Abstract
Bone and mineral disorders are highly prevalent in solid organ transplant recipients. These patients are at high risk for osteoporosis and fragility fractures due to several pre- and post-transplant factors, including end-stage organ disease leading to chronic malnutrition and osteomalacia, as well as chronic immunosuppressive therapy that has direct adverse effects on bone remodeling. Low pre-transplant bone mineral density is associated with an increased risk for fragility fracture post-transplant. Furthermore, there is a precipitous loss of bone density within 6–12 months post-transplant due to a myriad of causal factors. In this review, we will elaborate on the treatment options and challenges in management of osteoporosis in solid organ recipients using vitamin D, calcium, bisphosphonates, denosumab, and osteoanabolic agents. The greatest body of evidence discusses the use of bisphosphonates, with most patients benefiting from early treatment.
Plain language summary
Increased survival in transplant recipients has resulted in the recognition of long-term complications in this population, including osteoporosis and fractures. Due to existing pre-transplant bone disease and post-transplant immunosuppressive therapies, solid organ transplant recipients are at an increased risk for bone loss and fracture. Pre-transplant risk factors should be optimized by addressing secondary etiologies of osteoporosis including vitamin D and calcium insufficiency. Unique bone health factors in kidney, liver, heart, and lung transplant recipients will be reviewed. Early treatment prevents the accelerated bone loss that happens in the immediate post-transplant period. Options for treatment that will be discussed include vitamin D, calcium, bisphosphonates, denosumab, and osteoanabolic agents.
Keywords
Introduction
Solid organ transplantation (SOT) has been performed since 1954 with significant improvements in surgical technique and immunosuppression. 1 Accordingly, SOT recipients have increased life expectancy; median survival after kidney, liver, heart, and lung transplants are 15, 15, 12, and 6 years, respectively. 2 Prolonged survival underscores the importance of addressing bone health, as fractures are highly prevalent with implications on quality-of-life and survival. A retrospective analysis of 10,783 SOT recipients and age-matched controls from 2002 to 2015 showed SOT recipients had 46% higher risk of osteoporosis, and 19% higher fracture risk compared to the general population. Specifically, heart and lung transplant recipients had a 4-fold greater fracture risk. 3
Densitometric diagnosis of osteoporosis in SOT recipients is akin to that for the general population. In premenopausal women and men younger than 50 years old, dual-energy X-ray absorptiometry (DXA)
Pre-transplant considerations by organ transplant type
Risk factors for osteoporosis in patients with end-stage organ disease include older age, postmenopausal status, renal impairment, immobilization, nutritional deficiencies, sarcopenia, hypogonadism, tobacco use, alcohol use, genetic predisposition, family history of fracture, use of medications with adverse effects on bone (including loop diuretics, proton pump inhibitors, and anticonvulsants), and prior exposure to glucocorticoids and other immunosuppressive therapies.6–8 We will review additional unique risk factors by transplanted organ system.
Kidney transplant
There is a high prevalence of osteoporosis (42%–56%) in kidney transplant recipients.9,10 Chronic kidney disease-mineral and bone disorder (CKD-MBD) is a significant contributor to this burden of bone disease as it is prevalent in up to 90%–100% of kidney transplant recipients. 11 Progressive impairment in mineral homeostasis in CKD patients leads to a variable presentation of renal osteodystrophy, including high bone turnover due to hyperparathyroidism, osteomalacia due to reduced calcitriol synthesis, or adynamic bone disease. An added challenge is the confounding presentation of bone turnover, bone volume, and bone mineralization, which may require an iliac crest biopsy for definitive diagnosis of underlying skeletal pathology, as these factors cannot be distinguished by DXA alone. 12
