Abstract
Aims:
To assess the real-world impact of transitioning from the Dexcom G6® to G7® system in individuals with type 1 diabetes (T1D) using the Tandem t:slim X2™ with Control-IQ™. Primary outcomes included glycemic control changes, while secondary outcomes evaluated patient-reported outcomes (PROMs) and experiences (PREMs).
Methods:
A 3-month prospective, multicenter, observational study was conducted in individuals previously using Dexcom G6 and Control-IQ. Glycemic control was assessed via TIR70–180 and HbA1c. PREMs and PROMs were measured using validated questionnaires (Diabetes Quality of Life (DQoL), Type 1 Diabetes Life (ViDa1), Diabetes Distress Scale (DDS), and Diabetes Treatment Satisfaction Questionnaire). Statistical analyses were stratified by baseline TIR70–180 (>70% vs <70%) and sex.
Results:
The study included 92 participants (mean age 38 ± 13 years), with a baseline TIR70–180 of 76% ± 10%. The overall TIR70−180 remained stable throughout the study. However, among participants with baseline TIR70–180 <70%, a significant increase in TIR70−180 was observed, rising from 61% ± 9% to 65% ± 7% (p = 0.007). In consonance, HbA1c levels showed a significant decline, from 7.3% ± 0.9% to 7.0% ± 0.6% (p = 0.001). In addition, both glucose management indicator (GMI) and mean glucose levels significantly decreased over time, reflecting an overall improvement in glycemic control. These changes (GMI and glucose levels) were consistent across groups, with no significant differences based on baseline TIR70-180 stratification. Quality of life and diabetes distress improved (DQoL, ViDa1, and DDS), especially in participants with lower baseline TIR70-180 and in women. Some reported increased connectivity issues, but none led to treatment discontinuation.
Conclusion:
In Control-IQ users, the transition to G7® did not significantly impact glycemic control overall. However, a subgroup of patients with suboptimal baseline control (TIR70–180 <70%) may benefit from this change, and enhanced quality of life and diabetes distress, especially in women.
Plain language summary
People with type 1 diabetes need to carefully monitor their blood sugar levels to stay healthy. Many use advanced technologies like insulin pumps and continuous glucose monitors (CGMs). This study looked at what happens when people already using an automated insulin delivery system (Tandem Control-IQ with Dexcom G6) switch to a newer sensor model, the Dexcom G7. Over three months, 92 adults with type 1 diabetes in Spain were followed after switching to the G7 sensor. Researchers checked their blood sugar control and asked them how they felt about the new system using questionnaires. The results showed that overall blood sugar levels stayed stable. But for those who started with less optimal control, blood sugar levels improved significantly after the switch. These participants also had lower average glucose and HbA1c levels. In addition to better glucose control, people reported feeling less stressed about managing their diabetes and noted improvements in quality of life—especially women and those who initially had poorer control. Most people were satisfied with the new sensor, and very few stopped using it. Some people mentioned mild skin irritation or connection issues, but these did not interfere with the treatment. This study suggests that moving to the Dexcom G7 sensor can be safe and beneficial for people already using advanced diabetes technology, especially for those who need help improving their glucose levels and emotional well-being.
Keywords
Introduction
Type 1 diabetes mellitus (T1D) requires stringent glycemic control to prevent both acute and chronic complications.1,2 Automated insulin delivery (AID) systems have emerged as the preferred approach for T1D management, demonstrating efficacy in improving glycemic outcomes across randomized clinical trials and real-world evaluations, with a level A scientific evidence.1,3
Among AID systems, the Tandem t:slim X2™ insulin pump with Control-IQ™ technology has been widely adopted since its FDA approval in December 2019 and its subsequent availability in Europe in October 2020. The Control-IQ algorithm employs a model predictive control approach, forecasting glucose levels 30 min ahead to adjust insulin infusion rates and deliver automatic correction boluses when needed. In addition, it features specialized glycemic targets for different physiological states, such as a gradual tightening of control during sleep to 120 mg/dL. 4 These functionalities have been shown to significantly improve glycemic metrics, including a 12% increase in time in range (TIR70–180 mg/dL), reductions in mean glucose levels, a 0.38% decrease in HbA1c, and fewer hypoglycemic episodes.5,6
Initially, the Control-IQ system was integrated with the Dexcom G6® continuous glucose monitoring (CGM) system, 7 which provided real-time interstitial glucose measurements. However, advancements in CGM technology have led to the development of the Dexcom G7®, which offers improved accuracy (mean absolute relative difference (MARD) of 8.2% vs 9.0% for the G6), a reduced warm-up time from 2 h to just 30 min, and a more compact sensor design. 8 These enhancements are expected to optimize closed-loop performance by minimizing system downtime and enhancing real-time insulin delivery.
In early 2024, the European Medicines Agency approved the integration of the Tandem t:slim X2 Control-IQ system with the Dexcom G7. Given the potential benefits of this updated configuration, this study aims to prospectively evaluate the impact of transitioning from the Dexcom G6 to the Dexcom G7 in individuals using the Tandem t:slim X2 with Control-IQ. The primary objective was to assess improvements in glycemic control (TIR70–180), while secondary outcomes focused on evaluating patient-reported outcomes (PROMs) and experiences (PREMs) over a 3-month follow-up period.
Methods and ethics
Study design
This study was conducted in accordance with the principles of the Declaration of Helsinki and received approval from the Ramón y Cajal Ethics Committee (Madrid, Spain) on January 24, 2024 (Registration number: 022/24). Written informed consent was obtained from all participants after a comprehensive explanation of the study procedures.
