Abstract
Cancer negatively impacts bone health through various mechanisms, including treatment-induced bone loss and malignant bone lesions, often leading to increased fracture risk and higher morbidity and mortality. Antiresorptive agents (bisphosphonates and denosumab) are the current standard of care to reduce the risk of skeletal-related events and prevent treatment-related fragility fractures in patients with cancer. While there is strong evidence to support their benefits in cancer, there is potential room for further reduction in the risk of pathologic and fragility fractures. This narrative review explores the potential role and theoretical concerns regarding the use of osteoanabolic agents in cancer. We also discuss management challenges, such as recurrent pathologic fractures, fragility fractures, and osteonecrosis of the jaw that may arise in clinical practice, prompting consideration of the use of osteoanabolic agents in patients with a history of cancer. Preclinical studies have shown conflicting outcomes regarding the effects on cancer with parathyroid hormone treatment, but promising results with the use of anti-sclerostin antibody in cancer models. Definitive conclusions cannot be drawn from available preclinical and clinical data. Theoretical risks exist for both cancer survivors and patients with advanced cancer in the bone. Therefore, the risk-benefit ratio should be carefully considered when evaluating the use of an osteoanabolic agent in the cancer context.
Introduction
The current standard of care to preserve bone health across the cancer continuum from nonmetastatic cancer to advanced cancer involving the bone is antiresorptive therapy with bisphosphonates or denosumab (Dmab), both of which have high-level evidence to support their use in clinical settings. 1 In nonmetastatic settings, the goal is prevention of bone loss and fragility fractures. In advanced cancer involving the bone, the aim is to prevent skeletal-related events (SREs), a composite clinical endpoint that includes pathologic fracture, need for radiation to bone, need for surgery to bone, or spinal cord compression. 1 While approved osteoanabolic therapies (teriparatide, abaloparatide, and romosozumab) have advanced osteoporosis treatment, their role in cancer-related bone health remains relatively unexplored. Here we address challenging clinical scenarios such as treatment-related osteoporosis with recurrent fragility fractures and osteonecrosis of the jaw, which may prompt the clinician to consider the use of osteoanabolic agents. 1
This narrative review provides an overview of the impact of cancer on bone health and discusses the current standard of care for optimizing bone health in individuals with cancer. Importantly, this review will explore the rationale for using antiresorptive therapy as the current standard of care, focusing on its mechanism of action in addressing the underlying pathophysiology in cancer. We will also examine the potential role of osteoanabolic agents, for example, parathyroid hormone (PTH) analogs and the anti-sclerostin antibody romosozumab, in modulating this pathophysiology and address potential future use in individuals with cancer.
Impact of cancer and cancer treatment on bone health
Cancer treatment-induced bone loss
Treatment for many cancers induces a hypogonadal state, often a result of oophorectomy, chemotherapy, and hormone deprivation therapy. Estrogen and androgen deficiency disrupts bone remodeling by increasing bone resorption, leading to accelerated bone loss, compromised bone quality, and increased risk of fragility fractures. 2 In men with prostate cancer, osteoporotic fractures in patients receiving androgen deprivation therapy are more common than pathologic fractures due to metastatic bone disease.3,4
Hormone deprivation therapy may be administered for years. For example, aromatase inhibitors are first-line therapy for postmenopausal women with estrogen receptor-positive breast cancer as an adjuvant endocrine therapy for 5–10 years, and in some patients, it may be continued lifelong. 5 Therefore, efforts to optimize bone health and prevent fragility fractures in patients with cancer on hormonal deprivation therapy are of great importance. 6 Due to a long track record of efficacy, the drug of choice is either bisphosphonates or Dmab, both of which increase bone density and lower the risk of fractures. 7
A large meta-analysis of randomized controlled trials has shown that bisphosphonates improve survival in postmenopausal breast cancer patients via a reduction in the development of metastatic bone disease. 8 The American Society of Clinical Oncology recommends discussing adjuvant bisphosphonates as a potential treatment option in all postmenopausal women with primary breast cancer. 9 The “seed and soil” hypothesis attributes increased bone turnover to a more fertile soil for cancer cells to take route. Thus, one can speculate that adjuvant bisphosphonates lower the risk of bone metastases as dormant cancer cells are less likely to be stimulated by the release of bone matrix-derived growth factors, angiogenesis, or various other mechanisms in the setting of potent antiresorptive therapy. 10 In early stage breast cancer patients without bone metastases, elevated bone resorption at baseline (as demonstrated by the marker serum collagen type I (CTX)) predicted a higher risk of the development of metastatic bone disease. 11