Liver transplant
Nearly 75% of patients with chronic liver disease (CLD) have osteoporosis and/or fractures, known as hepatic osteodystrophy. 13 A recent meta-analysis of 30 studies evaluating liver transplant recipients from 12 countries found a 5-fold increase in incidence of both osteoporosis and fractures. 14
Liver transplant recipients have additional risk factors for skeletal fragility based on the etiology of CLD. Cholestatic liver disease is associated with osteoblast dysfunction from retained bilirubin and bile acids leading to deficiencies in fat-soluble vitamins D and K, which are important for bone remodeling. CLD patients additionally experience growth hormone resistance and reduction in IGF-1 levels, causing reduced osteoblast signaling. Iron deposition in hemochromatosis exerts toxic effects on bones and causes hypogonadism via pituitary iron deposition, compounding the insult on bone remodeling. Moreover, vitamin D hydroxylation is attenuated, and vitamin D degradation is enhanced via CYP24A1 in patients with cirrhosis. 15 Inflammatory bowel disease is another comorbid condition in those with primary sclerosing cholangitis which increases risk of bone disease, mostly via nutritional deficiencies.16,17
Heart transplant
Heart transplant candidates have a prevalence of osteoporosis between 7% and 23%. 18 Risk factors for osteoporosis in these patients include renal impairment from cardiorenal syndrome, loop diuretic-related hypercalciuria, angiotensin-converting enzyme inhibitors, glucocorticoid use, and vitamin D deficiency.18–20
Lung transplant
For lung transplant candidates, estimated prevalence of osteoporosis ranges from 54% to 61% with vertebral fractures present in up to 29% of patients.21–23 Cystic fibrosis patients have additional risk factors including chronic inflammation, glucocorticoid exposure, malnutrition, low body mass, pancreatic insufficiency leading to fat malabsorption and vitamin D deficiency, and hypogonadism. Due to delayed puberty from secondary hypogonadism, chronic inflammation, and malnutrition, peak bone mass accrual can be diminished.16,24
Post-transplant considerations
Rapid bone loss is noted within the first 6–12 months after SOT, primarily mediated by glucocorticoids as well as post-transplant renal dysfunction, malnutrition, immobility, vitamin D deficiency, and use of other immunosuppressive agents. 25 Of note, vitamin D deficiency prevalence remains high post-transplant with reported vitamin D insufficiency in 51%–97% of transplant recipients and severe vitamin D deficiency in 26%–33% of transplant recipients, which stresses the importance of adequate vitamin D supplementation in this high-risk population. 26 Beyond 1 year, the impact of transplant on bone health is variable as the factors noted above may resolve. Generally, the rate of bone loss decelerates 1 year after transplant and in some cases, bone density can recover.11,27 It is believed there is a late phase of recovery 12 months and beyond post-transplant; however, this can be blunted by impairment in osteoblast-mediated bone formation. 19 Early glucocorticoid withdrawal protocols have been increasingly implemented across transplant centers which could aid in the delayed recovery phase post-transplant.28–30
Impact of immunosuppressive medications on bone health
Supraphysiologic glucocorticoid exposure decreases osteoblast-mediated bone formation by causing osteoblast apoptosis via upregulation of Wnt inhibitors like sclerostin. Moreover, glucocorticoids promote osteoclast differentiation and action by suppressing osteoprotegerin. Glucocorticoid-mediated stimulation of receptor activator of nuclear factor kappa-B ligand also increases osteoclastogenesis. These changes lead to bone resorption. In addition, glucocorticoids reduce intestinal calcium absorption and increase urinary calcium excretion. Glucocorticoid excess also leads to secondary hypogonadism which compounds bone loss. 31