This multicenter, prospective, observational, and longitudinal study was conducted across six Diabetes Units in tertiary care hospitals in Spain. All investigators were active members of the Diabetes Technology Group of the Spanish Diabetes Society (SED; https://www.sediabetes.org/sobre-el-grupo/sobre-el-grupo-tecnologias-aplicadas-a-la-diabetes/). The study period extended from March to December 2024, employing a competitive enrollment strategy until the required sample size was achieved.
The primary objective of this study is to evaluate the real-world impact of transitioning to the Dexcom G7 CGM system in individuals with T1D using the Tandem t:slim X2 insulin pump with Control-IQ technology. The primary outcome is the change in time in range (TIR70–180 mg/dL). Secondary outcomes include PROMs, PREMs, HbA1c levels, and other CGM-derived parameters.
Participants were followed for a total of 3 months post-transition, with two scheduled visits: a baseline visit, coinciding with the switch from Dexcom G6 to Dexcom G7, and a final follow-up visit at 3 months. The transition was conducted as a routine technological upgrade provided by the manufacturer, in accordance with standard clinical practice. All participating centers adhered to their usual training protocols, providing technical guidance on Dexcom G7 usage without additional structured diabetes education reinforcement. If deemed necessary, adjustments to the system settings (such as insulin delivery parameters or CGM alert thresholds) were made throughout the study period, based on clinical judgment or patient preference, ensuring individualized optimization of glycemic control. 4 These configuration changes were documented during the final follow-up visit, recording whether adjustments had been made in the last 3 months (Yes/No).
The study incorporates both retrospective and prospective data collection. The study timeline is outlined as follows (see Figure 1): Baseline visit (Day 0): (i) Transition from Dexcom G6 to Dexcom G7; (ii) Retrospective CGM data collection from three time points: −90, −30, and −14 days prior to the transition, concluding the day before the switch; (iii) Anthropometric measurements and HbA1c assessment; (iv) Administration of baseline PREMs and PROMs questionnaires. Final visit (+90 days post-transition): (i) Anthropometric measurements and HbA1c assessment; (ii) Prospective CGM data collection at +14, +30, and +90 days post-transition, with the transition day included in the prospective dataset; (iii) Administration of final PREMs and PROMs questionnaires.

Study timeline. The schematic illustrates the study timeline, including retrospective and prospective data collection periods surrounding the transition from Dexcom G6® to Dexcom G7® (Day 0). Retrospective data collection: CGM data were retrieved at three time points before the transition (−90, −30, and −14 days), concluding the day before switching to Dexcom G7. Prospective data collection: at three time points after the transition (+14, +30, and +90 days), incorporating the day of the G7 transition in the analysis. Participants underwent two key study visits: Baseline Visit (Day 0): Included baseline questionnaires, anthropometric measurements, and HbA1c assessment. Final Visit (+90 days): Included final questionnaires, anthropometric measurements, and HbA1c assessment.
Study population
The study recruited individuals with T1D from diabetes care centers who met the following inclusion criteria: (i) Diagnosis of T1D; (ii) Age ⩾18 years; (iii) Current use of the Tandem t:Slim X2 insulin pump with Control-IQ; (iv) Prior use of the Dexcom G6 CGM system for at least 3 months before study enrollment; (v) Willingness to transition to the Dexcom G7 system; (vi) Ability to provide informed consent and comply with study procedures; (vii) Adequate use of the system and adherence to the Control-IQ automatic mode.
Participants were excluded from the study if they met any of the following criteria: (i) Presence of any significant medical condition or comorbidity that, in the investigator’s judgment, could interfere with study participation, affect glycemic management, or compromise safety; (ii) Current or planned use of AID systems other than the Tandem t:slim X2 with Control-IQ; (iii) Pregnancy or intention to conceive during the study period; (iv) Any condition or circumstance that, according to the investigator’s assessment, could pose a risk to the participant’s safety or confound study outcomes.
Study protocol
At the baseline visit, eligibility criteria were verified, and participants provided written informed consent before enrollment. Demographic and clinical data relevant to T1D management were recorded, including diabetes duration, time on Tandem t:slim X2 with Control-IQ therapy, and history of severe hypoglycemia. A comprehensive medical history was obtained, documenting current medications, smoking status, cardiovascular risk factors, and the presence of microvascular complications. Macrovascular complications were also recorded. Participants underwent a complete physical examination, including measurements of blood pressure, heart rate, height, and weight (measured in light clothing and without shoes). Body mass index (BMI) was calculated (kg/m2) and categorized accordingly. Laboratory assessments included renal function, lipid profile, and HbA1c, which were analyzed both at baseline and after 3 months of follow-up.
Data collection and measures
Glycemic control
Glycemic control was assessed through CGM metrics and HbA1c levels. The following CGM-derived parameters were collected: percentage of time spent in hypoglycemia (time below range: TBR<70, TBR<54, and total TBR<70), time in range (TIR70–180), and hyperglycemia (time above range TAR>180, TAR>250, and total TAR>180); mean glucose levels (mg/dL), glucose management indicator (GMI, %), coefficient of variation (CV, %), and glucose standard deviation (SD, mg/dL).
These parameters were analyzed according to the International Consensus on Time in Range. 9 Additional recorded metrics included total daily insulin dose (U/day), basal-to-bolus insulin ratio, and time spent in Sleep or Exercise Mode (in hours). Outcomes were analyzed per participant and stratified by times of interest (−14, −30, and −90 days before and +14, +30, and +90 days after the transition from Dexcom G6 to Dexcom G7). Data was extracted from the CGM and Tandem t:slim X2 Control-IQ system using the Glooko™ platform (https://my.glooko.com/).