Two randomized controlled trials investigating the use of adjuvant benefit of Dmab in patients with breast cancer have demonstrated conflicting results.12,13 Therefore, the adjuvant benefits of Dmab remain a matter of debate in women with early breast cancer. The fact that the ABCSG-18 study of adjuvant denosumab yielded an adjuvant benefit similar to that of bisphosphonates is noteworthy and suggests that the common denominator for potential adjuvant benefit may be the potent antiresorptive effect both bisphosphonates and denosumab possess. These results support the hypothesis that the “soil” of bone can be modified by potent antiresorptive drugs. In addition, receptor activator of nuclear factor kappa-B ligand (RANKL) is a targetable pathway in BRCA1 mutation carriers, suggesting that RANKL blockade is a promising strategy for preventing breast cancer associated with this gene abnormality. 14 A randomized controlled trial of men with nonmetastatic castration-resistant prostate cancer at high risk for progression (baseline prostate-specific antigen (PSA) ⩾8.0 ng/mL and/or PSA doubling time ⩽10.0 months) showed that Dmab increased bone metastasis-free survival. However, since there was no change in mortality and an increase in the incidence of osteonecrosis of the jaw, the United States Food and Drug Administration (FDA) approval was not granted for this indication. 15
Immune checkpoint inhibitors are widely used in treating cancer due to their efficacy profile. Recent observational studies have raised concerns about their impact on bone health. These studies have shown that the use of immune checkpoint inhibitors is associated with an increased risk of fractures within the first year, though this risk appears to decrease after the initial year. 16 These findings are supported by preclinical studies showing that PD-1 blockage reduces bone mass in mice due to increased bone resorption. While T cells are proposed to contribute to the observed bone loss, the exact mechanisms have yet to be elucidated. T cell activation can promote bone resorption by increasing the production of cytokines such as RANKL and tumor necrosis factor alpha.17–19
Bone metastasis
Pathophysiology of bone metastases
Bone is the third most common site for metastasis, after the lung and liver. In breast and prostate cancer, bone is the most common secondary site, affecting 70%–90% of patients with metastatic cancer.20,21 The progression of metastatic tumor cells in bone is determined by the bone microenvironment and tumor-intrinsic factors. 22 Bone remodeling is a dynamic process in which the balance between bone-forming osteoblasts that synthesize bone matrix and bone-resorbing osteoclasts that degrade matrix is tightly regulated. Cancer cells disrupt physiological bone remodeling by producing factors that activate the bone cells. For instance, cancer cells may secrete PTH-related peptide (PTHrP) that stimulates osteoblasts. Osteoblasts play an important role in creating a favorable environment for bone metastasis through various mechanisms, including the release of interleukin 6 and RANKLTN, both of which promote osteoclast differentiation and bone resorption. Increased bone resorption leads to elevated levels of transforming growth factor beta-1 and other bone-derived cytokines released from the bone matrix, further stimulating tumor cell proliferation. This process has been called the “vicious cycle of bone metastasis” (Figure 1).22,23 Conversely, the bone microenvironment may be unfavorable for cell proliferation, causing disseminated tumor cells (DTCs) to enter a dormant state. DTCs occupy niches in the bone after arrival from the bloodstream. Osteoblasts interact with DTCs in both the perivascular and osteogenic/endosteal niches, respectively, and can drive cancer cell colonization and proliferation.22,24 Osteoblasts and bone marrow vasculature are implicated in creating a dormancy-promoting microenvironment through expression of pro-dormancy factors, including thrombospondin-1 (TSP-1) and C-X-C motif chemokine 12 (CXCL-12).25–27 Gap junctions formed between the DTCs and osteoblast lineage cells allow for direct calcium transfer to the cancer cells. This “feeding” of the DTCs by the osteoblast appears to promote DTC proliferation and chemoresistance. Thus, the role of osteoblasts in tumor cell dormancy is complex, and the effect of osteoanabolic agents in this context remains incompletely understood. Cancer cells can remain dormant in the bone marrow for years before relapse. Although the mechanisms triggering dormant tumor cells to awaken are relatively unknown, osteoclasts and osteoblasts are implicated in this process. 22 Strikingly, dormant tumor cells have even been shown to be present in a state of ostensibly localized cancer, ductal carcinoma in situ. 28 Active bone metastases are often complicated by morbidities, such as SREs or admission for hypercalcemia, all of which can be debilitating and can severely interfere with the quality of life.29,30 Of even greater concern, bone metastases are associated with considerable reductions in overall survival.31–33