Calcineurin inhibitors (CNI) include tacrolimus and cyclosporine which may be used in steroid-sparing regimens. In rat models, CNIs were found to have rapid cortical and trabecular bone loss due to high turnover. 32 CNIs can cause hypomagnesemia and hypercalciuria, further contributing to hyperparathyroidism and bone resorption. 33 However, in SOT recipients, identifying the individual effects of CNI on bone loss serves difficult as contributing factors to bone loss include post-transplantation status, glucocorticoid use, and the effect of other immunosuppressive medications prescribed. Tacrolimus and cyclosporine have not been compared in head-to-head trials. Some trials have compared CNI monotherapy to glucocorticoid immunosuppression regimens post-transplant with similar rates of bone density loss. 34 However, CNIs may overall be protective for bone health by reducing glucocorticoid dosing. Other immunosuppressive agents like mycophenolate mofetil, azathioprine, and sirolimus are neutral for skeletal fragility.34,35
Kidney transplant
Kidney transplant recipients can lose up to 14.5% of BMD within 6 months, with a 34% higher risk of fracture compared to pre-transplant.11,36 Those who receive kidney–pancreas transplant have an even higher risk of fracture of up to 49%. 11 Although some pre-transplant osteoporosis risks improve after kidney transplant with parathyroid hormone (PTH) reaching nadir 1 year post-transplant, PTH can remain inappropriately elevated in 20% of patients 1 year post-transplant and contribute to ongoing risk for fracture.11,37
Liver transplant
In a meta-analysis, liver transplant recipients had a median hip BMD decrease of 7% by 6 months. 14 Gradually post-transplant, liver transplant recipients may have resolution of cholestasis, improvement in vitamin D levels, and return to eugonadal status that can improve bone metabolism and facilitate recovery.25,30,38
Heart transplant
Heart transplant recipients lost 3%–10% BMD at the lumbar spine (LS) and 6%–11% at the femoral neck (FN) within 1 year, and 12%–36% of patients experienced a new fracture within 1 year post-transplant. There is evidence this BMD loss may slow and even recover after 6 months post-transplant, at a time when glucocorticoids may be tapered off. 20 Issues contributing to ongoing bone density loss include ongoing use of angiotensin-converting enzyme inhibitors and loop diuretics, which may be used still in the post-transplant period. 19
Lung transplant
Lung transplant recipients lost 2%–5% BMD within 1 year, and up to 14% of patients sustained vertebral fractures within 1 year post-transplant.16,22,39 Lung transplant recipients remain on chronic glucocorticoids for immunosuppression for longer duration than seen with kidney, liver, and heart transplant recipients, in whom research has demonstrated rationale for early glucocorticoid withdrawal.40–43
Treatment of osteoporosis in solid organ transplant recipients
General principles of treatment of post-transplantation osteoporosis
Given early BMD loss post-transplantation, skeletal health should be addressed in all SOT recipients. Regular weight-bearing exercise is recommended as tolerated starting early post-transplant, which limited data suggests may hasten BMD recovery after initial losses post-transplant.44–46 All patients should be counseled regarding adequate calcium intake and vitamin D supplementation to maintain 25-hydroxyvitamin D (25OHD) greater than 30 ng/mL. The use of calcitriol post-transplantation was not found to be effective compared to placebo and conferred higher risk of hypercalciuria.6,16 Calcitonin is a possible adjunct to therapy but is insufficient as monotherapy to treat the rapid BMD loss post-transplant. 6 Replacement of sex hormone in hypogonadal patients addresses additional secondary factors to optimize bone health.6,16
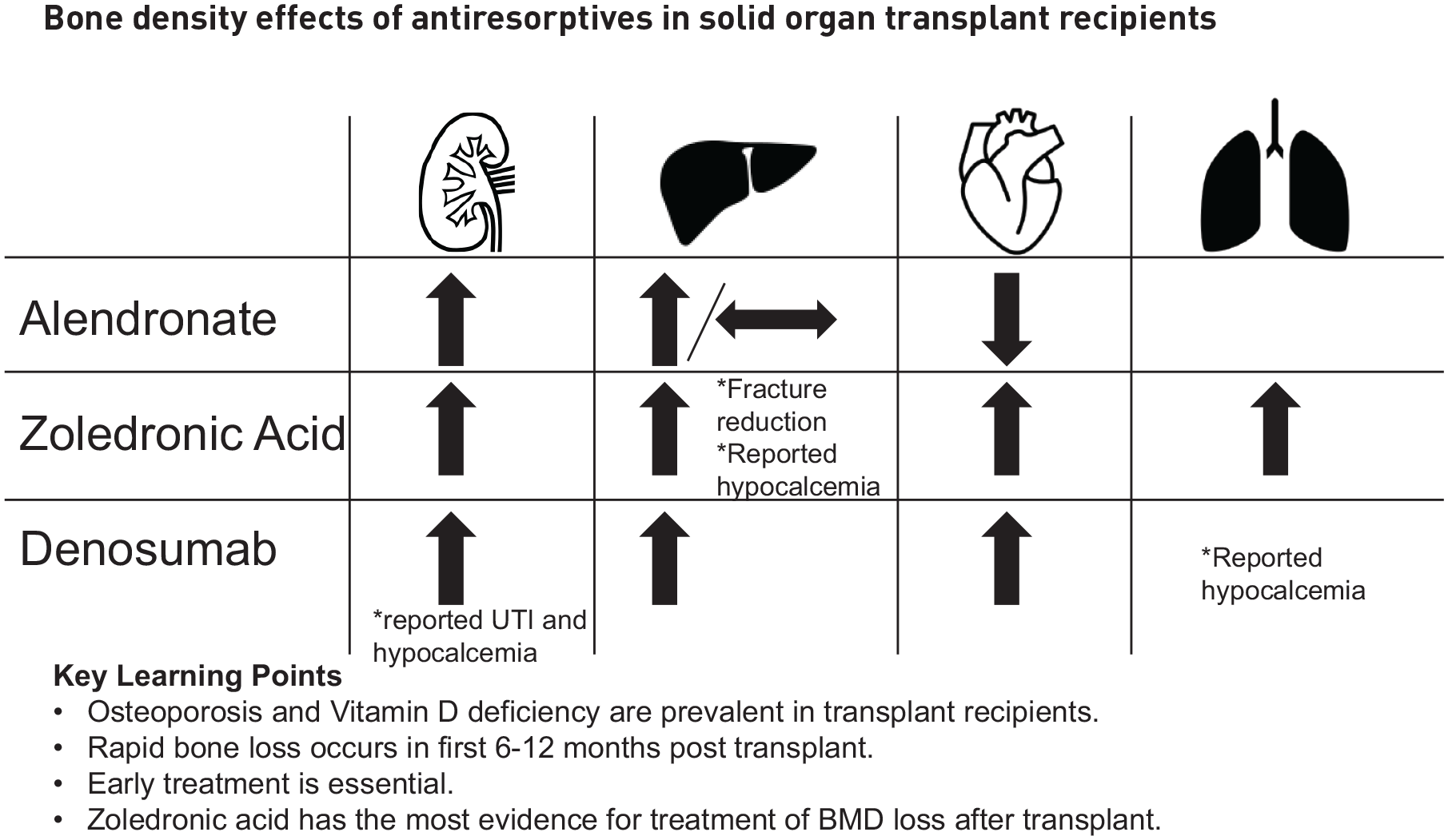
This review will focus further on treatment with bisphosphonates, denosumab, and osteoanabolic agents including PTH analogs. Studies were selected for review which exclusively studied SOT recipients with more recent randomized controlled trials prioritized for review. In review of therapeutics without randomized controlled trials published to this date, observational studies or case studies were included (Tables 1 and 2).
