Abstract
Background:
Recombinant human growth hormone (rhGH, somatropin) therapy is approved in children with Prader–Willi syndrome (PWS).
Objectives:
To report safety and effectiveness data for children with PWS treated with biosimilar rhGH (Omnitrope®, Sandoz) in the PAtients TReated with Omnitrope (PATRO) Children study.
Design:
PATRO Children was a multicenter, non-interventional, postmarketing surveillance study.
Methods:
Children with PWS received Omnitrope according to standard clinical practice. Adverse events (AEs) were monitored for the duration of Omnitrope treatment. Effectiveness outcomes were also assessed, including height standard deviation (SD) scores (HSDS).
Results:
As of July 2020 (study completion), 235 patients with PWS had been enrolled. At baseline, 95.7% (n = 225) of patients were prepubertal and 86.4% (n = 203) were rhGH treatment-naïve. At analysis, the median (range) treatment duration in the study was 56.8 (2.9–155.8) months. AEs were reported in 192 patients (81.7%) and were suspected as treatment-related in 39 patients (16.6%). Serious AEs (SAEs) were reported in 96 patients (40.9%) and were suspected as treatment-related in 22 patients (9.4%). The most frequent treatment-related SAEs were sleep apnea syndrome (n = 11; 4.7%), tonsillar hypertrophy (n = 4; 1.7%), and adenoidal hypertrophy (n = 4; 1.7%). Development of scoliosis was considered treatment-related in two patients; development of impaired glucose tolerance in one patient and type 2 diabetes mellitus in another patient were considered treatment-related. Effectiveness outcomes were primarily assessed in 153 patients who completed 3 years of treatment. Among the 151 prepubertal patients (135 treatment-naïve), mean (SD) change from baseline in HSDS after 3 years was +1.50 (1.07) across all patients and +1.57 (1.07) for treatment-naïve patients.
Conclusion:
These data suggest that biosimilar rhGH is well tolerated and effective in patients with PWS managed in real-life clinical practice. Patients with PWS should continue to be closely monitored for well-known safety issues (including respiratory, sleep, and glucose metabolism disorders, and scoliosis) during rhGH treatment.
Introduction
Prader–Willi syndrome (PWS) is a complex genetic disorder that arises from loss of expression of paternal alleles in the PWS region of chromosome 15 (15q11-q13). 1 The principal mechanisms responsible for PWS are paternal deletion of the 15q11-q13 region (around 60% of cases), maternal uniparental disomy of chromosome 15 (around 35% of cases), and imprinting defects.1,2 Clinical manifestations of PWS include initial feeding problems in infancy, followed by hyperphagia and early onset of severe obesity with associated comorbidities; growth failure; hypotonia and musculoskeletal problems; dysmorphic features; hypothalamic/pituitary hormonal abnormalities, such as hypogonadism; and cognitive/behavioral/psychiatric issues.3,4
Sleep disorders are also commonly reported in patients with PWS, including excessive daytime sleepiness and obstructive sleep apnea syndrome.5,6 Sleep-related respiratory dysfunction in PWS is associated with several clinical manifestations of the disorder, including obesity, hypotonia, and hypothalamic dysfunction.6,7 Other comorbidities associated with PWS include scoliosis (reported in up to 80% of patients), gastrointestinal disorders, and dental problems.5,6
Short stature and small hands and feet are characteristic features of PWS. Affected individuals typically demonstrate reduced height velocity (HV) in childhood, absence of pubertal growth spurt, and compromised adult height (AH).1,2 In addition to compromised growth, abnormal body composition (increased fat mass, decreased lean body mass, and low bone density) is consistent with a growth hormone (GH)-deficient status. 8 Recombinant human GH (rhGH, somatropin) therapy for PWS was approved in the United States in 2000 and in Europe in 2001. 9 Data from numerous studies have shown that rhGH therapy improves linear growth and body weight/composition in infants and toddlers as well as older children with PWS.10–14 Additionally, data are emerging that show the benefits are maintained with long-term treatment. 15 Although rhGH therapy has a favorable safety profile in patients with PWS, sudden deaths have been reported in both rhGH-treated and untreated patients, most commonly caused by respiratory disorders.16,17
In 2006, Omnitrope® (somatropin; Sandoz GmbH, Kundl, Austria), an rhGH, was the first biosimilar medicine to be approved by the European Medicines Agency.18,19
The PAtients TReated with Omnitrope (PATRO) Children study was a postmarketing surveillance program for Omnitrope, started in 2006.20,21 The main objectives of PATRO Children were to assess the safety and effectiveness of rhGH in real-world clinical practice across all approved pediatric indications. 19 This article reports safety and effectiveness data from the subgroup of patients with PWS who were included in PATRO Children as of July 2020.