Vicious cycle of bone metastases and bone-targeting therapies. Upon extravasation in the bone marrow, DTC may lodge close to OBs and enter a stage of dormancy. Once cancer cells proliferate in bone, they secrete several cytokines, including PTHrP, which stimulates osteoblasts to produce excessive RANKL. RANKL activates the OCs and upon increased bone resorption, growth factors, such as TGF-β1, are released from the bone matrix. These factors stimulate cancer cell proliferation, creating the “vicious cycle” of bone metastases (black arrows). BP and Dmab induce osteoclast apoptosis and inhibit differentiation, respectively, and are approved for the treatment of metastatic bone disease. These antiresorptive agents also reduce osteoblast activity due to coupling. In contrast, PTH analogs exert their bone anabolic action by increasing bone formation and subsequently resorption. In addition, preclinical data suggest they act on cancer cells via PTH receptors and reduce PTHrP expression. Anti-Scl-Ab increases bone formation while reducing bone resorption. The effect of bone anabolic agents on DTC dormancy and activation, as well as cancer cell proliferation, is largely unknown. Although not noted in this figure, the role of immune cells in metastatic bone disease is an area of active investigation.
Treatment and prevention of SREs
The goals of SRE therapy include pain control, prevention of skeletal morbidity, and local tumor control. Options include analgesics, antiresorptive agents, systemic cancer therapy, and/or radiation therapy. The use of antiresorptive agents in patients with bone metastases has been a major advancement in clinical care, shifting from a palliative to a more proactive approach aimed at delaying and preventing complications. Antiresorptive agents, such as zoledronic acid (ZOL) and Dmab, are approved by the FDA and the European Medicines Agency for the prevention of SREs. Dmab is a fully humanized monoclonal antibody that specifically binds to human RANKL to inhibit osteoclast maturation and activity, resulting in reduced bone resorption, tumor-induced bone destruction, and SREs. However, RANK and RANKL are not specific to bone metabolism; they are expressed in a wide variety of immune cells and are involved in various immune processes. 34 Dmab, as opposed to bisphosphonates, has no bone binding affinity and its effects on bone turnover and bone mineral density are reversible upon discontinuation. Dmab was FDA approved in 2010 to be administered at the dose of 120 mg subcutaneously (SC) every 4 weeks in patients with solid tumors with documented bone metastases and patients with multiple myeloma to delay and decrease SREs,35–38 with a randomized study demonstrating non-inferiority with every-12-week dosing. 39
ZOL decreases bone resorption by exerting an apoptotic effect on mature osteoclasts and prolonging the secondary mineralization of bone. It attaches to hydroxyapatite-binding sites on sites of active bone remodeling, impairing the ability of osteoclasts to adhere. ZOL was FDA approved in 2001 to be administered at the dose of 4 mg intravenously every 4 weeks in patients with solid tumors with documented bone metastases and patients with multiple myeloma to delay and decrease SREs,35,36 with a randomized study demonstrating non-inferiority with every-12-week dosing. 36 Randomized placebo-controlled trials demonstrated that parenteral antiresorptive agents lower the incidence of SREs in patients with advanced cancer involving the bone. The reduction in bone turnover, as measured by biochemical markers of bone turnover induced by antiresorptive therapy, correlates with protection against SREs and with reduced mortality.40,41 Two head-to-head studies of ZOL and Dmab, in breast and prostate cancer, respectively, demonstrated superiority for Dmab in terms of SRE prevention 42 but at the expense of a numerically although not statistically significantly higher risk of osteonecrosis of the jaw (Dmab: 52/2841 (1.8%) vs Zol: 37/2836 (1.3%), p = 0.13).43,44 Mechanistically, this benefit of Dmab is likely due to its more global inhibition of the osteoclast lineage cells and more potent antiresorptive efficacy as reflected by greater suppression of bone turnover markers. However, no significant differences were observed in overall survival and disease progression. Thus, the clinical relevance of the “superiority” demonstrated by denosumab over ZOL is debatable, and in clinical practice, guidelines do not prefer one agent over the other. 35
Common adverse effects (AE) for ZOL include transient acute-phase reaction or flu-like symptoms. ZOL may also cause renal toxicity in susceptible individuals. Hypocalcemia is a serious AE associated with both ZOL and Dmab, which may be particularly severe in those with risk factors such as a large burden of osteoblastic metastases, chronic renal failure, vitamin D deficiency, or impaired calcium absorption due to gastric bypass or other surgeries.45,46 Other rare serious AE common to both agents are atypical femoral fractures and osteonecrosis of the jaw.37,38 An important and concerning AE unique to Dmab is multiple vertebral fractures after drug discontinuation, which is likely related to a rebound increase in bone resorption after termination of the treatment. This AE has been mostly described in osteopenia/osteoporosis patients, but cases in patients being treated for metastatic bone disease have been described as well. 47 In patients with advanced cancer involving the bone, it remains to be determined whether this increase in bone resorption could increase the risk for SREs.
