Abstract
Cystic fibrosis-related bone disease (CFBD) is a common endocrinopathy in people living with cystic fibrosis (CF) that is complex and multifactorial in origin. People with CF experience high rates of progressive bone density loss and increased fracture risk. Focus on prevention and treatment of CFBD is of increasing importance in a now aging CF population. This review will discuss current practices in CFBD, gaps in knowledge, and potential future studies with the goal of advancing the clinical care of patients with CFBD.
Keywords
Introduction
Cystic fibrosis (CF) is an autosomal recessive disease affecting an estimated 160,000 people globally and over 30,000 people in the United States. CF is caused by pathogenic variants in CF transmembrane conductance regulator (CFTR), which encodes the CFTR protein, altering the chloride channel on epithelial surfaces.1,2 Cystic fibrosis-related bone disease (CFBD) is a common extrapulmonary manifestation of CF. Approximately one-quarter of people with cystic fibrosis (PwCF) will be diagnosed with CFBD during their lifetime, with some studies finding up to 78% of PwCF have early bone mineral density (BMD) loss.3–7 CFBD is characterized by lower rates of bone mass accrual in childhood and young adulthood as compared to healthy peers, early bone density loss, and increased risk for fractures.7–11 Bone disease in CF is a product of the direct effects of CFTR dysfunction and indirect factors such as undernutrition and glucocorticoid use.12–14 Osteoporosis and premature bone loss in CF are associated with impaired pulmonary function and increased mortality.15–17
There are still several unknown clinical questions regarding bone health in PwCF. 18 Areas that require further investigation include the true prevalence of fracture in children and adults with CF, efficacious therapies in fracture prevention in CF, and the impact of highly effective modulator therapy (HEMT) on bone density and fracture. While some interventions have been shown to increase BMD, evidence that these treatments reduce the risk of fracture is lacking. 19
The scope and purpose of this review is to survey present knowledge surrounding CFBD. We will identify clinical practice gaps and describe further investigations that are needed in the screening, diagnosis, and treatment of CFBD. Lastly, we will discuss the future of CFBD clinical care based on recent and ongoing studies.
Etiology
CFBD is multifactorial in etiology. 12 Underlying causes include dysfunctional CFTR, direct physiologic disruption of nutritional absorption, and external factors (Table 1). Investigation of underlying factors contributing to bone loss is fundamental in creating an effective treatment plan.
Summary of the etiological mechanisms of cystic fibrosis-related bone disease.

BMD, bone mineral density; CFTR, cystic fibrosis transmembrane conductance regulator; RANKL:OPG, receptor activator of nuclear factor kappa-B ligand to osteoprotegerin ratio.
Genetics and cellular mechanisms
Human osteoclasts, osteoblasts, and osteocytes express CFTR. 20 Dif et al. 21 found that both BMD and bone mineral content (BMC) were decreased in CFTR knockout mice as compared to CFTR (+/−) and CFTR (+/+) littermates. Furthermore, certain CFTR genotypes have shown a strong association with low BMD.22,23 King et al. 22 found that adults with F508del mutations, the most common CFTR mutation in CF, had significantly lower BMD Z-scores at the lumbar spine and femoral neck. Similar findings of low BMD were also reflected in mice with F508del mutations. 24 The findings of low BMD were independent of age, weight, pancreatic insufficiency (PI), and vitamin D status-matched controls, suggesting that the CFTR inherently influences bone homeostasis.21,22
Several studies have focused on examining the exact cellular mechanisms that instigate decreased BMD in CF. Findings include decreased osteoblast differentiation and maturation, increased osteoclast numbers activity, and increased bone turnover.23,25–27 Inflammation from bacterial infection and the pH change from chloride dysfunction are directly involved in the disruption of bone homeostasis, affecting the nuclear factor kappa-B (NF-kB) pathway and downregulating the Wnt pathway activity and vital osteogenic pathways.26–29 This results in a high receptor activator of NF-kB ligand to osteoprotegerin ratio (RANKL:OPG), and ultimately increased osteoclast activity, favoring bone resorption.25–29
Malnutrition
Undernutrition, a major sequela of CF, is a known risk factor for poor bone health. 12 Increased energy expenditure from inflammation and infection, decreased intake, PI, decreased bile acid secretion, and alterations in intestinal pH and gut motility lead to malabsorption of nutrients.12,30 Low body mass index (BMI), a surrogate for poor nutritional status, has long been recognized as a risk factor for low bone density in PwCF.22,31–35 Grey et al. examined BMD and body composition in adults with CF as compared to non-CF controls and found BMI correlated positively with total body BMD. Mean total body BMD was lower in the CF group versus healthy controls (−10.5%) as well as at the lumbar spine (−12.5%) and femoral neck (−11.1%). 32 Sheikh et al. 33 found a significant correlation between BMI and low weight with low BMD Z-scores in both adults and adolescents with CF. Frangolias et al. 35 also found similar findings with low BMI associated with decreased BMD in adults with CF. Overall, malnutrition has a strong association with low bone density.