Methods
Study design and patient population
PATRO Children was a multicenter, open-label, non-interventional study. The study design has been described in detail previously. 19 Briefly, the eligible patient population consisted of all infants, children, and adolescents who required rhGH treatment and received at least one dose of Omnitrope. Patients who were rhGH treatment-naïve and patients who were previously treated with another rhGH medicine were eligible for inclusion. All patients (and/or their parents/guardians) provided written, informed consent before participating in the study. The decision to prescribe Omnitrope was at the discretion of the treating physician and independent from participation in the PATRO Children study. Omnitrope was administered as per standard clinical practice and doses were given according to country-specific prescribing information.
The main objective of this study was to assess the safety of rhGH in children with PWS treated within routine clinical practice. A specific focus within the primary objective was to detect adverse events (AEs) that were unexpected and/or unique to patients with PWS. Effectiveness outcomes were also assessed as a secondary objective.
The PATRO Children study protocol was approved by the ethics review committee of participating centers in accordance with national laws and regulations. The study was conducted according to the ethical principles of the Declaration of Helsinki (1964) and its later amendments. All patients (and/or their parents/guardians) provided written informed consent. Patients were permitted to withdraw their informed consent at any time or discontinue Omnitrope treatment for any reason.
Data collection and statistical analysis
Data collection started in September 2007 and ended (final database lock) in July 2020. Patient data were entered into an electronic case report form (eCRF) at each routine visit. eCRFs were reviewed by data management and on-site monitoring was performed by a contract research organization (ICON plc, Dublin, Ireland) to ensure data quality. Standard descriptive statistics were used to describe categorical (e.g. sex) and continuous variables (e.g. age).
Safety assessments
The safety population comprised all patients with PWS enrolled and treated with Omnitrope before the analysis cut-off date (July 2020). Patients who did not have a recorded visit date or Omnitrope treatment start date were excluded from the safety population. All AEs were monitored and recorded for the complete duration of Omnitrope treatment. The intensity of AEs was assessed by investigators’ clinical judgment. The seriousness and relationship of AEs to Omnitrope treatment were independently evaluated by investigator and sponsor assessment and classified according to the worst case. Laboratory assessments and vital signs were requested to be documented at least once a year, according to routine clinical practice. Reasons for treatment discontinuation, including loss of follow-up, were also collected.
Effectiveness assessments
The effectiveness population comprised all patients from the safety population with documented height measurement at baseline (start of Omnitrope treatment) and at least one measurement of height under Omnitrope treatment before the analysis cut-off date (July 2020). Auxological data and rhGH dose were recorded at each visit. Missing data were not included in the analysis. HV (cm/year), height standard deviation (SD) score [SDS (HSDS)], height velocity standard deviation score (HVSDS), and body mass index (BMI) SDS were derived from height and weight measurements and country-specific reference tables from the general population. 22 AH SDS (AHSDS) was calculated using the observed AH (in cm) and the height reference data at age 18 years from the general population. AHSDS relative to reference values from a population of patients with PWS was also calculated. 23 Patients were considered to have reached AH if the investigator identified one of the following reasons for patient discontinuation: reaching AH/bone age maturation; reaching near-AH; referral to adult endocrinologist; HV <1 cm/year plus bone age >14 years for girls and >16 years for boys; HV <1 cm/year if start of puberty is documented and any height measurement is available ⩾4 years after puberty onset in cases where bone age was not known.
Results
Patient characteristics and diagnostic details – safety population
As of July 2020 (study completion), 235 patients with PWS had been enrolled from 12 countries and were included in the safety population. Patient baseline characteristics are shown in Table 1. In total, 203 patients (86.4%) were rhGH treatment-naïve at study entry and 32 patients (13.6%) had been pretreated with another rhGH. At baseline (start of Omnitrope treatment), 225 patients (95.7%) were prepubertal. In total, information on the specific genetic abnormality present was reported for 225 patients (95.7%; Table 2).
Patient baseline characteristics.

BMI, body mass index; HSDS, height standard deviation score; HV, height velocity; HVSDS, height velocity standard deviation score; PC, peak-centered; rhGH, recombinant human growth hormone; SD, standard deviation; SDS, standard deviation score.
Genetic abnormalities identified.

‘Other’ genetic abnormalities included imprinting abnormalities and methylation abnormalities.
Includes four patients who did not have their DNA mutation analyzed (or had missing information for this variable) and six patients who had their DNA mutation analyzed, but for whom a specific genetic abnormality was not recorded.
The mean/median (SD/range) age at baseline was 3.54/1.98 (3.56/0.3–22.5) years in the full safety population, 2.85/1.50 (3.08/0.3–22.5) years in treatment-naïve patients, and 7.96/8.12 (3.31/2.0–13.0) years in pretreated patients.