Multiple myeloma
Myeloma bone disease affects 80%–95% of patients with multiple myeloma and typically manifests as osteolytic lesions, with or without diffusely reduced bone density. This has significant clinical implications, as 50% of patients with multiple myeloma develop pathologic fractures. Notably, the impact on bone health persists beyond active disease, with 13% of patients in remission experiencing fragility fractures.48,49 Monoclonal gammopathy of undetermined significance, although considered an asymptomatic condition, has also been associated with an increased risk of fractures. 50
Myeloma bone disease is characterized by uncoupling of bone turnover: increased bone resorption and decreased bone formation. Increased bone resorption in multiple myeloma is driven by several pathways, including increased expression of RANKL due to direct interaction between multiple myeloma cells and bone marrow stromal cells. In addition, interaction between multiple myeloma and the immune system stimulates the release of cytokines that increase osteoclast activity and decrease osteoblast activity.51,52 Bone formation is also reduced by suppression of the Wnt pathway. 53 Levels of the osteocyte-derived Wnt inhibitor sclerostin are elevated in the serum of patients with multiple myeloma and correlate clinically with advanced disease. 54 Thus, there is rationale to suggest that osteoanabolic agents could be beneficial for patients across the myeloma spectrum.
The standard of care for the treatment of myeloma bone disease is antiresorptive therapy, that is, bisphosphonates or Dmab. Intravenous bisphosphonates reduce the risk of vertebral fractures and SREs by 26% compared with placebo in patients with multiple myeloma. 55 The drug of choice is determined based on side effects, cost, and route of administration. Antiresorptive agents are effective in reducing bone resorption and ultimately also reduce bone formation. Cancer patients who continue to experience SREs despite treatment with antiresorptive agents present a significant clinical challenge.
Diamond and colleagues performed a 12-month open-label pilot study in 12 myeloma patients who received SC teriparatide daily (20 mcg) in combination with anti-myeloma therapy and previous intravenous bisphosphonates. The subjects had severe osteoporosis and recurrent osteoporotic vertebral compression fractures. The serum bone formation marker propeptide of type 1 collagen (P1NP) increased by 4.8-fold, and lumbar spine quantitative tomography bone mineral density increased by 43.8%. No new fractures or lytic lesions were seen, suggesting favorable anabolic effects of teriparatide in patients with myeloma and severe osteoporosis. 56 In addition, sclerostin antibody treatment in preclinical models protects bone from multiple myeloma-induced bone destruction, maintains bone mass, and lowers the number of osteolytic lesions.57,58 These encouraging preclinical results have already been followed by an ongoing pilot clinical trial at Memorial Sloan Kettering Cancer Center using romosozumab in patients with multiple myeloma, osteoporosis, and lytic lesions (unpublished results).
The potential role of osteoanabolic agents in managing complex cases
Solid tumors with bone involvement
While antiresorptive agents delay the time to first SRE and reduce the overall risk of developing an SRE, there is potential for further reduction of risk. Thus, a clinically relevant question is whether osteoanabolic agents could, in select patients with advanced cancer in the bone, improve the bone protection afforded by antiresorptive agents. There are currently no published clinical trials on the impact of osteoanabolic agents on SRE risk in patients with metastatic bone disease. Preclinical research on the use of osteoanabolic medications in advanced cancer in the bone is currently limited, but growing. Given the strong clinical benefits of antiresorptive agents, a strong preclinical rationale would be necessary to pursue osteoanabolic therapy as an avenue of study in the nonmetastatic and metastatic settings.