Pancreatic insufficiency
Approximately 85% of PwCF are diagnosed with PI. 36 Both exocrine and endocrine PI can influence bone health.36,37 Exocrine PI causes malabsorption of fat-soluble vitamins A, D, E, and K. 36 Vitamin D plays an important role in calcium absorption and bone homeostasis. 38 Vitamin K is also needed at multiple points in the bone metabolic pathway, serving as a coenzyme for molecular pathways and regulating osteoblast and osteoclast formation, modulating both bone formation and bone resorption. 39
Vitamin D deficiency
Vitamin D deficiency is diagnosed in up to 90% of PwCF.40–44 Furthermore, PwCF experience high rates of both vitamin D deficiency (<50 nmol/L or <20 ng/mL) and insufficiency (<75 nmol/L or <30 ng/mL).40–44 In addition to malabsorption and exocrine PI, PwCF may have less sunlight exposure and lower fat mass, which also contribute to low levels of vitamin D, as sunlight is the major source of vitamin D and vitamin D is, in turn, stored in adipocytes.42,43 Decreased fat mass results in a lack of vitamin D storage, as opposed to the sequestration of vitamin D in excess adipose stores that is associated with obesity. Research has also shown that PwCF may have impaired absorption and metabolism of vitamin D.41,44–47 A study by Lark et al. 45 illustrated that PwCF absorbed significantly less vitamin D as compared to healthy controls. Similarly, Rovner et al. 41 found that 7% and 90% of participants with CF remained vitamin D deficient and insufficient, respectively, despite 800 IUs of vitamin D supplementation and concurrent pancreatic enzyme replacement. Boyle et al. 47 reported that despite two 8-week rounds of weekly vitamin D 50,000 IUs, only 5 out of 66 adults with CF reached vitamin D sufficient levels. Donovan et al. 34 found that BMD T-scores at the lumbar spine, radius, and total hip were significantly lower in participants with 25-hydroxyvitamin D (25 (OH)D) levels <10 ng/dL. Wolfenden et al. analyzed the prevalence of vitamin D deficiency and associated factors in a single CF center cohort: 27% of participants who had a lateral chest X-ray had evidence of vertebral fracture. Of patients with prevalent vertebral fractures, 80% were vitamin D insufficient, and 17% were vitamin D deficient. 43 The Cystic Fibrosis Foundation (CFF) guidelines recommend yearly screening of vitamin D levels in PwCF and supplementation with cholecalciferol to maintain vitamin D levels in sufficient range, 75 nmol/L (>30 ng/mL). 48 In summary, PwCF have high rates of vitamin D deficiency, which is associated with lower bone mass and a higher prevalence of fractures.
Cystic fibrosis-related diabetes
Up to 50% of PwCF are diagnosed with cystic fibrosis-related diabetes (CFRD), with the incidence increasing with age.49,50 Hyperglycemia is thought to inhibit osteoblast maturation and activity, increase factors involved in bone resorption, and decrease bone formation, in addition to altering the quality of bone architecture by oxidative stress and inflammation. 51 CFRD also correlates with poor nutritional status.49,50 Rana et al. 52 examined BMD in a single CF center cohort based on glycemic status and found that the cohort with CFRD or impaired glucose tolerance had significantly lower BMD and BMC than those with normal glucose tolerance. In addition, those with hyperglycemia tended to possess lower bone area for height and lower and BMC adjusted for bone area, representative of thin and narrow bone structure. 52 Lei et al. measured incretin hormones gastric inhibitory polypeptide (GIP), glucagon-like peptide-1 in parallel with markers of bone turnover c-terminal telopeptide (CTX), and procollagen type I N-terminal propeptide (P1NP) during an oral glucose tolerance test (OGTT) in a cohort of PwCF who were exocrine pancreatic insufficient. The study showed that participants who had a rise in GIP during the first 30 min and then a decrease over the remainder of the OGTT (appropriate incretin response to glucose) had a concurrent decrease in levels of CTX, indicative of attenuated bone resorption. 53 This is significant as incretin dysregulation is a plausible contributor to the pathogenesis of CFRD. 50 It is clear from studies conducted in people with and without CF that diabetes and worse glycemic control can impact bone health.
