 As a common systemic autoimmune disease, RA is characterized by inflammation and extra-articular involvement, and affects millions of individuals worldwide, corresponding to approximately 1% of the population.2,3 Atherosclerosis is a chronic inflammatory disease of the arterial wall that involves the accumulation of lipid-laden foam cells. 4 Driven by the underlying chronic inflammatory process rather than traditional cardiovascular risk factors, RA is regarded to be an independent risk factor for cardiovascular disease (CVD). 5 Thus, patients with RA are at high risk of developing CVDs.
Acetylsalicylic acid (aspirin [ASA]), an antipyretic and analgesic, was initially used 100 years ago and became the cornerstone of antiplatelet therapy for the secondary prevention of CVD during the second half of the 20th century. 6 Before the advent of disease-modifying antirheumatic drugs (DMARDs), high-dose ASA was widely used to treat RA. 7 However, whether use of ASA is effective in the primary prevention of CVD in patients diagnosed with RA remains unclear. As such, the aim of the present study was to investigate the efficacy of ASA in the primary prevention of major ischemic vascular events in patients diagnosed with RA.
Patients and methods
Study population and study design
This observational study included data from patients diagnosed with RA who were sourced from the population-based Kailuan cohort study. Participants enrolled into the Kailuan study were employees of the Kailuan Group and their relatives, and the study protocol was approved by the Ethics Committee of the Institutional Review Board of Kailuan Hospital, Hebei, China. Participants in the Kailuan cohort study have been continuously registered since June 2006 and have been followed-up biennially to the current date. All participants provided written informed consent, have been anonymized, and cannot be identified in any way. Due to the retrospective design of the present study, requirements for institutional review board approval were waived. The present retrospective observational study was conducted in accordance with the 2013 Declaration of Helsinki, and the study conformed to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. The current study analyzed data from 118,500 participants enrolled in the Kailuan cohort study between 2014 and 2016, and sequential patients with RA, aged ≥18 years, were selected for study inclusion. Exclusion criteria comprised missing data, and/or former medical histories of myocardial infarction (MI) and/or ischemic stroke (IS). Patients who underwent ASA therapy were propensity score matched at a ratio of 1:4 with patients with RA who did not undergo ASA therapy. The clinical endpoint of this study was major ischemic vascular events, including MI and IS, during a follow-up period of 3.3 years from baseline.
Demographic parameters were extracted from medical records, including age and sex. Data on the following parameters were also extracted: biochemical indices, comprising total cholesterol (TC), triglycerides (TG), high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), fasting blood glucose (FBG), and high-sensitivity C-reactive protein (hs-CRP). Medical history, including hypertension and diabetes mellitus, and lifestyle factors, including smoking and alcohol consumption, were documented. These parameters had been measured and/or recorded at initial enrollment and updated during follow-up for all participants.
Statistical analysis
Normally distributed continuous data are presented as mean ± SD and were analyzed by Student’s t-test for between-group comparisons. Skewed continuous data are presented as median with interquartile range (25th percentile [P25]–75th percentile [P75]), and were analyzed by Wilcoxon rank-sum test for between-group comparisons. Between-group comparisons of categorical variables were performed using χ2-test. A propensity score matching technique was employed to mitigate the bias arising from other variables (age, sex, BMI, blood pressure, FBG, TC, TG, HDL-C, LDL-C, hs-CRP, history of hypertension, history of diabetes, and smoking status). The cumulative incidence of major ischemic vascular events in each group was analyzed using the Kaplan–Meier method and compared using log-rank test. Cox proportional hazard models were used to evaluate the relationship between ASA therapy and major ischemic vascular events and its subtypes. Data were analyzed using SAS software, version 9.4 (SAS Institute Inc., Cary, NC, USA), and differences with P < 0.05 were considered to be statistically significant.
