Abstract
Background:
Stress-induced hyperglycemia ratio (SHR) has shown strong correlations with diabetes risk. However, its relationship with gestational diabetes mellitus (GDM) remains an area of limited research.
Objective:
Based on the National Health and Nutrition Examination Survey (NHANES) database, this cross-sectional study aims to explore the association between SHR and GDM.
Methods:
The study included 461 pregnant women from NHANES between 1999 and 2016. Logistic regression analysis was performed to investigate the relation between SHR and GDM, as well as the weighted tertiles of SHR. Stratified and subgroup analyses assessed the association between SHR and GDM. Restricted cubic splines (RCS) delineated the nonlinear relationship between SHR and GDM. Moreover, a two-segment linear regression model was employed to test the threshold effect of SHR on GDM. Stratified analysis was conducted by stratifying by months of pregnancy.
Results:
Among the 461 eligible women included in the study, SHR was a critical risk factor for GDM. Higher SHR values were associated with an elevated likelihood of developing GDM after accounting for other variables (OR: 1.77, 95% CI: 1.37–2.28,
Conclusion:
SHR is significantly positively correlated with an increased risk of GDM in early pregnancy. These results highlight the potential utility of SHR as a clinical marker for early identification and intervention in GDM risk cases.
Keywords
Introduction
Gestational diabetes mellitus (GDM) is a pregnancy-related glucose intolerance that typically emerges from insulin resistance and insufficient pancreatic β-cell function. 1 It is a major health concern for the mother and the developing fetus. 1 In 2021, the IDF Diabetes Atlas reported that 16.7% of all live births, amounting to 21.1 million women, faced hyperglycemia during pregnancy, with the vast majority (80.3%) being diagnosed with GDM. 2 GDM is a condition that significantly impacts both maternal and fetal health. It remarkably elevates the incidence of pregnancy complications such as hypertensive disorders and preeclampsia.3,4 In addition, GDM contributes to adverse perinatal outcomes, including macrosomia and neonatal hypoglycemia.3,4 GDM is linked to a higher risk of type 2 diabetes mellitus in both affected mothers and their offspring over an extended period.5,6 The diagnosis of GDM still depends on the oral glucose tolerance test (OGTT). This method, though widely used, is far from ideal—it is cumbersome, time-consuming, and its results can be skewed by various factors, 7 which has thus prompted a search for simpler and more reliable early prediction indicators for GDM.
Stress-induced hyperglycemia ratio (SHR), a new tool for assessing blood glucose regulation under stress, is defined as the ratio of admission blood glucose to estimated average glucose, which is calculated from HbA1c. 8 SHR offers a holistic view of glycemic control, combining short-term fluctuations with long-term trends. For instance, higher SHR levels are found to be associated with an elevated risk of type 2 diabetes mellitus in a Chinese population. 9 In addition, every increase of one standard deviation in SHR is related to a 20% increase in the probability of nonalcoholic fatty liver disease in patients with diabetes or prediabetes. 10 Although SHR has shown potential predictive value in various metabolic diseases and adverse outcomes, its role in GDM risk assessment has not been fully studied.
This study aims to systematically evaluate the association and potential thresholds between SHR and GDM using the National Health and Nutrition Examination Survey (NHANES) database. Through this study, we hope to provide scientific evidence for early screening and intervention of GDM, thereby improving maternal and infant health.
Methods
Data source and study population
This study is a cross-sectional study based on the NHANES database. NHANES adopted a complex stratified multistage probability sampling design to obtain a representative sample of American residents. NHANES is a biennial survey that enrolls approximately 7000 unique participants who provide valuable information through questionnaires, physical exams, and biological samples for biomarker analysis. Our study utilizes this publicly available data. Written informed consent was obtained from all respondents, and the study protocol was approved by the Institutional Review Board of the Centers for Disease Control and Prevention. This study followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement, 11 as outlined in Table S1.
We pooled data from nine NHANES cycles (1999–2016) and identified 1809 pregnant women with positive hCG urine tests. Participants with incomplete fasting plasma glucose data (

The flowchart of participants.
