Abstract
Lenalidomide is a derivative of thalidomide and belongs to the class of drugs known as the immunomodulatory drugs (IMiDs). The IMiDs have played a large role in improving the survival outcomes of patients with multiple myeloma. In particular, lenalidomide is currently standard of care in the newly diagnosed setting, in the maintenance setting post-autologous stem cell transplant, as well as in the relapsed/refractory setting. While the combination of lenalidomide and various proteasome inhibitors has proven particularly effective, there are emerging data demonstrating the effectiveness of lenalidomide in combination with other important classes of drugs including the monoclonal antibodies. Recent studies have provided insight into the molecular target of lenalidomide and the other IMiDs, although there is still much to be learned regarding the mechanisms by which lenalidomide affects the myeloma cell and the immune system. Here we review the molecular mechanisms of action, side effects, and the results of the clinical trials which have led to the widespread incorporation of lenalidomide into the myeloma therapeutic armamentarium.
Introduction
Multiple myeloma is a bone marrow cancer in which malignant plasma cells produce monoclonal protein and disrupt bone remodeling. Historically, the median survival of patients with myeloma was only 2–3 years in the 1980s and prior to the 2000s treatment options were quite limited, consisting primarily of the VAD regimen (vincristine, doxorubicin, dexamethasone), oral melphalan, corticosteroids, and autologous stem cell transplant (ASCT). However, the development of the immunomodulatory drugs (IMiDs) and the proteasome inhibitors (PIs) in the 2000s has resulted in significant improvements in survival rates. Lenalidomide, which is the second-in-class IMiD, is now widely used for induction therapy in both transplant-eligible and transplant-ineligible patients, as maintenance therapy following ASCT, and in the relapsed/refractory setting. Here we present an overview of the use of lenalidomide in myeloma.
Approved myeloma indications
Lenalidomide was first approved in the United States in combination with dexamethasone for the treatment of relapsed/refractory myeloma in 2006 (Figure 1). In 2015, the combination of lenalidomide and dexamethasone was approved for use in patients with newly diagnosed myeloma. In 2015 and 2016, four different lenalidomide-containing triplet regimens were approved for relapsed/refractory myeloma: carfilzomib/lenalidomide/dexamethasone (https://www.accessdata.fda.gov/drugsatfda_docs/label/2015/202714s009lbl.pdf), ixazomib/lenalidomide/dexamethasone (https://www.accessdata.fda.gov/drugsatfda_docs/label/2016/208462s001lbl.pdf), elotuzumab/lenalidomide/dexamethasone (https://www.accessdata.fda.gov/drugsatfda_docs/label/2015/761035s000lbl.pdf) and daratumumab/lenalidomide/dexamethasone (https://www.accessdata.fda.gov/drugsatfda_docs/label/2016/761036s004lbl.pdf). Finally, in 2017, lenalidomide maintenance therapy following ASCT was approved (https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/021880s049lbl.pdf).

Timeline of key events in the development of lenalidomide for the treatment of myeloma.
Mechanism of action
IMiDs are small molecules derived from glutamic acid and phthalic anhydride which share common phthalimide and glutarimide moieties and there are only subtle differences in the glutarimide portion that distinguish lenalidomide from thalidomide and pomalidomide. Despite their very similar chemical structures, the IMiDs have distinct pharmacological properties which include half life, metabolism, clearance, and side effects (reviewed in Holstein and McCarthy 1 ). Lenalidomide undergoes minimal metabolism and is largely excreted in the urine unchanged. 2 While thalidomide is technically not considered an IMiD based on trademarking, we will consider these drugs together as IMiDs.
The IMiDs were so named because of the recognition that they induce a variety of immunomodulatory effects in addition to direct cytotoxic effects on myeloma cells. The cytotoxic effects were attributed to multiple mechanisms, including increased expression of proapoptotic factors and decreased expression of antiapoptotic factors, inhibition of nuclear factor kappa-light-chain-enhancer of activated B cells (NFkB), disruption of the phosphoinositide 3-kinase/protein kinase B (PI3K/Akt) pathway, and downregulation of CCAAT/enhancer binding protein beta (C/EBPβ) with resulting decrease in interferon regulatory factor 4 (IRF-4) production.3–6 An effect on osteoclastogenesis has been reported, with lenalidomide decreasing the secretion of osteoclastogenic factors including macrophage inflammatory protein-1 alpha (MIP-1α), B cell-activating factor (BAFF), a proliferation-inducing ligand (APRIL), and receptor activator of nuclear factor kappa-B ligand (RANK-L).7,8 A variety of effects on the immune system have been reported, including decreasing tumor necrosis factor (TNF)-α, interleukin (IL)-1β, IL-6, and IL-12 production and increasing IL-2 and interferon (IFN)-γ synthesis, increasing T-cell priming, enhancing tumor antigen uptake by dendritic cells with increased antigen presentation efficacy, inhibiting T-regulatory cells, and enhancing the activity of natural killer (NK) and NK T cells.9–13 The mechanisms of action for these effects and the molecular targets to which IMiDs bind were not initially understood.
