Abstract
Lenalidomide in combination with dexamethasone is a treatment for patients with relapsed or refractory myeloma. Although this combination demonstrates a high level of efficacy, it further exacerbates the hypercoaguable state that exists within myeloma. Thromboprophylactic regimen require careful selection and if warfarin is chosen, assiduous monitoring is required to ensure it will be clinically effective. We report the case of one patient who experienced multiple thrombotic events despite anticoagulant or antiplatelet thromboprophylaxis and review the contributing factors.
Keywords
Introduction
Lenalidomide is an analogue of thalidomide, which has been designed to be more potent but with fewer side effects [Armoiry et al. 2008; Hideshima et al. 2008]. It is a third-line treatment in myeloma, licensed for patients who have failed on two previous treatments and has been approved for use within NHS Scotland for such a purpose [Scottish Medicines Consortium, 2010]. However, there are concerns about the thrombotic risk associated with this medication, especially when used in conjunction with dexamethasone [Medicines and Healthcare Products Regulatory Agency, 2011]. This article outlines an illustrative case and discusses in detail practical thrombotic risk management in such patients.
Case report
Overview
A 77-year-old man presented to hospital with a 4-day history of productive cough and shortness of breath, the severity of which worsened when lying flat. There was no associated chest pain or purulent sputum. The patient had a past medical history of relapsed immunoglobulin G myeloma, hip replacement and recent myocardial infarction [ST elevation myocardial infarction (STEMI)] 3 weeks previously. The patient was taking the prescribed medication listed in Table 1. On examination his respiratory rate was 24 rpm, temperature 36.2°C, blood pressure 148/83 mmHg and pulse 65 bpm. An electrocardiogram revealed multiple atrial ectopics. Urea and electrolytes, liver function tests and full blood count results were within the normal ranges. A large volume bilateral pulmonary embolus (PE) was confirmed on computed tomographic pulmonary angiography.
List of medications on admission of 77-year-old man to hospital.
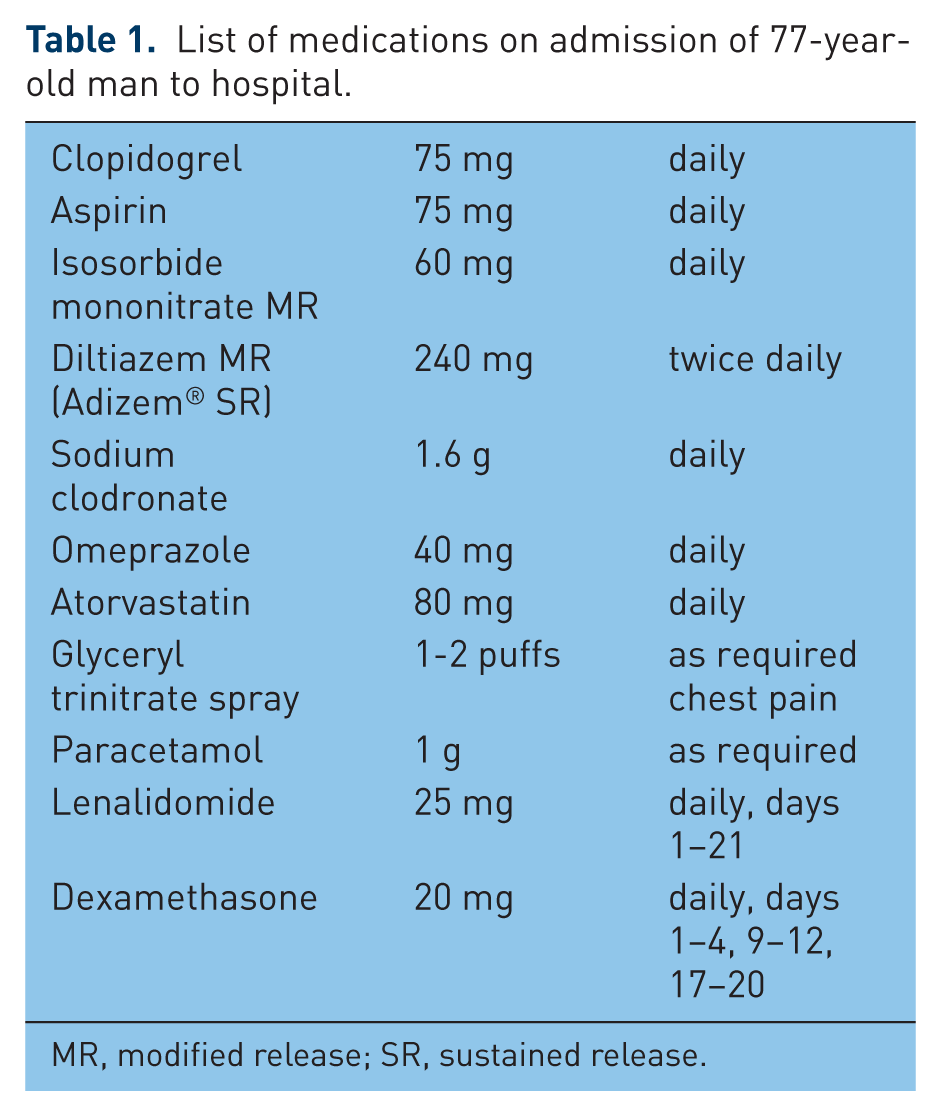
MR, modified release; SR, sustained release.