The dose of Omnitrope at baseline is shown in Table 1. Omnitrope dosing distribution over 6 years for patients is shown in Figures 1 and 2. In naïve patients, the median dose increased over the first year, before leveling off and then steadily decreasing from year 2 to 6 (Figure 2). The median (range) rhGH daily dose during the treatment period of 1–1.5 years after baseline was 33.0 (2.0–46.0) µg/kg in naïve patients (change of +10.5 µg/kg from baseline dose) and 24.6 (7.0–41.0) µg/kg in pretreated patients (change of +2.1 µg/kg from baseline dose). During the treatment period of 1–1.5 years after baseline, 29 patients (13.2%) received Omnitrope within the recommended dosing range (between 85% and 120% of the Summary of Product Characteristics recommended dose of 35.0 µg/kg/day), 47 patients (21.5%) received Omnitrope above the recommended dosing range, and 132 patients (60.3%) received Omnitrope lower than the recommended dosing range. This trend continued over 6 years, with >70% of naïve patients receiving a mean dose of ⩽30.0 µg/kg/day in each treatment interval (Figure 1). At study discontinuation, the median (range) rhGH daily dose was 29.0 (2.0–48.0) µg/kg in naïve patients and 23.0 (7.0–40.0) µg/kg in pretreated patients. The median (range) treatment duration in the study was 56.8 (2.9–155.8) months (equaling approximately 4.7 years). In total, 180 patients (76.6%) completed 3 years of treatment in the study.

Omnitrope dosing distribution over 6 years (patients in the full safety population who were rhGH treatment-naïve at study entry).

Median Omnitrope dosing over 6 years (full safety population).
Past and concomitant illnesses
At PWS diagnosis, 47 patients (20.0%) had documented concomitant respiratory dysfunction. Of these patients, sleep apnea was documented in 66.0% (n = 31) of patients (current or chronic condition, n = 29; past condition, n = 2); snoring in 40.4% (n = 19) of patients (current or chronic condition, n = 16; past condition, n = 3); hypertrophy of tonsils/adenoids in 19.1% (n = 9) of patients (current or chronic condition, n = 5; past condition, n = 4); obesity-hypoventilation syndrome in 12.8% (n = 6) of patients (current or chronic condition, n = 5; past condition, n = 1); aspiration in 8.5% (n = 4) of patients (current or chronic condition, n = 1; past condition, n = 3); and pneumonia in 8.5% (n = 4) of patients (past condition in all four patients).
Endocrine disorders recorded as a current or chronic condition included hypothyroidism/primary hypothyroidism [n = 16; 6.8% (one additional patient had primary hypothyroidism recorded as a past illness)], secondary hypothyroidism (n = 5; 2.1%), GH deficiency (n = 4; 1.7%), cortisol deficiency/secondary adrenocortical insufficiency (n = 3; 1.3%), gonadotropin deficiency/secondary hypogonadism (n = 2; 0.9%), precocious puberty (n = 2; 0.9%), delayed puberty (n = 1; 0.4%), hypoparathyroidism (n = 1; 0.4%), and hypothalamo-pituitary disorder (n = 1; 0.4%).
Study discontinuation
In patients who discontinued treatment prior to study completion (n = 80), the primary reasons for discontinuation were site closure (n = 20), loss to follow-up (n = 17), or change of hospital (n = 5); reaching AH/bone age maturation (n = 14), reaching near-AH (n = 2), or referral to adult endocrinologist (n = 2); switching to another rhGH product (n = 6); AE (n = 6); patient noncompliance (n = 4) or patient not wishing to continue the injections (n = 1); nonresponse (n = 1); and physician decision (n = 2) or parent decision (n = 1). In the six patients who discontinued the study due to an AE, the reported AEs were sleep apnea syndrome (n = 2, one moderate and one severe; both events considered treatment-related), upper airway resistance syndrome (n = 1, severe and treatment-related), abnormal polysomnography (n = 1, moderate and treatment-related), aggression (n = 1, moderate and treatment-related), and injection-site pain (n = 1, mild and not treatment-related).
Adverse events
A summary of patients with AEs is shown in Table 3. Most events were considered to be mild or moderate in intensity, based on investigator clinical judgment. AEs considered to be related to treatment were reported in 39 patients (16.6%, n = 56 events); 44.6% (25/56) of these AEs occurred within the first year of treatment. The most frequently reported treatment-related AEs were sleep apnea syndrome (n = 15 patients; 6.4%); tonsillar hypertrophy (n = 5; 2.1%); adenoidal hypertrophy (n = 4; 1.7%); snoring (n = 2; 0.9%); insulin-like growth factor (IGF) increased to inappropriately high level (n = 2; 0.9%); and scoliosis (n = 2; 0.9%). In total, 22 patients (9.4%) had serious AEs (SAEs) that were considered to be related to treatment (n = 34 events); 50% (17/34) of these SAEs considered to be related to treatment occurred within the first year of treatment. The most frequent treatment-related SAEs were sleep apnea syndrome (n = 11; 4.7%), tonsillar hypertrophy (n = 4; 1.7%), and adenoidal hypertrophy (n = 4; 1.7%). Treatment-related SAEs of moderate or severe intensity are shown in Table 4. There were no SAEs reported with a fatal outcome.
