Abstract
With the 1997 filing of an investigational new drug application for the first agent to target angiogenesis, bevacizumab entered into phase I clinical trials and has now become a mainstay in the treatment of several cancers. Bevacizumab has changed the treatment approach for cancers due to its efficacy as well as toxicity. This article serves as a review of current efficacy data including recently published safety analyses and the direction of future pharmacodynamic evaluation to hopefully better guide its utilization.
Introduction
In 2004, bevacizumab (Avastin®, South San Francisco, Genentech, CA, USA), a humanized monoclonal antibody directed against circulating vascular endothelial growth factor (VEGF) A, became the first anti-angiogenic agent approved by the US Food and Drug Administration (FDA) for the treatment of cancer. After this initial FDA approval for use as first-line therapy in metastatic colorectal cancer (CRC) patients, it subsequently was approved for use in first-line metastatic, nonsquamous, non-small cell lung cancer (NSCLC), second-line glioblastoma therapy, and in combination with interferon alpha for metastatic renal cell carcinoma (RCC). In 2008, bevacizumab was given accelerated approval for use in combination with paclitaxel in patients with HER2-negative breast cancer. On 16 December 2010, the FDA recommended removal of breast cancer from bevacizumab labeling based on a lack of evidence demonstrating an overall survival efficacy in this disease. Herein, we describe the current role of bevacizumab in cancer therapy with a focus on its emerging adverse event profile and the ongoing search for reliable biomarkers predictive of its efficacy.
Colorectal cancer
Based on promising phase II data, Hurwitz and colleagues investigated the addition of bevacizumab to bolus 5-fluorouracil and irinotecan (IFL) with IFL alone as the comparator arm [Hurwitz et al. 2004]. With an unprecedented median survival of 20.3 months in the experimental arm, the efficacy of bevacizumab when combined with the IFL regimen ushered in a new era in targeted cancer therapeutics. Subsequent integration of bevacizumab into oxaliplatin/ fluoropyrimidine-based regimens resulted in median survival times of up to and beyond 2 years [Hochster et al. 2008] in first-line regimens and 13 months in second-line therapy [Giantonio et al. 2007]. Unfortunately, the introduction of bevacizumab into the adjuvant treatment of patients with stage II and III CRC has not been successful as yet. No statistically significant improvement in disease-free or overall survival was found at the 3-year timepoint when bevacizumab was administered in combination with 6 months of infusional/bolus fluorouracil, leucovorin and oxaliplatin (modified FOLFOX6) and followed by an additional 6 months of bevacizumab alone in the National Surgical Adjuvant Breast and Bowel Project (NSABP) C-08 trial [Allegra et al. 2011]. While a transient effect was apparent in the cohort of bevacizumab exposed patients as evidenced by an initial lower event rate, this effect did not persist beyond the 15-month landmark. Also investigating the activity of bevacizumab in the adjuvant setting, the Adjuvant FOLFOX4 Versus Bevacizumab and FOLFOX4 Versus Bevacizumab, Oxaliplatin, and Capecitabine in Patients With High-Risk Stage II or Stage III Colon Cancer (AVANT) trial enrolled nearly 3500 patients who had undergone resection. Patients were randomized to receive FOLFOX4 alone for 24 weeks or FOLFOX4 or XELOX with bevacizumab for 24 weeks followed by bevacizumab alone for an additional 24 weeks. The primary endpoint was disease-free survival, which was not met with a median duration of follow up of 48 months. Efficacy analysis results favored the chemotherapy alone cohort. At the 2011 Annual American Society of Clinical Oncology (ASCO) meeting, subgroup analysis results were presented and all favored the chemotherapy alone arm. Overall survival analysis has not yet matured. While a transient bevacizumab effect was demonstrated in patients with N2 disease, this effect did not persist.
When utilized in a neoadjuvant regimen including 5-fluorouracil with radiation in 32 patients with rectal cancer, the addition of bevacizumab yielded a 5-year disease-free survival rate of 75% with no local recurrences [Willett et al. 2009]. No grade 4 or 5 toxicities were reported. While postoperative complications including anastomotic leak, delayed healing of incision and wound infection were reported, the relationship of bevacizumab to these morbidities was unclear.