In 2010 it was reported that thalidomide-induced teratogenicity was dependent on cereblon (CRBN). 14 Subsequently it was demonstrated that binding of IMiDs to CRBN is responsible for the anti-myeloma effects as well. 15 CRBN forms a complex with three other proteins Cullin-4 (CUL4), DNA damage-binding protein 1 (DBB1) and Regulator of Cullins-1 (ROC1) to produce the cullin-4 RING E3 ligase (CRL4) complex which has E3 ubiquitin ligase activity.14,16,17 IMiDs can stabilize CRBN and inhibit its own ubiquitination, which leads to an increase in CRL4-mediated degradation of target proteins. 18 An ubiquitin-independent function of CRBN has also been reported in which CRBN has chaperone-like activity and facilitates the formation and activation of the CD147 (Basigin) monocarboxylate transporters (MCT1) transmembrane complex. 19 In the presence of IMiDs, there is competition with CD147 and MCT1 for CRBN binding, resulting in destabilization of the CD147-MCT1 complex. 19
Further understanding of the mechanism of action of lenalidomide and other IMiDs was achieved following the identification of Ikaros (IKZF1) and Aiolos (IKZF3) as key CRBN-interacting proteins.20–22 These proteins are zinc finger transcription factors involved in B- and T-cell development. 23 Upon IMiD-CRBN binding, there is enhanced affinity of CRBN for IKZF1/3 which results in their ubiquitination and degradation.20–22 The degradation of IKZF1/3 results in significant changes in gene transcription, including decreased expression of IRF4 and increased expression of IL-2. IMiDs can also block substrates such as myeloid ecotropic insertion site 2 (MEIS2) from binding the CUL4-Roc1-DDB1-CRBN complex while IKZF1 or IKZF3 are being recruited, thus demonstrating the complexity inherent in the interaction between IMiDs and the CRBN complex. 24
The ability of lenalidomide to potentiate the activity of monoclonal antibody therapy was first reported in preclinical studies evaluating the combination of lenalidomide with the anti-CD20 antibody rituximab in a mouse lymphoma model.
25
This activity appeared to be dependent on NK cells, as
Finally, while it is evident that IKZF1/3 play key roles in mediating IMiD effects, studies have demonstrated numerous other CRBN-binding proteins whose expression is decreased following treatment with lenalidomide. 20 Gene expression profiling studies of myeloma cells showed at least 600 genes that were upregulated (at least twofold) and 600 genes that were downregulated following lenalidomide treatment. 15 Furthermore, in CRBN-knockdown cells, lenalidomide treatment induced an upregulation of 150 genes and downregulation of 30 genes. 30 These results suggest that some of the effects of lenalidomide may be exerted through non-CRBN-mediated pathways. In addition, both lenalidomide and pomalidomide recently were reportedly to directly bind p53-related protein kinase (TP53RK) and its binding protein TPRKB. 31 Binding of these IMiDs to TP53RK results in inhibition of its kinase activity, ultimately resulting in induction of apoptosis. 31 These effects were independent of CRBN. Thus further studies are needed to fully elucidate the mechanisms of action of lenalidomide and other IMiDs.