Exercise
Weight-bearing exercise is essential in the accrual and maintenance of bone density. Weight-bearing activates bone mechanoreceptors, which leads to a cascade of osteogenic reactions, including promotion of osteoblastic activity and stimulation of osteocytes to reinforce structural bone integrity. 54 The studies regarding the relationship between exercise and bone health in PwCF have been mixed.55–57 PwCF may engage in low rates of exercise due to exercise intolerance, which is thought to be multifactorial in origin brought on by cardiopulmonary limitations and metabolic and oxygen utilization impairment. 58 One study examined changes in BMD between an intervention cohort of PwCF that performed a prescribed exercise regimen three times a week and a cohort that performed their usual physical activity routine over the course of 1 year, finding no differences found in BMD between the exercise group and control group. 55 Alternatively, Garcia et al. 56 studied BMD based on physical activity duration and found that increased physical activity had a positive correlation with greater BMD. Frangolias et al. 35 examined VO2max as a physiologic marker for exercise capacity in a cohort of PwCF and found an association between decreased exercise capacity and decreased BMD. The differing results may be reflective of the heterogeneity in exercise interventions between studies, making it difficult to draw generalized conclusions surrounding the relationship between exercise and bone health in PwCF. At this time, it is uncertain what frequency, intensity, or type of exercise is optimal to promote bone accrual in children and young adults or maintenance of bone mass in adults with CF.
Exogenous glucocorticoids
Historically, up to 50% of PwCF were treated with exogenous glucocorticoids for their anti-inflammatory effects although this clinical practice may be changing.5,11,12,59,60 Supraphysiologic glucocorticoids are known for their toxic effects on bone which include increased osteoclastic activity, attenuation of osteoblasts, inducement of osteocyte apoptosis, and altering the RANKL:OPG ratio tilting the equilibrium of bone metabolism toward bone loss. 61 Glucocorticoids also exert indirect effects on bone by dampening the hypothalamic-pituitary-gonadal axis, decreasing IGF-1 action on bone, and disrupting calcium homeostasis through the impairment of intestinal absorption and renal reabsorption. 61 Studies demonstrated that glucocorticoid users with CF have lower BMD, lower BMD T-scores at the spine and femur, lower BMD Z-scores at the hip, and whole body and lower annual bone density accrual compared to matched controls who did not use glucocorticoids.11,62,63 Alternatively, Donovan et al. 34 did not find a difference in BMD in those treated with oral glucocorticoids (>7.5 mg oral prednisone for >3 months) with a mean treatment duration of 1.5 years. Glucocorticoids are known for their toxic influence on bone and most studies regarding steroids and bone in CF have illustrated this concept.
Delayed puberty and hypogonadism
Delayed puberty and hypogonadism contribute to decreased bone mass accrual and bone loss via deficiency of sex steroid influence.64,65 Estrogen is osteo-protective, involved in the regulation of osteoclasts and osteoblasts in bone metabolism, and overall exerts an anabolic, antiresorptive influence on bone. 64 Bianchi et al. 66 illustrated high rates of bone accrual during puberty in PwCF. 65 This suggests that those who experience delayed puberty may fail to achieve peak bone mass due to the loss of critical time necessary to potentiate bone gain. Rossini et al. 67 re-enforced this concept, finding that BMD correlated negatively with increased age at puberty.
Hypogonadism has been described in both men and women with CF.67–69 The true prevalence is unknown; however, a recent review of testosterone levels in men with CF found that roughly a quarter of men who had a testosterone level checked exhibited a level <300 ng/dL. 68 Another study found that 23% of females and 40% of men with CF exhibited estradiol levels below the reference range and that there was a significant correlation between estradiol levels and femoral BMD. 67 Additionally, low estradiol and testosterone levels were associated with an increased prevalence of vertebral fracture in men. 67
Currently, there are no large-scale studies examining the effects of hormone therapy (HT) on PwCF. Wu et al. conducted a small study examining BMD in premenopausal women taking oral estrogen supplementation and those who were not. The study showed that women on oral estrogen demonstrated both lower mean BMD and lower mean Z-scores at the lumbar spine as compared to those not taking estrogen. 70 This differs from findings in the Women’s Health Initiative, which illustrated increased spine and hip BMD in postmenopausal women on HT. 71 However, the doses of estrogen and population age between the two studies differed. This illustrates additional important questions for investigation: the optimal age to institute hormone therapy and effective dosage to preserve bone health in PwCF. Given PwCF are living longer, it would be expected that more females with CF will start to experience menopausal bone loss through a decline in estrogen and that more men will start to experience age-related hypogonadism, although this has not yet been studied extensively.