Based on data suggesting the potential for long-term survival with resection of liver metastases in patients with a history of colon cancer [Choti et al. 2002; Fong and Salo, 1999], a phase II, single institution trial combining bevacizumab with oxaliplatin and capecitabine prior to liver resection was performed [Gruenberger et al. 2008]. In this study, 56 patients were enrolled and received bevacizumab therapy until 5 weeks prior to surgical resection. The response rate was 73.2% with a pathologic complete response rate of 8.9%. There was no increase in perioperative adverse events including bleeding or wound complications, and no treatment-related deaths were reported. Postoperative liver regeneration and function were assessed with blood tests and computed tomography (CT) scans, and 98% of those enrolled exhibited full recovery further suggesting that bevacizumab can be safely administered prior to liver resection. A single patient who did not demonstrate normal liver function following bevacizumab exposure had been noted to have concurrent steatohepatitis by pathology from liver resection specimen.
Non-small cell lung cancer
The incorporation of bevacizumab into the treatment of patients with advanced, nonsquamous lung cancer was based on the results from a trial (E4599) conducted by the Eastern Cooperative Oncology Group (ECOG) [Sandler et al. 2006]. Nearly 900 treatment-naïve patients were randomized to receive chemotherapy consisting of carboplatin with paclitaxel for six cycles or the same chemotherapy with the addition of bevacizumab. In the latter arm, patients received bevacizumab maintenance until disease progression or toxicity after completion of the six cycles of chemotherapy with bevacizumab. A statistically significant and clinically meaningful survival benefit (12.3 months versus 10.3 months, p = 0.003) was found in the patients who received bevacizumab with chemotherapy. Patients receiving the combination also demonstrated a significantly higher response rate of 35% compared with 15% in the chemotherapy-only treatment group (p < 0.001). While patients with high-risk features were excluded from trial participation (e.g. squamous cell histology, history of hemoptysis), clinically significant bleeding events were higher in the bevacizumab-treated patients compared with those who received chemotherapy alone (4.4% versus 0.7%; p < 0.001). A subsequent retrospective analysis of the tumor histologies included in the E4599 cohort confirmed a survival advantage imparted by the inclusion of bevacizumab in patients with adenocarcinoma but could not draw any definitive conclusions with regard to other histologies secondary to low patient numbers [Sandler et al. 2010]. In a similar design, the AVAiL trial sought to investigate the utility of bevacizumab in patients with nonsquamous NSCLC in combination with a chemotherapy regimen of cisplatin with gemcitabine (CG) [Reck et al. 2009]. Enrolling approximately 1000 patients, Reck and colleagues tested not only the 15 mg/kg dose as had been used in the E4599 trial but also randomized a cohort of patients to receive a 7.5 mg/kg dose of bevacizumab. The lower dose arm of bevacizumab was included secondary to lack of extensive safety data for the agent’s use in combination with CG. With a primary endpoint of progression-free survival, both bevacizumab containing arms demonstrated a statistically significant advantage compared with the CG arm alone 6.7 months versus 6.1 months (p < 0.003) and 6.5 months versus 6.1 months (p < 0.03) for the low-dose and high-dose bevacizumab containing arms, respectively. Adverse events were similar to those observed in the E4599 trial with no novel safety signals reported. Although the primary endpoint of progression-free survival was achieved, the overall survival failed to demonstrate a benefit in the bevacizumab containing arms when compared to CG plus placebo [Reck et al. 2010]. That patients with an inherently better prognosis (8% were stage IIIB without pleural effusion) were enrolled, the increased use of second-line therapy prior to evidence of disease progression as well as the high percentage of patients (60%) who received subsequent therapy, a rate higher than other contemporary lung cancer treatment trials, were all cited as potential reasons for the disparate results when compared with the positive E4599 outcomes. The subsequently published survival analysis of the AVAiL trial did not yield any new safety events.
Based on data suggestive of synergism between angiogenesis inhibitors and the anti-folate agent, pemetrexed, a phase II study of the combination was undertaken in bevacizumab-naïve patients with advanced NSCLC whose disease had progressed after one line of therapy [Adjei et al. 2010]. The primary objective of the study was to assess the rate of progression-free survival at 3 months, an objective which was met by 57% of the first 42 patients enrolled. Median overall survival was reported to be 8.6 months (95% confidence interval [CI] 6.6–13.7 months). In a phase II study of the combination as first-line therapy, patients received carboplatin and pemetrexed with bevacizumab for six cycles followed by pemetrexed with bevacizumab until toxicity or progression [Patel et al. 2009]. With median progression-free and overall survival rates of 7.8 months (95% CI 5.2–11.5 months) and 14.1 months (95% CI 10.8–19.6 months), respectively, a randomized phase III trial of this combination compared with six cycles of carboplatin/paclitaxel/bevacizumab followed by bevacizumab alone was performed and results are pending.