Lenalidomide side effects
The first phase I study in patients with myeloma determined the maximal tolerated dose to be 25 mg. Notably, the side effects associated with thalidomide such as somnolence, constipation, and neuropathy were infrequently observed with lenalidomide. 32 In subsequent phase II–III studies, the most common toxicities were hematological, including neutropenia (grade 3/4, 25–40%), anemia (grade 3/4, 9–26%), thrombocytopenia (grade 3/4, 11–15%).33–35 Nonhematological toxicities included diarrhea, fatigue, muscle cramps, rash, and infection. 36 In addition, an increased risk of venous thromboembolic events (VTEs) was noted (reviewed by Musallam and colleagues 37 ). Thereafter, studies began to include thromboprophylaxis, including aspirin.38–43 A phase III study randomized patients to low-dose aspirin (100 mg/day) or to low molecular weight heparin (LMWH) (enoxaparin 40 mg/day) in newly diagnosed patients receiving lenalidomide/dexamethasone induction and melphalan/prednisone/lenalidomide consolidation. 41 The incidence of VTE was 2.27% with aspirin and 1.20% with LMWH. In general, aspirin is considered the standard of care for patients receiving lenalidomide who do not have other risk factors for VTE, while for patients at higher risk, LMWH or full-dose anticoagulation is recommended. 44
Lenalidomide therapy in newly diagnosed patients
The initial studies assessing lenalidomide/dexamethasone in the newly diagnosed setting reported response rates of 68–91%.45,46 The Eastern Cooperative Oncology Group (ECOG) E4A03 randomized newly diagnosed patients to ‘high-dose’ dexamethasone (40 mg days 1–4, 9–12, 17–20 of a 28-day cycle)
The FIRST trial randomized 1623 newly diagnosed transplant ineligible patients to lenalidomide/dexamethasone (continued until progression)
The ECOG E1A06 and HOVON87/NMSG18 trials were similarly designed randomized phase III studies which compared MPT with melphalan/prednisone/lenalidomide (MPR) in transplant-ineligible patients. In the EOCG study, 306 patients were randomized to MPT
For a period of time, the doublet therapy of lenalidomide/dexamethasone was widely used in both the transplant-eligible and transplant-ineligible populations. However, more recently, triplet induction therapy consisting of lenalidomide, bortezomib, and dexamethasone (RVD) has become the standard of care for newly diagnosed patients. The initial phase I/II study demonstrated a 100% overall response rate (ORR).
50
Subsequently, the SWOG S0777 phase III study compared lenalidomide/dexamethasone with lenalidomide/bortezomib/dexamethasone.
51
The median PFS was significantly better in the triplet arm (43 months) than in the doublet arm (30 months) (HR 0.712,
There are multiple ongoing studies that are assessing the addition of novel agents to the lenalidomide/dexamethasone backbone as well as to the lenalidomide/PI/dexamethasone backbone (summarized in Table 1). Preliminary results from a phase II study assessing elotuzumab RVD have been reported. While the ORR was 100% after four cycles of therapy, two patients died while taking part in the study (septicemia and respiratory failure).
54
A phase Ib study assessing the addition of daratumumab to KRD reported an ORR of 100% with no grade 5 adverse events, although one cardiac grade 3 adverse event was noted.
55
A phase III study (KEYNOTE-185) evaluating the addition of the checkpoint inhibitor pembrolizumab to the RD backbone in newly diagnosed transplant-ineligible patients has been stopped prematurely by the US Food and Drug Administration (FDA) (https://www.fda.gov/Drugs/DrugSafety/ucm574305.htm). An interim analysis revealed an increased risk of death in the pembrolizumab-containing arm. At a median follow up of 6.6 months, there were 19 deaths in the pembrolizumab arm
Ongoing phase II or III studies assessing the incorporation of a novel agent to the lenalidomide/dexamethasone backbone in newly diagnosed patients.

CR, complete response; Dara, daratumumab; Isa, isatuximab; KRD, carfilzomib, lenalidomide, dexamethasone; MRD, minimal residual disease; ORR, overall response rate; PFS, progression-free survival; RD, lenalidomide, dexamethasone; RID, lenalidomide, ixazomib, dexamethasone; RVD, lenalidomide, bortezomib, dexamethasone; sCR, stringent CR.
Lenalidomide maintenance therapy following ASCT
There have been four large randomized phase III studies evaluating lenalidomide maintenance following ASCT (Table 2). All four studies have demonstrated very consistent improvements in PFS. A meta-analysis which included the CALGB, IFM, and GIMEMA studies revealed that at a median follow up of 79.5 months, lenalidomide maintenance significantly improves OS with a HR of 0.75 (95% CI 0.63–0.90,
Summary of randomized trials assessing lenalidomide maintenance after ASCT.

Combining ASCT and chemotherapy groups.
Transplant eligible only (total number in the study was 1970).
Bor, bortezomib; CTD, cyclophosphamide, thalidomide, dexamethasone; CVD, cyclophosphamide, bortezomib, dexamethasone; Dex, dexamethasone; Len, lenalidomide; MEL200, melphalan 200 mg/m2; MPR, melphalan, prednisone, lenalidomide; PFS, progression-free survival; OS, overall survival; RCD, lenalidomide, cyclophosphamide, dexamethasone; Thal, thalidomide.