Results
Characteristics of the study participants
A total of 1243 patients with RA were initially selected for study inclusion. Following review of data, 243 patients were excluded due to missing data, and a further 61 patients were excluded due to former medical histories of MI and/or IS. Ultimately, 939 patients with RA were enrolled (79.87% male), comprising 189 patients who received ASA and 750 patients who did not receive ASA (Figure 1). The mean (±SD) age of the participants was 57.8 ± 11.8 years. There were no statistically significant differences between the ASA and non-ASA groups in any of the variables. Detailed characteristics of the study participants are summarized in Table 1.

Flow chart of patient selection for study inclusion. ASA, aspirin; IS, ischemic stroke; MI, myocardial infarction; RA, rheumatoid arthritis.
Baseline characteristics of 939 patients with rheumatoid arthritis at the beginning of the follow-up, categorized into the those who received ASA treatment (ASA group) or did not receive ASA treatment (non-ASA group).
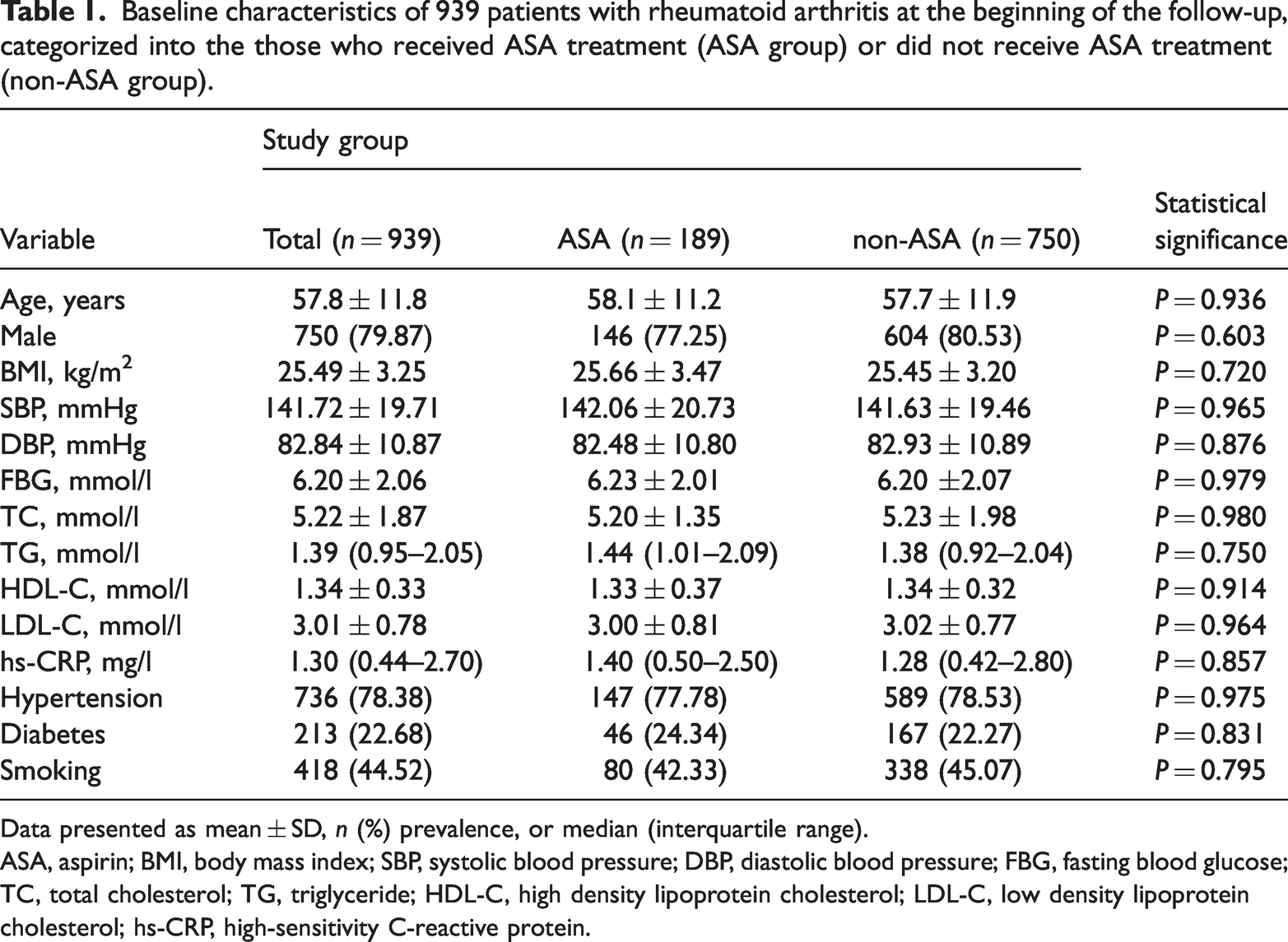
Data presented as mean ± SD, n (%) prevalence, or median (interquartile range).