A phase I/II trial of erlotinib in combination with bevacizumab demonstrated the safety of this combination in patients with relapsed or refractory NSCLC [Herbst et al. 2005]. With 85% of patients demonstrating at least stable disease in this preliminary study, the combination was tested in a phase III, placebo-controlled study. Over 650 patients were randomized in a blinded manner to erlotinib with or without bevacizumab. No overall survival benefit was detected with the addition of bevacizumab. While progression-free survival and overall response rate appeared to favor the bevacizumab group, a prespecified overall survival benefit had to be demonstrated before any secondary analyses could be determined.
Renal cell carcinoma
The inactivation of the tumor suppressor gene von Hippel Lindau, characteristic of RCC with subsequent downstream activation of angiogenesis, has provided mechanistic support for the efficacy of angiogenesis-targeting agents in the treatment of RCC. The prolongation of survival using single-agent bevacizumab in a randomized trial of treatment refractory patients with RCC led to two phase III trials investigating its use in treatment-naïve patients [Yang et al. 2003]. In CALGB 90206, over 700 patients with previously untreated RCC with a clear cell component were randomized to receive interferon (IFN)-α-2b with or without bevacizumab [Rini et al. 2008]. Bevacizumab was administered at a dose of 10 mg per kg every 14 days of each 28-day cycle. IFN-α-2b at a dose of 9 million units thrice weekly was administered alone or at the identical dose and administration in combination with bevacizumab. Results published in 2008 demonstrated a progression-free survival in patients who received the combination therapy with a median progression-free survival of 8.5 months (95% CI 7.5–9.7 months) compared with 5.2 months (95% CI 3.1–5.6 months, p < 0.001) in the IFN-alone group with a near doubling of the response rate in the bevacizumab-treated cohort [Rini et al. 2008]. However, subsequent overall survival analysis did not meet criteria for significance, likely secondary to the treatment of patients with tyrosine kinase inhibitors (TKIs) after disease progression on the trial. In a similarly designed trial known as AVOREN, nearly 650 patients with advanced RCC were randomized to receive either bevacizumab plus IFN or IFN alone in identical dosage and administration schedule as in the CALGB 90206 trial [Escudier et al. 2010]. As with the CALGB trial, overall survival analysis yielded no significant difference between the two arms, an outcome also confounded by the availability of multiple active agents upon progression. At the 2011 ASCO Genitourinary Cancers Symposium, Likun and Ba presented a meta-analysis of the CALGB and AVOREN trials and did demonstrate a statistically significant overall survival benefit for the patients who received bevacizumab containing therapy (hazard ratio [HR] 0.86, 95% CI 0.76–0.97, p = 0.02) [Likun and Ba, 2011].
In a recent retrospective analysis of the AVOREN trial, subsequent survival data of patients who went on to receive TKI therapy after disease progression on either bevacizumab plus IFN or IFN alone was undertaken [Bracarda et al. 2011]. Median overall survival of patients who received any TKI was 38.6 months in those who had received bevacizumab plus IFN versus 33.6 months in the IFN plus placebo treated patients (HR 0.80, 95% CI 0.56–1.13, p = 0.203), a finding which did not reach statistical significance. Although not sufficiently powered or prospectively defined, this analysis suggested ongoing benefit from anti-angiogenesis exposure even with subsequent therapy.