Randomized controlled trials evaluating therapeutic options for treatment of low bone density in solid organ transplant recipients.

BMD, bone mineral density; CI, confidence interval; FN, femoral neck; LS, lumbar spine; UTI, urinary tract infection; ZOL, zoledronic acid.
Nonrandomized studies evaluating therapeutic options for the treatment of low bone density in solid organ transplant recipients.

BMD, bone mineral density; FN, femoral neck; LS, lumbar spine; UTI, urinary tract infection; ZOL, zoledronic acid.
Treatment of osteoporosis in the kidney transplant recipient
Kidney Disease Improving Global Outcomes (KDIGO) 2017 Clinical Practice Guideline Update for Diagnosis, Evaluation, Prevention, and Treatment of CKD-MBD recommends that kidney transplant recipients undergo early DXA to guide treatment. Moreover, they discuss the role of iliac crest biopsy in guiding treatment given DXA alone cannot delineate the underlying mechanisms of renal osteodystrophy. Currently, there is insufficient clinical and objective data to help guide osteoporosis treatment choices or duration due to the complex and multifaceted nature of CKD-MBD, which can evolve post-transplant. KDIGO guidelines recommend considering treatment with vitamin D, calcitriol, and/or pharmacologic therapy for osteoporosis in the first 12 months post-transplantation. 47 Below are potential treatment options for kidney transplant recipients with osteoporosis.
Vitamin D
As discussed previously, the prevalence of vitamin D deficiency in SOT recipients is high and in part contributes to the magnitude of bone loss post-transplant. As vitamin D aids in calcium and phosphorus absorption which are essential building blocks for bone, it is imperative to ensure patients receive adequate supplementation to goal 25OHD levels >30 ng/mL. However, for those with post-transplantation osteoporosis, vitamin D monotherapy is insufficient and other osteoporosis medications are indicated.
Researchers such as Battaglia et al. have evaluated the impact of vitamin D monotherapy in preventing bone loss post-transplant. In a 2022 study, 100 kidney transplant recipients with Vitamin D insufficiency not on antiresorptive therapy were treated with cholecalciferol 25,000 international units (IU) weekly for 12 weeks and then 1500 IU daily. In addition, 30% of patients also took calcitriol. Over 2–3 years of follow-up, 25OHD levels raised to sufficient levels in 45% of patients; yet there was no significant change in BMD. 48 In 2023, Battaglia et al. conducted another study of 130 long-term kidney transplant recipients with Vitamin D insufficiency/deficiency to compare the impact of new start of vitamin D supplementation in treatment-naïve individuals (no vitamin D, bisphosphonates, or calcimimetics in the past) following the same protocol of cholecalciferol replacement above. They found kidney transplant recipients who did not receive vitamin D supplementation in the past were younger and with a lower prevalence of diabetes, which could represent a population intrinsically at lower risk for osteoporosis and BMD loss. BMD at LS, but not at FN, improved in patients with new start vitamin D supplementation compared to those previously treated with vitamin D. There were no reported fractures during follow-up period of 2 years, but it is unclear if this finding is due to chance. These two studies indicate that vitamin D monotherapy does not appear to be a sufficient treatment to address the rapid onset of BMD loss post-transplant. 49
Bisphosphonates
Several studies have evaluated the efficacy of zoledronic acid (ZOL) in kidney transplant recipients. Marques et al. conducted a randomized control trial (RCT) of 34 kidney transplant recipients with 17 receiving ZOL 5 mg at time of transplant and 17 receiving no treatment except ergocalciferol 50,000 IU monthly. Patients randomized to ZOL treatment underwent iliac crest bone biopsy with tetracycline labeling at time of transplant and again at 1 year. At time of transplant, 19% of patients had osteoporosis, with half of patients in both control and ZOL groups with low bone turnover. Patients were given ZOL at time of transplant regardless of bone turnover status. There was no significant difference in the rates of adynamic bone disease on iliac crest biopsy 1 year after transplant in the control versus ZOL group (63% vs 53%,
Haas et al. evaluated 20 renal transplant recipients who received ZOL 4 mg twice versus placebo within 3 months after transplant. Bone biopsies were measured at transplant and after 6 months. On iliac crest biopsy, which was not tetracycline labeled, osteoid surface increased from 47% to 64% in the ZOL group (
Oral bisphosphonates have also been shown to prevent BMD loss in kidney transplant recipients though there are no studies comparing efficacy of oral to intravenous (IV) formulations. Okamoto et al. conducted a prospective controlled trial of 12 kidney transplant recipients with 5 randomly assigned to alendronate 35 mg weekly and 7 patients to no treatment. Whole body BMD increased 2% (
Denosumab