Intermittent PTH increases bone mass through a remodeling-based mechanism, which raises the concern of increased cancer cell proliferation in response to resorption-related cues. Indeed, in a preclinical model of prostate cancer, PTH has been shown to increase the number of metastatic prostate cancer cells in the bone. 59 In contrast, in breast cancer, preclinical studies have demonstrated that intermittent administration of PTH for 4 weeks reduced spontaneous metastasis from breast to bone. 60 Furthermore, intermittent PTH treatment reduced breast cancer cell engraftment, alleviated tumor growth in bone, and protected bone integrity in an intratibial model of tumor growth in bone and osteolytic disease. Consequently, the survival of breast cancer-bearing mice was prolonged upon PTH treatment. 60 This was due to reduced metastatic burden in the bones since PTH did not affect metastasis to visceral organs. PTH signals through PTH 1 receptor (PTH1R), which is expressed by osteoblasts and breast cancer cells. Animal studies have shown that PTH1R expression in both cell types is important for the protective effect of PTH in bone metastasis. Further studies showed that PTH decreases the expression of PTHrP in breast cancer cells—one of the drivers of the vicious cycle 61 —suggesting a possible mechanism of PTH action in the metastatic environment. In another study where PTH was administered only for 5 days prior to MDA-MB-231 breast cancer cell injection, no effect on cancer cell homing to hind limbs was found. 62 However, in this preclinical model, tumor burden in skeletal sites other than the hind limbs was increased. Although these conflicting outcomes may be explained by the different experimental conditions used in the studies (e.g., duration of PTH intervention and/or continuous vs intermittent PTH), the findings highlight the complexity of PTH treatment in cancer with bone involvement (Table 1).
Preclinical and clinical studies of osteoanabolic agents in cancer.

Ab, antibody; BMD, bone mineral density; LS, lumbar spine; MRONJ, medication-related osteonecrosis of the jaw; P1NP, propeptide of type 1 collagen; PTH, parathyroid hormone; QCT, quantitative computed tomography.
Since PTHrP is one of the drivers of the vicious cycle of metastatic bone disease, the potential use of abaloparatide, a synthetic peptide analog of PTHrP, in the treatment of osteolytic disease is partially counterintuitive and has been under-investigated. However, a preclinical study was conducted very recently using two different breast cancer cell lines, triple-negative MDA-MB-231 and ER+ MCF7 cells, in a bone metastases mouse model. Interestingly, in both models, abaloparatide increased bone mass in bone metastases-bearing mice due to enhanced bone formation. No effect on breast cancer growth in bone or metastasis to soft tissue was observed. 63 These results suggest that abaloparatide might be a valid approach to protect the bone from osteolytic disease, a hypothesis requiring further preclinical and/or clinical research.
Romosozumab is an agent with dual actions: it begins with a rapid but transient anabolic effect, followed by a sustained antiresorptive phase. This unique pharmacodynamic effect makes it an attractive osteoanabolic medication to consider in cancer patients with complex clinical histories. 64 Of note, a history of cancer is not a contraindication for romosozumab. Preclinical studies have shown that treatment of established bone metastases in breast cancer with anti-sclerostin antibody reduces tumor growth in bone and protects bone from cancer-induced destruction.65,66 Similar effects were seen in studies using a small-molecule inhibitor against sclerostin.57,58,67 In contrast, Wnt signaling is also implicated in tumor progression and metastasis in various cancers, including breast, raising some concerns about using romosozumab in cancer indications. 68 Indeed Wnt pathway inhibitors have been studied as anticancer therapy for various tumor types. However, sclerostin is an inhibitor of Wnt1 ligands only and therefore should be selective for pre-osteoblasts. 69 Preclinical studies with sclerostin inhibition in myeloma and breast cancer have shown either no effect or a reduction in tumor growth with sclerostin inhibition. Although there is no contraindication for the use of romosozumab in cancer patients, one of the exclusion criteria in the pivotal clinical trial was a history of cancer (Table 1). 70 Regarding its safety profile, romosozumab demonstrated a higher incidence of serious cardiovascular events in one head-to-head randomized trial versus alendronate. It should be avoided in patients at a high risk of myocardial infarction or stroke. 71
PTH analogs stimulate bone remodeling and could improve osseous wound healing in the oral cavity. 72 Based on this rationale, teriparatide has been used to manage challenging cases of medication-related osteonecrosis of the jaw (MRONJ) in individuals with active cancer. In a randomized controlled trial of 47 MRONJ lesions in 34 patients (13/15 (86.7%) of teriparatide-treated subjects had active cancer as the etiology of MRONJ), the use of teriparatide for 8 weeks compared with placebo was associated with a greater rate of resolution of MRONJ at 12 months (45.4% vs 33.3%). There were “no cases of new malignancy or worsening of preexisting malignancy” among participants who received teriparatide. However, the trial was of short duration, did not specifically address changes within metastatic bone lesions, and was not powered to demonstrate safety in patients with cancer. 73 There were two prostate cancer patients (it was not stated whether these subjects had active osteoblastic metastases) treated with teriparatide in this study. In clinical practice, teriparatide is not the standard of care to treat MRONJ and is an expensive medication, often difficult to obtain.