Glioblastoma
Based on the demonstration of high VEGF expression in glioblastoma (GBM) cells as well as in vivo bevacizumab activity in a murine model, a phase II trial was undertaken in patients with recurrent disease [Vredenburgh et al. 2007]. The response rate of 57% in the 35 patients enrolled with nearly half of patients progression free at 6 months was a significant improvement over historical controls with published response rates of approximately 10% and 6 month progression-free interval rates of approximately 20% [Ballman et al. 2007; Yung et al. 2000]. A more extensive, multicenter, phase II randomized, noncomparative trial involving 167 patients supported bevacizumab’s approval by the FDA for use in patients with recurrent glioblastoma [Friedman et al. 2009]. In this trial, patients in first or second relapse who had progressed on temozolomide were randomized to receive either bevacizumab (10 mg/kg) alone or in combination with irinotecan in every 2-week cycles. Objective response rates of 28% in the single-agent arm and 38% in the combination arm were reported. Six-month progression-free and overall survival rates were reported as 43% and 50% and 9.2 months and 8.7 months in the bevacizumab alone and combination arms, respectively. Treatment was well tolerated with hypertension, seizure, neutropenia and fatigue being the most frequently reported events. Of the five patients with intracranial hemorrhage, one was grade 4 and the remaining cases were grade 1 or 2. Updated survival data presented at the annual ASCO meeting in 2010 demonstrated a 30-month survival rate in 16% of patients treated with bevacizumab plus irinotecan with no new safety signals reported [Cloughesy et al. 2010]. In an effort to investigate the use of bevacizumab as part of first-line therapy, a phase II trial was undertaken in 70 patients with newly diagnosed GBM. Bevacizumab was administered biweekly in combination with daily temozolomide and radiation therapy [Lai et al. 2010]. Outcomes were compared with a control cohort of patients who received temozolomide with radiation as front-line therapy followed by bevacizumab at the time of progression. An improved progression-free survival was noted in the patients receiving bevacizumab as part of first-line treatment; however, this did not translate into an overall survival benefit when compared with the control cohort.
Breast cancer
On 22 February 2008, bevacizumab gained accelerated approval from the FDA for the treatment of patients with HER2-negative, metastatic, treatment-naïve breast cancer in combination with paclitaxel. This accelerated approval was based on results from the E2100 trial in which patients who received the combination of agents demonstrated a statistically significant prolongation in progression-free survival when compared with patients who received paclitaxel alone, although no overall survival advantage was noted [Miller et al. 2007]. Patients treated with the combination regimen demonstrated a median progression-free survival of 11.8 months versus 5.9 months in the patients treated with paclitaxel alone (HR 0.60, p < 0.001). Median overall survival was 26.7 versus 25.2 months (HR 0.88, p = 0.16). With disappointing results from subsequent trials investigating the use of