Abstract
Background:
Charcot foot is a severe complication of diabetes mellitus. Amputation is associated with 5-year mortality rates as high as 70%, and the overall treatment cost for diabetic foot surpasses that of conditions such as cancer or depression.
Objectives:
To compare clinical, quality-of-life, and cost outcomes related to Charcot foot management through two distinct treatments: amputation and resection with stabilization using circular external fixation (CEF).
Methods:
This retrospective study included all adult patients treated at our unit between 2008 and 2022 for acute diabetic foot with infected ulcers. The allocation to treatment groups was based on the timing of patient enrollment. We gathered anthropometric, diagnostic, and surgical data, documenting individualized costs for preoperative, postoperative, and rehabilitation phases. Health status was assessed using the EQ-5D-3L questionnaire, and recorded data included mortality.
Results:
A total of 31 patients (18 amputations; 13 CEF) were included. Amputees exhibited significantly higher mortality compared to those with a CEF (44.8% vs 7.7%, p = 0.045). The estimated 3-year survival was 60.8% for amputees and 90% for the CEF group (log-rank test, p = 0.096). In terms of quality of life (EQ-5D-3L), amputees reported a reduction of 14.67 points while CEF patients reported an increase of 40.39 points (p < 0.001). The EQ-5D-3L index improved by 1.8 points for amputees, as compared with 62.3 points in the CEF group (p < 0.001). The total mean cost of managing an amputated patient was €222,864, practically identical to the €224,438 incurred in the CEF group (p = 0.767). No statistically significant differences were found in the time distribution of costs. However, some specific expense items demonstrated statistical significance.
Conclusion:
In treating infected diabetic foot ulcers, external fixation leads to a better quality of life compared to amputation. There’s also a trend suggesting higher survival rates with external fixation, and both approaches have similar costs.
Plain language summary
Background
- One of the most severe complications of diabetes is the occurrence of infected ulcers on the foot.
- The reduced sensitivity of diabetic patients, coupled with their low defense capacity against infections, makes amputation of the foot one of the few viable treatments.
- However, amputation in these patients is associated with higher mortality rates than many cancers. And surviving patients experience greatly reduced quality of life.
- The economic costs of their treatment are very high.
- In recent years, a new treatment option has become popular, consisting of removing all dead or infected tissue and stabilizing the foot with a device known as a circular external fixator.
Objectives
- To compare the clinical outcomes, mortality, and quality of life of the two treatment methods: amputation and circular external fixation.
- To compare the economic costs associated with each treatment.
Methods
- We divided patients with acute diabetic foot with infected ulcers into two groups:
○ Amputation.
○ Circular external fixation.
- We compared the mortality, clinical results, quality of life, and economic costs.
Results
- 31 patients were included in the study. Of these, 18 were treated by amputation and 13 by circular external fixation.
- The mortality rate in the amputee group (8 out of 18) was higher than in those treated by circular external fixation (1 out of 13).
- The quality of life of amputated patients decreased, while that of patients treated by circular external fixation increased.
- The average total cost of treatment for an amputated patient (€222,864) and for a patient with circular external fixation (€224,438) was very similar.
Conclusion
- Patients with infected diabetic foot ulcers treated by circular external fixation have lower mortality rates and better quality of life than those treated by amputation.
- The costs of treatment by amputation and by circular external fixation are practically identical.
