Abstract
Heparin and heparan sulfate, a variety of negatively charged highly sulfated polysaccharides, can influence the biological functions of human bone morphogenetic proteins (BMPs). Notably, BMPs control numerous essential biological activities and processes, such as bone formation, bone turnover, brain development, tumor initiation, and progression. BMPs also enhance the repair of bone tissue injuries and are used in bone remodeling alongside implantable prosthetic devices. BMPs either potentiate or inhibit the growth of cancer stem cells (CSCs). This dual biological effect appears to depend upon the cell type, underlying cytogenetic and biochemical aberrations in various distinct malignancies. Similarly, heparins may modulate CSCs positively or negatively through BMPs. The primary aims of this review are to investigate whether heparin prophylaxis would likely stimulate the propagation of a chemotherapy-resistant subpopulation of CSCs and aggravate tumor response to treatment, and result in tumor expansion, tumor recurrence and metastasis. The secondary aim is to document whether such detrimental effects surpass their beneficial effects as anticoagulants in primary bone cancers such as osteosarcoma. The current state of scientific knowledge based on key published articles from the standpoint of rigidity of data and identification of data gaps is discussed.
Keywords
Introduction
The biological impact of heparin prophylaxis on cancer stem cells (CSCs) and bone tumorigenesis has not previously been reported. Most studies have focused on potential beneficial health and antitumor effects of the heparins in nonorthopedic tumors, rather than both safety and efficacy. Heparin and other related heparan-sulfate molecules are increasingly being recognized as important modulators of many signaling pathways. Heparin-like molecules may also exert their effects on cancer and/or CSCs by modulating the signaling of growth factors that interact with extracellular matrix proteins located in the microenvironment of bone and other tumors, as discussed (later) in the text. In this review, the roles of bone morphogenetic proteins (BMPs) were evaluated because of their potential interactions with heparan-sulfate proteoglycans and stem cells in the pathogenesis of bone cancer.
Heparin use in patients with orthopedic cancer is generally intended to block the formation of blood clots triggered by surgical procedures and device implants, curtail cancer-associated vascular thrombosis, and ameliorate tumor metastasis. As a result, people expect to see significant event-free survival benefits. A review of the literature published between 1980 and 2001 concludes that the effects of heparins on cancer progression are uncertain because heparins elicit diverse biological activities [Smorenburg and van Noorden, 2001]. To date, this uncertainty is largely unresolved. The authors in a recent review concluded the following: ‘the anticoagulants (such as heparins and warfarin) are more effective in inhibition of stages of the metastatic cascade than in the influence on primary tumors’ [Bobek and Kovarik, 2004]. Others stated that heparin has direct antineoplastic effect and asserted that ‘Improved survival cannot simply be explained by a reduction in fatal pulmonary embolism’ [Cunningham et al. 2009]. Although these explanations are logical and may be valid, they are an incomplete collection of thoughts and observations remotely related to the plausibility of an exogenous heparin affecting vital signaling pathways in CSCs.
BMPs are a group of signal transduction ligands that belong to a larger family of growth factors, including transforming growth factor beta cytokines. Some BMPs, such as BMP-2 and BMP-7, are available as treatment options in orthopedics. BMPs may promote or inhibit tumorigenesis and/or progression depending on the tumor types. It is possible that BMPs’ effects on bone cancers are distinct from other types of tumors, including solid tumors, because BMPs regulate the osteogenic differentiation pathway. BMPs control a variety of cellular processes such as proliferation, differentiation, apoptosis, and migration of cancer cells, as well as new bone formation during embryogenesis and postnatal bone development (reviewed by Alarmo and colleagues [Alarmo and Kallioniemi, 2010]). The question is whether the heparin pressure increases a BMP-driven induction of tumor growth and/or metastases in osteosarcoma.
The premises of this review are that heparins bind to BMPs and other cellular growth/signaling proteins that stabilize and modify their functions, which may in turn modulate the biological properties of cancerous cells, be they the least differentiated cancer stem-like cells or the most differentiated committed progenitor cells, and thus potentially affect clinicopathological parameters and outcomes. Heparins exhibit positive and negative effects on tumorigenesis; the long-term survival disadvantage of patients with primary bone cancers may partly be rooted in heparin use for anticoagulant benefits. The objective includes a risk-to-benefit profile assessment of heparins in primary bone cancers, such as osteosarcoma, based on a weight-of-evidence approach. The current findings and knowledge gaps in the plausible impact of exogenous heparins on tumorigenesis are discussed: from CSCs to self-renewal capacity, growth, and expansion of residual tumor to formation of micrometastasis in primary bone cancers.
Biological observations, publications, and assessments
The PubMed database was searched and the selected articles were reviewed to evaluate whether heparin prophylaxis stimulates the propagation of chemotherapy-resistant subpopulations of CSCs through osteogenic BMPs, aggravates tumor response to treatment, and/or leads to tumor expansion, tumor recurrence and metastasis. The selected original research articles and several key reviews mostly on cells in cultures and xenograft models in animals are cited because controlled clinical trials on this particular topic on human subjects are unavailable. Some articles were excluded due to scope constraints or if the explanation is widely known. In addition, key findings on other signaling molecules that are associated with bone cancers, especially in primary bone malignancies, that appear to demonstrate a causal relationship with pathological growth (osteoblastic, osteogenic) and destructive (osteolytic, osteosclerotic) outcomes were reviewed, and discussed below in this article.