bevacizumab in combination with other chemotherapy agents in this setting (see Table 1), the FDA recommended removal of the breast cancer indication from product labeling on 16 December 2010 after the Oncology Drug Advisory Committee (ODAC) voted 12 to 1 in July 2010 in favor of such action. The FDA granted a public hearing in June 2011 after Genentech/Roche submitted an application to appeal this ruling. After a 2-day hearing, ODAC unanimously voted again to uphold the FDA’s earlier decision to remove the accelerated approval of bevacizumab in combination with paclitaxel in patients with HER2-negative advanced disease. A final decision by FDA commissioner Margaret Hamburg is awaited.
Bevacizumab trials in HER2-negative metastatic breast cancer.
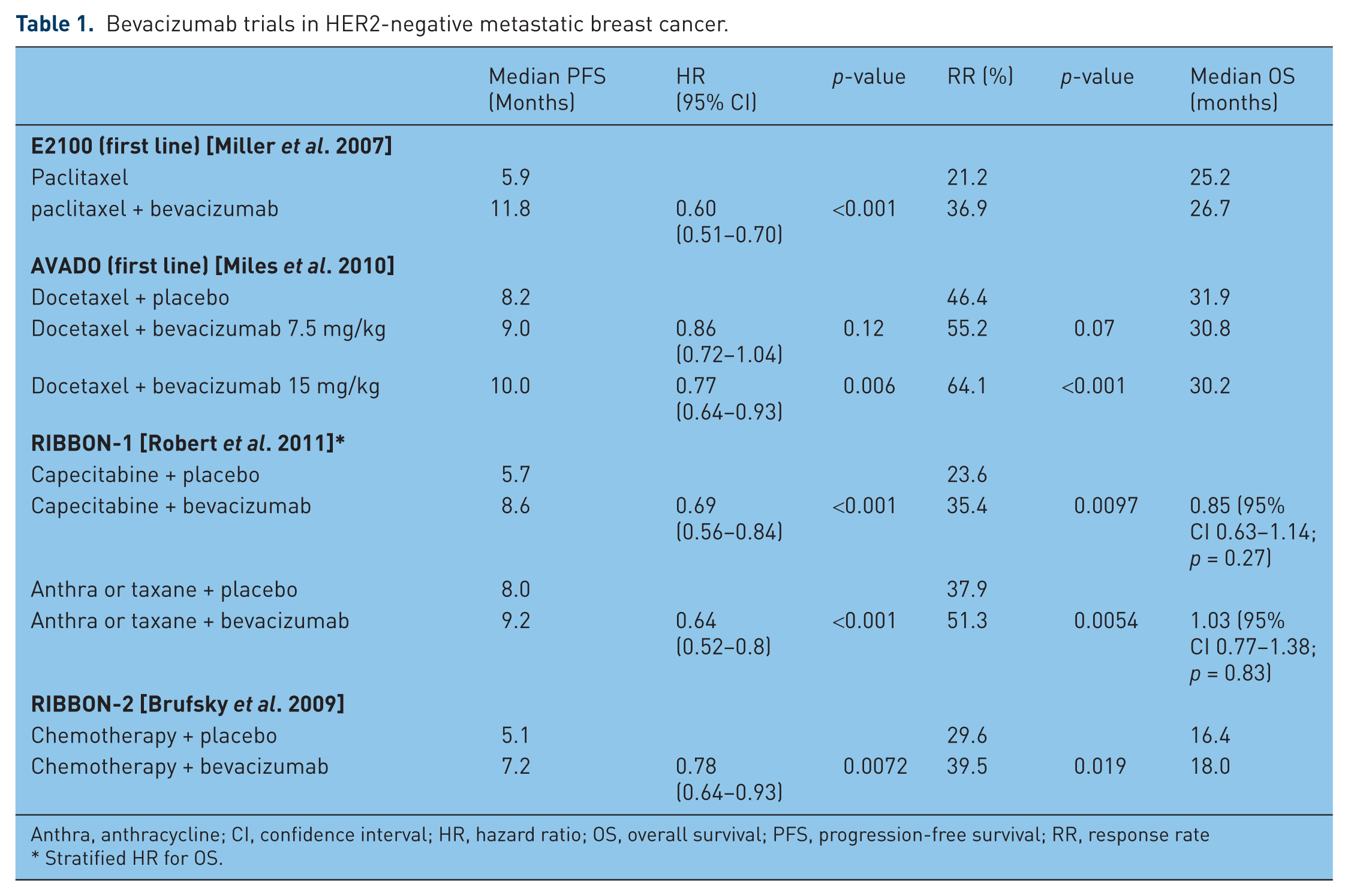
Anthra, anthracycline; CI, confidence interval; HR, hazard ratio; OS, overall survival; PFS, progression-free survival; RR, response rate * Stratified HR for OS.
Histologies without bevacizumab activity
While bevacizumab has demonstrated activity in a variety of tumor histologies, two prominent examples of cancers in which it has not shown efficacy based on current data include prostate cancer and pancreatic cancer. Based on clinical data demonstrating increasing levels of VEGF in the plasma and urine of patients with metastatic prostate cancer (both independent predictors of decreased survival) as well as the correlation of increased VEGF expression with progression of disease [Bok et al. 2001; Duque et al. 1999; Ferrer et al. 1997; George et al. 2001], CALGB 90006 was undertaken [Picus et al. 2011]. This multi-institution study enrolled 79 patients with castration-resistant prostate cancer. Subjects were treated with a combination of estramustine, docetaxel and bevacizumab. Activity was promising with three quarters of patients demonstrating a 50% or greater prostate-specific antigen (PSA) decline and 59% of patients with measurable disease exhibiting a partial or complete response. The median overall survival of 24 months lent further support to its subsequent investigation. Toxicity, however, was not minimal with 25% of patients experiencing grade 3 or 4 fatigue and a thromboembolic complication rate of 9%.
Based on these phase II results, CALGB 90401 was initiated, which was a randomized, double-blind, placebo-controlled trial of docetaxel and prednisone with or without bevacizumab [Kelly et al. 2010]. Presented at the annual ASCO meeting in 2010, results were disappointing. Over 1000 patients with metastatic, castration resistant, treatment-naïve prostate cancer were enrolled. The study did not meet its primary endpoint to detect an increase in median survival from 19 months to 24 months. Patients who received bevacizumab demonstrated a statistically significant increase in median progression-free survival at 9.9 months compared with 7.5 months in the docetaxel/prednisone alone arm (HR 0.77, 95% CI 0.68–0.88, p < 0.0001). A statistically significant increase in the rate of treatment-related death was observed in the bevacizumab containing arm (4.4% versus 1.1%, p = 0.0014). In their presentation, investigators stressed the longer than expected survival rate in the docetaxel/ prednisone alone arm as possibly contributing to the observed results.