Introduction
Current increases in life expectancy have resulted in a higher prevalence of certain chronic conditions such as diabetes mellitus (DM). The overall prevalence of DM, which in 2019 stood at 9.3% of the world population, is expected to rise to 10.9% in 2030, with the initial figure soaring by up to 51% by 2045. 1 This surge in the incidence of DM is bound to be accompanied by an increase in the number of associated complications, diabetic foot being among those with a more profound effect on patient quality of life. 2 Charcot foot, which consists of a process of abnormal, rapidly developing bone resorption occasionally causing spontaneous fractures, deformities, and infections, is the most severe manifestation of this complication. All of this typically leads to a significant health problem, which not only impairs patient quality of life but also results in exorbitant costs for health systems.2–10
Initial treatment of patients with infected ulcers is often conservative and combines long-term use of antibiotics with boot casts or offloading footwear designed to prevent the formation of pressure ulcers that may contribute to the aggravation of existing deformities or the appearance of new ones.11,12 Despite these preventive measures, many cases evolve toward severe osteomyelitis 6 which, until recently, could only be successfully treated by amputation of the limb.10,13,14 However, rehabilitation of amputated patients is an unpredictable process, with high morbidity and mortality rates, similar to those associated with the most aggressive types of cancer.7,10,12,15
In the last few years, new surgical alternatives have been explored that are based on the resection of the infected bone and the reconstruction of the limb by means of internal fixation or circular external fixation (CEF).7,16–21 External fixation has the advantage that it allows the reconstruction to be conducted in one single surgical stage and permits correction of complex multiplanar deformities.22,23 Although these procedures do not eliminate the need for amputation, they may reduce its incidence and improve the morbidity and mortality resulting from the operation.17,20
Apart from the strong functional and economic impact it has on patients’ lives, diabetic foot is also associated with a major socioeconomic impact. Between 12% and 15% of annual healthcare spending in developed countries goes to the management of the complications resulting from diabetes.1,4,24 Considering that between 15% and 34% of diabetic patients present with an ulcer in the foot at some point in their lives, and that approximately 20% of patients end up undergoing an amputation, the cost of treating diabetic foot works out higher than that associated with other severe conditions such as cancer or depression.24–27
The complexity of diabetic foot is such that its management requires a multidisciplinary approach.7,26,28 It is for this reason that our hospital boasts a dedicated diabetic foot unit, which is responsible for defining strategies aimed at optimizing health outcomes and minimizing the economic cost associated with treating the disease. The aim of the present study is to compare the cost of reconstructive treatment by means of CEF with that of amputation, as well as to evaluate the quality of life in both patient groups.
Materials and methods
This is a retrospective study including a consecutive series of patients treated at the Diabetic Foot Unit of the Consorci Sanitari Integral between 1 January 2008 and 31 December 2022. The study was approved by the hospital’s Ethics Committee (code CEIC HUB PR367/21) and all patients gave their informed consent before they were included.
All patients with DM who were over 18 years of age, presented with chronic diabetic foot (Brodsky type 1/Eichenholtz stage III midfoot Charcot neuroarthropathy) 29 with MRI-diagnosed infected deep midfoot ulcers; had been treated means of CEF or amputation surgery; and had been followed up for at least 1 year; were included in the study. Patients who received conservative treatment and who progressed favorably without requiring surgery were excluded (Figure 1).

Patient management and inclusion algorithm in the study.
Diagnosis of the condition was made by means of imaging techniques, the main goals being identification of the affected areas, assessment of bone quality, and determination of the presence of infection. Findings were classified using the Wagner–Meggitt scale, 30 the SINBAD classification, 31 and the PEDIS score. 32 Samples were also taken for pathological and microbiological analysis.
Given that the goal of our analysis was to evaluate a new treatment strategy, it was decided to use a single-arm before-and-after design. 33 Before 2012, all cases of bone infection used to be treated by means of amputation; reconstructive management started being applied from that year onward. Patients were divided into two groups:
- An amputation group: infracondylar amputation, use of an external prosthesis, and gait retraining.
- A CEF group: resection of necrotic and infected tissue, accompanied by stabilization of the foot using a circular external fixator (TrueLok, Orthofix Srl, Italy). Patients were kept from weight-bearing for 6 weeks and were then allowed to progress to normal gait as tolerated.
In addition to anthropometric and diagnostic data, all available data on the costs associated with treatment were collected, grouped into the categories described in Table 1, and divided into a pretreatment and a posttreatment phase. The costs associated with each category correspond to the year 2022 and were obtained from the hospital’s management.
Costs included in the analysis.