Organ transplant
Transplant patients and those on immunosuppressant therapy are at risk for accelerated bone loss from the sequelae of advanced illness severity and the use of certain immunosuppressant drugs.72–74 Transplant patients may warrant special considerations for treatment of osteoporosis to mitigate perioperative bone loss and optimize bone health prior to transplant. 72
Screening
The CFF and other professional societies provide screening guidelines for CFBD (Figure 1). Currently, the CFF recommends screening all adults starting by age 18 for CFBD via dual-energy X-ray bone absorptiometry (DXA). The interval of subsequent DXA studies is determined by the Z/T-scores (Figure 2). Screening should start earlier at age 8 those with increased risk factors for CFBD including positive clinical fracture history, delayed puberty, undernutrition, and poor lung function. 18 Alternatively, the European Cystic Fibrosis Society (ECFS) recommends the commencement of routine DXA screening in childhood at the age of 8–10, and similarly, the French Cystic Fibrosis Society guidelines recommend DXA starting at age 8.75,76
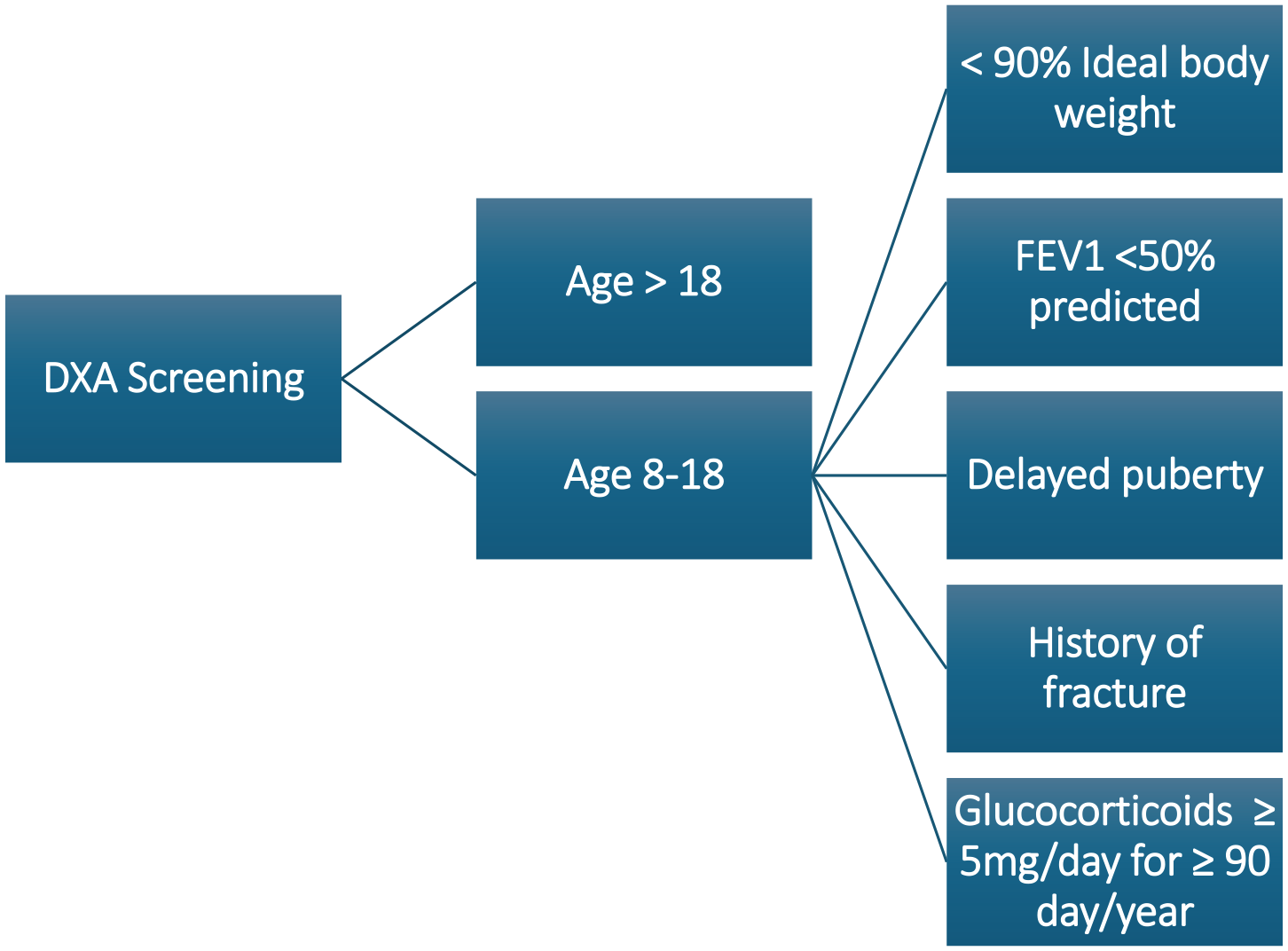
Criteria to institute DXA screening in PwCF.