Ovarian cancer
Although not approved for use in patients with ovarian cancer, the use of bevacizumab in this gynecologic malignancy has been the topic of extensive investigation. Preclinical and clinical studies have supported the role of VEGF in disease progression and ascites formation with evidence demonstrating its overexpression as prognostic for inferior outcomes [Ferrara, 1999; Gasparini et al. 1996; Paley et al. 1997; Yoneda et al. 1998]. In a phase II trial conducted by the Gynecologic Oncology Group (GOG), single-agent bevacizumab in patients with refractory epithelial ovarian cancer or primary peritoneal cancer yielded median progression-free survival of 4.7 months and median overall survival of 17 months [Burger et al. 2007]. Of the 62 patients treated, 13 demonstrated a clinical response with 25 patients surviving beyond 6 months without progression. Also published in 2007, Cannistra and colleagues published their results of single-agent bevacizumab in a more heavily pretreated population of patients with ovarian cancer or primary peritoneal cancer [Cannistra et al. 2007]. The median progression-free survival and overall survival were 4.4 months and 10.7 months, respectively. Of the 44 patients who received bevacizumab, 7 patients (15.9%) had a partial response.
Both presented at the annual ASCO meeting in 2011, the OCEANS trial and ICON7 lent further support to the activity of bevacizumab in patients with advanced ovarian cancer. The OCEANS trial enrolled 484 patients with recurrent, platinum-sensitive ovarian cancer [Aghajanian et al. 2011]. Patients were randomized to carboplatin with gemcitabine and either bevacizumab or placebo for six cycles followed by bevacizumab or placebo until disease progression or toxicity. The primary endpoint of the trial was primary progression-free survival as by investigator assessment. Secondary endpoints included overall survival. Median progression-free survival in the bevacizumab containing arm was reported to be 12.4 months (95% CI 11.4–12.7 months) compared with 8.4 months (8.3–9.7 months) in the placebo arm (HR 0.484, 95% CI 0.388–0.605, p < 0.0001). Review of results by an independent review committee was concordant. Overall survival events were not yet mature.
The ICON7 study sought to determine the activity of bevacizumab in combination with carboplatin/paclitaxel in patients with newly diagnosed epithelial ovarian, primary peritoneal or fallopian tube cancer [Kristensen et al. 2011]. As requested by regulatory authorities, an interim overall survival analysis was performed and presented at the 2011 ASCO annual meeting. Initially presented at the 2010 European Society of Medical Oncology (ESMO) meeting, a progression-free survival benefit was demonstrated with a trend for overall survival improvement. Over 1500 women with high-risk early stage disease or advanced disease were enrolled. Patients were randomized to receive six cycles of carboplatin with paclitaxel with or without concurrent bevacizumab followed by bevacizumab to complete 1 year of therapy. The results presented demonstrated a continued progression-free survival advantage with a trend in overall survival improvement. Final overall survival results are anticipated in 2013.
Bevacizumab toxicity
While early trials of bevacizumab brought much excitement to the oncology community, this enthusiasm was tempered with the observation of rare, but serious adverse events. In AVF0757g, a randomized phase II study of patients with NSCLC treated with carboplatin and paclitaxel with or without bevacizumab, six patients developed major, life-threatening hemorrhage [Johnson et al. 2004]. Four of the six patients were noted to have squamous cell carcinoma, a finding which led to the halting of bevacizumab testing in patients with squamous histology. Hypothesizing that delayed inclusion of bevacizumab until after initial cycles of chemotherapy are administered may prevent pulmonary hemorrhage; the BRIDGE study investigated such a treatment approach in patients with predominantly squamous histology. [Hainsworth et al. 2011]. Patients with cavitary thoracic lesions, gross hemoptysis in the past 3 months, and tumor impingement or involvement of major vessels or vascular structures, were excluded from trial participation. The primary endpoint of the trial was the incidence of grade 3 pulmonary hemorrhage in patients treated with two cycles of carboplatin and paclitaxel with subsequent addition of bevacizumab for cycles three through six and followed by bevacizumab alone for cycle seven and onward. The incidence of grade 3 or greater pulmonary hemorrhage was 3.2% in patients who received at least one dose of bevacizumab. The incidence of any grade pulmonary hemorrhage was 6.5%. Pulmonary hemorrhage in these patients appeared to be associated with the development of cavitation subsequent to treatment initiation. Although no definitive conclusions could be drawn regarding the safety of bevacizumab in patients with squamous histology, the rate of pulmonary hemorrhage was lower than in prior series when this delayed bevacizumab approach was utilized. Reliable clinical markers to predict for bevacizumab toxicity remain to be defined such that use of bevacizumab in patients with lung cancer of squamous histology remains investigational.