Summary of AEs in patients with Prader–Willi syndrome.

Includes the following MedDRA preferred terms: respiratory tract infection, upper respiratory tract infection, lower respiratory tract infection, respiratory tract infection viral, or respiratory tract infection bacterial.
AE, adverse event; MedDRA, Medical Dictionary for Regulatory Activities; rhGH, recombinant human growth hormone; SAE, serious adverse event.
Moderate and severe treatment-related SAEs in patients with Prader–Willi syndrome.

Time to SAE onset after start of rhGH treatment (all patients with treatment-related SAEs were naïve to rhGH therapy).
Outcome recorded at the time of analysis (July 2020).
MedDRA, Medical Dictionary for Regulatory Activities; NS, not stated; rhGH, recombinant human growth hormone; SAE, serious adverse event.
Reported AEs included impaired glucose tolerance (n = 3 patients, considered treatment-related in one patient), hyperinsulinism (n = 2, considered treatment-related in one patient), insulin resistance (n = 2, considered unrelated to treatment), dyslipidemia (n = 1, considered unrelated to treatment), and type 2 diabetes mellitus (n = 1, considered treatment-related). This latter patient was classified as having obesity at time of type 2 diabetes mellitus diagnosis (BMI of 46.6 kg/m2). Increased blood alkaline phosphatase was reported as an AE in three patients (all considered unrelated to treatment).
Scoliosis was reported as an AE in 48 patients (20.4%) during the course of the study (incidence 41.08 patients per 1000 patient years); four of these patients had a medical history of scoliosis. Scoliosis AEs were considered mild in 25 patients (10.6%), moderate in 13 patients (5.5%), and severe in 8 patients (3.4%). Event intensity information was missing in the remaining two patients. The occurrence of scoliosis was considered to be treatment-related in two rhGH treatment-naïve patients (1.0%), neither of whom had a medical history of scoliosis; one event was severe in intensity and intensity information was missing for the other event.
In total, 26 patients (11.1%) reported sleep apnea syndrome as an AE during the study (incidence 22.25 patients per 1000 patient years); the condition was newly occurring in 15 patients and recorded as a current, chronic, or past condition in the medical history of 11 patients. Sleep apnea AEs were considered to be of mild intensity in 6 patients (2.6%), moderate in 13 patients (5.5%), and severe in 7 patients (3.0%). Sleep apnea was considered treatment-related in 15 patients (6.4%), all of whom were rhGH treatment-naïve; sleep apnea was considered serious in 11 of these patients (4.7%). The intensity of these events was mild in one patient, moderate in seven patients, and severe in three patients.
Of the 26 patients who reported sleep apnea syndrome during the study, 13 patients developed the condition in the first year of rhGH treatment, 4 patients in the second year of treatment, 5 patients in the third year of treatment, and the remaining 4 patients after ⩾4 years of treatment.
Benign skin papilloma was recorded in a female patient; the event was considered mild, unrelated to treatment, and nonserious.
The incidence of SAEs and treatment-related SAEs by age of the patient at baseline was assessed in the full safety population (Table 5). Overall, 55% of the 120 patients <2 years of age at baseline experienced an SAE, compared with 26.1% of the 115 patients ⩾2 years of age at baseline. Treatment-related SAEs were experienced by 11.7% of patients <2 years of age at baseline, compared with 7.0% of patients ⩾2 years of age at baseline. The incidence rate of SAEs was also higher in patients <2 years of age at baseline compared with patients in older age groups.
Incidence of SAEs and treatment-related SAEs by age group at baseline.

PY, patient years; SAE, serious adverse event.
IGF-I values
IGF-I values were categorized as below, within, or above normal range, according to the local laboratory reference ranges. In all treatment-naïve patients with available data, 48.1% of patients had IGF-I levels within normal range at baseline, rising to 70.7% after 1 year of treatment and 73.4% after 2 years (Figure 3). Concurrently, the number of patients with IGF-I levels below normal dropped from 49.6% at baseline to 3.8% after 1 year of treatment, and 1.6% after 2 years. Beyond 1 year of treatment, the proportion of patients within the different IGF-I categories (low, normal, high) was broadly stable, with 70.4–73.4% of patients within the normal range at each subsequent timepoint up to Year 4.