ASA, aspirin; BMI, body mass index; SBP, systolic blood pressure; DBP, diastolic blood pressure; FBG, fasting blood glucose; TC, total cholesterol; TG, triglyceride; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; hs-CRP, high-sensitivity C-reactive protein.
Cumulative incidence of major ischemic vascular events
During the 3.3-year follow-up, major ischemic vascular events occurred in six patients (3.2%) in the ASA group (MI [n = 1] and IS [n = 5]) and 35 (4.7%) in the non-ASA group (MI [n = 2], IS [n = 33]), with no statistically significant difference in the cumulative incidence of major ischemic vascular events between the two groups (Figure 2).
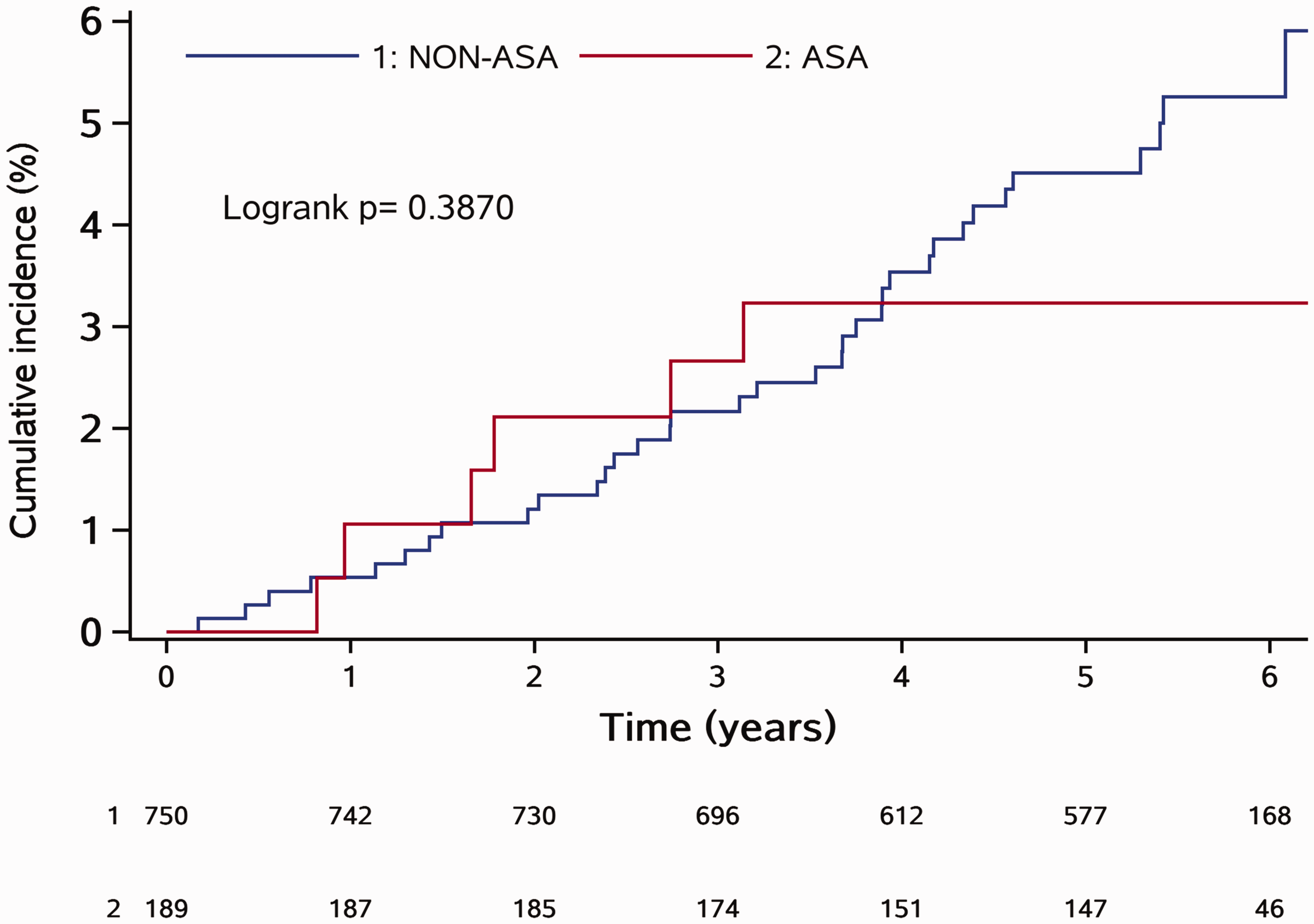
Cumulative incidence of major ischemic vascular events in patients with rheumatoid arthritis categorized into those who received aspirin (ASA group) and those who did not receive aspirin (non-ASA group).
Cox proportional hazards regression analysis