Safety assessment in tumor-specific trials including lung cancer and breast cancer were recently published and incorporate data from a broad range of treated patients. The safety of bevacizumab in the treatment of lung cancer patients in a community setting was the subject of a phase IV study entitled SaIL, which was initiated in 40 countries worldwide [Crino et al. 2010]. Patients with nonsquamous, NSCLC received six cycles of bevacizumab-containing therapy with investigator-chosen first-line chemotherapy. The primary objective of the study was assessment of bevacizumab safety in combination with first-line chemotherapy with secondary objectives including efficacy analysis as well as safety outcomes in patients who developed CNS metastases during or within 6 months of bevacizumab therapy. The rate of adverse events was as expected based on prior clinical trials. The incidence of febrile neutropenia, grade 3 neutropenia, thrombosis, and at least grade 3 hypertension were all less than 10%. The incidence of clinically significant pulmonary hemorrhage (≥grade 3) was 1% and only 5 of 281 patients who developed CNS metastases suffered from any grade of cerebral hemorrhage. Median overall survival was 14.6 months, which corroborated prior results in a population of patients with mainly adenocarcinoma. This study confirmed the safety of bevacizumab in a population more representative of that encountered in daily clinical practice.
Also with the intent of determining safety of bevacizumab in a patient population typically encountered in an oncology practice, the primary endpoint of the ATHENA trial was safety of bevacizumab in combination with first-line therapy in patients with metastatic breast cancer [Smith et al. 2011]. In this study of 2251 patients, bevacizumab was combined most commonly with taxane-based therapy. No new safety signals were observed. Of the 205 patients who developed CNS metastases, 2.0% of these patients developed intracranial bleeding during study follow up. Incidence of bevacizumab-related toxicities were similar to previously reported rates. The median time to progression of 9.1 months observed in this trial supported median progression-free survival reported in other phase III trials.
Ranpura and colleagues set out to determine the incidence of fatal adverse events in a meta-analysis of all published randomized clinical trials across a variety of tumor types [Ranpura et al. 2011]. Over 10,000 patients were included from trials in which bevacizumab with chemotherapy was compared with chemotherapy alone. The incidence of fatal events with bevacizumab use was 2.9% (95% CI 2.0–4.2%) resulting in an absolute risk increase of 0.7% (relative risk [RR] 1.33, 95% CI 1.02–1.73, p = 0.04). The association of bevacizumab with higher treatment-related mortality was significant for type of chemotherapy agent, but not tumor type or bevacizumab dose. Of the chemotherapy agents utilized, taxane-based therapy was most highly associated with fatal adverse event with a RR of 3.49 (95% CI 1.82–1.66). Of the fatal events associated with bevacizumab, those with highest rates were hemorrhage followed by neutropenia. Although the incidence of treatment-associated mortality was not an endpoint in the individual studies, this meta-analysis provided the most extensive analysis of bevacizumab safety to date and confirmed the importance of not only early monitoring for adverse events but also exercising caution in patient selection.