Cancer stem cells and bone morphogenetic proteins
Appendicular sarcomas, including primary bone cancers, for example osteosarcoma, by definition manifest various abortive steps in the differentiation of mesenchymal stem cells (MSCs) caused by genetic and epigenetic changes. Appendicular sarcomas represent the least differentiated stem-like cells and/or most differentiated ‘atypical’ osteogenic tumor cells with characteristics partially shared with committed progenitor-like cells [Tang et al. 2008]. These cells arise from a putative reservoir of CSCs in vivo that, presumably originating from mutated MSCs or susceptible osteoprogenitor cells, may survive and develop into a pool of treatment-resistant CSCs capable of transitioning to local relapse and/or metastasis [Siclari and Qin, 2010]. Moreover, malignant CSCs in primary bone cancers may be defective in their differentiation pathways and produce ectopic osteoids or bone-like substances [Siclari and Qin, 2010; Luo et al. 2008; Tang et al. 2008].
Some BMP subtypes either promote or inhibit tumor cells. The dual roles of BMPs in malignancies, and their potential to modulate the growth and/or mobilization of stem cells, are largely unsolved. BMP-4 and BMP-7 were found to induce the differentiation of brain tumor stem cells and inhibit human glioblastoma tumor growth in a mouse intracerebral xenograft model. Moreover, BMPs exhibit antitumor effects in human brain cancer cells [Chirasani et al. 2010; Piccirillo et al. 2006]. Conversely, BMP-2 substantially enhances the viability of most-differentiated osteoinductive osteosarcoma tumor cells (e.g. SaOS-2) [Turhani et al. 2007], and migration of both murine osteosarcoma [Sotobori et al. 2006], and human chondrosarcoma cells in cultures [Fong et al. 2008]. In rat osteosarcoma cells (ROS 17/2/8), BMP-7 inhibits cell proliferation [Maliakal et al. 1994] whereas BMP-12, which induces tendon-like tissues, enhances proliferation of these cells [Furuya et al. 1999]. Furthermore, several different types of osteogenic BMPs (BMP-2, BMP-6, BMP-7, and BMP-9) promote the growth of the least-differentiated stem-like subpopulation of human osteosarcoma tumor cells (e.g. MG63) in intratibially injected athymic mice [Luo et al. 2008].
Consistent with these results, histopathological examination of other human malignancies and functional imaging studies on tumor xenografts in animals support the notion that BMPs promote the invasion of cancerous cells, from prostate and breast to skeletal tissues, and subsequent development of the tumor-induced pathological bone lesions [Alarmo and Kallioniemi, 2010; Rentsch et al. 2009; Katsuno et al. 2008]. In addition, studies using a human breast tumor model in mice suggest that BMP-2 promotes bone metastasis [Takada et al. 2003]. In contrast, treatment of a subpopulation of human malignant osteosarcoma (OS99-1) CSCs with BMP-2 downregulates stem-cell markers (Oct3/4, Nanog, and Sox-2) and inhibits their growth in a xenograft model in mice [Wang et al. 2011]. These conflicting observations are reminiscent of diverse characteristics of osteosarcoma tumor cells and the opposing biological effects of heparins.
Heparins and bone morphogenetic proteins
BMPs contain a heparin-binding site that modifies their biological activities [Ruppert et al. 1996]. Soluble heparins chemically react with BMPs to form stably bonded heparin–BMP molecular complexes. All negatively charged heparin polysaccharides, including heparan sulfate and related pharmaceutical grade, low-molecular-weight heparins (LMWHs), are a highly sulfated group of linear sugar glycosaminoglacans (GAGs) (Figure 1). Heparins can bind to the basic amino acid stretches residing on BMPs. In BMP-2 and BMP-4, the basic amino acid clusters are located outside of their conserved receptor binding sites. Heparin, which binds to both the active homodimer (BMP-2 or BMP-4) and less active heterodimer (BMP-2/6 or BMP-2/7) forms of BMPs, enhances the activity of BMPs ‘by continuously serving the ligands’ from the extracellular side (the culture media) to their signaling receptors located in the cell membranes [Takada et al. 2003]. The heparin attachments to BMPs increase the stability of BMPs and protect them from degradation [Zhao et al. 2006; Ruppert et al. 1996]. Together, these observations suggest that heparin-stabilized BMPs may increase their bioavailabilities in both bone tissue and the tumor microenvironment.