Denosumab has also been studied in kidney transplant recipients. Bonani et al. completed a prospective RCT of 90 kidney transplant recipients randomized to denosumab or no treatment within weeks of transplant. All patients received vitamin D 800 IU daily and calcium 1000 mg daily and had an average glomerular filtration rate (GFR) 53.5 ± 15.4 mL/min/1.73 m2. After 12 months, denosumab significantly increased BMD at 5% at LS and 2% at total hip compared to <1% change in BMD at both sites in control group (LS
Sayed et al. conducted a RCT of denosumab or alendronate versus no treatment. Patients started treatment on average 4 months after transplant and continued therapy for 1 year. After 1 year of therapy, absolute median change in LS BMD
Brunova et al. also studied the use of denosumab in a retrospective analysis of 34 patients with kidney transplant, 15 patients with combined kidney/pancreas transplant, and 14 patients with liver transplantation. On average, denosumab was started 6 ± 6 years after transplant for a mean duration of 2 ± 1 years. Mean GFR prior to denosumab was 33 ml/min/1.73 m2 (95% confidence interval, 28.8–36.6). Before starting denosumab, 43.2% of patients had 25OHD <30 ng/mL, increasing their risk of hypocalcemia. Despite this, only one patient suffered mild hypocalcemia, likely due to standardized calcium 500–1000 mg and vitamin D 800–1000 IU daily repletion during the study. Prior to treatment, 82% of kidney transplant recipients had osteoporosis at LS with a mean pretreatment
Osteoanabolic agents
Cejka et al. studied teriparatide use in kidney transplant recipients in a 6 month double-blind RCT of 24 patients randomized to daily teriparatide 20 mcg versus placebo. On average, treatment started 21 days after transplant. Iliac crest biopsy (without tetracycline labeling) and DXA were checked at time of transplant and after 6 months of therapy. Patients with persistent severe hyperparathyroidism with less than 50% reduction of PTH post-transplant, bone biopsy diagnosed high turnover disease, or pre-transplant PTH >300 pg/mL were excluded. After 6 months, change in LS BMD did not meet significance with median percentage change +0.07% in teriparatide-treated patients compared to +0.8% in placebo group. At FN, the placebo group had a significant median percentage change of −7% in BMD from baseline (
Limitations and future directions
The studies of bisphosphonates in kidney transplant recipients describe the impact of treatments on BMD change, however, due to short-term follow-up and low volume of patients, no studies found a significant reduction in fracture incidence. In addition, lack of tetracycline labeling with bone biopsy limits the exploration of bone formation. Moreover, not all studies reported evaluation for adynamic bone disease, which is an important consideration given the high prevalence of adynamic bone disease in renal transplant recipients.
Further research is needed to explore the comparative efficacy of oral versus IV bisphosphonates in the treatment of kidney transplant recipients. We believe IV bisphosphonates are preferable to oral due to less frequent dosing interval, which can improve adherence. In addition, IV bisphosphonates are not subject to issues with gastric absorption, have a longer duration of action, and allow providers to treat bone disease early post-transplant before any further renal injury, which could otherwise interrupt courses of oral bisphosphonates.
Furthermore, identifying adynamic bone disease and clarifying bone turnover status in CKD-MBD can be especially challenging in SOT recipients in whom bone turnover markers have not been validated and may be unreliable in the case of low renal clearance. Data on the indications for iliac crest biopsy to guide treatment with antiresorptives are limited and should be further explored. Future research could define patient selection criteria for iliac crest biopsies and explore whether there is a significant benefit in performing bone biopsy to guide treatment compared to empiric antiresorptive treatment without biopsy.
Further research comparing ZOL and denosumab efficacy should also be pursued to guide treatment in those with creatinine clearance >35 ml/min who may still be candidates for bisphosphonate therapy.
Studies with denosumab had a short follow-up duration of 1–2 years and did not find any change in fracture outcomes. Moreover, studies did not discuss how long denosumab was anticipated to be continued or any long-term outcomes/safety on therapy. To date, the FREEDOM open-label extension trial provided data of only up to 10 years of data in non-transplant recipients which prompts further reflection on whether denosumab is an appropriate therapy for younger kidney transplant recipients who may require decades of antiresorptive therapy. 59 In addition, subsequent therapies or fractures from rebound BMD loss after denosumab discontinuation were not discussed. In patients in whom denosumab discontinuation is anticipated, we would recommend sequential therapy with bisphosphonate to avoid rebound BMD loss and fractures, though this could be limited in kidney transplant recipients if creatinine clearance remains below 35 mL/min.