Patient-reported outcomes
To assess the impact of transitioning to the Dexcom G7 system on patient experience, multiple validated patient-reported outcome measures (PROMs) were administered at two time points: (i) Baseline assessment (prior to Dexcom G7 initiation) and (ii) Follow-up assessment (after 3 months of Dexcom G7 use). The following PREMs and PROMs were used to evaluate key psychosocial and treatment-related aspects of diabetes management:
Hypoglycemia awareness: Evaluated using the Clarke Hypoglycemia Awareness Score,10,11 which identifies impaired awareness of hypoglycemia and the associated risk of severe hypoglycemic episodes.
Quality of life: Assessed through two diabetes-specific instruments: - The Diabetes Quality of Life (DQoL) Questionnaire, which measures the perceived impact of diabetes on daily life, social relationships, and emotional well-being.12,13 - The Type 1 Diabetes Life (ViDa1) Questionnaire, designed to evaluate the specific challenges and lifestyle implications associated with T1D.
14
Diabetes-related distress: Measured using the Diabetes Distress Scale (DDS), which quantifies the emotional burden and stress related to diabetes management. 15
Treatment satisfaction: Assessed using the Diabetes Treatment Satisfaction Questionnaire (DTSQ), which evaluates patient satisfaction with diabetes therapy, including ease of use, perceived effectiveness, and overall treatment experience following the switch to Dexcom G7. 16
All questionnaires are validated for use in adult populations. The questionnaires were used with appropriate permission for academic, non-commercial purposes and are available in Supplemental File 1.
Sample size calculation
The sample size was calculated using the Granmo program (https://apisal.es/Investigacion/Recursos/granmo.html) to detect a clinically significant difference of 4% in time in range (TIR70–180), assuming a common SD of 11%. 5 Accepting an alpha risk of 0.05 and a beta risk of 0.2 in a two-sided test, 90 patients transitioning to the Dexcom G7 CGM system integrated with the Tandem t:slim X2 with Control-IQ are required to detect a 4% difference in TIR70–180. A 10% loss to follow-up was accounted for in the calculation. A 4% absolute difference in TIR70–180 was selected for the sample size calculation. This threshold was chosen based on three key considerations: (i) the greater interindividual variability and potential confounding inherent to real-world observational studies, which may obscure smaller effects; (ii) the need to ensure feasibility of recruitment and follow-up within a defined time frame and clinical setting; and (iii) the intention to reduce the risk of type II error by targeting an effect size large enough to be both clinically relevant and statistically detectable in a heterogeneous population.
Statistical analysis
Continuous variables are presented as mean ± SD, while categorical variables are expressed as absolute counts and percentages (%). The normality of continuous variables was assessed using the Kolmogorov–Smirnov test. If normality assumptions were not met, a logarithmic transformation was applied as necessary.
Analyses were stratified by sex. To compare baseline characteristics between subgroups (men and women), we used an unpaired t test for normally distributed continuous variables, ANOVA for multiple group comparisons, and χ2 or Fisher’s exact test, as appropriate, for categorical variables. Longitudinal changes in CGM-derived metrics across study visits were analyzed using a repeated-measures general linear model (GLM). Baseline TIR70–180 (>70% vs <70%) was included as a between-subjects factor, while visit time points (−90, −30, −14 days prior to transitioning to Dexcom G7 and +14, +30, +90 days post-transition) were considered within-subjects factors. Analyses were adjusted for diabetes duration, and both main effects and interactions were examined. A stratified analysis based on baseline TIR70–180 (<70% vs ⩾70%) was conducted to explore differential responses according to initial glycemic control. Although not pre-specified as a primary endpoint, this threshold is supported by clinical consensus and was selected a priori as a relevant subgroup for exploratory purposes. p-Values are reported for completeness, but the results should be interpreted accordingly.
To assess longitudinal changes in HbA1c levels, PREMs and PROMs scores between the baseline and +90-day visits, a repeated-measures GLM was employed. In this analysis, sex (male/female) and baseline TIR70–180 (>70% vs <70%) were included as between-subjects factors, while study visits were considered within-subjects factors.
An intention-to-treat approach was used, including all enrolled participants. Missing data were handled using the last observation carried forward (LOCF) method. The LOCF method was applied independently within each treatment period (G6 and G7), without carrying forward values across periods. A two-sided p-value < 0.05 was considered statistically significant. Statistical analyses were performed using IBM SPSS Statistics v.29 (IBM España S.A., Madrid, Spain).