Prior multiple myeloma therapy
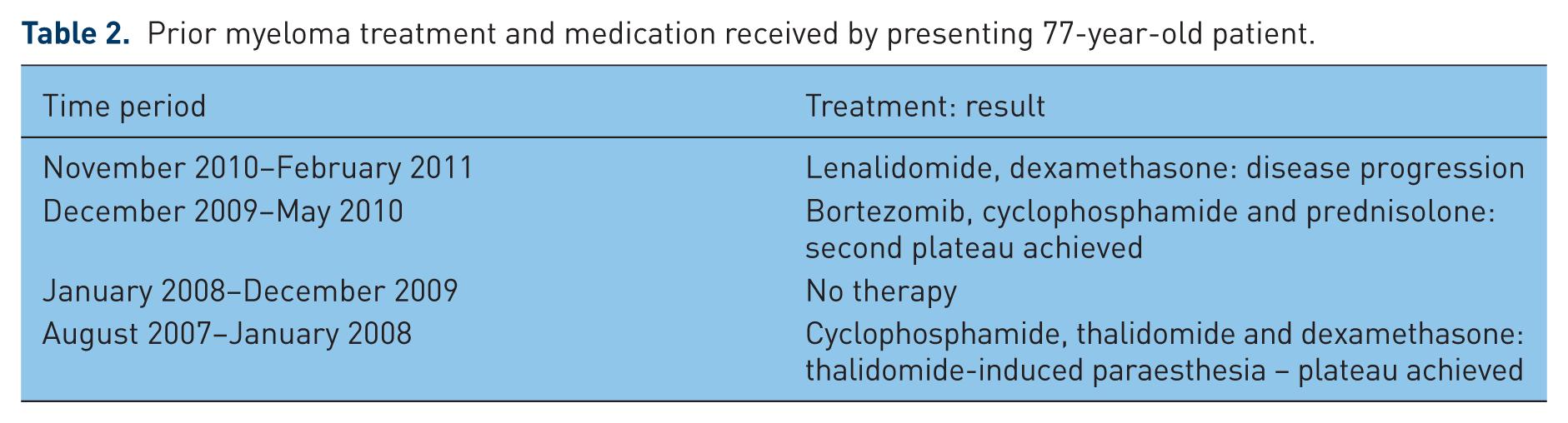
Between June 2007 and January 2011, the patient had been prescribed a total of four different regimens for multiple myeloma, three of which had to be stopped due to toxicity or lack of efficacy (Table 2). When seen at clinic (November 2010) the patient was started on a new medication regimen – lenalidomide and dexamethasone. At this time, the thrombotic risk associated with the therapy was covered by prescribing and titrating warfarin as prophylaxis to reduce the risk of thromboembolism. However, on presentation with chest pain during the previous admission the patient’s international normalized ratio (INR) was noted to be 1.2. Adherence to medication was discussed with the patient and thought to have been good. It is therefore most likely that the subtherapeutic INR was the result of an interaction between warfarin and dexamethasone, making stabilization difficult to achieve in this particular patient and resulting in fluctuations in INR. Considering the recent STEMI, the improvement in the multiple myeloma since starting the dual lenalidomide and dexamethasone regimen and issues with attaining a stable warfarin dose it was thought appropriate to use dual antiplatelet agents as alternative thromboprophylaxis to warfarin. Therefore, post-STEMI warfarin was stopped and the patient started on dual antiplatelet therapy with aspirin and clopidogrel for a minimum of 3 months. However, 3 weeks after the STEMI the patient presented with a PE as described above. It is likely that this patient experienced an arterial then venous thrombotic event while taking a combination of lenalidomide and dexamethasone.