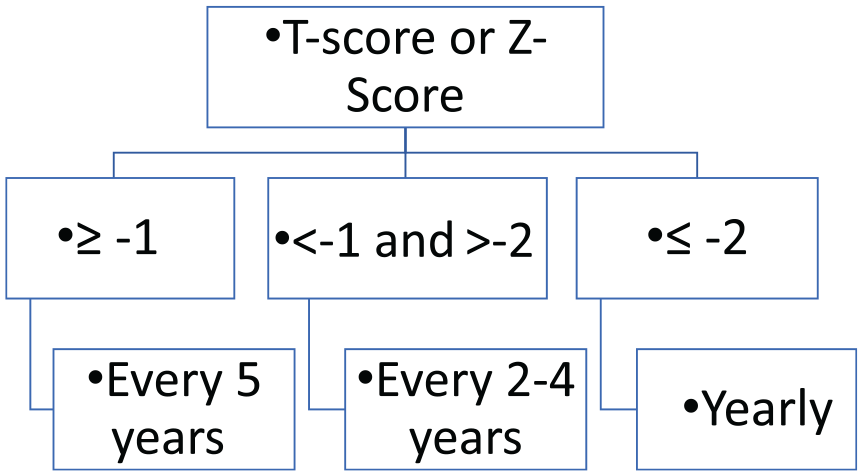
Interval DXA screening based on Z-score (for premenopausal women and men <50) or T-score (for postmenopausal women and men >50). 18
Diagnosis
Osteoporosis is a clinical diagnosis in children and young adults, coupling clinically significant fracture history and low bone density for age. In postmenopausal women and men >50 years of age, the diagnosis of osteoporosis can be made by dual-energy X-ray absorptiometry (DXA) BMD alone.
Fracture is a critical part of the diagnostic criteria for osteoporosis in CF (Figure 3).18,75 Significant osteoporotic fractures in CF include vertebral compression fracture, low trauma fracture of long bones of the lower limb, or ⩾2 upper long bone limb fractures.16,75

Diagnostic algorithm for the diagnosis of osteoporosis in cystic fibrosis.
BMD by DXA is also used. BMD is reported in terms of Z-scores and T-scores. T-scores are a standard deviation calculation that compares the BMD of an individual against the BMD of gender-matched controls aged 25–29 (peak bone mass) and is generally used for postmenopausal women and men over 50. Z-scores are used for premenopausal women and men under 50 and calculate the standard deviation of an individual’s BMD against gender and age-matched controls. In children, Z-scores are adjusted for height to account for growth during the analysis of bone size.77,78
Biochemical evaluation
Multiple sources describe biochemical screening for secondary causes of osteoporosis.75,77,78 There are no unified recommendations for when to screen for secondary etiologies of osteoporosis in CF per se. Current suggestions include a general screening workup for all patients diagnosed with osteoporosis, with extended investigations in premenopausal women and men under 50 and patients with Z-scores ⩽ −2 (Table 2).77,79 Comorbid disease states in PwCF have been described as contributors to bone loss. Putman et al. 80 described a case series in which two patients were diagnosed with celiac disease during evaluation for low bone density. A recent case study described a man with CF who was ultimately diagnosed with prolactinoma. The patient exhibited increased BMD upward of 10.5% after 2 years of treatment with cabergoline. 81 These cases illustrate the importance of identifying secondary contributors when assessing bone disease in PwCF to ensure appropriate treatment.
Biochemical evaluation for secondary causes of bone disease (not an exhaustive list).

FSH, follicular stimulating hormone; LH, luteinizing hormone; P1NP, procollagen type I N-terminal propeptide; SPEP, serum protein electrophoresis; TSH, thyroid-stimulating hormone; UPEP, urine protein electrophoresis.