In cases of Dmab-induced MRONJ, clinicians face a difficult dilemma: continuing treatment to avoid risk for bone loss, SREs, and/or multiple vertebral fractures may hinder MRONJ healing, whereas withdrawing Dmab to support healing may increase the risk of vertebral fractures. With both options carrying significant risks, alternative strategies need to be considered. Available data suggest that when teriparatide is given to patients who were pretreated with Dmab, transient bone loss results, followed by an increase in bone density at the lumbar spine (though the overall bone density benefit is blunted) and a small decrease at the total hip. 74 Therefore, this strategy is not the standard of care and is usually avoided. Romosozumab is a potential option in this situation since its anabolic action may theoretically be favorable in the context of active MRONJ and its antiresorptive effect benefits the increase in bone resorption induced by Dmab withdrawal.75,76
In addition, PTH analogs can lead to increases in serum calcium. This is a concern for their potential use in metastatic bone disease, as metastatic cancer in the bone may be complicated by hypercalcemia of malignancy (HCM) in 20%–30% of patients. HCM is mediated via a variety of mechanisms, including osteolytic lesions, secretion of PTHrP, and overproduction of 1,25-dihyroxyvitamin D. 77 This is not a concern with romosozumab, which can rarely lead to hypocalcemia. In contrast to PTH analogs, antiresorptive agents are invaluable in the treatment of HCM. 77
PTH increased the number of metastatic lesions in preclinical studies on prostate cancer, a cancer associated with blastic bone lesions. Therefore, we generally avoid all osteoanabolic therapy in the solid tumor patient with active osteoblastic metastases, given that these lesions already demonstrate an overactivity of osteoblasts. 78 For future research on this patient population, romosozumab presents a theoretically more attractive option than PTH or PTHrP analogs due to its ability to decrease, rather than increase, bone resorption. While preclinical studies on romosozumab are promising, the lack of published clinical trials means its potential remains speculative.
Treatment-induced bone loss without advanced cancer involving the bone
Fragility fractures secondary to osteoporosis are associated with significant morbidity and mortality.29,30,79,80 Advances in the use of osteoanabolic agents have updated the osteoporosis treatment paradigm. Osteoanabolic agents are recommended as the first-line approach for individuals at a very high risk of fracture (e.g., T-score ⩽−3.5 or multiple recent fractures). 81 Clinical data on the efficacy and safety of osteoanabolic agents in cancer survivors or patients with active cancer are scarce, and their use in these patients may represent a clinical dilemma. The landmark clinical trials of approved anabolic drugs excluded patients with cancer and more recent studies specifically excluded subjects with a history of recent cancer within the past 5 years.71,82 As mentioned earlier, PTH and PTHrP may theoretically create a favorable environment for bone metastasis by increasing bone resorption and/or stimulating osteoblasts. Therefore, the use of osteoanabolic agents in this population is generally avoided. However, individuals with cancer and osteoporosis who are suffering fractures despite treatment with the current standard of care antiresorptive therapy have limited options. The decision to begin an osteoanabolic therapy should consider the individual patient’s risk of recurrence at that point in time. For example, after 10 years of remission, one could expect the risk of recurrence to be relatively low in early stage breast cancer. However, since cases of bone recurrence have been reported even beyond a decade, a multidisciplinary discussion with the oncologist is essential to carefully weigh the risks and benefits before proceeding with osteoanabolic treatment.