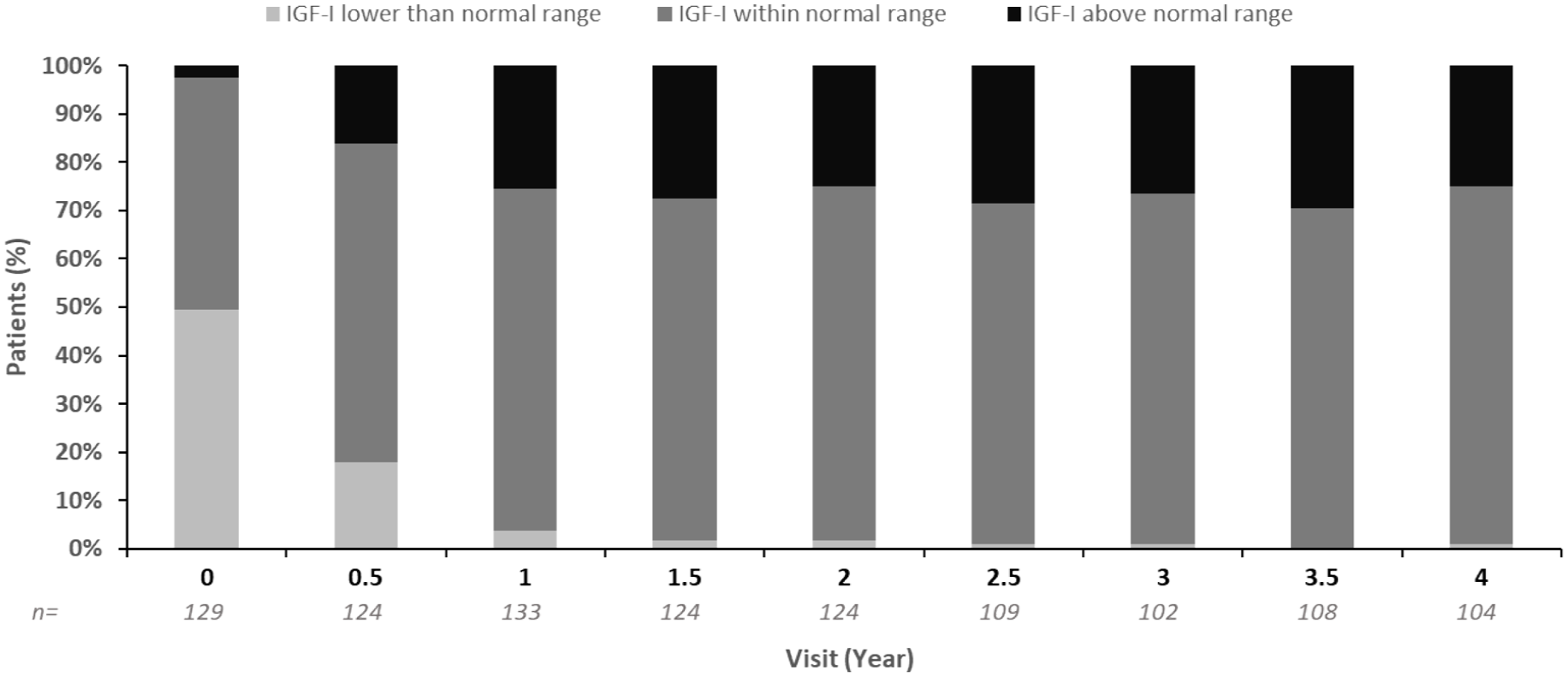
Percentage of patients at each visit with IGF-I values lower than, within, or above normal range (treatment-naïve patients in the safety population with available data).
Effectiveness population
The effectiveness population included 206 patients as of July 2020; 197 of these (95.6%) were prepubertal at baseline. Additional baseline characteristics are shown in Table 1. Thirty-one (15%) of these patients had previously received rhGH treatment before entering the study. The mean (SD) Omnitrope treatment duration was 60.5 (34.5) months. The median (range) prescribed dose at baseline was similar in rhGH-naïve [23.0 (6.0–51.0) µg/kg/day] and pretreated [22.5 (9.0–36.0) µg/kg/day] patients (Table 1). At study discontinuation, the median (range) rhGH dose was 29.0 (10.0–48.0) µg/kg/day in rhGH-naïve patients and 23.0 (7.0–40.0) µg/kg/day in pretreated patients. A total of 153 patients in the effectiveness population had completed 3 years of study treatment (3-year effectiveness cohort); effectiveness data presented in the subsequent section are based on patients in this cohort with available data, unless otherwise stated.
Growth parameters
At baseline, mean (SD) HV was 8.1 (4.2) cm/year across all patients in the 3-year effectiveness cohort, 8.5 (4.3) cm/year in treatment-naïve patients, and 5.9 (2.5) cm/year in pretreated patients. In patients who were prepubertal at baseline (n = 151 in the 3-year effectiveness cohort), mean (SD) HV measured at baseline was 8.2 (4.2) cm/year, 8.6 (4.3) in treatment-naïve patients, and 5.9 (2.7) cm/year in pretreated patients. Mean (SD) HV measured at 3 years’ follow-up was 7.4 (2.0) cm/year across all patients in the 3-year effectiveness cohort, 7.5 (2.0) cm/year in treatment-naïve patients, and 6.0 (1.4) cm/year in pretreated patients. In patients who were prepubertal at baseline, mean (SD) HV measured at 3 years was 7.5 (1.9) cm/year, 7.6 (2.0) cm/year in treatment-naïve patients, and 6.6 (0.8) cm/year in pretreated patients.