CALGB 100104 randomized 460 patients to lenalidomide
IFM 2005-02 randomized 614 patients to lenalidomide
RV-MM-209 enrolled 402 patients in a study comparing chemotherapy (MPR) with ASCT consolidation following completion of induction therapy (lenalidomide/dexamethasone) as well as a second randomization to lenalidomide maintenance
The Myeloma XI study has been presented in abstract form. This study included treatment pathways for transplant-eligible (
One question of interest is whether lenalidomide maintenance improves outcomes regardless of cytogenetic risk status. The CALGB study did not collect cytogenetic information. The IFM study had cytogenetic data on approximately 80% of patients and it was reported that the 3-year PFS was higher in patients with or without a 13q deletion who received lenalidomide.
60
In an ASH abstract it was reported that the median PFS for patients with t(4;14) was 27 months in the lenalidomide arm
Although it is evident that lenalidomide maintenance post ASCT significantly improves outcomes, the majority of patients will still relapse. Therefore studies are underway to determine whether the addition of a novel agent to lenalidomide in the maintenance setting will further improve outcomes. The preliminary results of a phase II study investigating the combination of lenalidomide and ixazomib as post-ASCT maintenance therapy were recently presented. 66 The initial dose of ixazomib was 4 mg on days 1, 8, 15 of a 28-day cycle, however the protocol was later amended to reduce the dose to 3 mg. Of the 64 patients enrolled, 29 were reported to have an improvement in response during maintenance therapy. With a median follow up of 37.8 months, the median PFS has not yet been reached. Another study with preliminary results is a phase II study of lenalidomide and elotuzumab maintenance post ASCT. 67 In this study elotuzumab was administered 10 mg/kg intravenously weekly for two cycles (28-day cycle), then every other week for cycles 3–6 and then at 20 mg/kg monthly for cycles 7 and beyond. Thus far, 55 patients have been treated. The authors reported that 44% of patients had an improvement in their response and that 13 of 14 patients in complete response (CR) who had been tested for MRD were MRD negative. Studies assessing the use of daratumumab in combination with lenalidomide are planned or ongoing. Longer follow-up and larger studies are needed to determine whether the addition of any of these novel agents to lenalidomide improve survival and have any effects on SPM risk.
Lenalidomide in the relapsed/refractory setting
The initial phase II/III studies demonstrated that the doublet combination of lenalidomide/dexamethasone induced ORRs of 48–61% and 46–57% response rates in patients who had received prior treatment with thalidomide.33–35 Since then, numerous studies have evaluated the addition of other agents to the lenalidomide/dexamethasone backbone, including PIs (bortezomib,68,69 carfilzomib70,71), alkylating agents (low-dose cyclophosphamide,72,73 bendamustine74,75), histone deacetylase inhibitors (panobinostat, 76 ricolinostat, 77 vorinostat 78 ), monoclonal antibodies (elotuzumab, 79 daratumumab, 80 isatuximab, 81 pembrolizumab 82 ), liposomal doxorubicin, 83 and the nuclear export protein exportin 1 (XPO1) inhibitor selinexor. 84 These studies are summarized in Table 3.
Summary of phase I/II studies assessing lenalidomide-containing regimens in the relapsed/refractory setting.

Patients treated at the maximum tolerated dose.
Patients treated in the dose-expansion cohort.
NR, not reported.
Randomized phase III studies comparing lenalidomide/dexamethasone with lenalidomide/dexamethasone plus carfilzomib, daratumumab, elotuzumab, or ixazomib have led to the approval of all four triplet therapies as in all cases there was significant PFS benefit for the triplet compared with the doublet.85 –88 The results of these studies are summarized in Table 4. It should be noted that all four of these studies primarily involved lenalidomide-sensitive patients, and in fact, the majority of the patients were lenalidomide naïve. Thus, the applicability of the results of these studies to the standard US myeloma population which has not only received lenalidomide in the induction setting, but also in the maintenance setting, is unknown.
Summary of randomized phase III trials comparing lenalidomide/dexamethasone with lenalidomide/dexamethasone/novel agent in relapsed/refractory myeloma.