Dual-energy X-ray absorptiometry
DXA is the gold standard for BMD evaluation. DXA produces two-dimensional images, measuring BMC over a designated area (g/cm2).16,78 DXA is also used for trabecular bone score (TBS) and vertebral fracture analysis (VFA). TBS estimates the microarchitecture of bone by analyzing the pixel content of DXA images and is an independent risk factor for fragility fracture in postmenopausal osteoporosis.82,83 One study examining TBS in PwCF did not demonstrate an independent association with fracture as compared to femoral neck BMD; however, it was more useful when used in conjunction with other risk factors. 82 The sensitivity of VFA examination for the identification of vertebral fractures has not been studied extensively in CF.
Other imaging modalities
Although DXA remains the gold standard for screening and the diagnosis of low BMD and CFBD, other imaging modalities can be utilized depending on the clinical context, including plain-film X-rays, quantitative CT, MRI, and quantitative ultrasound. Other than the utilization of X-rays in the diagnosis of vertebral fracture, none of these modalities have been studied extensively in the evaluation of CF-related bone loss. Lateral chest X-rays have been employed in multiple studies to aid in the identification of vertebral fractures in PwCF.11,43,63
Technological advances
Future technologies for osteoporosis screening, risk determination, and predictive response to therapy are imminent. A recent study looked at 10,000 patients, predicting the risk of osteoporosis based on a set of chronic disease data, demographics, and risk factors using a variety of machine learning algorithms. Age, gender, lipid metabolism disorders, cancer, and COPD status were associated with an increasing prediction risk for osteoporosis. 84 Multiple studies are emerging examining the utility of artificial intelligence (AI) to augment CT scans and plain-film X-rays in osteoporosis screening.85,86 Machine learning and AI have not been utilized in CF bone studies specifically. However, research is underway regarding the use of machine learning in other CF arenas and has been posited as a useful tool in the care of PwCF.87–89
Treatment
Nutrition
BMD has a positive association with optimized nutrition status.12,75 Current recommendations emphasize maintaining a BMI ⩾50th percentile in children aged 2–20 and BMI ⩾22 in adult women and ⩾ 23 in adult men in addition to monitoring growth parameters. 90 The CFF recommends maintaining a 25 (OH)D level of >30 ng/mL (75 nmol/L) and the ECFS guidelines recommend a level of at least 20 ng/mL (50 nmol/L).48,75 Both organizations advocate for supplementation with cholecalciferol (D3). The CFF recommends an annual screening of 25 (OH)D levels. Recommended calcium intake is graded by age, with 1000–1200 and 1300–1500 mg of dietary calcium referenced in the ECFS and CFF guidelines, respectively, for adults.18,75 Both organizations also endorse monitoring and supplementation with vitamin K if needed, particularly in exocrine pancreatic insufficiency patients.
Exercise
Studies have found a positive association between exercise capacity and BMD. Both the European and CFF guidelines recommend exercise as part of the treatment of CF bone disease.18,75 The ECFS recommends that children with CF engage in 20–30 min of activity 3 times/week, which should include weight-bearing exercise, and that adults should also engage in weight-bearing activity and resistance training. 75
Pharmacologic anti-osteoporosis treatment
Bisphosphonates
Antiresorptive medication can be employed in the treatment of CF bone disease (Table 3). To date, bisphosphonates remain the most studied therapy in PwCF and are the only recommended anti-osteoporosis medication in the treatment of CFBD when T/Z-scores fall ⩽−2.18,75 Several studies have demonstrated efficacy in BMD gains with bisphosphonate treatment in PwCF.19,91–99 Early studies focused on pamidronate infusions. Aris et al. 91 examined the use of pamidronate in PwCF post-lung transplant and found that patients treated with pamidronate every 3 months exhibited higher gains in BMD at both the spine and femur than the control group treated with oral calcium and vitamin D supplementation over a 2 year period. Haworth et al. 92 found similar results with increased BMD in a cohort of CF participants treated with pamidronate over 6 months at the spine and hip. Zoledronic acid has also been studied. Chapman et al. 99 demonstrated significant increases in lumbar and femoral neck BMD in a treatment group with zoledronic acid versus normal saline placebo over a 2-year time period.
FDA-approved anti-osteoporosis drugs.

RANKL, receptor activator of nuclear factor kappa-B ligand.