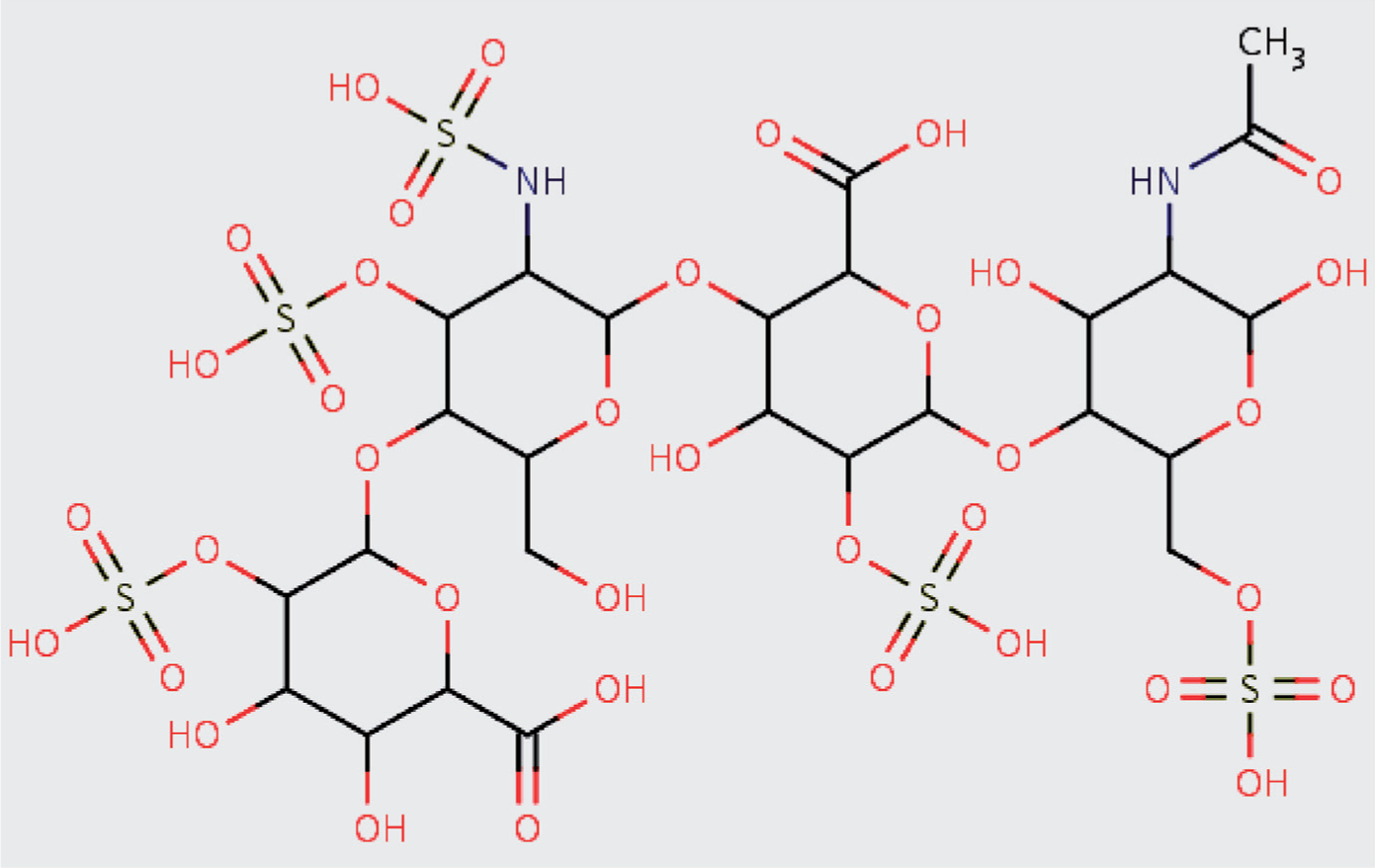
A representative monomeric chemical structure of an almost fully sulfated glycosaminoglycan (GAG) heparin and low-molecular-weight heparin (LMWH) [the image is reproduced with permission from DrugBank (DB01225), Wishart et al. 2008; Hirsh and Raschke, 2004] indicates two attached asymmetrical disaccharide units and five sulfur groups (S-domains). The molecular backbone consists of repeating hexose (six-carbon sugar)/glucuronic acid (GlcA) and hexosamine/N-acetyl glucosamine (GlcNAc) residues. The molecular chain is enzymatically modified by de-N-acetylation and N-sulfation of selected GLcNAc residues to form N-sulfoglucosamine (GlcNS), epimerization of certain GlcA residues to iduronic acid (IduA) and addition of sulfate groups at the 2-O position of GlcA/IduA and 6-O and seldom at 3-O position of GlcNSA/GlcNAc. Distinct pharmaceutical-grade LMWH agents vary from each other in their structural features and extent of modifications, including the types of sugars, geometry of linkages, and/or density/position and patterning of sulfations within the polymeric chains.
Further, in the presence of BMP-2, exogenously added heparin increases the differentiation of cultured osteoblasts in a dose-dependent manner [Takada et al. 2003]. In line with this, simultaneous treatment with heparin and BMP-2 may induce ectopic bone formation in vivo [Zhao et al. 2006]. These studies demonstrated that not only heparin, but also various types of negatively charged polysaccharides (dextran sulfate, heparan sulfate, and various sulfated LMWH derivatives) might tip the balance in favor of osteoblast-differentiating activity. The effects on inhibition of cell proliferation were modest, though considerable. Others indicate that heparins can inhibit the proliferation of both normal osteoblasts and human osteosarcoma cells [Nikitovic et al. 2005]. Nonetheless, these test systems include a few established osteoblast cell lineages, and exclude cancer drug studies on treatment naïve and resistant high-grade osteosarcoma cells.
Anticoagulant heparins and cancer
Exogenous heparins can bind to numerous different signaling proteins that are vital to carcinogenesis and local recurrence as well as bone remodeling and distant metastasis. Many different fractionated heparin products, including the sulfated heparan or pharmaceutical grade LMWHs, such as dalteparin and enoxaparin, have increasingly replaced the widely used coumarin derivatives (warfarin) in thromboprophylaxis of major orthopedic surgeries in many countries worldwide [Ettema et al. 2009]. The international guidelines with respect to the prevention of blood clotting in patients undergoing orthopedic surgery are evolving as new data, insights, and better anticoagulants are developed [Struijk-Mulder et al. 2010; Hull et al. 2009]. The most frequently noted concerns in previous assessments and guidance documents have focused on bleeding side effects rather than all other risk-to-benefit parameters.