Mean HSDS and HVSDS over 3 years are shown in Figures 4 and 5. Mean (SD) ΔHSDS after 3 years was +1.46 (1.10) across all patients (n = 153 at baseline) and +1.57 (1.05) for treatment-naïve patients (n = 136 at baseline). Among the prepubertal group, mean (SD) ΔHSDS after 3 years was +1.50 (1.07) across all patients (n = 151 at baseline) and +1.57 (1.07) for treatment-naïve patients (n = 135 at baseline). After 3 years, mean (SD) ΔHVSDS was +1.43 (3.45) across all patients and +1.59 (3.28) for those who were treatment-naïve. The mean (SD) ΔHVSDS after 3 years was +1.58 (3.12) for all prepubertal patients and +1.55 (3.19) for treatment-naïve prepubertal patients.

HSDS for patients with PWS over 3 years of Omnitrope treatment (3-year effectiveness cohort). (a) All patients. (b) Prepubertal patients.

Peak-centered HVSDS for patients with PWS over 3 years of Omnitrope treatment (3-year effectiveness cohort). (a) All patients. (b) Prepubertal patients.
AH was reached by 10 patients (10/153; 6.5%) from the 3-year effectiveness cohort; 5 of these patients were treatment-naïve at study entry (4 patients were prepubertal) and 5 were pretreated (4 patients were prepubertal). The mean (SD) AH was 157.3 (11.3) cm in treatment-naïve patients and 164.6 (12.5) cm in pretreated patients.
Mean (SD) patient age when reaching AH was 14.3 (1.84) years in treatment-naïve patients and 15.2 (0.35) years in pretreated patients (mean age at baseline is provided in Table 1). The mean (SD) patient age when reaching AH was 13.95 (1.35) years among female patients (n = 4) and 15.24 (1.17) years among male patients (n = 6). Among the treatment-naïve patients who reached AH, mean (SD) HSDS (calculated using reference data from the general population) was −1.49 (1.42) at the start of Omnitrope treatment; these patients achieved a mean (SD) AHSDS of −1.89 (1.57). The mean (SD) AHSDS achieved by the pretreated patients was −1.74 (1.81). Mean (SD) AHSDS relative to reference data from a population of patients with PWS 23 was +0.39 (1.64) among treatment-naïve patients and +0.37 (1.55) among pretreated patients. The mean (SD) AH was 152.3 (12.7) cm in female patients (n = 4) and 164.7 (9.8) cm in male patients (n = 6). The mean (SD) AH in the reference population of patients with PWS was 150.2 (5.5) cm for female patients and 161.6 (8.1) cm for male patients. 23
In the full effectiveness population (n = 206), mean HSDS over 3 years of Omnitrope treatment was comparable between patients regardless of genotype (deletion of 15q11-q13 or maternal disomy; Figure 6).

HSDS by genetic abnormality (full effectiveness population).
Body composition data
Mean BMI SDS over time for the whole and prepubertal effectiveness groups in the 3-year effectiveness cohort are shown in Figure 7. After 3 years, mean (SD) ΔBMI SDS was +1.24 (1.65) across all patients, and +1.33 (1.68) in those who were treatment-naïve. Across prepubertal patients, the ΔBMI SDS after 3 years was +1.32 (1.65) in all patients and +1.39 (1.67) in those who were treatment-naïve.

BMI SDS for patients with PWS over 3 years of Omnitrope treatment (3-year effectiveness cohort). (a) All patients. (b) Prepubertal patients.
Among all enrolled patients with data available (safety analysis set), mean (SD) total fat mass decreased from 33.6% (10.1) at baseline (n = 16) to 21.9% (10.7) at 3 years (n = 53). Mean (SD) lean body mass increased from 42.5% (23.4) at baseline (n = 9) to 60.6% (31.7) at 3 years (n = 12).
Discussion
The PATRO Children study was designed to gather valuable data on the safety and effectiveness of Omnitrope treatment in pediatric patients in a real-life clinical setting. In this analysis of patients with PWS, the median (range) treatment duration was 56.8 (2.9–155.8) months (median approximately 4.7 years). Results from this analysis demonstrated that Omnitrope is well tolerated in the majority of these mostly very young patients with PWS. Most AEs were considered to be unrelated to rhGH therapy and were mild or moderate in intensity. No new safety concerns regarding rhGH therapy in PWS were raised from this analysis. The most frequently reported treatment-related AEs and SAEs were sleep apnea syndrome, adenoidal hypertrophy, and tonsillar hypertrophy; these events were expected in this population as breathing and sleep disorders are well-documented features of PWS. In terms of the timing of treatment-related AEs and SAEs, around 50% occurred in the first year of treatment.