Unadjusted Cox proportional hazards regression analysis revealed no significant association between ASA therapy and ischemic vascular events (hazard ratio [HR] 0.684; 95% confidence interval [CI] 0.288, 1.627; P = 0.391). In addition, no statistically significant association was found between ASA therapy and MI (HR 0.575; 95% CI 0.180, 21.900; P = 0.575) or IS (HR 0.603; 95% CI 0.235, 1.544; P = 0.291; Table 2). Further analyses of patients categorized according to age, sex, BMI, history of hypertension, history of diabetes mellitus, and smoking status also revealed no statistically significant interactions across the diverse population (Table 3).
Cox proportional hazards regression analysis of cardiovascular events in 939 patients with rheumatoid arthritis with did or did not receive ASA treatment.
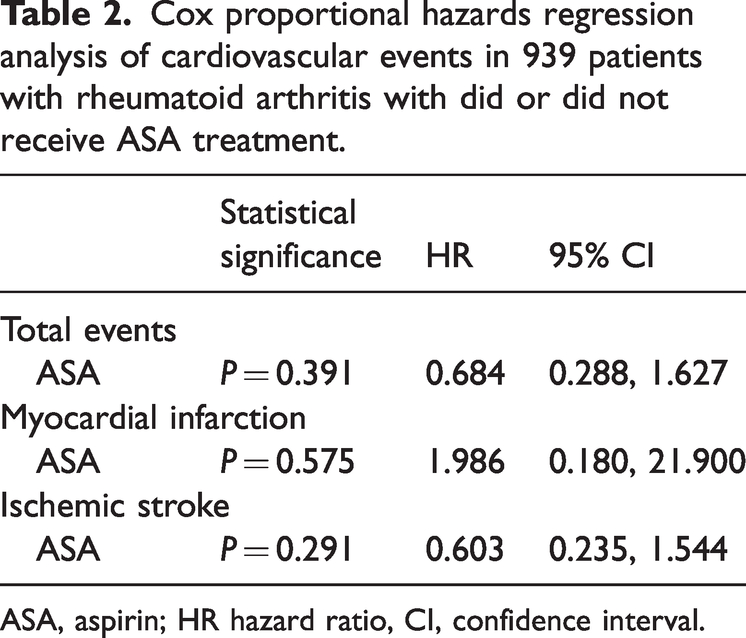
ASA, aspirin; HR hazard ratio, CI, confidence interval.
Relative risks for various subgroups of 939 patients with rheumatoid arthritis with did or did not receive ASA treatment.

Data presented as n (%) prevalence.
BMI, body mass index; ASA, aspirin; HR hazard ratio, CI, confidence interval.