Prior myeloma treatment and medication received by presenting 77-year-old patient.
Outcome
Following the diagnosis of PE, the decision was taken to continue treating the myeloma with lenalidomide and dexamethasone as it was continuing to control the myeloma but also to prescribe enoxaparin, which is a low molecular weight heparin (LMWH), at a treatment dose of 1.5 mg/kg/day for 3 months and aspirin 75 mg daily as secondary prevention of cardiovascular disease.
Discussion
Disease overview
In myeloma, changes to the bone marrow microenvironment and alterations in a multistep genetic process result in the excess proliferation of neoplastic monoclonal plasma cells [Palumbo and Anderson, 2011]. These cells can then accumulate in bone-producing osteolytic lesions while dysfunctional antibody production and increased paraprotein production cause immunosuppression and renal impairment respectively [Raab et al. 2009].
Multiple myeloma remains an incurable condition and relapse is likely, even after response to the initial treatment [Kastritis et al. 2009]. In the UK, the incidence of myeloma is around 60–70 per million [Bird et al. 2011]. This accounts for around 10% of all haematological malignancies and 1.5–2% of mortality in patients with cancer [Palumbo et al. 2009]. One study of 1027 newly diagnosed patients with myeloma between 1985 and 1998 noted the median age at diagnosis was 66 years old, while the median survival was 33 months [Kyle et al. 2003].
Thrombotic risk associated with haematological malignancies
There is an increased risk of thrombotic events in all patients with cancer; the increased risk varies according to cancer type [Palumbo et al. 2009]. A subclinical hypercoaguable state is common in patients with haematological malignancies and follows a linear relationship with disease progression [Falanga and Marchetti, 2009].
Patients with multiple myeloma have been found to have increased levels of Von Williebrand’s factor, factor VIII and fibrinogen [Kristinsson, 2010]. There are also a number of raised inflammatory cytokines in multiple myeloma, including interleukin (IL)-6 and tumour necrosis factor-α [Uaprasert et al. 2010]. This is suggestive that patients who are diagnosed with multiple myeloma are already at an increased risk of thromboembolism prior to initiation of treatment, regardless of the propensity for that treatment to cause thrombotic events.
The incidence of venous thromboembolism (VTE) in multiple myeloma varies from 3% to 10% depending on the drug regimen prescribed [Gay and Palumbo, 2010]. It is associated with a reduction in quality of life and an increase in treatment delays and ultimately mortality [Lyman, 2011].
Management of relapsed multiple myeloma
Medication choice within relapsed multiple myeloma is predominantly based on two factors: previous resistance, which is commonplace, developing over time and excluding a therapy from future use; and toxicity profile for an individual therapy. Assuming neither resistance nor toxicity is an issue then there are three principal medications used for relapsed multiple myeloma. They are normally prescribed with a corticosteroid, pulsed or weekly, and may be prescribed in combination with cyclophosphamide. These novel medications are thalidomide, bortezomib and lenalidomide [Bird et al. 2011]. Thalidomide is an inhibitor of tumour necrosis factor-α, a stimulator of IL-2 and interferon-α, and has antiangiogenic, apoptotic and regulatory effects over adhesion molecule expression [Hideshima et al. 2000], whereas bortezomib elicits its action as via protease inhibition. Lenalidomide is discussed below.
Pharmacokinetics and pharmacodynamics of lenalidomide
Lenalidomide has a rapid absorption after oral administration in healthy volunteers and the time to maximum plasma concentration (T max) is 0.625–1.5 h [Davies et al. 2009]. Lenalidomide is not metabolized by, nor does it affect, the cytochrome 450 hepatic enzyme systems and is excreted via the renal system with an elimination half life of 3 h.
Lenalidomide is an analogue of thalidomide and although it shares many of its pharmacologic properties, it has greater potency and distinctly different toxicity profile to the parent compound. Lenalidomide is not associated with constipation, somnolence and neuropathy, which are associated with thalidomide, but is linked to neutropenia and thrombocytopenia [Kastritis et al. 2009]. Both agents are protagonists in the generation of thrombotic events.