Results
Study population characteristics
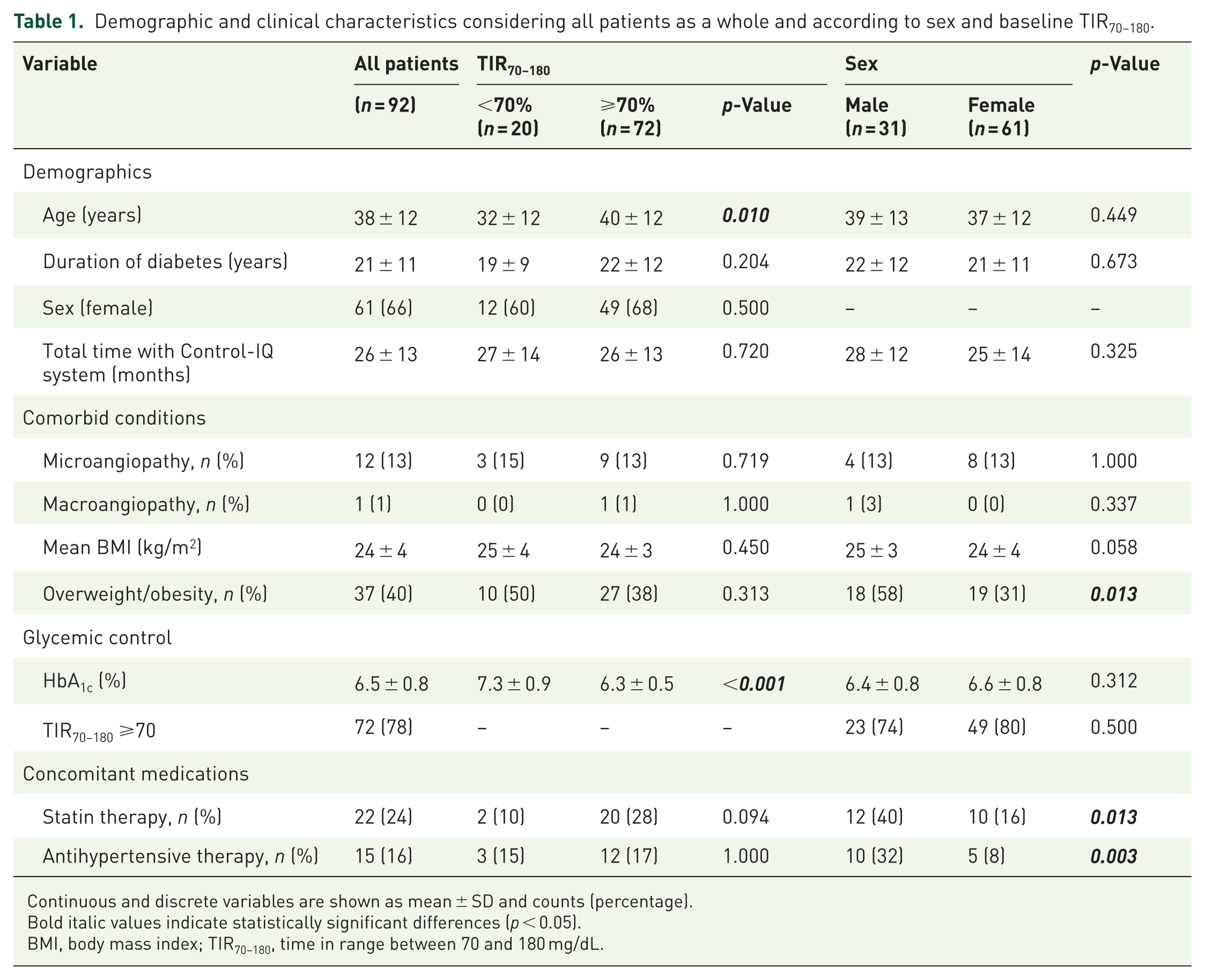
Of the 92 participants with T1D included in the analysis, 31 (34%) were male and 61 (66%) were female. The proportion of patients with TIR70−180 ⩾70% was comparable between sexes. At baseline, the mean age was 38 ± 13 years, with a mean diabetes duration of 21 ± 11 years. The mean HbA1c was 6.5% ± 0.8% (47 ± 9 mmol/mol). Table 1 summarizes the demographic and clinical characteristics of the study population, both overall and stratified by sex and baseline TIR70–180 (<70% vs ⩾70%). No significant differences were observed between males and females in terms of age, diabetes duration, or total time using the Control-IQ system. Males had a significantly higher prevalence of overweight/obesity compared to females (58% vs 31%, p = 0.013). They were also more likely to receive statin therapy (40% vs 16%, p = 0.013) and antihypertensive treatment (32% vs 8%, p = 0.003). In contrast, no significant differences were observed between sexes in the prevalence of microangiopathy, macroangiopathy, mean BMI, or HbA1c levels.
Demographic and clinical characteristics considering all patients as a whole and according to sex and baseline TIR70–180.
Continuous and discrete variables are shown as mean ± SD and counts (percentage).
Bold italic values indicate statistically significant differences (p < 0.05).
BMI, body mass index; TIR70–180, time in range between 70 and 180 mg/dL.
When stratifying participants by baseline TIR70−180, individuals with baseline TIR70−180 <70% (n = 19) were significantly younger (32 ± 12 vs 40 ± 12 years, p = 0.010) compared to those with TIR70−180 ⩾70% (n = 72). No significant differences were found in sex distribution, total time using the Control-IQ system, or mean BMI between the two groups. As expected, patients with baseline TIR70−180 <70% exhibited a significantly higher HbA1c (6.3% ± 0.5% vs 7.3% ± 0.9%, p < 0.001). The prevalence of microangiopathy and macroangiopathy did not differ significantly between groups.
Notably, all participants completed the full 3-month follow-up period, with no withdrawals recorded.
Glycemic control and use of the system
At baseline, the study cohort exhibited high adherence to sensor use, with CGM active for 93% ± 12% of the time. The Control-IQ AID system was engaged 91% ± 13% of the time, while Sleep Activity Mode was activated for 30% ± 15% of the time. The mean sensor glucose level was 148 ± 21 mg/dL, with a CV of 33% ± 6%. The overall baseline GMI was 6.8% ± 0.4%.