UTI in kidney transplant recipients was found to occur in patients on denosumab, though no long-term follow-up has been conducted on this to determine if UTIs continue to occur years into denosumab therapy post-transplant. Discovering the etiology of this finding and evaluating if there is any impact on long-term kidney function or rejection rates should be pursued to guide risk/benefit discussions regarding denosumab therapy.
Further studies are needed to compare the efficacy of osteoanabolic therapies like teriparatide, abaloparatide, and romosozumab in kidney transplant recipients. Although there is limited data for romosozumab benefit for patients with end-stage renal disease on dialysis to improve spine BMD, there is a paucity of data in kidney transplant recipients. 60 However, osteoanabolic agents may be essential for patients with severe osteoporosis and/or adynamic bone disease, and studies evaluating patient selection for these treatments will be invaluable.
Treatment of osteoporosis in the liver transplant recipient
The American Association for the Study of Liver Diseases 2012 guidelines for long-term management of adult liver transplant recipients recommend early glucocorticoid withdrawal, daily calcium 1200 mg supplementation, and vitamin D supplementation to maintain 25OHD >30 ng/mL. Bisphosphonate therapy is recommended for patients with
Bisphosphonates
Alendronate treatment in liver transplant recipients was studied in a nonrandomized prospective study of 136 liver transplant recipients who were followed for a median of 28 months. A total of 98 patients with osteopenia or osteoporosis pre-transplant were given alendronate 70 mg weekly after transplant (the remainder of patients with normal BMD received calcium/vitamin D supplementation). Only the 32 patients with pre-transplant osteoporosis who received alendronate had a significant increase in BMD after treatment (
Several studies show the impact of ZOL on treatment of post-transplant bone loss. Crawford et al. conducted a RCT of 62 liver transplant recipients and randomized 32 patients to receive ZOL 4 mg or placebo IV saline (30 patients) for a total of five doses in the first year post-transplant. Both control and ZOL groups experienced statistically significant BMD gain at the LS over 12 months but without a significant difference between treatment groups. At the FN and total hip, the mean percentage difference in BMD change between treatment groups was significant at +3% favoring ZOL (
Bodingbauer et al. also conducted a RCT evaluating ZOL in liver transplant recipients. In this study, 47 patients received eight infusions of ZOL 4 mg during the first-year post-transplant while 49 patients received vitamin D and calcium supplementation only. DXA and iliac crest biopsies (not tetracycline labeled) were conducted pre-transplant and at 6 months post-transplant. DXA was repeated at 1-year post-transplant. By year 2 of follow-up, significantly fewer patients in ZOL group fractured compared to control (9% compared to 23%,
Shane et al. compared the effects of single dose ZOL 5 mg versus alendronate 70 mg weekly versus placebo for 12 months in a RCT of 80 heart transplant recipients and 31 liver transplant recipients. Of all participants, liver transplant recipients received one infusion of ZOL 5 mg (14 patients) or alendronate 70 mg weekly (17 patients) or calcium/vitamin D supplementation only (10 patients) for 1 year, starting within 1 month of transplant. Of note, the placebo group had higher BMD prior to treatment compared to alendronate and ZOL groups (
Pennisi et al. conducted a prospective controlled study of 85 liver transplant recipients demonstrating the effects of pamidronate. Forty-three patients with low BMD pre-transplant received pamidronate 30 mg every 3 months for 1 year after transplant. Patients with normal BMD pre-transplant served as the control, receiving only calcium/vitamin D supplementation. Both groups had significant losses in BMD at total hip compared to baseline, though less in the pamidronate group compared to control (5% in pamidronate vs 6% in control,
Denosumab
There are limited data on the use of denosumab in liver transplant recipients. As mentioned above in the treatment of kidney transplant recipients section, Brunova et al. studied 14 liver transplant recipients in addition to kidney and kidney/pancreas transplant recipients who were treated for 1 year with denosumab starting on average 6 years after transplant. All the liver transplant recipients in the study had osteoporosis before denosumab treatment. Liver transplant recipients had an average LS
Osteoanabolic agents
No RCTs on the use of teriparatide, abaloparatide, or romosozumab in liver transplant candidates have been published. However, some preliminary published data describes use of teriparatide in liver transplant recipients with severe osteoporosis. 66
Limitations and future directions
There is insufficient evidence to compare efficacy of alendronate versus ZOL in liver transplant recipients. Follow-up is short, which limits interpretation of fracture outcomes from treatment. It was also noted in studies that patients on ZOL had rates of hypocalcemia of 15%–40% which was not noted as frequently in other studies of other SOT recipients. It is unclear if factors unique to liver transplant, such as the high rates of vitamin D deficiency post-transplant, contributed to higher rates of hypocalcemia.