To determine the effectiveness of each treatment and the time to resolution of the patient’s symptoms, ulcers were considered healed when they showed complete epithelialization with no drainage and presented with no clinical or radiologic signs of bone infection. Patients were followed up according to the hospital protocol, and asked about their health status in person at their first visit to the hospital and at their last follow-up appointment, using the EQ-5D-3L health questionnaire. 34 The death rate of each group was also recorded.
A descriptive analysis was carried out of the data, calculating measures of central tendency and dispersion. The groups were compared using either parametric or nonparametric means difference tests, depending on whether the normality of the samples could be assumed. Qualitative variables were analyzed using Pearson’s Chi-squared test or the Fisher exact test, depending on the magnitude of the expected values. Paired comparisons were used when repeated (preoperative and follow-up) measurements were obtained for each patient. Survival was analyzed by means of the Kaplan–Meier methodology, using death as an endpoint. Differences in survival were evaluated with the log-rank test. In all cases, statistical significance was set at a p-value equal to 0.05. The data was analyzed using R software, version 4.1.3. (R Development Core Team). 35
Results
Overall results
The study included a total of 31 patients (24 men and 7 women), whose demographic data is summarized in Table 2. Eighteen patients (58.1%) were treated with major (infracondylar) amputation and 13 (41.9%) with CEF. It should be noted that six of the latter had already undergone minor (digital, transmetatarsal) amputations before the application of the external fixator. All ulcers were classified as Wagner–Meggitt grade III lesions 30 and they all obtained a score of 5 points on the SINBAD, 31 suggesting a high risk of amputation. 36 As far as the PEDIS score is concerned, 32 all ulcers were classified according to perfusion as P1 (no signs of peripheral arterial disease); to size as being larger than 1 cm2; to depth as D3 (deep ulcers affecting the bone); to the presence of infection as I3 (osteomyelitis); and to sensation as S2 (loss of protective sensation). The mean length of follow-up from diagnosis was 63.2 ± 35.2 months, mean patient age at the time of treatment was 64.1 ± 13.2 years, and mean length of follow-up after treatment was 37.2 ± 31.8 months.