Cancer is associated with an increased risk of both arterial and venous thromboembolism complications when heparins are used for their antithrombotic and potential anticancer effects [Falanga and Marchetti, 2007]. A recent epidemiological survey points out that the appropriate practices of thromboembolism prophylaxis in accordance with the American College of Chest Physicians guidelines are poorly implemented in cancer patients [Amin et al. 2008]. The sufficiency of the evidence-based guidelines for thromboprophylaxis for all types of cancers, especially for primary bone cancers, with respect to risk-to-benefit profile of heparins is questionable.
Bone morphogenetic proteins in primary bone cancer
The carcinogenesis of primary bone cancers, for example osteosarcoma, results in aberrant repetitive cycles of synthesis and destruction of local bone and surrounding tissues. Distinct human osteosarcoma cells in cultures appear to selectively express multiple subtypes of BMPs, their receptors, and many other auxiliary osteogenic factors [Yu et al. 2004; Anderson et al. 2002; Gobbi et al. 2002; Raval et al. 1996], but only extracts of the osteoinductive cells form ectopic new bone in athymic/nude mice [Yu et al. 2004; Gobbi et al. 2002]. Ample amounts of BMP-1, BMP-3, BMP-4 are expressed in malignant osteoinductive SaOS-2 cells, but not in nonosteoinductive U-2OS cells [Anderson et al. 2002; Raval et al. 1996]. Using sensitive reverse transcriptase polymerase chain reaction and in situ hybridization assays, BMP-2/BMP-4 were also readily detected [Yu et al. 2004]. These data suggest that more than one BMP subtype, probably in concert with unknown osteoinductive factor(s), may influence the disease process and outcome.
Consistent with these data, BMPs are active in cancerous cells of animal models of osteosarcoma. For instance, an elevated expression of BMP-2 correlated with the metastatic phenotype of murine osteosarcoma (M2K2) cells [Weiss et al. 2006]. One should recognize that while a subset of different BMPs (BMP-2/BMP-4/BMP-6/BMP-7/BMP-9) induces osteogenic differentiation of the (normal) mesenchymal stem-like cells [Kang et al. 2009], certain key cell-cycle transcriptional regulators (RB and P53) and differentiation pathways may be defective in malignant osteosarcoma cells [Luo et al. 2008; Tang et al. 2008]. Furthermore, suppression of BMPs’ activities by BMP antagonists (such as noggin) leads to a marked apoptosis in cultured tumor cells [Kang et al. 2009]. These findings suggest BMPs and other interactive elements are operative amid tumorigenesis and may be essential to the disease pathogenesis in primary bone cancer.
Discussion
The mechanism of action is unknown or poorly understood for many marketed pharmaceuticals, which is acceptable as long as the drugs work and their adverse effects are tolerated. Earlier publications pointed out that heparins can interfere with the adherence of cancer cells to the vascular endothelium and have both inhibitory and stimulatory effects on angiogenesis [Smorenburg and van Noorden, 2001]. Could anticoagulant heparins positively influence the outcome of disease in all varieties of bone cancers? To the best of our knowledge, the short answer is that we do not know. Could anticoagulant heparins negatively influence the outcome of disease in various primary bone cancers such as osteosarcoma? The answer is maybe.
On the up side, the anticoagulant benefits of heparins seem undoubtedly associated with survival benefits in humans [Falanga and Marchetti, 2007; Cosgrove et al. 2002], but most such studies were retrospective and conclusive evidence was extracted from the curtailment of cancer-associated vascular thrombosis [Falanga and Marchetti, 2007], rather than reduction of tumor expansion/metastasis as demonstrated in animals [Li, 2008]. Is heparin amelioration of tumor metastasis linked to disruption of platelets and microvesicle aggregates near the tumor site? The simplest explanation for the rationale of using heparin in tumor thromboprophylaxis traditionally rests on its inhibition of stable blood clots triggered by tissue factors and microparticles shed from tumor cells. Heparin polymers bind and accelerate the protease activity of the antithrombin enzyme (AT) or form a ternary complex of heparin–AT–thrombin, which in turn can inhibit the activation of coagulation proteins (such as thrombin, factor IX, Xa) and platelets in the blood coagulation cascade. The multifunctionality of heparin, including pharmaceutical grade LMWHs, are more than that and appears expectedly convoluted.
Supported by studies on animal tumor models, the heparin curtailment of mortality rates in patients with cancer could be because of the ability of heparins to sustain the structural integrity of cell surface and other extracellular matrix proteins through inhibition of heparanase [Yang et al. 2010] or modification of cellular growth factors [Sanderson et al. 2004]. There is no compelling evidence supportive of the previously asserted antiangiogenic effect of soluble heparins; instead, the antiheparanse notion takes renewed momentum. Recent data indicate that the elevated expression and secretion of heparanase and other matrix-degrading enzymes released by tumor cells and other cytokines such as receptor activator of nuclear factor-κB ligand (RANKL) enhance bone destruction within the tumor microenvironment in myeloma [Yang et al. 2010]. This plausibility could push a localized tumor toward remission by suppressing the tumor expansion/metastasis through deaggregation of platelets and microvesicles or microparticles released from cancerous and normal cells. In this sense, it is tempting to also classify heparins as anticancer drugs. Consistent with this, a nonclinical study demonstrated that heparins strongly inhibit proliferation of transformed osteoblastic cells at concentrations of at least 1 µg/ml, but it was also inhibitory to normal osteoblasts [Nikitovic et al. 2005]. Nevertheless, this remotely supportive evidence stems from observations on two osteosarcoma cell lines. Osteosarcoma exhibits a diverse histological variability, including osteoblastic, chonodroblastic and fibroblastic cells. The sufficiency of evidence requires testing large sample sizes, including cells with documented multipotent potential. Clinically relevant evidence for intent-to-prevent tumor-induced emboli or metastasis in primary bone cancers is scarce or unpublished. Would long-term use of heparin or heparin-like molecules negatively influence the outcome of osteosarcoma in humans?