Lenalidomide has been shown in vitro and in vivo models to potentiate its antineoplastic effects via a number of different mechanisms [Kotla et al. 2009]. First, lenalidomide has been shown to have immunomodulatory effects which alter cytokine production, natural killer lymphocyte cytotoxicity and T-cell activation. In turn, this has a number of physiological outcomes, including priming of apoptosis of the myeloma cell due to a decrease in IL-6, antibody-dependent cell-mediated cytotoxicity and upregulation of T-cell activity, and T-cell colonal proliferation resulting in tumour cytotoxicity. Second, antiangiogenesis activity with lenalidomide is considered to be independent of the immunomodulatory effects. Tumour cells require a blood supply to aid development and growth and lenalidomide has been shown to be two to three times as potent as thalidomide in vivo at reducing angiogenesis. This is principally modulated by a decrease in vascular endothelial growth factor and IL-6. Third, lenalidomide can arrest growth of myeloma cells between G0 and G1, resulting in death of the cell while increasing the expression of the p21 gene (tumour suppressor gene). Finally, the formation and activation of osteoclasts is downregulated by lenalidomide, resulting in a change to the microenvironment, and the beneficial relationship between the osteoclast and the myeloma cell is interrupted.
Lenalidomide in multiple myeloma
The licence for the use of lenalidomide and dexamethasone for relapsed or refractory multiple myeloma was given initially based on the results of two parallel randomized, double-blind, placebo-controlled phase III studies [Dimopoulos et al. 2007; Weber et al. 2007]. Both of these studies used lenalidomide 25 mg daily or matching placebo on days 1–21 of a 28-day cycle given concomitantly with dexamethasone 40 mg daily on days 1–4, 9–12 and 17–20 for the first four cycles. The thrombotic adverse event rates reported by Weber and colleagues were 14.7% versus 3.4%; p<0.001 between the lenalidomide and placebo group [Weber et al. 2007]. Both arms of the study received active dexamethasone. Dimopolous and colleagues reported thrombotic events to be 11.4% versus 4.5% for the lenalidomide versus placebo group [Dimopolous et al. 2007].
The British Society for Haematology recently published guidelines on the treatment of multiple myeloma [Bird et al. 2011]. These guidelines concur with the National Institute for Health and Clinical Excellence (NICE) guidance that the combination therapy of lenalidomide and dexamethasone can be used for treating patients who have received at least one prior therapy for multiple myeloma [National Institute for Health and Clinical Excellence, 2010]. In Scotland, the Scottish Medicines Consortium has stipulated that the place of lenalidomide in therapy should be as a third-line agent in patients whose condition has failed to respond to two previous therapies [Scottish Medicines Consortium, 2010]. These guidelines influenced the choice of therapy for the patient in November 2010 (as described in this case report).
Thrombotic risk associated with multiple myeloma treatments
Monotherapy with thalidomide or lenalidomide is not considered to increase the risk of thrombosis until prescribed with dexamethasone or other chemotherapies known to increase thrombosis risk, that is doxorubicin [Gay and Palumbo, 2010; Richardson et al. 2006]. The risk of VTE is also reduced in a patient who has relapsed disease compared with a newly diagnosed patient [Palumbo et al. 2008]. There are no studies investigating lenalidomide and dexamethasone for myeloma which compare the efficacy of different thromboprophylaxis regimens [Zangari et al. 2009; Musallam et al. 2009]. The majority of data available to support clinical decision making regarding thromboprophylactic choice are dependent on trials of thalidomide or lenalidomide in myeloma which use single prophylactic agents [Zangari et al. 2009]. However, from these data, choices can be made as to the risk reduction associated with specific prophylactic regimens.