Patient demographics.

Statistically significant differences between the groups.
Given that, in the case of amputation, ulcers were considered healed at the time of the procedure, mean time to ulcer healing was 0 months in the amputation group and 6.5 ± 5 months in the CEF group.
Quality-of-life outcomes
The overall quality-of-life outcomes and their comparative evolution (paired analysis) are shown in Table 3 and in Figures 2 and 3.
EQ-5D-3L questionnaire.
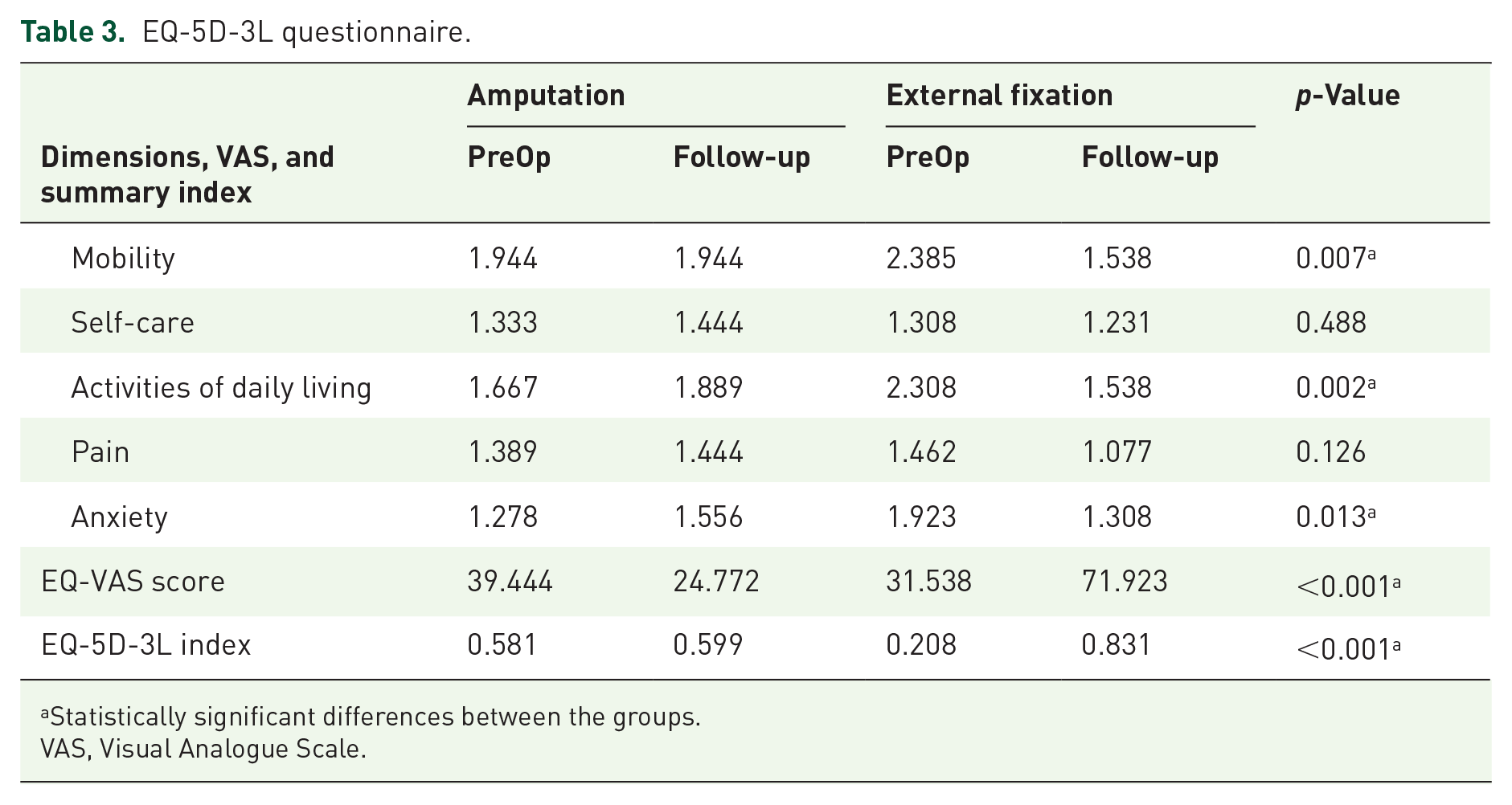
Statistically significant differences between the groups.
VAS, Visual Analogue Scale.

Variation between preoperative and follow-up scores on the EQ-5D-3L subscales.

Variation between preoperative and follow-up scores on the EQ-5D-3L subscales, VAS score, and EQ-5D-3L index.
The postoperative quality of life of amputees deteriorated on all the different criteria evaluated in the EQ-5D-3L questionnaire except for mobility, where no changes were observed although all patients wore a prosthesis. Conversely, patients treated with CEF reported an improvement in their quality of life across all the questionnaire’s subscales. Moreover, statistically significant differences in favor of CEF were observed in the degree of improvement reported in terms of mobility (p = 0.007), activities of daily living (p = 0.002), anxiety (p = 0.013), overall health (p < 0.001), and the EQ-5D-3L index (p < 0.001).
Some of these differences are of great clinical significance, such as the improvement in quality of life reported by patients treated with CEF (40.39 points higher than baseline) as compared with a decrease by 14.67 points for amputees; or an improvement of 62.3 points on the EQ-5D-3L index among CEF patients as compared with an amelioration of only 1.8 points for amputees.
As regards mortality, our study found that eight (44.4%) patients in the amputation group died as compared with one (7.7%) in the CEF group (Fisher exact test, p = 0.045). Survival estimations, performed using Kaplan–Meier plots (Figure 4), yielded a 3-year survival rate of 60.8% in the amputation group and 90% in the CEF group. This difference did not reach statistical significance on the log-rank test (p = 0.096).