The overall TIR70–180 remained stable throughout the study, with no significant differences observed between baseline (mean TIR70–180 76% ± 11% at −90 days) and the end of follow-up (mean TIR70–180 77% ± 10% at +90 days; p = 0.446). However, following the transition from Dexcom G6 to G7 in users of the Tandem t:slim X2 Control-IQ system a reduction in mean glucose levels (Δ = −6 mg/dL; p = 0.010), a decrease in GMI (Δ = −0.1%; p = 0.012), and an increase in the proportion of auto-correction boluses from 14.1% to 16.4% (Δ = +2.3 percentage points; p = 0.026), were observed. Table 2 summarizes longitudinal changes in CGM-derived metrics, HbA1c levels, and insulin dosing across the study period for the overall cohort. No significant differences were observed in sensor wear time or time spent in Control-IQ mode between the Dexcom G6 and G7 periods (Table 2).
Longitudinal changes in continuous glucose monitoring metrics, HbA1c levels, and insulin dosing throughout the study period in whole group.

Data are mean ± SD. Longitudinal changes in CGM metrics and insulin dosing were analyzed at each study time point (−90, −30, −14 days before transitioning to Dexcom G7® and +14, +30, +90 days post-transition). A repeated-measures general linear model was used to evaluate differences over time, with adjustments for diabetes duration.
Bold italic values indicate statistically significant differences (p < 0.05).
Significant differences between −90 days with +90 days.
CGM, continuous glucose monitoring; CV, coefficient of variation; GMI, glucose management indicator; SD, standard deviation; NS, not statistically significant (p ≥ 0.05); TAR, time above range; TBR, time below range; TIR, time in range; TTIR, time in tight range (70–140 mg/dL).
Among participants with baseline TIR70−180 <70%, a significant improvement in glycemic control was observed over the study period. TIR70−180 increased from 61% ± 9% at day −90 to 65% ± 7% at day +90 (p = 0.007), accompanied by a significant rise in time in tight range (TTIR70−140), from 34% ± 8% to 39% ± 7% (p = 0.027). This group also experienced a significant reduction in TAR>250, from 13% ± 7% to 10% ± 6% (p = 0.003), and a decrease in total TAR>180, from 37% ± 9% to 33% ± 8% (p = 0.024). In line with these improvements, HbA1c levels declined significantly from 7.3 ± 0.9 at baseline to 7.0% ± 0.6% at day +90 (p = 0.001). Table 3 shows longitudinal changes in CGM metrics, HbA1c levels, and insulin dosing throughout the study stratified by baseline TIR70−180 (<70% vs ⩾70%). In addition, both GMI and mean sensor glucose levels significantly decreased over time, reflecting an overall improvement in glycemic control. These changes were consistent across groups, with no significant differences based on baseline TIR70–180 stratification (Table 3). Figure 2 shows the changes in glycemic metrics following the transition from Dexcom G6 to Dexcom G7 in participants with baseline TIR70–180 <70%.
Longitudinal changes in continuous glucose monitoring metrics, HbA1c levels, and insulin dosing throughout the study period, stratified by baseline TIR70−180 (>70% vs <70%).
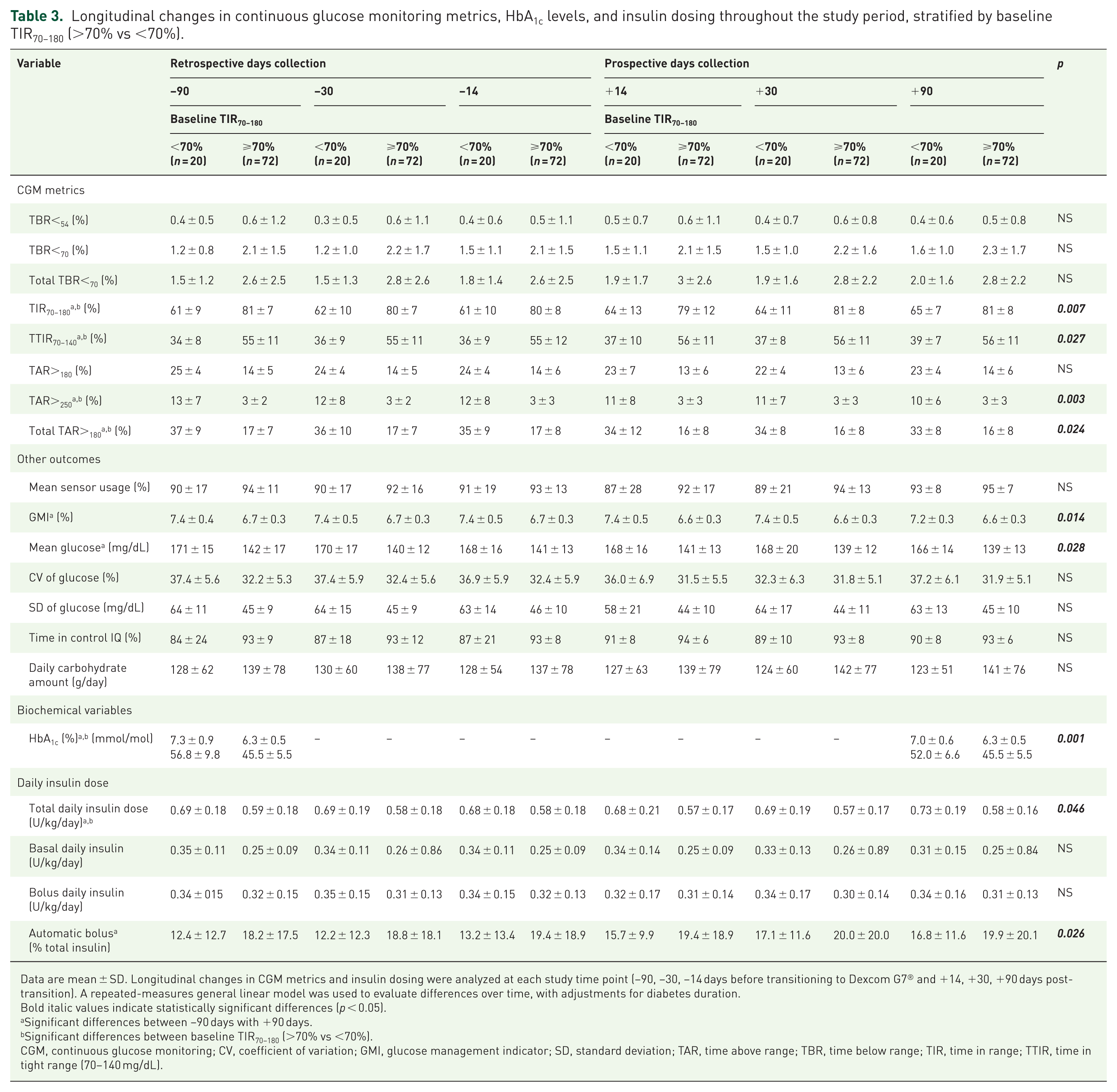
Data are mean ± SD. Longitudinal changes in CGM metrics and insulin dosing were analyzed at each study time point (−90, −30, −14 days before transitioning to Dexcom G7® and +14, +30, +90 days post-transition). A repeated-measures general linear model was used to evaluate differences over time, with adjustments for diabetes duration.