On the downside, the anticoagulant effects of heparins might encourage an excessive and long-term (off-label) use that may increase the likelihood of their adverse effects. Soluble sulfated heparins released near tumor sites could also act to promote tumors and lead to the growth and metastasis of tumors [Li, 2008; Sanderson et al. 2004]. These situations could deteriorate in certain subtypes of bone cancers and tip the balance in favor of tumor growth and metastasis by heparin treatments, which might bind and stabilize matrix protumorigenic BMPs and other growth factors. These unexplained inconsistencies are conundrums. In addition to stimulating BMPs, anticlotting heparins, including certain LMWHs, can arguably exacerbate tumorigenic events by promoting the formation and maintenance of tumor vasculature. The only currently available, yet indirect, evidence in support of this plausibility is afforded by a vascular endothelial growth factor (VEGF)-mediated angiogenesis study of dalteparin on rats. Unlike tinzaparain, dalteparin promotes the formation of new vasculature [Norrby and Nordenhem, 2010]. The proangiogenic function of dalteparin was attributed to the differences in manufacturing and chemical backbone structures, and not to the density of sulfations, molecular weights, and source. That is, ‘while dalteparin is produced by nitrous acid depolymerization [of porcine gut mucosal unfractionated heparin (UFH)], which results in the formation of anhydromannose (a five-member ring) in the molecule, tinzaparin is produced by controlled enzymatic depolymerization by heparinase digestion [of porcine gut mucosal UFH], which adds a double bond to the end group of the molecule’ [Norrby and Nordenhem, 2010]. The LMWHs cannot then be regarded as nonselective agents. The insufficiency of evidence and in vivo examinations based on one brand of heparin make the inferences to other heparins remote.
The brief use of heparin during surgery to prevent thrombosis is unlikely to have any relevant angiogenic effect in the overall tumor growth. The observations were in tumor-free animals, which were treated for a few days with heparin in combination with very low doses of VEGF. Since VEGF expression in bone tumors may indicate poor prognosis [Rossi et al. 2010], heparin use can stimulate angiogenesis to surviving tumor cells. Furthermore, the extended use for thromboprophylaxis maintenance of endoprosthetic implants may increase the odds of stimulating remaining microtumor lesions. Whether heparins here might either promote or prevent the growth and mobilization of CSCs during/after surgery is a different topic. The totality of in vivo evidence for risk-to-benefit profile of heparins opens up more questions, partly addressed below.
Bone morphogenetic proteins in appendicular sarcomas
The levels of BMP-7 and BMP-8 proteins are higher than other BMPs in osteosarcoma, but neither they nor other subtypes were predictive of the disease outcome when 47 biopsy specimens were examined immunohistochemically [Sulzbacher et al. 2002]. This assay is generally less reliable than the modern gene expression assays, and can include several drawbacks. For instance, BMP subtypes may be misfolded, inappropriately localized and inaccessible in certain types of osteosarcoma; some epitopes may be sequestered, due to heterodimers formation and/or binding to as yet unidentified BMP-reactive osteogenic and/or osteosuppressive factors, as partly addressed previously [Yu et al. 2004; Anderson et al. 2002]; antibodies may cross-react with different subtypes and/or cellular components; and the methods used to process biopsies may harm the BMPs. Which BMP(s) influence osteoblastic and osteolytic alterations and which ones influence bone overgrowth and metastasis?
Supported by examples in other tumors, BMP-2 is both inhibitory and neutral to tumor cell proliferation, and its stable RNA levels are either decreased or unchanged. Novel pharmacogenomic and protein therapeutic approaches have just begun to elucidate the biological impact of BMPs on the pathogenesis of skeletal malignancies. Gene expression profiling of the most popularly researched BMPs (BMP-2, BMP-6 and BMP-7) in addition to the limited set of functional studies in patient specimens and cultured cells were evaluated [Alarmo and Kallioniemi, 2010]. The authors’ conclusion that BMP-2 mRNA levels are downregulated is a rational thought, though the weight of evidence is insufficient to determine whether it is a specific transcriptional or post-transcriptional event linked to an external stimulus. However, whether the observed reductions in stable RNA levels are beneficial or detrimental to the clinicopathological parameters is currently unknown. An increase in BMP-7 expression, as supported by exogenously added BMP-7, stimulates the migration of cancerous cells in cultures, but it also inhibits cell division in a cell-line-specific manner [Alarmo and Kallioniemi, 2010; Alarmo et al. 2009]. Using a bone metastasis model and whole-body bioluminescent reporter imaging, a decrease in BMP-7 expression in the tumor cell specimens correlates with bone metastases in patients with at least 10 years of follow up [Buijs et al. 2010, 2007]. These conflicting observations have not yet been reconciled.