Survival curves of patients treated with amputation and patients treated with external fixation estimated using the Kaplan–Meier method.
Cost-related outcomes
The costs associated with each treatment are summarized in Table 4 and Figures 5 and 6.
Costs.

Statistically significant differences between the groups.

Box plots comparing cost between groups. (a) Preoperative. (b) Postoperative. (c) Total.

Comparison of all cost components included in the study.
The mean total cost of managing an amputated patient came to €222,864, while the mean cost of managing a patient treated with external fixation was €224,438, with no statistically significant differences between both groups (p = 0.767). Nor were there any statistically significant differences between preoperative and postoperative costs, although the former was somewhat higher among amputees (€161,459 vs €152,510; p = 0.767) and the latter was higher in the CEF group (€61,406 vs €71,928; p = 0.708).
Statistically significant differences were found, however, with respect to certain specific expense items such as preoperative diagnostic tests (p = 0.022), preoperative walking aids (p < 0.001), preoperative medical consultations (p = 0.008), realignment surgeries performed prior to treatment (p < 0.001), amputations (p < 0.001), surgical equipment (p < 0.001), preoperative rehabilitation (p < 0.001), posttreatment diagnostic tests (p = 0.033), postoperative antibiotics (p = 0.006), postoperative medical consultations (p = 0.025), other posttreatment surgeries (p < 0.001), and postoperative rehabilitation (p = 0.002).
Discussion
The most significant finding of the present study was the fact that the clinical results of treatment with a circular external fixator are clearly superior to those of amputation, the cost of both options being practically identical.
The management of infected foot ulcerations in the context of DM remains a significant challenge for both patients and the community at large. Patients have to bear the burden of pain, infection, lengthy hospital stays, amputations, and, in some cases, death. They inevitably experience a deterioration of their quality of life, which also takes a toll on their families. From a community perspective, management of this condition is associated with very high costs, which could probably be brought down if evidence-based guidelines for the treatment of these chronic ulcers were available.37,38
It has been reported that 25% of patients with DM develop a foot ulcer at some point in their lives, 39 and that 85% of amputations in diabetics are preceded by an ulcer. 40 Amputation was for many years regarded as the treatment of choice for patients with severely infected ulcerations, 41 up to 45%–70% of lower limb amputations being performed in diabetic patients.42–44 Nonetheless, mortality among diabetic amputees is extremely high. Between 41% and 70% of diabetic patients undergoing a major amputation do not survive past 5 years from surgery,44–47 which entails a mortality rate comparable to, or even higher than, that of some types of cancer. 27 Even patients who do not end up undergoing an amputation have been found to face a 3-year mortality rate of 28%. 48 This data is in line with the results of our series, where overall mortality was 29%, and the mortality of amputated patients was 44.4%.
It is therefore not surprising that considerable efforts have gone into exploring new treatments that might prevent the onset of the disease or, at the very least, reduce the amputation rate.39,49–53 Prevention, evaluation, and treatment of diabetic foot ulcers require a multidisciplinary team,20,40,54–56 where the role of the orthopedic surgeon has become increasingly important given their ability to provide a biomechanical perspective, which is essential to reduce the risk of complications. 54 There is a general consensus that prevention is the most effective strategy to fight the disease and reduce the expenses associated with its management. Implementation of several glycemic monitoring and ulcer severity control strategies has resulted in a reduction in the number of complications associated with diabetes.56,57 In addition, realignment surgeries, arthroplasties, and offloading shoes have been shown to reduce the percentage of patients who end up undergoing an amputation.40,56 In this regard, Van Acker et al. 58 found that preventive treatment of patients costs 5.9 times less than the healing of mild cases and up to 36 times less than amputation surgery.