Bold italic values indicate statistically significant differences (p < 0.05).
Significant differences between −90 days with +90 days.
Significant differences between baseline TIR70–180 (>70% vs <70%).
CGM, continuous glucose monitoring; CV, coefficient of variation; GMI, glucose management indicator; SD, standard deviation; TAR, time above range; TBR, time below range; TIR, time in range; TTIR, time in tight range (70–140 mg/dL).

Changes in glycemic metrics following the transition from Dexcom G6® to Dexcom G7® in participants with baseline TIR70–180 <70%. Stacked bar graphs represent the percentage of time spent in different glycemic ranges at each time point, comparing retrospective data collected with Dexcom G6 (days −90, −30, and −14) and prospective data collected with Dexcom G7 (days +14, +30, and +90). Green: TIR70–180; Yellow: TAR>180; Orange: TAR>250; Red: TBR<70. Statistically significant improvements were observed in TIR70–180 (p = 0.007), TAR>250 (p = 0.003), and total TAR>180 (p = 0.024) at day +90 compared to baseline measurements.
Regarding insulin therapy, both the total daily insulin dose (expressed in units per kilogram of body weight) and the percentage of automatic boluses showed significant changes over time. However, no significant differences were observed in the distribution between basal and bolus insulin (Table 2). In addition, at the final study visit, 27 participants (29%) reported having made changes to the Control-IQ system settings.
PROMs and PREMs
All participants completed the baseline PREMs and PROMs questionnaires, with a final response rate of >95%, ensuring robust data collection. Questionnaire scores, including overall and subscale results at baseline and after 3 months of Dexcom G7 use, are presented in Table 4. After 3 months of transitioning from the Dexcom G6 to the Dexcom G7 using the Control-IQ system, significant improvements were observed in diabetes distress and quality of life, while diabetes treatment technology satisfaction and hypoglycemia perception remained unchanged.
Patient-reported outcomes.

Questionnaire scores, including overall and subscale results at baseline and after 3 months of Dexcom G7 use. Data are presented for all participants and stratified by baseline TIR70−180 (>70% vs <70%) and sex. Scoring Interpretation: Clarke Score: >3 points indicate impaired hypoglycemia awareness. Response Rate: >95% questionnaire response rate. Data are expressed as mean ± SD. DDS: lower scores signify less diabetes-related distress. DQoL: lower scores reflect better quality of life. DTSc: higher scores indicate greater satisfaction with treatment. ViDa1: lower scores denote better quality of life.
Bold italic values indicate statistically significant differences (p < 0.05).
Significance indicators: ap < 0.05: Significant change at 3 months compared with baseline for all participants.
p < 0.05: Significant interaction between baseline TIR70–180 (>70% vs <70%).
p < 0.05: Significant interaction between sex.
p < 0.05: Significant interaction between baseline TIR70–180 (>70% vs <70%) and sex.
DDS, Diabetes Distress Scale; DQoL, Diabetes Quality of Life; DTSc, Diabetes Treatment Satisfaction Questionnaire; TIR70–80, time in range between 70 and 180 mg/dL.
Diabetes distress (DDS)
Diabetes distress scores significantly decreased from 2.4 ± 1.1 at baseline to 2.2 ± 1.0 at 3 months (p < 0.005), indicating a reduction in the emotional burden of diabetes management. Specifically, significant improvements were observed in the subdomains of “emotional burden” (Δ = −0.3 ± 0.5, p < 0.05), “regimen-related distress” (Δ = −0.2 ± 0.4, p < 0.05), and “interpersonal distress” subdomains (Δ = −0.2 ± 0.5, p < 0.05), while no significant changes were found in the “physician-related distress” subdomains.
Quality of life (DQoL and ViDa1 questionnaires)
Overall quality of life improved significantly. The DQoL total score decreased from 90 ± 20 to 87 ± 21 (p < 0.005). A significant interaction was observed in the Satisfaction subdomain, where participants with a baseline TIR70–180 <70% experienced greater improvements (p < 0.005). Additionally, women showed a significantly higher decrease in Satisfaction scores compared to men (p < 0.005), suggesting a more pronounced benefit in perceived treatment satisfaction among female participants. Similarly, the ViDa1 total score reduced from 112 ± 11 to 109 ± 10 (p < 0.005), confirming an enhancement in perceived well-being. Women reported significantly greater improvements in ViDa1 scores compared to men (p < 0.05 for interaction).