The findings from this analysis are consistent with previous reports of rhGH treatment safety in patients with PWS from observational studies. In a retrospective, observational study of 41 prepubertal rhGH-treated patients with PWS, the most frequently reported AEs were respiratory tract infection (14.6%) and scoliosis (19.5%), 24 both well-documented comorbidities of PWS. These were also the most frequent AEs in our analysis, with 20.4% of patients reporting scoliosis and 16.2% of patients reporting respiratory tract infections. In an analysis of 2332 patients with PWS treated with rhGH in the Pfizer International Growth Database, scoliosis was also the most frequently reported AE, although reported by fewer patients compared with our analysis (6.6%). 13 Also consistent with our findings, PWS-specific features were reported as AEs, including sleep apnea syndrome and psychiatric disorders. 13
Patients with PWS have a higher risk of developing type 2 diabetes mellitus compared with the general population, likely linked to obesity. 25 In this analysis, which included a median treatment duration of 4.7 years, only three patients with PWS developed impaired glucose tolerance (one case considered by investigators to be treatment-related) and one patient developed type 2 diabetes mellitus (considered by investigators to be treatment-related). The patient who developed type 2 diabetes mellitus was classified as having obesity at time of this diagnosis (BMI of 46.6 kg/m2). Our findings are consistent with previous studies that demonstrated rhGH therapy in children with PWS has little or no impact on glucose metabolism.11,26,27 In one study, glucose metabolism improved over 3 years of rhGH treatment in 36 pediatric patients with PWS. 28 One explanation could be that GH’s effect to decrease insulin sensitivity is counteracted by the improvement in body composition (decreased fat mass, increased lean mass), which itself improves insulin sensitivity.
Patients with PWS often have reduced pulmonary function linked to physical characteristics of the syndrome, such as obesity, scoliosis, hypotonia, and respiratory muscle weakness.25,29 Pulmonary function impairments, together with other factors, contribute to the frequent occurrence of respiratory sleep disorders found in patients with PWS. 29 In our analysis, 12.3% of patients (n = 29) had sleep apnea syndrome recorded as a current or chronic condition at the start of treatment and a further 6.4% of patients (n = 15) reported newly occurring sleep apnea during the study. No data were available on whether sleep apnea improved during Omnitrope treatment in this study. Improvements in respiratory function and body composition due to rhGH treatment may have a positive effect on sleep disorders; however, further studies are required to confirm this effect. 27
In this analysis, no AEs with a fatal outcome were reported. However, a risk of unexpected death in patients with PWS has previously been suggested. One review evaluated 486 deaths reported in patients with PWS over a 40-year period. 30 Most deaths occurred in adults, with only 20% of deaths reported in patients younger than 18 years of age. Overall, the most common causes of death were respiratory failure (31% of deaths) and cardiac disease (16% of deaths). 30 Similar results were seen in a study from France, which reported 104 deaths among patients with PWS over an 11- year period, 16% of which occurred in patients under 18 years of age. 31 The primary cause of death was respiratory in 53% of cases (n = 55/104). 31 Another review considered 64 PWS deaths in rhGH-treated (n = 28) and untreated children (n = 36). 16 Respiratory disorders accounted for most of the deaths in both groups. In rhGH-treated children, the majority of deaths occurred in the first 9 months of therapy, suggesting that this may be a high-risk period. 16 Although a causal relationship between rhGH therapy and the reported fatalities is not confirmed, close monitoring of patients with PWS in the first few months of therapy is recommended.32,33 Additional precautions recommended in PWS treatment guidelines include starting patients at a low rhGH dose and monitoring IGF-I to avoid overtreatment. 33
In this study, the percentage of treatment-naïve patients with IGF-I levels in normal range rose from 48% at baseline to 71% after 1 year of rhGH treatment. Thereafter, the percentage of patients within the different IGF-I categories was stable up to Year 4. This increase in IGF-I over the first year of rhGH treatment, followed by stabilization, is in-line with IGF-I data observed in previous studies in patients with PWS treated with rhGH.24,26 A treatment-related AE of IGF-I increased to inappropriately high level was observed in two patients (0.9%) in this study, comparable with the rate observed for the treatment-related AE IGF-I increased in the Pfizer International Growth Database (16/2332 patients = 0.7%). 13
Compared with the overall population of patients enrolled in PATRO Children (which includes the PWS cohort), a higher proportion of patients in the PWS subpopulation reported AEs (81.7% versus 45.8%), SAEs (40.9% versus 10.6%), and treatment-related SAEs (9.4% versus 0.6%). 21 Patients with PWS require closer monitoring during the first years of rhGH therapy compared with other pediatric indications due to the risk of respiratory sleep disorders. To reduce this risk, consensus guidelines recommend carrying out repeat polysomnography (and/or oximetry) during the first 3–6 months of rhGH treatment. 32 If respiratory sleep disorders or related symptoms appear (or worsen), the guidelines strongly recommend further investigation by ear, nose, and throat examination, polysomnography if needed, and IGF-I assessment. 32
Consensus guidelines also recommend that patients with PWS are assessed by a multidisciplinary team prior to starting rhGH treatment, to identify and treat comorbidities that may impact rhGH safety and clinical response.32,33
The starting dose recommended by some consensus guidelines is 0.5 mg/m2 body surface area with subsequent adjustments to 1.0 mg/m2; 32 it has also been advised that GH treatment should be started at a low dose, such as 0.25–0.30 mg/m2/day (or 9.0–12.0 µg/kg/day), increasing in the initial months to reach a standard replacement GH dose of 1.0 mg/m2/day (or 35.0 µg/kg/day). 33 Despite these recommendations, there was wide variation in the baseline dose reported in our analysis (range 2.0–51.0 µg/kg/day in patients who were rhGH treatment-naïve at study entry). Furthermore, the median dose reported in this group at baseline (22.5 µg/kg/day) was considerably higher than the recommended starting dose. As most patients with PWS are overweight or have obesity, weight-based dosing calculations can result in rhGH doses that are too high. The most recent consensus guidelines therefore recommend calculating the appropriate dose using body surface area or, if using patient weight, basing calculations on a non-obese weight corresponding to the patient’s height. 32 Despite the higher-than-recommended starting dose, over the course of the study the majority of patients stayed below the standard GH dose of 35.0 µg/kg/day, with >70% of naïve patients receiving mean doses of ⩽30.0 µg/kg/day over the first 6 years. This may reflect an attempt to avoid, or the fear of, side effects in patients with PWS treated with GH.