Other studies analyzed the effect of oral bisphosphonates on bone in PwCF. A placebo-controlled randomized double-blind trial (RCT) comparing daily alendronate to placebo over a 1-year period revealed significant gains in BMD at the spine and femur in the treatment group while the placebo arm cohort lost BMD. 93 Another RCT examined the effects of weekly alendronate compared to placebo and again found significantly higher bone accrual at the lumbar spine and total hip BMD in the alendronate treatment group. 94 Bianchi et al. conducted a multi-center two-phase trial. Over the first phase of 12 months, all patients were treated with calcium and calcifediol, with approximately 25% of participants achieving ⩾5% gains in bone mineral apparent density. The second phase was a double-blind, randomized, placebo-controlled, parallel-group study with weekly alendronate treatment in the participants that had not achieved ⩾5% in bone density. BMD increased by 16.3% on average in the treatment group versus 3.1% in the placebo group. 98
Although many studies have demonstrated that bisphosphonates can effectively increase BMD in the treatment of CFBD, the data regarding fracture reduction is not yet clear. A 2023 Cochrane Review, Bisphosphonates for osteoporosis in people with CF, found no significant difference between fractures in the treatment groups compared to the control groups. 19 It was noted that limitations on study size and duration may limit findings regarding fracture reduction.
Other anti-osteoporosis pharmacologic therapies
Recently, there has been increased interest in the use of additional postmenopausal anti-osteoporosis treatments in PwCF (Table 3). Some of these drugs are being used in practice with significant improvements in bone density; however, the data is limited to case series and reports surrounding the use outside of age-related osteoporosis.
Anabolic therapy
Anabolic anti-osteoporosis medications stimulate bone building and formation via activation of osteoblasts. 100 Siwamogsatham et al. discussed their experience treating CFBD with anabolic therapy, teriparatide, through a case series. Three out of four patients successfully completed a treatment 2-year course of teriparatide and exhibited BMD gains up to 11% at the lumbar spine and 10.2% at the total hip. 101 Crow et al. 102 shared a case in which a 46-year-old premenopausal female undertook 12 months of treatment with romosozumab, a dual anabolic, antiresorptive medication, with a 12.8% increase in BMD from the start to the end of therapy. 103 Z-scores also increased from −2.5 to −1.1 at the lumbar spine and from −1.7 to −0.5 at the total hip. 102 To date, anabolic therapy has not been well studied in PwCF.
Antiresorptive monoclonal antibody
Denosumab is a monoclonal antibody that blocks RANKL from binding to RANK, decreasing the maturation and activation of osteoclasts and ultimately inhibiting bone resorption. 104 This medication may be particularly useful given the known alteration in RANKL:OPG in PwCF. However, there are no published studies regarding the use of denosumab for the treatment of CFBD yet. A clinical trial is underway (NCT03921060).
Further studies are needed regarding fracture reduction rates in PwCF, rather than an increase in BMD alone. This should include an examination of the optimal timing for the initiation of anti-osteoporosis medication in CFBD in terms of benefits in fracture versus risks of prolonged medication exposure, side effects, and additional burden/medical complexity on already burdened populations. Given increasing age, PwCF that experience bone loss may benefit from sequential anti-osteoporosis therapy or periodic iterations of anti-osteoporosis treatment to attenuate early bone loss throughout the lifespan. Rigorous studies are needed on this topic.
Future directions
Highly effective modulator therapy
The future trajectory of CFBD remains ambiguous with both the integration of CFTR HEMT and an aging CF population. HEMTs are medications that restore the function of the CFTR protein in certain PwCF. 105 Possible outcomes of HEMT on bone disease include: direct influence on bone architecture, indirect influence through mediation of CFBD contributors, and increased life span. Studies have shown that HEMT is associated with improved lung function along with less pulmonary exacerbations and decreased airway inflammation.105–108 In terms of bone health, hypotheses include if HEMT attenuates PI which could in turn lead to better nutrient absorption, specifically fat-soluble vitamins including vitamin D.109,110 So far, the results from studies examining vitamin D status and HEMT use have differed and there have not been any long-term studies analyzing this relationship.110–113
HEMT may also exert direct effects on bone through the normalization of CFTR-mediated chloride transport. Le Henaff et al. 23 found that mice treated with medication to promote the normal function of the CFTR chloride channel demonstrated increased BMD through re-calibration of bone formation and resorption as well as improved bone microarchitecture. Putman et al. 114 studied this outcome further by analyzing bone microarchitecture and BMD in 26 participants with CF treated with ivacaftor, a HEMT medication, and found increased cortical bone microarchitecture over 2 years. However, no significant changes were noted in the areal BMD or trabecular makeup. This constellation of potential outcomes could herald preservation of bone mass and decrease early bone loss in those who are eligible for HEMT. The preliminary effects of HEMT on bone health are currently being investigated (NCT04038047). 115