Discussion
Rheumatoid arthritis and atherosclerosis share many common pathological mechanisms in which inflammation plays a critical role. RA-induced inflammation leads to atherosclerosis via multiple pathways. The pathogenesis of RA involves cytokines, such as tumor necrosis factor-alpha and interleukin-6, which are also independently predictive of subsequent CVD. RA-induced inflammation alters lipid metabolism and oxidative processes, thereby accelerating atherosclerosis and cardiovascular events. 8 Endothelial dysfunction in RA is an early marker of atherosclerosis, 9 and increased intima-media thickness is an early indicator of atherosclerosis. 10 Both endothelial dysfunction and increased intima-media thickness of the common carotid artery in patients with RA have been documented.11–14
Incidences of subclinical and clinical atherosclerosis have been reported to be higher in patients with RA, 1 and the risk of CVD in patients diagnosed with RA is up to 2 times higher than that in the general population. 15 In patients with RA, the risk of MI doubles, whereas the increased risk of cerebrovascular incidents is approximately 1.5-fold. 16 Chung et al. 17 reported that the risk of acute MI increased by 38% in patients compared with that in the general population. In particular, comorbidities, such as hypertension, diabetes, hyperlipidemia, cerebrovascular accidents, chronic obstructive pulmonary disease, and end-stage renal disease, are associated with an increased risk of acute MI. Palomäki et al. 18 reported that patients with RA have a poor prognosis after MI, and the duration of RA and usage and dosage of corticosteroids have been reported to be independent predictors of mortality in these patients. In contrast, RA was found to be an independent risk factor for stroke. The risk of different types of stroke has been shown to be higher in patients with most rheumatic diseases compared with the general population, particularly in patients <50 years of age, 19 and seropositive RA has been positively associated with IS. 20 Patients with RA and serious infections and insufficient treatment for CVD are at an increased risk of subsequent stroke(s). 21
Aspirin is a non-steroidal anti-inflammatory drug that relieves pain, reduces inflammation, and inhibits platelet aggregation. ASA irreversibly acetylates the serine residue of cyclooxygenase 1, which is required for prostaglandin and thromboxane synthesis. 22 ASA has been considered the mainstay of antiplatelet therapy for secondary prevention of CVD since the second half of the 20th century. 6 ASA administration for secondary prevention of atherothrombotic events in patients with known atherosclerotic disease has shown a favorable benefit-to-risk profile.23–25 In contrast, the overall benefits of ASA in the primary prevention of atherothrombotic events in patients without a history of atherosclerotic disease have been controversial. The reduction in atherothrombotic events is largely counterbalanced by an increase in bleeding events. 26 Recent studies have demonstrated that ASA, as a primary prevention strategy, neither lowers the risk for CVD nor prolongs the disability-free survival period in older adults, but results in a significantly higher risk for major bleeding events.27,28 Unexpectedly, healthy older adults who received ASA experienced higher all-cause mortality than those who received placebo. 28
Based on cumulative data, the United States Preventive Services Task Force (USPSTF) updated the guidelines on ASA for the primary prevention of CVD,
29
as follows: The decision to initiate low-dose aspirin use for the primary prevention of CVD in adults aged 40 to 59 years who have a 10% or greater 10-year CVD risk should be an individual one (grade C recommendation). The USPSTF recommends against initiating low-dose aspirin use for the primary prevention of CVD in adults 60 years or older (grade D recommendation).