Diabetes technology satisfaction and hypoglycemia perception (Clarke Test)
The diabetes technology satisfaction (DTSc) score remained unchanged, with baseline and 3-month (values of 30 ± 7 vs 29 ± 7, p = NS) indicating stable satisfaction with diabetes technology despite the sensor transition. Similarly, the Clarke test did not show significant changes (2.1 ± 2.0 vs 2.1 ± 1.9, p = NS), suggesting that the perception of hypoglycemia unawareness was not affected by the transition to the new sensor.
Safety
All participants successfully completed the study, and no discontinuations of the Dexcom G7 system due to adverse events were reported. After the 3-month study period, only two participants opted to revert to the Dexcom G6 sensor while continuing with the Control-IQ system; however, this decision did not lead to early withdrawal from the study.
Seven participants (7.6%) experienced mild dermatological reactions, none of which warranted discontinuation of sensor use or further medical evaluation.
Additionally, 27 out of 92 participants (29.3%) reported issues related to the Dexcom G7 system. Among these, 22% noted inadequate glucose measurement accuracy, 22% reported connectivity loss with the insulin pump, 11% experienced sensor detachment, 7% reported a sensor lifespan of less than 10 days, and 37% did not specify a particular reason. In 22% of participants, subjective perceptions of inaccurate glucose readings were reported in the final survey, although these were not confirmed by CGM performance data. Despite these reports, none of the issues led to early study withdrawal or significant disruptions in glycemic management.
Overall user satisfaction with the Dexcom G6 sensor, assessed on a numerical scale from 1 to 10, was 8.26 at baseline. After 3 months of using the Dexcom G7, the score remained virtually unchanged at 8.32 (p = NS), indicating a comparable user experience with no significant differences observed.
One severe hypoglycemic episode requiring third-party assistance was reported. Upon review, the event was attributed to a carbohydrate counting error during a meal bolus, rather than a malfunction of the sensor or algorithm. Notably, no hospitalizations or fatalities were recorded throughout the study period.
Discussion
This study evaluated the real-world impact of transitioning from the Dexcom G6 to the Dexcom G7 sensor in individuals with T1D using the Tandem t:slim X2 insulin pump with Control-IQ technology. The results reveal that overall glycemic control remained stable after transitioning from the G6 to the G7 sensor. However, participants with suboptimal baseline TIR70–180 (<70%) showed significant improvements in glycemic outcomes (increase in TIR70–180, TTIR70–140, and a reduction in TAR >250, in total TAR>180 and HbA1c levels), suggesting a potential benefit in this subgroup. In addition, the transition from Dexcom G6 to Dexcom G7 in Control-IQ users led to significant improvements in diabetes distress and quality of life (both DQoL, ViDa1 scores, and DDS questionnaire), particularly in participants with lower baseline TIR70–180 and in women. However, no significant changes were observed in DTSc or hypoglycemia perception (Clarke test). The high questionnaire response rate (>95%) underscores the strong adherence and feasibility of this technological upgrade in real-world settings, supported by a high adherence to the protocol and the absence of early dropouts.
Our findings align with previous real-world studies evaluating the Tandem Control-IQ system, which have consistently shown improvements in TIR70–180 and HbA1c, particularly in individuals with baseline suboptimal glycemic control.17–19 The observed increase in TIR70–180 and reduction in TAR>180 in previous real-world data from the One-Year Real-World Use of Control-IQ study reported sustained glycemic benefits over 12 months. 20 The efficacy of the Control-IQ system in glycemic control is supported by a previously published meta-analysis of randomized controlled trials, which reported an approximate 12% increase in TIR, along with reductions in mean glucose levels and HbA1c. 5 However, most prior studies used the Control-IQ technology integrated with the Dexcom G6 sensor, whereas our study specifically assessed the transition to the Dexcom G7. To our knowledge, this is the first study to evaluate the transition from Dexcom G6 to G7 and to assess the efficacy of the Control-IQ system in combination with Dexcom G7 in a real-world setting, while also providing a comprehensive assessment of quality of life, treatment satisfaction, and safety.
The improved accuracy of the Dexcom G7 (particularly in the hyperglycemic range TIR>250) 8 could significantly affect the performance of the Control-IQ system, especially in patients with a baseline TIR70–180 outside recommended targets. The reduction in MARD in hyperglycemia observed on the Dexcom G7 compared to the G6 (6.3% vs 5.4%, in the arm) suggests greater accuracy in detecting elevated glucose levels, which would allow for a more accurate response of the insulin automation algorithm.7,8 Because Control-IQ adjusts insulin delivery based on MCG data, the lower inaccuracy at higher glucose levels could lead to better correction of hyperglycemia, optimizing TIR70–180 for individuals with suboptimal basal glycemic control. These findings would support the hypothesis that integration of Dexcom G7 with Control-IQ technology could provide additional clinical benefits for populations with a higher hyperglycemic burden. However, we recognize that these changes may be at least partly attributable to regression to the mean, a phenomenon commonly observed in repeated-measures studies. This possibility should be considered when interpreting these subgroup findings. These findings remain exploratory and require confirmation in future controlled studies.