Heparin–bone morphogenetic proteins–cancer stem cells
The probability of heparins influencing the release and expansion of bone CSCs is tenable because heparin polymers stabilize BMPs/growth factors. These agents promote the growth and migration of osteosarcoma cells in vivo and might even potentiate formation of tumor microvasculature. One tangentially related research project questioned the interrelationship between heparin-influenced BMP-4 in SaOS-2 osteosarcoma cells with a proven osteogenic phenotype and intact endogenous GAG heparins. The study demonstrated that heparin affects cellular response to BMP-4 30 min after treatment, probably mediated by the specific sulfate residues on heparin saccharides [Khan et al. 2008]. It is unclear whether the benefits outweigh the costs of any heparin use, be it single dose, multiple/repeat dose or long-term treatment. Several BMPs are probably involved in the conflicting cycles of pathological growth (osteoblastic, osteogenic) and destructive (osteolytic, osteosclerotic) outcomes. The subtypes and differential effects of BMPs and how they influence CSCs in distinct primary and metastatic bone cancers are currently unknown. Some of these key steps are illustrated in Figure 2.
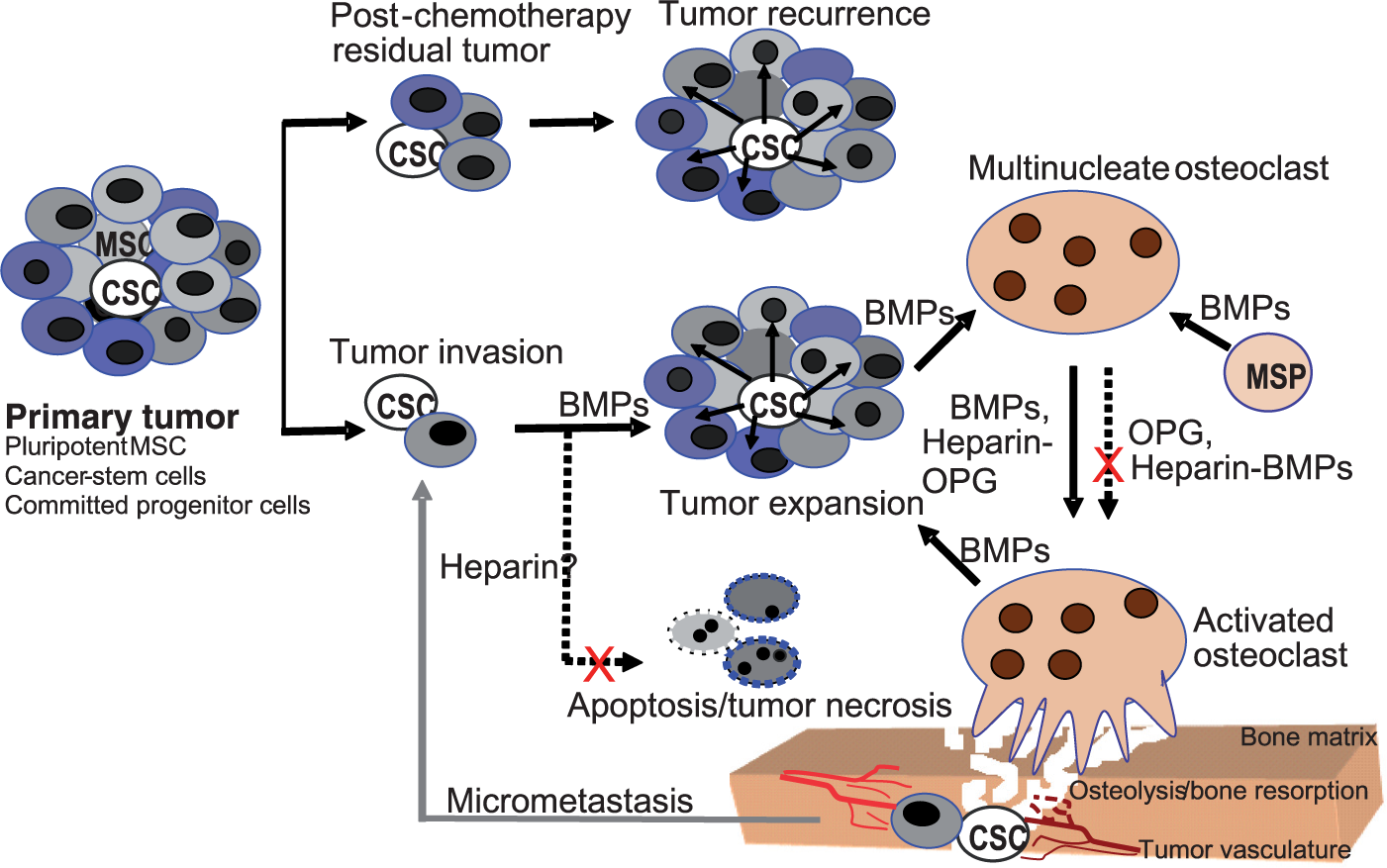
Schematic representation of tumorigenic situations (drug resistance, metastasis/recurrence) and networks involving cancer stem cells (CSCs) affected by tumor- and nontumor-cell produced bone morphogenetic proteins (BMPs), the osteoclasts maturation/osteolysis mediator, osteoprotegerin (OPG), and exogenous heparins is proposed. MSC, mesenchymal stem cell; MSP, monocytic osteoclast precursors.