The one clinical scenario where there is clinical experience with PTH analogs in cancer survivors is in individuals with hypoparathyroidism. An approved agent in the US and EU from 2015 to 2024 (no longer marketed) was recombinant human parathyroid hormone (rhPTH) 1–84. In clinical trials, 76%–85% of participants had postsurgical hypoparathyroidism, a common complication of head and neck surgeries, including those for cancer. Autoimmune disease only accounted for 2%–6% of cases.83,84 Palopegteriparatide is a recently approved PTH analog for hypoparathyroidism; it has a sustained kinetic profile that differs markedly from the pulse in PTH created by teriparatide. As expected, bone turnover markers increase with this PTH replacement, although in hypoparathyroidism, bone remodeling is underactive, given the deficiency in PTH. Theoretically, the pharmacokinetic profile that stimulates bone resorption to a lesser extent would, in turn, be less likely to stimulate dormant tumor cells, but in lieu of animal data, this is speculation. Furthermore, as per the drug label, palopegteriparatide, like other PTH analogs, is not recommended in patients with “bone metastases or a history of skeletal malignancies.” Ongoing registry data with this agent may be helpful in determining long-term bone outcomes in cancer survivors, including the development of metastatic bone disease in survivors of bone-tropic cancers.
In individuals with osteoporosis without cancer, the combination of PTH analogs with Dmab effectively reduced bone resorption and improved bone density. For example, when teriparatide was given with Dmab, bone resorption as measured by serum CTX was lower in those who received the combination compared with those who received teriparatide alone. 85 Such an approach might be beneficial for patients with cancer since, as mentioned, increased bone resorption has been associated both with increased bone-only recurrence in breast cancer and with increased SREs and mortality in various advanced cancers involving the bone. Trials of teriparatide combined with ZOL also demonstrated that ZOL restrains the increases in bone resorption brought about by teriparatide, although conflicting bone mineral density results were seen with the combination.86,87 This strategy of combining ZOL or Dmab with PTH analogs could theoretically reap the benefits of osteoanabolic therapy while mitigating the potential risks in cancer (Table 2).85,88,89 Similarly, preclinical studies have demonstrated additive effects of combining sclerostin inhibition with ZOL in terms of restoring multiple myeloma-induced bone loss. Of note, in multiple myeloma, sclerostin antibodies did not suppress bone resorption, unlike in osteoporosis and breast cancer bone metastases. More research is needed to investigate the effect of combined antiresorptive and anabolic therapy in solid tumor bone metastases.57,58,66