It is also important to consider potential confounding factors, such as behavioral modifications induced by study participation (Hawthorne effect) and the use of different CGM systems (Dexcom G6 vs G7) across study phases, which may introduce measurement variability or affect glycemic outcomes independently of true metabolic changes. Nonetheless, the parallel improvement observed in HbA1c—particularly among individuals with suboptimal baseline control—supports the interpretation of a genuine improvement in metabolic status.
Despite reports of more frequent connectivity issues with Dexcom G7 compared to its predecessor, these did not lead to discontinuation or treatment disruption. Overall, these findings support the safety and tolerability of Dexcom G7 in Control-IQ users, with no significant barriers to adoption and a high level of user retention.
Our findings on quality of life and satisfaction with the Control-IQ system align with previous studies. For example, Meade et al. 21 reported high overall satisfaction with the system, along with significant reductions in HbA1c levels at 3, 6, and 12 months of use. In addition, a study by Pinsker et al. 22 highlighted improvements in diabetes-related quality of life, ease of use, and seamless connectivity with the CGM system. These results further support the effectiveness of Control-IQ in improving both clinical outcomes and psychological personal outcomes (PREMs and PROMs) in real-world settings.23,24 Although changes in some PROMs and PREMs reached statistical significance, the magnitude of change cannot always be considered clinically relevant. Minimal variations could reflect an improvement in perceptions of usability rather than a significant change in self-management behaviors. Moreover, we recognize the possibility of a placebo effect, given that participants were aware of switching to a newer and widely perceived as superior technology. This expectancy bias could partially account for the positive evaluations observed, particularly in subjective experience metrics.
This study has several strengths. First, to our knowledge, it is the first study to evaluate the transition from Dexcom G6 to Dexcom G7 and assess the efficacy, security, PREMs, and PROMs of the Control-IQ system in combination with Dexcom G7 in a real-world setting. Second, the study was prospective and multicenter, enhancing the generalizability of the findings. Third, a formal sample size calculation was conducted during the study design phase, ensuring that the study was adequately powered to detect significant changes in glycemic control. In addition, the high response rate to quality-of-life questionnaires strengthens the reliability of PROMs, providing valuable insights into the user experience.
However, this study has several limitations that must be acknowledged. First, its observational, non-randomized design limits the ability to infer causality and introduces potential confounding factors such as visit frequency, behavioral adaptation, and regression to the mean. The absence of a control group limits our ability to interpret whether improvements in PROMs reflect actual glycemic changes, a novelty effect, or user perception, and these findings should therefore be considered exploratory. Second, although the sample size was based on a formal power calculation, it remains relatively small, and the follow-up period of 3 months may not capture long-term effects. Third, the use of selection criteria, informed consent, and structured questionnaires, while necessary for ethical and methodological rigor, may reduce the generalizability of findings to broader real-world populations. Fourth, approximately 29% of participants underwent adjustments in Control-IQ settings during the study, which may have influenced outcomes. Prior randomized data suggest that Control-IQ performance is largely robust across standard configuration approaches and does not require extensive individualization to achieve effective glycemic control. 25 However, recent evidence suggests that adjustments to parameters such as glucose target or correction factor are significantly associated with time in range. 26 Therefore, we cannot exclude the possibility that the observed glycemic changes may be more attributable to algorithm reconfiguration than to the transition from Dexcom G6 to G7. Unfortunately, we did not collect detailed information on the exact nature of the configuration changes made by each participant, which limits our ability to assess the specific impact of these adjustments. Finally, the subgroup analysis based on baseline TIR70–180 was not pre-specified and should be considered exploratory, although it was informed by clinically relevant thresholds. These limitations highlight the need for future randomized studies with larger and more diverse populations. Some participants experienced mild skin-related issues, such as irritation or allergic reactions, which were not systematically assessed and warrant further investigation. Future research should consider these aspects to better isolate the specific impact of the technology.
In conclusion, the transition from Dexcom G6 to G7 in users of the Control-IQ system did not result in a significant change in glycemic control. However, in a subgroup of patients with baseline TIR70–180 <70%, an improvement was observed across several glycemic parameters. To our knowledge, this is the first study to assess this transition and to evaluate the efficacy of the Control-IQ system in combination with Dexcom G7 in a real-world setting. However, these results should be interpreted with caution due to the observational study design and its inherent limitations. These findings support the safety and efficacy of Dexcom G7 integration with hybrid closed-loop insulin therapy. Future research should explore long-term adherence, connectivity performance, and the impact of Dexcom G7 on broader diabetes populations.
Supplemental Material
sj-pdf-1-tae-10.1177_20420188251407515 – Supplemental material for Prospective multicenter evaluation of glycemic and patient-reported outcomes following transition to a next-generation continuous glucose monitoring system in users of advanced hybrid closed-loop technology
Supplemental material, sj-pdf-1-tae-10.1177_20420188251407515 for Prospective multicenter evaluation of glycemic and patient-reported outcomes following transition to a next-generation continuous glucose monitoring system in users of advanced hybrid closed-loop technology by Lía Nattero-Chávez, Mar Lorenzo, María Soledad Ruiz de Adana, Olga Simó-Servat, Mercè Abad, Carmen Quirós, María Durán Martínez, Alejandra Quintero Tobar, Ane Bayona, Teresa Ruiz Gracia, Esther de la Calle de la Villa and María José Picón in Therapeutic Advances in Endocrinology and Metabolism
Footnotes
Acknowledgements
We thank the participants and the clinical teams involved in the study.
Declarations
Supplemental material
Supplemental material for this article is available online.
Declaration of generative AI and AI-assisted technologies in the writing process
During the preparation of this work, the authors used ChatGPT to enhance the language and clarity of the text. After using this tool/service, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