Can both fractionated (LMWH) and nonfractionated heparins modify BMP signaling and activity? Various pharmaceutical-grade anticoagulant heparins are heavily modified with sulfate salts (Figure 1). There is no evidence to indicate that fractionated LMWHs bind with equivalent strength and/or modify BMPs more than their nonfractionated counterparts. As their sulfate residues were found essential for binding BMPs [Zhao et al. 2006; Nikitovic et al. 2005; Takada et al. 2003], and propagation of osteosarcoma cells [Nikitovic et al. 2005], compared with nonfractionated heparin, most if not all sulfated LMWHs probably would modulate BMP signaling and activity in vivo. The positions of sulfurs, but apparently not their concentrations, determine their impact on binding to different BMPs [Khan et al. 2008; Takada et al. 2003]. This appears to imply that all commercial grade heparins with different density and/or sulfation patterning may variably bind and modify BMPs. Therefore, the molecular geometry, configuration and density of sulfate groups in heparins are collectively important in influencing the functionalities of BMPs in cancer cell microenvironments compared with the linear, unmodified or less-sulfated heparins. Furthermore, other bone matrix proteins presumably in concert with BMPs are involved in CSC-induced formation of osteoids and the disease progression in primary bone cancers.
Heparin–osteoprotegerin–cancer stem cells
Heparins interact with another molecule that acts as a biological modifier involved in tumor growth and apoptosis as well as bone remodeling. In normal murine osteoblast/osteoclast assays, heparin blocked the binding of osteoprotegerin (OPG), thus suggesting it enhances osteoclastic bone resorption [Irie et al. 2007]. Heparin and heparin-like molecules bind to OPG, a key homologue of receptor for tumor necrosis factor kappa B (RANK) that acts as a decoy receptor for RANKL on osteoblasts [Irie et al. 2007; Lamoureux et al. 2007; Theoleyre et al. 2006]. It can also bind and inhibit the apoptotic effect of tumor necrosis factor-related apoptosis inducing ligand (TRAIL) on tumor cells [Wittrant et al. 2004; Grimaud et al. 2003]. OPG produced by tumor cells increases tumor growth and survival probably by neutralizing TRAIL, which induces apoptosis of tumor cells [Emery et al. 1998]. In osteosarcoma, there is an imbalance in expression and activities of OPG and other cytokines, although OPG is active in perpetuating the repetitive cause–effect cycle of tumor cell proliferation and bone destruction/resorption [Lamoureux et al. 2009]. While the interaction of heparin with OPG attenuates tumor apoptosis, it also diminishes osteoclasts in a tumor environment. Interestingly, a recent independent research study indicates that osteosarcoma cells inhibit osteoclastogenesis, and suggests, ‘the metastatic potential of osteosarcoma is determined early in tumor development and that loss of osteoclasts in the primary lesion enhances metastasis’ [Endo-Munoz et al. 2010].
In the bone matrix, the physical proximity of BMPs with the ostegenic/osteloytic protein OPG, including several levels of intervening and interfering reactions involving different cytokines (reviewed by Lamoureux, Velasco and colleagues [Lamoureux et al. 2010; Velasco et al. 2010]), may influence the biology of CSCs. The unifying elements of the opposing pathological changes in primary bone cancers (tumor growth, formation of osteoid/bone-like substances/calcification versus osteolysis/osteoporosis) are the consequences of a hyperactive state of heparin–BMP and heparin–OPG complexes. These molecules enable both protumoral and bone resorption outcomes. In other words, although OPG inhibits tumor cells through TRAIL-induced apoptosis, the tumor growth-promoting heparin–BMPs can act cooperatively with the heparin–OPG complex, which prevents OPG-mediated inhibition of osteoclastic bone resorption [Lamoureux et al. 2009]. A heparin prophylaxis could also generate a tipping point in favor of metastasis (Figure 2). Would a short-term use of heparin affect the release and migration of CSCs into the circulation through osteogenic BMPs and, additionally, key osteolytic cytokines such as OPG? The strength of associations for either or both of these mediators in the growth and decay cycle of bone in clinical situations are unpublished, although studies in cells and in animals allow both possibilities to be raised.
The options of using heparins in primary bone cancers call into question their effects on residual tumor expansion and metastasis post chemotherapy. Heparins are likely used more in major bone surgeries because the risk of bleeding and the need for blood transfusion is high. In osteosarcoma heparins are used as anticoagulants in both biopsy and during/after prosthesis-implanting operations. There are positives and negatives to the use of heparins. Whether the positive outcome of increased survival by restricting the formation of blood clots or cancer-induced thrombosis outweighs the negative outcomes of increased tumor growth and metastasis is unknown. These analyses have a number of limitations and compromises. The data quality assessment is inadequate because the most relevant clinical data are unpublished. These hypotheses may be linked and require prospective investigations to determine if there are any significant consequences of heparin prophylaxis influencing the critical components of BMP activities or other cell/tissue factors in clinical situations in primary bone cancers. These apparent data gaps conjure up the adage that ‘the absence of evidence is not the evidence of absence’. The plausibility of heparins disturbing the risk-to-benefit balance must be further assessed in primary as well as metastatic bone cancers, for instance, in high-risk (survival of 0%), low-risk (survival >70%) groups, and intermediate pathological endpoints.