Participants were excluded if they met any of the predefined criteria for diabetes: (1) a self-reported diagnosis of diabetes by a physician, (2) taking diabetes medications to control blood sugar, or (3) current insulin use. 12
Evaluation of SHR
Fasting plasma glucose (FPG) measurements were conducted at the Fairview Medical Center Laboratory of the University of Minnesota. Glycated hemoglobin (HbA1c) was measured using a Tosoh A1c2.2 Plus analyzer at the University of Minnesota Diabetes Laboratory. The SHR was calculated using the formula: [FPG(mg/dL)]/[1.59*HbA1c(%)–2.59]. 13 Participants were then grouped into three categories following SHR tertiles (Q1, Q2, Q3).
GDM
GDM was determined based on the diagnostic strategy recommended by the International diabetes and Pregnancy Study Group (IADPSG), that is, fasting blood glucose ⩾ 5.1 mmol/L (92 mg/dL). 14 In addition, during the 2005–2016 NHANES cycle, participants also underwent a one-step OGTT, with a diagnostic criterion of 2-h blood glucose ⩾ 8.5 mmol/L (153 mg/dL). 15 Participants who met any of the above criteria are considered to have GDM
Covariates
Demographic, examination, laboratory, and questionnaire data were downloaded from NHANES. The variables included age, race (non-Hispanic White, non-Hispanic Black, Hispanic, or other races/ethnicities (including multiracial individuals), 16 education (high school diploma or less, or above high school), 17 marital status (unmarried, married or cohabitating, widowed, or divorced/separated), 18 poverty-income ratio (⩽1.3: low income, 1.3-3.5: middle income, >3.5: high income), 19 and body mass index (BMI) (<25, 25–30, or ⩾30 kg/m2). 20 Smoking status was determined based on the question “Have you smoked at least 100 cigarettes in your life?” Respondents who answered “no” were classified as never smokers. For those who answered “yes,” they were further categorized into former smokers and current smokers based on their response to the question “Do you now smoke cigarettes?”. 21 Alcohol use was defined as having had at least 12 standard alcoholic drinks in the past year. 22 One standard drink was equivalent to 12 ounces of regular beer, 5 ounces of wine, or 1.5 ounces of 80-proof distilled spirits. 23 Hypertension was defined by self-reported high blood pressure, or systolic blood pressure of ⩾140 mmHg, or diastolic blood pressure of ⩾90 mmHg, or the use of antihypertensive medications. 24 In the 1999–2006 cycles, vigorous physical activity was defined as engaging in any form of vigorous physical activity lasting at least 10 min in the past 30 days, which led to heavy sweating, elevated heart rate, or accelerated breathing. In the 2007–2016 cycles, vigorous physical activity was indicated if participants reported evident increases in breathing or heart rate during work time or leisure activities. 25 Cholesterol measurements included low-density lipoprotein cholesterol, triglycerides, and high-density lipoprotein cholesterol. 26
Statistical analysis
In this study, fasting sub-sample weights were used for adjustment, with weights calculated as (2/9) × WTSAF4YR from 1999 to 2002 and (7/9) × WTSAF2YR from 2003 to 2016. Baseline tables were constructed using the
Results
Baseline profile of respondents
A cohort of 461 female participants from the NHANES database (1999–2016) was included (mean age: 28.87 ± 5.84 years). Specific characteristics are detailed in Table 1. Significant differences were identified between GDM and non-GDM groups in terms of hypertension, low-density lipoprotein cholesterol (LDL-C), triglycerides, and the SHR index, including its tertile distribution (
Characteristics of NHANES participants between 1999 and 2016.

Mean (SD) and
Subgroup analysis
Table 2 shows the findings on the relationship between SHR and GDM. In overall population, SHR was positively associated with GDM in all models: the unadjusted model (OR: 1.75, 95% CI: 1.33–2.31,
The odds ratios between SHR and gestational diabetes mellitus, NHANES 1999–2016.