That bevacizumab is associated with an increased risk of bleeding and the inherent elevated risk of venous thromboembolism (VTE) in patients with malignancy has led to concern for the safety of anticoagulation in the patients undergoing active treatment with bevacizumab. Most clinical trials of bevacizumab exclude patients who are receiving anticoagulation from participation, thus limiting existing data on the safety of therapeutic anticoagulation in this treatment setting. Leighl and colleagues recently published results of a retrospective analysis of three randomized, placebo-controlled trials in which patients who developed VTE after initiation of protocol therapy were allowed to continue on trial [Leighl et al. 2011]. The trials included in the analysis included two trials of bevacizumab use in CRC and one trial in lung cancer. The overall risk of bleeding in the pooled placebo-treated group was 4.1% versus 4.2% in the pooled bevacizumab-treated cohort. No fatal bleeding events were reported in any of the patients who received therapeutic anticoagulation. Similar results from the NSCLC patients treated on the phase IV SaIL trial revealed a bleeding rate of 17.2% (none grade 3 or higher) in patients who received therapeutic anticoagulation compared with a 17.0% incidence in the overall bevacizumab-treated population. These results have provided preliminary data suggesting the feasibility of anticoagulation for VTE in the setting of bevacizumab treatment.
Bevacizumab pharmacodynamics
The disappointing efficacy results of bevacizumab trials in advanced prostate and pancreatic cancer, in adjuvant colon cancer treatment and in the advanced breast cancer setting emphasize the importance of developing predictive markers to determine which patients will not respond and can be spared the economic and health-related costs of bevacizumab treatment. While the development of hypertension has been proposed as a potential predictor of clinical activity [Maitland et al. 2006; Scartozzi et al. 2009], in a meta-analysis of over 12,000 patients conducted to assess the risk of hypertension in bevacizumab treated patients, no improvement in clinical outcome was associated with the development of hypertension in the studies analyzed [Ranpura et al. 2010]. Based on the hypothesis that genotypic differences in VEGF predict for breast cancer prognosis, an analysis of E2100 outcomes based on differential VEGF and VEGFR-2 expression was undertaken [Schneider et al. 2008]. Analyzed from paraffin-embedded tissue, those subjects with VEGF-2578 AA and VEGF-1154 AA displayed median overall survival times of 37.0 (p = 0.035) and 46.5 (p = 0.047) months, respectively, in the experimental arm, a result statistically significantly longer when compared with the control arm. The genotypes associated with the lowest risk of grade 3 or 4 hypertension included VEGF-634 CC and VEG-1498 CC. These two genotypes which appeared to impart a resistance to the development of hypertension were mutually exclusive of those portending an improved outcome with experimental therapy suggesting that development of hypertension may be a pharmacodynamic marker of efficacy. When analyzed for treatment outcome based on the development of hypertension, a statistically significant improvement in median overall survival time was detected in those patients who demonstrated an increase in blood pressure. No association between VEGF protein expression by immunohistochemistry and outcomes was found.
Analysis of single-nucleotide polymorphisms (SNPs) from germline DNA of metastatic pancreatic cancer patients who were treated with gemcitabine and erlotinib with or without bevacizumab as part of the AVITA trial revealed a differential sensitivity to anti-angiogenesis therapy [Lambrechts et al. 2009]. Median survival increased from 4.8 months to 10.3 months in patients with a particular SNP in the VEGFR-1 gene coding for the essential receptor tyrosine kinase domain. Of interest, no association with the above-described VEGF-2578 AA polymorphism and survival prolongation was detected in this data set from pancreatic cancer patients. With the hopes of developing an easily accessible biomarker, circulating VEGF levels have been hypothesized as predictive of bevacizumab efficacy. However, in an analysis of over 1800 patients from four randomized trials in colorectal, lung and renal cell carcinoma, measurement of plasma VEGF predicted for worse prognosis but was not predictive of anti-angiogenesis treatment response.
Trials such as the Biomarker-integrated Approaches of Targeted Therapy for Lung Cancer Elimination (BATTLE) in which core biopsies are performed in order to assess an individual patient’s tumor profile, may expedite the discovery of predictive biomarkers [Kim et al. 2011]. A front-line study, BATTLE-FL, is now open and is attempting to study which biologic drug with chemotherapy may benefit patients the most as initial therapy.
Conclusion
Bevacizumab is a safe and effective drug utilized in several cancers. The quest for reliable predictive markers remains of utmost importance as its indiscriminate use presents an untenable situation when taking into consideration survival benefits, toxicity and cost. Although evidence exists to suggest that prolonged therapy with bevacizumab yields improved outcomes as somewhat supported by ovarian and lung cancer data, the key to the agent’s longevity as a viable treatment modality lies in current and future endeavors to appropriately identify potentially responsive patient subpopulations.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
The authors declare no conflict of interest in preparing this article.