Table 3 highlights thrombosis rates, lenalidomide and dexamethasone doses, and antithombotic prophylaxis if used in patients with multiple myeloma. The results of the studies on newly diagnosed patients with myeloma are interesting and display a wide range of the incidence of thromboembolic events. The incidence spans from 3% of those reported by Rajkumar and colleagues [Rajkumar et al. 2005] to 75% of the patients initially treated by Zonder and colleagues [Zonder et al. 2006]. Firstly, it is important to identify the differences in these two trials. The doses vary slightly. Zonder and colleagues gave 25 mg daily for 28 days for the first three cycles, each lasting 35 days. They also did not use any prophylaxis until data from the first 12 patients revealed that 9 (75%) experienced thromboembolic events. Aspirin 325 mg daily was then given which resulted in the rate of thromboembolism dropping to 15% of patients recruited thereafter. Overall the rate of thromboembolic events is similar to that of patients with relapsed or refractory disease who were not given any prophylaxis [Dimopoulos et al. 2007; Weber et al. 2007]. However, the rate of thromboembolic events was decreased in one study which gave prophylaxis to patients with relapsed or refractory multiple myeloma in whom the dose of dexamethasone was decreased [Klein et al. 2009]. It is of interest to note that none of the studies in Table 3 used warfarin as prophylaxis, perhaps suggestive of the known variability of warfarin effectiveness with high-dose steroids.
Thromboprophylaxis regimens and thrombosis rates in patients with myeloma receiving lenalidomide and dexamethasone.
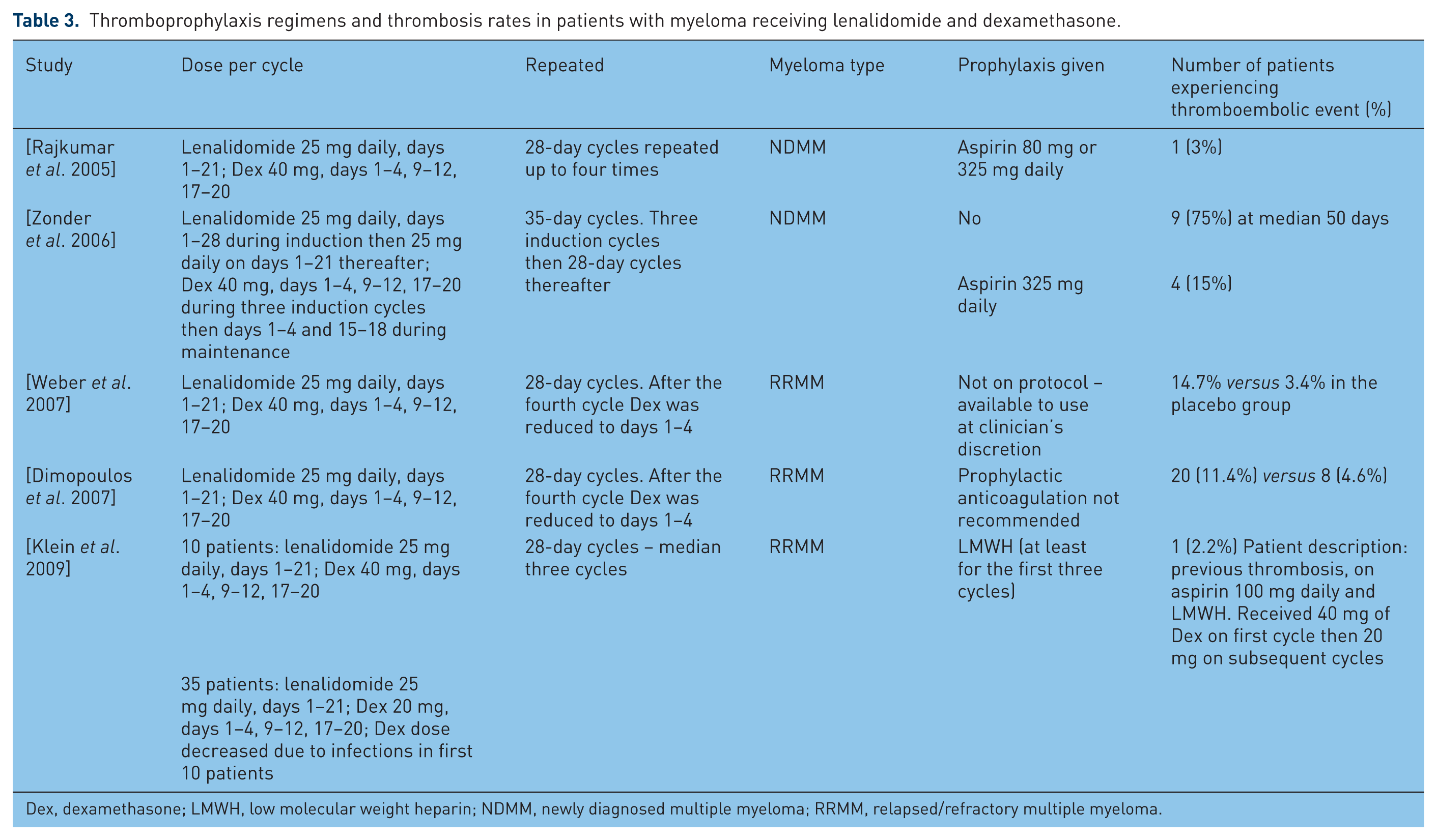
Dex, dexamethasone; LMWH, low molecular weight heparin; NDMM, newly diagnosed multiple myeloma; RRMM, relapsed/refractory multiple myeloma.