Once the infection has set in, the medical team must pursue a series of goals, including eradicating the infection through debridement of the wound and administering antibiotics, restoring blood flow to the affected area, correcting any existing deformities, and preventing the development of new ones.40,56 In these cases, it is mandatory to extend the tarsometatarsal fusion beyond the ulcer site, resect enough bone to allow a proper reduction without tightening the soft tissue envelope and use implants capable of maximizing the mechanical stability of the assembly, keeping the foot in plantigrade position.40,54 Although this can be achieved by means of either internal or external fixation, even the advocates of internal fixation agree that osteosynthesis should be avoided until the bone infection is resolved and the wounds are healed. CEF, for its part, makes it possible to correct the deformity at the same time as the infection is treated, 20 reduces the risk of iatrogenic damage to the tissue envelope, provides adequate stability, allows performance of secondary corrections, and facilitates dressing of the lesions. It must be said, however, that only patients who are able to follow the prescribed postoperative regimen may be considered for fusion or correction of the deformity. 54
Given that the traditional management of diabetic foot ulcers only contemplated surgery in the event of persistent bone infection or when the deformity could not be accommodated with an orthopedic device, treatment was considered successful if the infectious process could be resolved and the limb could be salvaged. Only one study reports functional or quality-of-life outcomes to describe the results of treatment, 20 making it difficult to compare the findings of our series with those of the existing literature.
Although not statistically significant (p = 0.096), the difference we found between the survival curves of both groups was extremely significant from a clinical perspective, with mortality rates of 7.7% in the CEF group and 44.4% in the amputation group (Fisher exact test, p = 0.045). This difference itself is probably justification enough for the use of CEF as a preferred treatment. However, it is the improvement observed in patient quality of life that provides indisputable grounds for the use of CEF. Patients treated with CEF experienced a clinically and statistically significant increase in their quality of life, as measured by the EQ-5D-3L questionnaire, which was particularly striking as these individuals were considerably worse off than those in the amputation group at the start of treatment. As mentioned above, the lack of studies comparing the quality of life of patients undergoing different kinds of treatment makes it impossible to contextualize the significance of our findings, which we nevertheless deem to be extremely enlightening. Our data bears out the findings of the few series of diabetic patients with osteomyelitis treated with CEF. In Pinzur et al., 20 58 out of the 73 patients analyzed achieved wound healing and did not show signs of recurrence of infection at 1 year. Only one patient in that series died, and another three required an amputation. Dalla Paola et al. 17 treated 39 of their 45 patients successfully, with one single amputation. Saltzman 16 achieved healing in all eight patients they treated with CEF, while Farber et al. 19 treated 10 out of their 11 patients successfully with this treatment modality.
Nonetheless, in addition to the burden that diabetic foot imposes on patients and their families, it is also associated with a significant economic cost.24–27,59,60 The total estimated cost of diagnosed diabetes cases in the United States in 2017 was US$ 327 billion, which includes US$ 237 billion in direct medical costs and another US$ 90 billion in productivity losses. 61 Moreover, it is estimated that between 8% and 25% of the direct medical costs of diabetes are attributable to the management of diabetic foot ulcers.62,63 Our study set out to find out the total cost associated with the management of a patient with osteomyelitis diabetic foot from diagnosis to ulcer healing or death.
The analysis conducted here showed that the overall cost associated with the two treatment modalities analyzed is identical, albeit the distribution of expenses into the different categories was slightly different. For example, the mean preoperative expenses incurred in the case of amputees are slightly (yet not statistically significantly) higher than in patients treated with CEF. This was the case even considering that the mean time elapsed between diagnosis and surgery was longer in the CEF group (29.2 vs 40.5 months; p = 0.082). Conversely, mean postoperative costs were higher, yet not statistically significantly so, in the CEF group. From a quantitative perspective, the expense items showing the greatest difference between the groups were hospital stay, which was longer in the amputation group during the preoperative period and in the CEF group in the postoperative period, and rehabilitation, CEF patients incurring much lower costs than amputees both before-and-after surgery.