Factors to consider in weighing the risks and benefits of osteoanabolic therapy in different clinical situations.
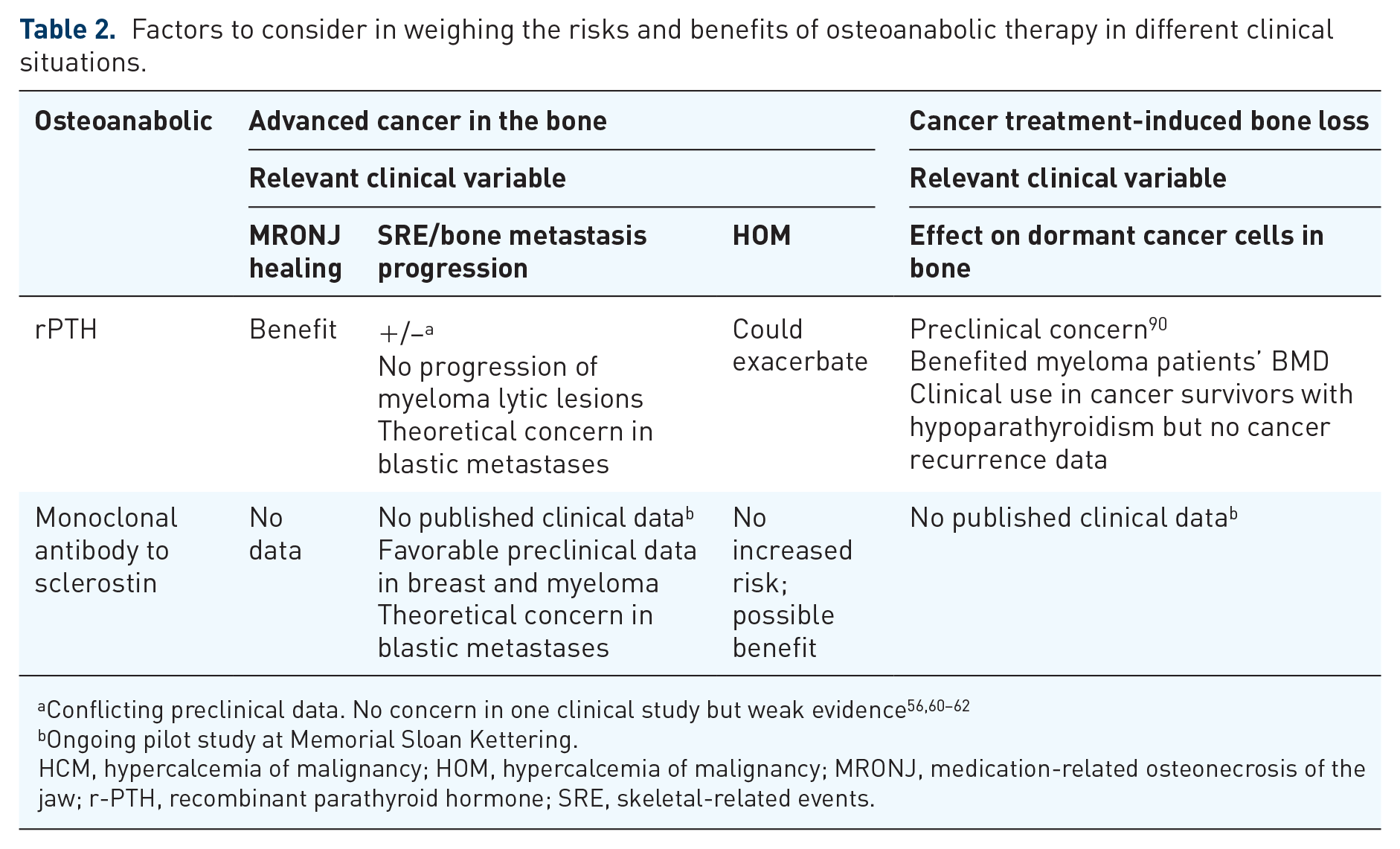
Ongoing pilot study at Memorial Sloan Kettering.
HCM, hypercalcemia of malignancy; HOM, hypercalcemia of malignancy; MRONJ, medication-related osteonecrosis of the jaw; r-PTH, recombinant parathyroid hormone; SRE, skeletal-related events.
Conclusion
Antiresorptive agents have strong clinical data on their efficacy in preventing SREs, treatment-induced bone loss, and fragility fractures. While antiresorptive agents delay the time to first SRE and reduce the overall risk of developing an SRE, there is potential for further reduction of risk. Certain clinical scenarios, including recurrent fractures despite antiresorptive therapy or ONJ, present unique challenges that may not be sufficiently addressed by osteoclast inhibition and may benefit from anabolic effects. Preclinical studies have shown conflicting outcomes regarding the effects on cancer with PTH analog treatment, but promising results with the use of anti-sclerostin antibody. The use of osteoanabolic agents in patients with cancer remains a complex area that requires further exploration. Future clinical research should utilize retrospective analyses of large national databases to carefully assess the efficacy and safety of using osteoanabolic agents in survivors of bone-tropic cancers. In patients with active cancer involving the bone, pilot studies to assess safety are required. For such pilot intervention studies, osteoanabolic approaches that restrain bone resorption (romosozumab or the combination of PTH analog with antiresorptive therapy) appear to be logical starting points. Furthermore, an area in need of further exploration is how stimulation of the Wnt pathway with osteoanabolic agents could interact with the underlying tumor. In summary, the clinician considering the use of osteoanabolic agents in a patient with cancer should be aware that definitive conclusions cannot be drawn from available preclinical and limited clinical data. Theoretical risks exist for both the cancer survivor (activation of dormant cancer cells) and the patient with advanced cancer in the bone (disease progression). Therefore, the risk-benefit ratio should be carefully considered when evaluating the use of an osteoanabolic agent in the cancer context. We generally limit their use to cases of significant morbidity from recurrent fragility fractures or severe MRONJ without active blastic bone metastases.