Implications for clinical practice
Prevention
The risk of thrombosis is further compounded by other disease-specific factors. Patients who are newly diagnosed are at a higher risk of thrombosis than those who have relapsed or refractory myeloma or those with a higher tumour burden [Kastritis and Dimopoulos, 2007]. The concomitant use of erythropoetin in addition to lenalidomide and dexamethasone has been shown to cause thrombosis in 23% of patients compared with 5% in those who are not on erythropoetin therapy [Knight et al. 2006]. When erythropoetin is clinically indicated in addition to lenalidomide and dexamethasone thromboprophylaxis with aspirin has been shown to be an effective prophylactic [Chen et al. 2009].
There is not enough evidence to recommend an optimum thromboprophylactic treatment. No head-to-head studies have compared different prophylactic regimens, although evidence has been published on the efficacy of individual prophylactic strategies. One such study reported the use of aspirin 81 mg daily as prophylaxis in an anthracycline-based regimen that included thalidomide, which is also known to have thrombotic side effects [Baz et al. 2005]. The incidence of thromboembolic events decreased.
Recognition and thrombotic risk stratification
Palumbo and colleagues have outlined a risk stratification that allows clinicians to assess patients with myeloma prior to prescribing lenalidomide therapy [Palumbo et al. 2008]. This stratification can be broken down into three categories. The first category covers the type and stage of malignancy. Patients with newly diagnosed disease are at higher risk. The second category examines prescribed therapy. Patients prescribed high-dose dexamethasone, erythropoetin or regimens containing chemotherapy agents, principally doxorubicin, are considered to have additional risk factors for developing thrombosis. Finally, concomitant medical conditions also require assessment. Patients who are immobile and have had previous VTE, diabetes or cardiac disease, or have undergone recent surgery are also considered to have additional risk factors. Patients with no risk factors or one risk factor should be prescribed low-dose aspirin; those with at least two risk factors should be prescribed LMWH or treatment dose warfarin. In addition to this, all patients who are prescribed high-dose dexamethasone or concomitant chemotherapy should be prescribed LMWH or treatment dose warfarin. Although LMWH is thromboprotective and its effects beneficial, it does not completely stop thrombotic events in regimens containing thalidomide [Niesvizky et al. 2007]. These results should be considered in terms of the difficulty of warfarin stabilization in patients on high-dose dexamethasone. As yet there is no published evidence of other orally active anticoagulant agents – dabigatran (direct thrombin inhibitor) or rivaroxaban (direct activated factor X inhibitor) – being used as thromboprophylaxis in this group of patients.
Management
LMWH (e.g. enoxaparin 1.5 mg/kg daily) should be the initial management for a venous thrombotic event in a patient with myeloma treated with lenalidomide and high-dose dexamethasone. The duration of treatment is less certain. However, patients who are continuing on lenalidomide therapy should receive at least a prophylactic dose of LMWH for the remainder of their treatment.
Conclusion
Anticoagulation with warfarin in all patients prescribed high-dose dexamethasone for multiple myeloma may be unreliable due to an interaction between the two drugs and subsequent difficulties in warfarin stabilization. Antiplatelet therapy, including dual antiplatelet administration, does not appear to provide optimal thromboprophylactic cover in all cases. Daily self-injection with LMWH has proved to be a more reliable thromboprophylactic option; however, patient acceptability of the route of administration also has to be considered. Appropriate counselling, including a discussion of the risks and benefits of this treatment, is also needed.
Footnotes
The authors received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
The authors declare no conflict of interest in preparing this article.