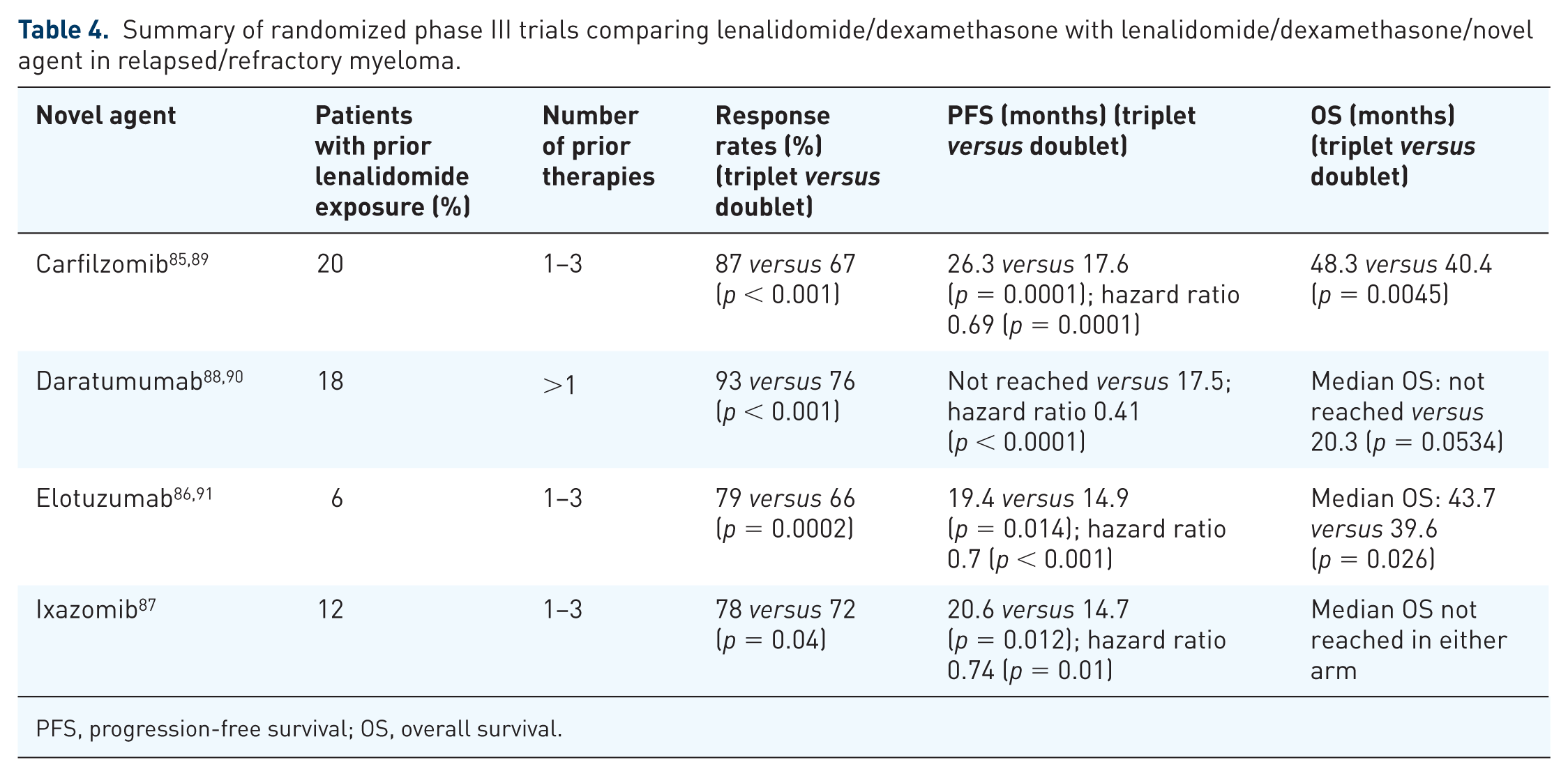
PFS, progression-free survival; OS, overall survival.
Lenalidomide and second primary malignancies
In 2010, preliminary reports from the CALGB 100104 and IFM 2005-02 studies highlighted an increased rate of SPMs in patients receiving lenalidomide compared with placebo maintenance after ASCT. These initial reports included a 2.6%
An analysis of SPMs in the Myeloma XI trial revealed that 104 SPMs were diagnosed in 96 of 2732 total patients. 96 This translated to a cumulative incidence of 3.8% at 3 years in the overall patient population and 2.7% and 5.2% in the transplant-eligible and transplant-ineligible subgroups. Approximately one third of those malignancies were noninvasive skin cancers. Lenalidomide maintenance was associated with an increased cumulative incidence of SPM regardless of whether patients were transplant eligible or transplant ineligible.