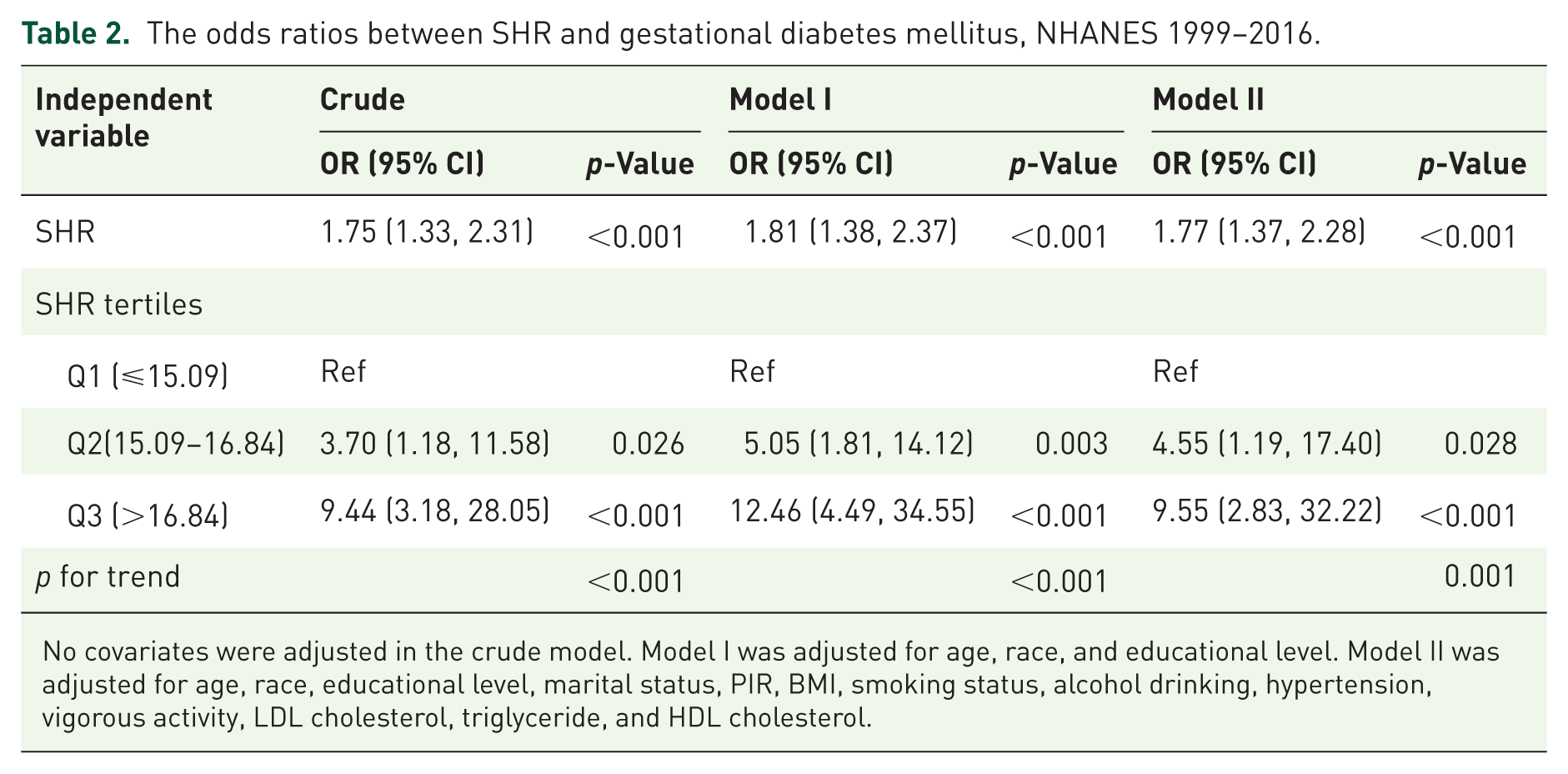
No covariates were adjusted in the crude model. Model I was adjusted for age, race, and educational level. Model II was adjusted for age, race, educational level, marital status, PIR, BMI, smoking status, alcohol drinking, hypertension, vigorous activity, LDL cholesterol, triglyceride, and HDL cholesterol.
Table 3 illustrates the SHR-GDM relationship across various population subgroups, considering potential interactions. The data revealed a significant link between SHR and GDM in all subgroups stratified by education and alcohol consumption status (OR > 1,
Subgroup analysis for the association between SHR and gestational diabetes mellitus, NHANES 1999–2016.