Tennvall and Apelqvist 59 and Raghav 60 explain the reasons why economic studies on diabetic foot are difficult to compare. Firstly, many of them do not differentiate between the expenses attributable to treating infected diabetic foot ulcers from those associated with managing diabetes and diabetes-related and other comorbidities suffered by patients. Secondly, not all authors consider the same types of costs in their analyses. Tennvall and Apelqvist 59 provide a list of all the costs that ought to be considered, most of which were as a matter of fact examined in the present analysis (Table 1). Thirdly, costs tend to be underestimated because the expenses incurred before patients are referred for treatment are not known or because the costs incurred by departments other than the ones performing the amputation are excluded. Finally, some publications mix diabetic with nondiabetic patients or infected with noninfected. To these reasons should be added the fact that the literature comprises studies from countries with very heterogeneous health systems and very different costs of living, and conducted at different points of time, some of them drawing on secondary or not very trustworthy sources and bundling together ulcers of various degrees of severity. As a result of the multiplicity of methodologies used, the reported costs for these treatments are unsurprisingly heterogeneous. In a systematic review of 2004, 59 the mean cost of treatment (updated to current values) ranged between €28,776 and €138,773, although it must be said that the article reporting the lowest costs only included hospital costs 64 and the one with the highest costs included both diabetic and nondiabetic patients. 65 The study most akin to ours is Apelqvist et al., 28 which included 50 patients with major amputations and analyzed all direct pre- and postoperative costs as well as the costs corresponding to other departments. The authors reported a per-patient cost of €115,907 (updated to current values). The striking disparity between that figure and the one reported in this study may be due to the 20-year period that elapsed between both analyses and to the fact that the studies were conducted in different countries and that only 15 of the patients in Apelqvist et al. presented with an associated infection. In their economic evaluation, Pinzur et al. 66 found results similar to ours, showing minimal differences between patients treated with amputation and those treated with external fixation. However, their absolute values are somewhat lower, likely due to several factors: they only included patients with successful amputations, they counted costs from the moment of surgery, and they combined patients with and without osteomyelitis. It must be said, however, that there seems to be a certain consensus28,67 that antibiotics account for only a small part (2%–4%) of the total cost. Although these drugs accounted for only 0.09% of the total expense in our series, their prolonged use can result in undesirable medical results.
Unlike previous studies, 67 the present analysis found no correlation between the length of treatment and cost (p = 0.776). This could be explained by the fact that, as mentioned earlier, the time-frames used by the different studies for computing the various expenses are highly heterogeneous. It has also been suggested that patient age could be related to the costs incurred, 59 the cost associated with younger patients being lower than that of older ones. This correlation, however, was not observed in our analysis (p = 0.308).
Our study is not without limitations. Firstly, and most evidently, ours is a non-randomized retrospective study with a small sample size. However, before-and-after designs, typically interventional, that is, non-observational, are warranted in cases where it could be considered unethical to deny a necessary treatment to patients in the control group, 33 as in our case. Also, these designs may be associated with high generalizability. 68 In addition, our small sample size, which is in line with that of previously published studies, is a reflection of the low prevalence of the disease.16,17,19 It should also be pointed out that we did not include the costs associated with the loss of productivity of patients of working age or the expenditure resulting from old age pensions or dependency benefits. However, the fact that the type of treatment administered varied depending on when each case was diagnosed could, to a certain extent, mitigate a potential assignment bias, as assignment was not decided according to the patient’s condition or personal characteristics. Moreover, the study was not based on secondary sources and took into consideration all the costs incurred by each patient from diagnosis to discharge or death. To the best of our knowledge, this is the only study to date that compares the clinical, quality-of-life, and cost-related outcomes of two alternative treatments for diabetic foot which, in addition, includes a cohort of relatively homogeneous subjects, with similar severity levels, where the different patients and conditions were classified into distinct groups. This makes it an innovative study that draws specific conclusions with significant clinical implications.
Conclusion
Patients suffering from infected diabetic foot ulcers treated with external fixation experience a significant quality-of-life improvement as compared with those managed with amputation. Moreover, patients treated with an external fixator tend to survive longer, the costs of both treatment modalities being comparable. The findings of this study demonstrate the efficacy and feasibility of external fixation as a beneficial therapeutic option for this patient population.