In the ECOG E1A06 study, the reported incidence rate (per 100 person years) was 3.46 in the MPT arm and 2.01 in the MPR arm with 10 hematologic SPMs in the MPT arm and four in the MPR arm.
42
In the HOVON87/NMSG18 study, the incidence rates (excluding non-melanoma skin cancers) were 2.9 (MPT) and 2.1 (MPR) per 100 patient years (
In aggregate, the studies thus far have demonstrated that the risk of SPMs associated with lenalidomide therapy are context specific. The recent International Myeloma Working Group review/consensus statement concluded that the highest risk of SPM with lenalidomide is in the context of oral low-dose melphalan, and to a lesser extent, high-dose melphalan. 100 Despite this increased risk, it was noted that the overall risk of death from myeloma remained much higher than the risk of death secondary to an SPM. 100 Thus while lenalidomide/oral melphalan regimens should be avoided, it is clear that the overall benefits achieved from lenalidomide maintenance in the post-ASCT maintenance setting outweigh the risks. However, improved understanding of the molecular mechanisms underlying lenalidomide-induced SPMs post ASCT may allow for the identification of a subset of patients for whom alternative maintenance strategies should be offered.
Future study considerations
Advances in the treatment of myeloma have resulted in increases in OS durations. With each incremental improvement in OS, it has become more challenging to assess the clinical benefit of experimental treatments due a number of reasons, including the large number of patients required to demonstrate a clinically meaningful improvement in OS, the amount of time required to enroll and follow the patients, and study-related costs. Furthermore, there are potential confounding factors related to either crossover from the standard treatment to the experimental treatment per study design or the widespread use of the experimental treatment as subsequent therapy.
It is therefore of critical importance that surrogate endpoints for OS be investigated in myeloma. Appropriateness of a surrogate endpoint may depend upon the line of treatment and the nature of the treatment (cytotoxic
There is increasing interest in the use of MRD status as a surrogate endpoint. It has thus far only been used as an exploratory endpoint (reviewed by Holstein and colleagues
103
). The use of MRD negativity as a primary endpoint could allow for study designs that utilize smaller numbers of patients and shorter follow-up durations, thus increasing the likelihood that the study outcomes would be reported within a clinically relevant time period. While promising, there are a number of uncertainties which have not yet been resolved, including the method of MRD analysis, the timing of the measurement, and whether the endpoint is achievement of MRD negativity at a single time point
Summary of current gaps in knowledge regarding the use of lenalidomide in myeloma.

ASCT, autologous stem cell transplant.
Recommendations
Outside of the context of a clinical trial, the current standard of care in the United States for newly diagnosed patients is RVD induction therapy. While it is likely that for transplant-eligible patients, the eventual new standard of care will be quadruplet therapy (monoclonal antibody + lenalidomide + proteasome inhibitor + dexamethasone), it remains to be determined what the optimal partner should be with lenalidomide/dexamethasone in transplant ineligible patients. In the post-transplant maintenance setting, single agent lenalidomide continued until progression is the clear standard of care based on substantial phase III clinical trial data. However, the optimal maintenance therapy for high-risk patients is not yet determined. Finally, in patients with lenalidomide-sensitive disease, there are a number of very effective triplet combinations available in the relapsed/refractory setting. However, it is not clear whether these regimens are efficacious in patients who are lenalidomide refractory.
Summary
Lenalidomide is now firmly established as the backbone of many induction, maintenance, and relapsed/refractory regimens and has contributed significantly to the overall improvement in outcomes of patients with myeloma. As novel agents such as monoclonal antibodies and other immunotherapeutics are being developed, it will be important to better understand how these new drugs can be incorporated into lenalidomide-containing regimens. Clearly what we have learned thus far is that there is both significant potential for benefit from these novel combinations as well as the potential for unexpected toxicity. Further understanding of the immune-modulating properties and mechanisms of action of lenalidomide and related compounds is therefore critical for the future success of these novel combinations.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
S.A.H. has served on advisory committees for Celgene, Takeda, and Amgen and has received consulting fees from Celgene; V.J.S. has nothing to disclose; P.L.M. has received honoraria from Bristol-Myers Squibb, Celgene, Sanofi-aventis, Takeda, Binding Site, research funding from Celgene, and has served on advisory committees/review panels/board membership for Bristol-Myers Squibb, Celgene, Sanofi-Aventis, Takeda, Binding Site, and Karyopharm.