Nonlinear association between SHR and GDM risk
RCS analysis was conducted to examine the relation between SHR and GDM (Figure 2). RCS curve demonstrates a significant overall trend (
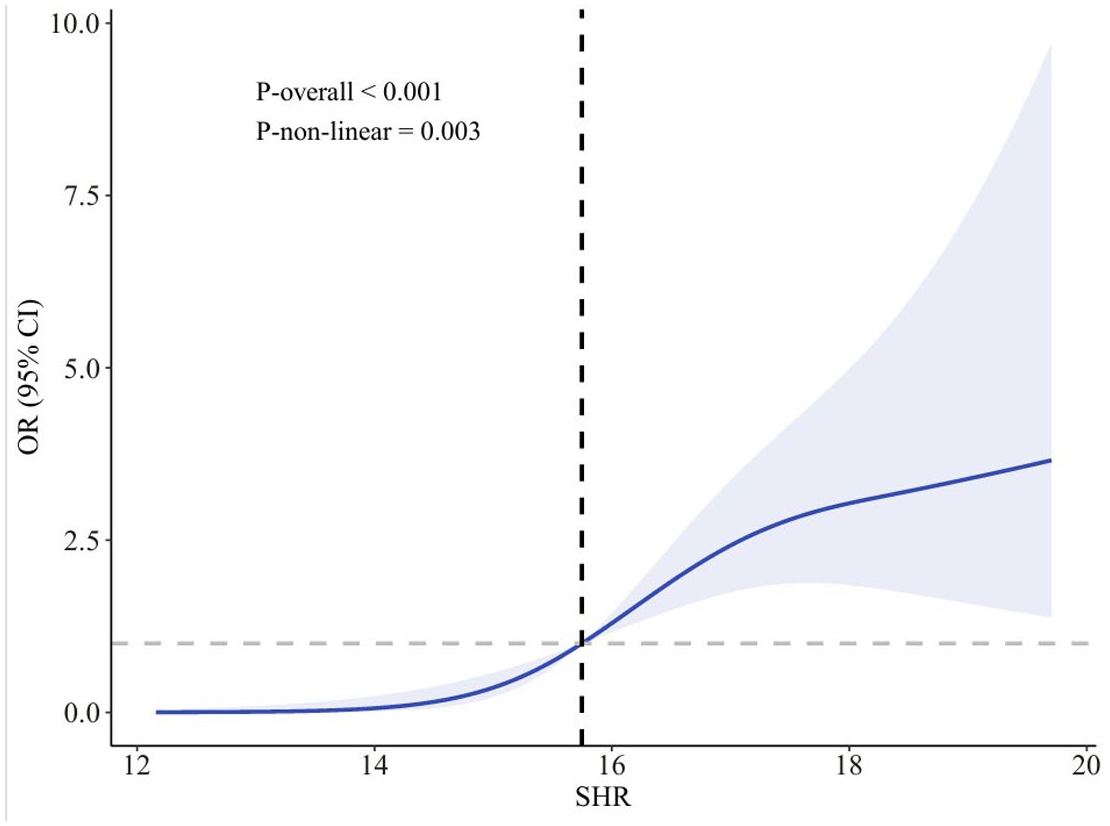
The OR of SHR and gestational diabetes mellitus adjusted by covariates, NHANES 1999–2016. RCS line is adjusted by age, race, educational level, marital status, PIR, BMI, smoking status, alcohol drinking, hypertension, vigorous activity, LDL cholesterol, triglyceride, and HDL cholesterol. The OR is represented by the blue line, and the shaded part represents the 95% CI.
Threshold analysis
To explore the threshold effect of SHR on GDM, we conducted a two-segment linear regression analysis. The breakpoint was found to be 15.75. Below this threshold, SHR was strongly associated with GDM (OR = 5.11, 95% CI: 1.56–16.75,
Threshold effect analysis of SHR on gestational diabetes mellitus by the two-piecewise linear regression.

Stratified analysis
We further stratified the association between SHR and GDM according to months of pregnancy. The results showed that the positive correlation between SHR and GDM remained significant (OR = 2.16, 95% CI: 1.12–4.16,
Association between SHR and GDM stratified by month of pregnancy.

Model II was adjusted for age, race, educational level, marital status, PIR, BMI, smoking status, alcohol drinking, hypertension, vigorous activity, LDL cholesterol, triglyceride, and HDL cholesterol.
Discussion
Using NHANES data, our cross-sectional study assessed the relationship between SHR and GDM. Results confirmed the existence of a positive correlation, which persisted after accounting for all potential confounders. This correlation was also observed in subgroup analyses. The RCS analysis shed light on a nonlinear relationship between SHR and GDM risk. When SHR is 15.75 or less, each unit increase in SHR is linked to a marked increase in GDM risk.