Aging in CF
Aging in CF is a new frontier in clinical practice. Therefore, not many studies exist regarding the relationship between CF bone disease and aging. The median predicted survival of a person born with CF in the United States in 2023 is now 68 years, largely due to HEMT. 116 In the general population, bone density remains relatively stable between the achievement of peak bone mass in emerging adulthood until the fifth decade, when estrogen levels start to fall. However, longitudinal studies have shown that PwCF exhibit bone loss during this time period and that PwCF exhibit vertebral fractures at the rate of or superseding that of postmenopausal women.4,9,11,43,117
Many questions remain unanswered in this realm, such as the prevalence of glucocorticoid use in people who receive HEMT, changes in nutritional status and absorption throughout the lifespan, alterations in exercise capacity, and how this might influence bone health in an aging population. People who receive HEMT have been shown to exhibit improved pulmonary function and, therefore, may have increased exercise capacity and decreased glucocorticoid use, which theoretically could result in preserved bone density throughout the lifespan. Improved nutritional status, especially starting at a young age, could contribute to achieving a higher peak bone mass and increased bone preservation. All of these questions require further long-term investigation, some of which are undergoing evaluation through the PROMISE study. 115
Another area that merits further investigation is the study of HT in PwCF, including the effects on bone health. Women with CF may go through menopause at an earlier age than the general population (48.5 vs 52.5). 118 Currently, HT is approved for the treatment of menopausal genitourinary and vasomotor symptoms. 119 There are no large studies examining the use of HT in peri/postmenopausal women with CF, including estrogen, progesterone, or testosterone. While HT is not recommended as a first-line treatment for postmenopausal osteoporosis, multiple studies have demonstrated that estrogen-containing HT is beneficial for bone health.120–122 This includes both the prevention of bone loss and fracture reduction with one meta-analysis finding that HT reduced fractures by up to 35%, 28%, and 22% in the spine, hip, and non-vertebral skeleton, respectively.120,122 Additional research is needed to discern if the same benefits are demonstrated in PwCF treated with HT. There has been some concern that estrogen may alter pulmonary function and worsen Pseudomonas aeruginosa infections in women with CF.123,124 However, the use of exogenous estrogen in the form of oral contraceptives did not correlate with increased pulmonary exacerbations.124,125 Future studies should examine the effects of HT on all organ systems, including pulmonary function, to evaluate the risks and benefits of HT in PwCF specifically. Other aspects that could be examined include the ideal dose, time, and indication to begin HT in women with CF to achieve maximal benefit and mitigation of the risks of HT.
Alternatively, men with CF may start to experience higher rates of age-related hypogonadism. The impact of testosterone therapy on bone health in the general male population is currently a question of interest. In terms of the skeletal system, testosterone-replacement has been demonstrated to increase bone density; however, it remains questionable if this also translates to fracture reduction. 126 In a recent randomized clinical control trial, testosterone-replacement did not correlate with fracture reduction, with 3.50% of participants on testosterone-replacement diagnosed with a clinical fracture versus 2.46% in the placebo cohort. 127 Furthermore, the general effects of testosterone-replacement, including bone health, in men with CF have not been well studied. Additional investigation regarding the risks and benefits of testosterone-replacement in men with CF is needed both in general and also for age-related hypogonadism. Special attention is needed as testosterone therapy can result in infertility superimposed with congenital absence of the vas deferens and can also create dependency since men with CF may start hormonal therapy at younger ages than the general population.
Ultimately, two possible scenarios with regard to the aging CF population are: HEMT will ameliorate or eliminate early, progressive bone loss in PwCF that are eligible for HEMT, or PwCF will continue to experience longitudinal bone loss and this will be superimposed on age-related bone loss from usual aging processes through the lifespan. Currently, there is currently no robust longitudinal data to support either situation. It should also be noted that a large percentage of the worldwide population remains ineligible or unable to obtain HEMT. 1
Conclusion
In conclusion, with the increased longevity of PwCF, further emphasis must be placed on optimizing bone health over the lifespan. Technological advances have the potential to make osteoporosis screening more accessible for patients, allowing for earlier diagnosis and intervention to prevent bone loss. Other anti-osteoporosis treatments in addition to bisphosphonates may be beneficial for fracture reduction; however, more research is needed in this realm. It is unknown if early implementation of HEMT or prolonged use of HEMT will decrease the incidence or severity of CFBD in those eligible for modulator treatment. Further attention is needed on bone health in this new era of CF care.