RCTs studying the impact of denosumab in treatment of bone loss post-transplant will be beneficial. Ideally, it would be helpful to gauge response to therapy started early after transplant to see if this helps blunt the rapid bone loss in the first-year post-transplant. Future studies should also compare efficacy and relative risk of hypocalcemia compared to bisphosphonates given high incidence of hypocalcemia in ZOL studies reported above. Long-term follow-up is also important in future studies given rapid reversibility of denosumab once discontinued.
Treatment of osteoporosis in the heart transplant recipient
The International Society for Heart and Lung Transplantation guidelines for the care of heart transplant recipients recommend DXA within 12 months of transplant. They recommend early initiation of bisphosphonates while awaiting transplantation for those with osteopenia with therapy continued the first year after transplantation. For those with osteoporosis pre-transplant, longer therapy for at least 3 years is recommended. They also recommend calcium and vitamin D supplementation to maintain 25OHD >20 ng/mL. Early glucocorticoid withdrawal protocols are described, and if glucocorticoids are withdrawn within the first-year post-transplant, bisphosphonate therapy could be stopped in those with
Bisphosphonates
Alendronate was studied in a RCT by Shane et al. of 149 heart transplant recipients. Patients were randomized to alendronate 10 mg daily, calcitriol 0.5 mcg daily, or no treatment with treatment starting within 1-month post-transplant. At 1 year, alendronate preserved BMD significantly at LS and FN compared to no treatment. Percentage BMD change at LS was −1% in alendronate (
Another study by Lange et al. studied the use of risedronate and alendronate in a prospective uncontrolled study of 33 heart transplant recipients. Patients were either treated with risedronate 35 mg weekly or alendronate 70 mg weekly along with calcium and vitamin D supplementation. Results were not stratified by risedronate or alendronate when reported. At time of transplant, 8/33 patients had osteoporosis, while by the end of 2 years, only 6/33 had osteoporosis, with BMD improving to osteopenia range for 2 patients. Mean changes in
Shane et al. compared ZOL 5 mg once versus weekly alendronate versus placebo in a RCT of 80 heart and 31 liver transplant recipients, treated for 12 months. Heart transplant recipients received one infusion of ZOL 5 mg (27 patients) or alendronate 70 mg weekly (26 patients) or calcium/vitamin D supplementation only (17 patients) for 1 year, starting within 1 month of transplant. 99% of heart transplant recipients were still on prednisone at 1 year. In the heart transplant cohort, LS BMD increased by 2% in the ZOL group (
Denosumab
A case series by Uzquiano et al. describes the use of denosumab in heart transplant recipients. Nine heart transplant recipients received denosumab 60 mg every 6 months for three doses, starting 1 year post-transplant. These patients also received 1000 IU vitamin D and 600 mg of calcium daily. Baseline DXA showed osteoporosis in 7/9 patients post-transplant. Follow-up DXA was repeated on average 8 months after starting treatment. The patients in this series had on average 6% ± 5% BMD gain at LS (
Osteoanabolic agents
One case report of teriparatide use in a heart transplant recipient with adynamic bone disease and 10 prior vertebral fractures was reported. Iliac crest biopsy with tetracycline labeling after 24 months of teriparatide treatment showed 20-fold increased trabecular bone volume, 4-fold increased osteoid surface, and 9% increase in trabecular thickness. BMD change was reported as nonsignificant but specific values were not reported, and the patient experienced no new fractures following treatment. The patient was treated for 24 months, followed by transition to denosumab. 71
Limitations and future directions
There are limited data comparing the efficacy of oral and IV bisphosphonate therapy on BMD and fracture outcomes. The only RCT evaluating ZOL treatment in heart transplant recipients has 27 patients, and further research with larger cohorts of patients will help determine the efficacy of ZOL treatment.
Future directions should include RCTs evaluating early post-transplant treatment options with a longer follow-up period to evaluate if the BMD gains noted by Shane et al. with ZOL therapy are evident in longer term and if there is any fracture reduction. Additionally, further research to evaluate the role of excluding adynamic bone disease prior to choosing a treatment would be beneficial to guide antiresorptive therapy for those with concomitant cardiorenal syndrome in the peri-transplant period.
RCTs are needed to compare denosumab to bisphosphonates. In specific, determining the frequency of hypocalcemia in heart transplant recipients with CKD stage 3 will help guide future treatment and choice between denosumab and bisphosphonates. Further studies exploring BMD/fracture outcomes with shorter versus longer term denosumab treatment and the ideal sequence/duration of bisphosphonate use after denosumab should be explored.
Further studies will be necessary to determine long-term impact of osteoanabolic treatment and the ideal sequence of antiresorptive following osteoanabolic therapy in heart transplant recipients. In addition, further research should be undergone regarding romosozumab therapy in heart transplant recipients, as romosozumab currently has a black box warning regarding a higher rate of major adverse cardiac events compared to alendronate and is contraindicated within 1 year post myocardial infarction or stroke. 72
Treatment of osteoporosis in the lung transplant recipient
There are limited published data and no formal transplant society guidelines on osteoporosis therapies specific to lung transplant. A few recent publications will be highlighted below.