In this study, the prevalence of hypertension was higher in the GDM group, which is consistent with the conclusion of a previous study showing that gestational hypertension can increase the risk of GDM. 30 It is worth noting that the GDM group had lower levels of low-density lipoprotein cholesterol and triglycerides. Lower levels of low-density lipoprotein are found to be potentially associated with a higher risk of GDM. 31 However, it is suggested that triglyceride levels in GDM patients are usually higher than those in the normal group, 32 which is inconsistent with the results of this study. This inconsistency may be related to differences in dietary management among pregnant women, small sample sizes, or sampling biases.
SHR is composed of the ratio of fasting blood glucose measured at the time of measurement to HbA1c. A higher SHR indicates a relative increase in short-term fasting blood glucose when considering long-term blood glucose levels, suggesting that an individual’s ability to regulate blood glucose under stress may be impaired. A previous cohort study demonstrated a positive correlation between SHR and type 2 diabetes.
9
On this basis, this study further found that higher levels of SHR are also associated with a higher risk of GDM. Our study revealed that when the SHR was at or below 15.75, it was closely related to GDM. SHR is indicative of the body’s acute glycemic response to stress, which could intensify existing insulin resistance—a crucial element driving the onset of GDM.
33
Pregnancy brings about marked changes in hormone levels, especially in placental hormones. These hormones can work against insulin, leading to heightened insulin resistance.
34
In addition, the function of pancreatic beta cells is often hindered, preventing the secretion of sufficient insulin to offset the resistance, which results in elevated blood glucose levels.
35
However, the correlation between SHR and GDM was less significant when SHR exceeded 15.75 (OR = 1.37, 95% CI: 0.93–2.01,
The subgroup analysis results of this study showed a significant positive correlation between SHR and GDM regardless of education level and alcohol consumption status. Although education level and alcohol consumption status have a certain impact on individual health,27,28 they do not fundamentally change the relationship between SHR and GDM. This discovery suggests that SHR may be a robust indicator that can effectively reflect the stress glucose metabolism status of pregnant women in different educational and lifestyle backgrounds.
This study further analyzes the association between SHR and GDM by stratifying months of pregnancy. The results showed that the positive correlation between the two mainly existed in the early-to-mid pregnancy period (1–5 months), while it was no longer significant in the mid-to-late pregnancy period (6–10 months). This phenomenon may be related to the time window of GDM screening and health management. Standard GDM screening is typically conducted between 24 and 28 weeks.
36
Meanwhile, the prenatal care guidelines released by the World Health Organization in 2016 recommend that the frequency of contact with healthcare providers during pregnancy should gradually increase as the gestational age progresses, with a significant increase in prenatal check-up frequency in the middle and late stages of pregnancy (https://www.who.int/news/item/07-11-2016-new-guidelines-on-antenatal-care-for-a-positive-pregnancy-experience; Access time: December 3, 2025). Therefore, the increased frequency of prenatal checkups in the middle and late stages of pregnancy, systematic screening of GDM, and standardized interventions after diagnosis collectively constitute a stricter health management process, which may explain the phenomenon of weakened association between SHR and GDM in the middle and late stages of pregnancy. Overall, these results emphasize that SHR may serve as a potential indicator for identifying GDM risk in early pregnancy. The study demonstrates a correlation between SHR and an elevated risk of GDM, yet several limitations should be noted. First, the NHANES period from 1999 to 2004 did not include OGTT testing, and the identification of GDM was only based on fasting blood glucose levels, which may lead to misclassification bias in some cases. Second, the sample size of GDM patients in this study was relatively small (
Conclusion
SHR is significantly associated with an increased risk of GDM, especially in early pregnancy. Its simplicity and reliability make it a promising tool for GDM early identification and intervention. Research could focus on exploring its practical applications in clinical settings.
Supplemental Material
sj-xlsx-1-tae-10.1177_20420188261431025 – Supplemental material for Stress-induced hyperglycemia ratio and gestational diabetes mellitus: a cross-sectional study based on NHANES database
Supplemental material, sj-xlsx-1-tae-10.1177_20420188261431025 for Stress-induced hyperglycemia ratio and gestational diabetes mellitus: a cross-sectional study based on NHANES database by Yina Piao and Mengxia Jin in Therapeutic Advances in Endocrinology and Metabolism