Thus, the USPSTF does not recommend the use of ASA for the primary prevention of CVD in any population, which is consistent with the 2019 American College of Cardiology/American Heart Association Task Force guidelines on the primary prevention of cardiovascular disease, 30 and the 2021 European Society of Cardiology guidelines on cardiovascular disease prevention in clinical practice. 31 Nevertheless, this shift also meant that the efficacy and safety of ASA for primary prevention in a particular population (40–59 years of age) could, to a certain extent, be confirmed. 32
Before the advent of DMARDs, high-dose ASA was widely used to treat RA. 7 However, frequent use of ASA may increase the risk for gastrointestinal complications in this patient population. 33 In a relatively large study of female healthcare professionals, low-dose ASA was not associated with a significant reduction in risk of RA. 34
Whether ASA can be used for the primary prevention of CVD in patients with RA remains unclear. The present study revealed no difference in the cumulative incidence of major ischemic vascular events between the ASA and non-ASA therapy groups in patients diagnosed with RA, suggesting that ASA does not confer protection against such events in this patient population. Consistent with the present findings, Linos et al. 35 followed 473 patients with RA who received aspirin treatment for 10 years: no reduction in the incidence of MI (classic angina pectoris, sudden unexpected death, or cerebral infarction) was confirmed. Furthermore, Durán et al. 36 demonstrated no protective association between ASA score and MI in patients with RA, and Solomon et al. 7 reported that patients with RA treated with low-dose ASA had a risk for major adverse cardiovascular events similar to those not treated. However, Iacono et al. 37 reported that the incidence of cardiovascular events was significantly lower in patients receiving ASA than in those without.
As described above, studies investigating the use of ASA for the primary prevention of CVD in patients with RA have reported conflicting results; as such, this topic warrants further investigation. These mixed findings indicate that primary prevention may be suitable only for a specific subset of individuals. Patients diagnosed with RA may potentially benefit from a lower threshold for prescribing ASA due to their faster development of atherosclerosis, which is linked to impaired endothelial function, higher levels of lipoprotein A, increased oxidation of LDL, and changes in HDL levels.36,38 Previous research has revealed that patients with early RA exhibit a thicker inner lining of arteries and a lower percentage of flow-mediated dilation, indicating an early stage of atherosclerosis. 39 Patients in their younger to middle-age years, who have low disease activity, no cardiovascular risk factors, and no existing heart disease exhibit altered endothelial reactivity, which appears to be mainly due to the chronic inflammatory condition of the disease. 40 Moreover, these patients tended to use non-steroidal anti-inflammatory drugs (NSAIDs) and corticosteroids more frequently, further increasing their risk for CVD. Research has demonstrated that, compared with placebo, rofecoxib is associated with the highest risk of heart attacks, followed by lumiracoxib; ibuprofen is associated with the highest risk of stroke, followed by diclofenac; and etoricoxib and diclofenac are associated with the highest risk for cardiovascular-related death. 41 Additionally, studies have shown that the amount of medication taken daily, total duration of use, and cumulative dose are all associated with a higher risk of MI. 42 However, these medications (such as steroids, NSAIDS, methotrexate, or anti-interleukin-6 drugs) simultaneously place these patients at a higher risk of events from ASA and also interact with ASA.36,43,44
Conclusions
There was no significant difference in the cumulative incidence of major ischemic vascular events in patients diagnosed with RA who received ASA therapy compared with those who did not receive ASA therapy, nor did ASA confer protection against major ischemic vascular events.
Limitations
Although the number of participants initially enrolled in the Kailuan study was relatively large, few participants fulfilled the inclusion criteria for the present study, which may mask the potential role of ASA in the primary prevention of major ischemic vascular events. Secondly, medication information (such as DMARDs, glucocorticoids, and statins), which may also have impacted the risk of major ischemic vascular events, was not considered. Thirdly, the lack of statistically significant between-group difference in the cumulative incidence of major ischemic vascular events may have been influenced by the limited observation period of the study. An extended follow-up duration may have resulted in more pronounced differences. In addition, because the average age of this cohort was <60 years, whether the present results can be extrapolated to a broader demographic remains uncertain. Finaly, because the current study was retrospective and observational in design, with a limited number of patients, it may have been underpowered to detect statistically significant differences.
Footnotes
Acknowledgements
We thank all subjects enrolled in this project.
Author contributions
Authors CG, QQH, HXC, CTL and XP drafted the manuscript, collected the data and performed statistical analyses. QH, SW and KL contributed equally to designing the rational and protocol of this study. All authors read and approved the final manuscript.
Data availability statement
The original research data supporting the findings of this study are available from the corresponding author upon request.
Declaration of conflicting interest
The Authors declare that there is no conflict of interest.
Funding
This study was supported by the Key Scientific Research Project of Health Commission of Hebei province, PR China (Project No. 20231775).