Bisphosphonates
A retrospective analysis by Ng et al. evaluated 60 lung transplant recipients, some of whom received early administration of ZOL around the time of transplant. Thirty-seven patients who received ZOL within 6 months before or after transplant were compared to 23 patients who did not. Patients were followed for a minimum of 3 years. The institutional protocol administered ZOL 4 or 5 mg within 6 months of transplant at 6-month intervals until 1 year post-transplant when repeat DXA was obtained. Subsequent doses of ZOL were continued past 1 year at 6-month intervals if BMD dropped ⩾4% at LS, ⩾5% at the femur, or if
Denosumab
No specific efficacy data have been published for the use of denosumab in lung transplant patients. However, Shrosbee et al. published a case series of 10 patients demonstrating the risk of hypocalcemia in lung transplant recipients treated with denosumab 60 mg every 6 months. Five patients developed hypocalcemia despite pretreatment 25OHD >60 ng/dL and supplementation with cholecalciferol 1000–2000 IU daily. These five patients had mean GFR 27 ml/min/1.73 m2 compared to 45 ml/min/1.73 m2 in the normocalcemic patients. Out of the five patients, three patients required hospitalization with serum calcium levels of 5.56, 6.24, and 6.76 mg/dL. Two out of the three hospitalized patients developed hypocalcemia within 2 weeks of denosumab, with the third patient developing hypocalcemia within 6 weeks after denosumab. Thus, close monitoring of calcium levels after denosumab is imperative, especially in those with GFR <30 ml/min/1.73 m2. 74
Osteoanabolic agents
One case series describes three patients with severe osteoporosis who were treated with teriparatide 20 mcg daily for 18 months post-lung transplant. All three previously received ZOL (last dose 12–24 months prior to teriparatide). Following completion of teriparatide, all patients received consolidation treatment with 4 mg ZOL. All patients experienced an increase in BMD at the LS (median +12%; range, 2%–14%) and total proximal femur (median +8%, range, 8%–10%). One patient developed a fracture after completion of teriparatide. No adverse effects were reported. 75
Limitations and future directions
RCTs evaluating the efficacy of ZOL in lung transplant candidates are necessary and have not been published to date. More up-to-date research on efficacy of oral bisphosphonates is also crucial.
In addition, in future directions, research could evaluate the role in trabecular bone score in SOT candidates to identify those at higher risk of BMD loss post-transplant who may benefit from early treatment with antiresorptives.
Bone health protocols such as the one discussed by Ng et al. have the potential to increase the rates of SOT recipients receiving timely antiresorptive therapy and should be strongly considered to preserve bone health in high-risk patients.
A retrospective review by Tungate et al. of 58 lung transplant recipients at a single institution suggested that the use of a bone health protocol could optimize outcomes in post-transplant osteoporosis. They described an institutional protocol outlining monitoring DXA within 6 months of transplant, ensuring adequate daily calcium 1200 mg and vitamin D 800–1000 IU repletion to goal 25OHD >30 ng/mL, and identifying patients who need antiresorptive therapy based on
At our institution, we are piloting a pre-transplant protocol using ZOL in lung transplant candidates to try and prevent the rapid bone loss after post-transplant in higher risk patients (Figure 1). The standardized protocol recommends all patients receive calcium 1200 mg daily and vitamin D supplementation to target 25OHD >30 ng/mL. Pre-transplant DXA with adjusted FRAX and LS X-ray (to screen for occult fractures) are obtained for all patients, regardless of risk category or age. In our protocol, all post-menopausal women and men over age 50 years with

Proposed algorithm for pre-lung transplant ZOL treatment for all postmenopausal female patients and men over the age of 50 years of age with
Conclusion
Across all SOT patients, there is rapid BMD loss and high fracture risk in the first 6–12 months post-transplant due to a myriad of risk factors including high doses of glucocorticoids, immobility, and nutritional deficiencies. All organ transplant recipients should receive comprehensive skeletal care and consideration for early osteoporosis pharmacologic therapy. DXA should be obtained prior to or immediately after transplant to risk stratify patients. All SOT recipients should receive counseling regarding adequate vitamin D and calcium intake and regular weight-bearing exercise. Although robust RCTs are lacking, early treatment with bisphosphonates (especially ZOL) preserves bone density and reduces fracture risk after SOT, and there are emerging data about the use of alternative therapies including denosumab and osteoanabolic therapies. There are additional organ-specific considerations, including in renal transplant patients who are at high risk for renal osteodystrophy, in whom determining bone turnover status prior to osteoporosis therapy may be beneficial. Protocolization may serve to improve metrics of calcium/vitamin D supplementation, rates of osteoporosis medication initiation, and consistency of DXA monitoring and provide support in treating bone loss in this high-risk population.