Key points and hypotheses
A rather common misconception among the advantages of heparin prophylaxis is the assumption that heparins increase cancer survival by reducing tumor-induced thrombosis and metastasis (as the sole possible mechanism).
Although heparin use mitigates cancer-associated thrombosis and may help in the suppression of certain tumors, it may hinder the disease outcomes in primary bone cancers by stimulating CSCs.
Heparin and heparin-like agents bind to BMPs, stabilize their structure function, and may promote the growth of bone CSCs, which may increase the plausibility of migration and/or recurrence of tumor cells.
Heparin/heparan sulfates bind to another signaling protein (heparin–OPG) which may act cooperatively with heparin–BMP complexes, thus increasing the risk of tumor-induced bone loss and the dissemination of CSCs.
In summary, these contradictory data matter because several levels of regulations intervene and interfere with each other: the heparin/heparan sulfate differences in the structures may induce different biological responses; BMPs may either potentiate or inhibit the growth of CSCs, for instance, because of variations in the expression of BMPs or BMP receptors [Hay et al. 2004] or related transcription factors (e.g. Smads, a group of several signal transducing molecules of the transforming growth factor beta superfamily) or target genes (reviewed by Yamaguchi and colleagues) [Yamaguchi et al. 2000]; heparins may modulate CSCs positively or negatively through BMPs; and all these effects may vary depending on the types of cells or cancers considered. The results of these regulation levels are uncertain and drawing unequivocal conclusions remains unrealistic. In osteosarcoma the growth and expansion of CSCs mediated by heparin–BMPs may in turn facilitate heparin–OPG-induced bone loss as the bone matrix/tumor vasculature disintegrates. These biochemical events (modulated by heparin/heparan sulfates) collectively may promote the recurrence and/or dissemination of CSCs and thus increase the likelihood of tumor invasion (Figure 2). Finally, the tumor-induced collateral bone destruction may compromise the durability of orthopedic reconstructions, which may require additional therapeutic interventions.
Perspective
Considering the seemingly homogeneous functionality of CSCs and the highly variable histopathological features in various primary bone cancers, the assessments described in this article are proposed to draw attention to whether linking the heparin–BMP–CSC and/or heparin–OPG axis acts as a double-edge sword in tumorigenesis of bone cancer. While heparin prophylaxis could reportedly increase the survival in thrombotic cancers, heparin-modified stimulation of BMPs and blocking of OPG might shift the balance versus chemotherapy in favor of apoptosis-resistant CSC-induced bone lesions by enhancing osteogenic bone destruction. This set up could allow renewal of the resistant subpopulation of CSCs to invade distant tissue sites and form micrometastatic foci, despite mitigation of (an observable or nonobservable [microemboli]) thrombosis. Potential signaling traffics partly mediated by BMPs and partly by OPG in tumorigenic situations that can be affected by heparins (Figure 2). To this end, no core evidence is available. Prospective clinical trials have not yet challenged the hypothesis that exogenous heparins are probably both beneficial and perilous in primary bone cancers.
Additional model validation and complementary studies are required to elucidate the underlying mechanisms in the above probabilistic clinicopathological outcomes. To address these issues and other points raised in this article and to obtain a three-dimensional perspective, one may delve into prospective controlled preclinical and clinical investigations to see whether there is indeed a link between heparin stabilization of the BMPs and adverse tumorigenic outcomes. Acceptable well designed test system(s) based on appropriate disease subtype-based studies could be built upon with specific answers to the perceptive conclusions based on available laboratory studies cited in this article. These investigations are, however, cost prohibitive and might be undesirable for sponsors/manufacturers of the marketed heparins. Although piggybacking ongoing pathological studies on patient biopsy specimens may seem a logical approach, unbiased new approaches could determine whether the administration of distinct LMWHs at relevant treatment configurations (dose frequency and intensity, cumulative dose exposure at effect site) would affect the BMP-induced bone alterations. Furthermore, to define all confounding variables from carcinogenesis to bone changes to distant metastasis would be challenging, but such obstacles may partly be surmountable by well controlled animal models of bone cancers.
Conclusion
The plausibility of a molecular network between heparin-stimulated, BMP-influenced, and CSC-inspired osteogenic and/or osteolytic alterations involving the impairment of osteoclastogenesis without mitigating tumor vasculature growth and/or tumor expansion/metastasis might bring out the worst of BMPs. The available data that BMPs promote the tumor growth of human osteosarcomas that harbor differentiation potential are unchallenged. Nonetheless, there is no hard evidence suggesting that any particular subtype of BMP expression is predicative of the disease outcome in osteosarcoma, although the involvement of these proteins in developing various histological features in primary bone cancers cannot be ruled out based on the current state of knowledge. Heparins can also interfere with bone remodeling by binding to other proteins and/or components of matrix proteins and act as either protumorigenic or antitumorigenic agents. The missing data links are important for further investigation, not just in metastatic bone cancers, but also in primary bone cancers with an emphasis on treatment-resistant CSCs. Further prospective (preclinical/clinical) studies are required to elucidate the significance of key biochemical changes and the risk-to-benefit ratio of using pharmacologic-grade heparins in primary bone cancer.
Footnotes
Acknowledgements
I am grateful to Dr Patricio E. Ray, MD, Professor of Pediatrics at the George Washington University and Children’s National Medical Center, Washington DC, for his critical reading of the manuscript and insightful discussions.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The author does not have a conflict of interest nor any financial relationship with the developers or marketers of heparins, BMPs or related products or their competitors.