These data demonstrate that rhGH (Omnitrope) therapy is effective in patients with PWS in real-world clinical practice. Despite observing a decrease for AHSDS parameters relative to height data from the general population, improvements were recorded in growth parameters, consistent with previous studies of rhGH treatment in these patients. Various factors, including variability in duration of therapy and dosing, as well as the heterogeneity of response, may contribute to this observed discrepancy. Low pubertal height gain observed in patients with PWS can result in reduced AHSDS relative to height data from the general population. Mean AHSDS values relative to height data from a population of GH-naïve patients with PWS were +0.39, demonstrating the effectiveness of Omnitrope in terms of height gain in this patient population.
In this study, the mean (SD) values for HV, HSDS, HVSDS, and BMI SDS in prepubertal patients after 3 years of Omnitrope treatment were comparable with those observed in previous studies in prepubertal children with PWS treated with GH.10,13,34 A retrospective study from Turkey also demonstrated the effectiveness of rhGH in improving HSDS over up to 2 years of treatment in 21 patients with PWS. 35
Considering that obesity in patients with PWS displays different characteristics compared with obesity in a healthy population, it is important to understand how weight changes over time in patients with PWS36,37; an observational study of weight changes in PWS reported small increases in weight and BMI over a 6-month period. 37 It is well documented that while BMI is correlated with fat mass, it is not a precise measure of overall body fat as it is also correlated with lean body mass and height. 38 Nonetheless, as BMI is easy to measure and calculate without requiring access to specialist equipment, it is the method used to measure fat by the majority of pediatric clinics and health practices globally. While BMI increased in patients in this study, mean total fat mass decreased from 33.6% at baseline to 21.9% at 3 years, and mean lean body mass increased from 42.5% at baseline to 60.6% at 3 years. It is important to note that these assessments are based on a relatively small cohort of patients, as data were collected according to routine clinical practice and facilities to measure fat mass and lean body mass are not available in all clinics. Improvements in total fat mass and lean body mass with GH treatment have also been observed in other studies in children with PWS.11,12,15,39
Response to rhGH therapy will vary according to age, the rhGH dose used, pubertal status, degree of growth impairment, and treatment duration. 32 Consensus guidelines for rhGH therapy in PWS suggest a successful first-year response in children includes Δ HSDS greater than +0.3, HV increment ⩾3 cm/year, or HVSDS greater than or equal to +1. 32 Response in PATRO Children was considered successful by all of these criteria.
Early initiation of rhGH (between 4 and 6 months of age) is recommended for children with PWS. 32 Among rhGH-naïve patients included in the current analysis, the mean age at start of treatment was 2.85 years; this difference likely reflects delays in specialist referral/diagnosis in the real-world setting. Alongside delayed access to specialist treatment centers and delayed diagnosis, fears of an increase in adenoid tissue in infants can also result in the late initiation of GH treatment. Treatment delays can result from the fact that some centers will wait until sleep apnea and other breathing problems have been excluded prior to initiating GH to reduce the risk of treatment complications. Difficulties in carrying out or accessing polysomnography studies can contribute to delays in these situations.
Results from this study suggest that the incidence of SAEs and treatment-related SAEs may be higher in younger patients with PWS treated with rhGH; however, it is important to note that these analyses are limited by the fact that 70% of patients in this study were aged ⩽3 years and more than 80% of SAEs occurred in this age group, along with the generally low incidence of SAEs, particularly treatment-related SAEs.
A topic of clinical interest that was outside of the scope of our study is the potential relationship between AEs and IGF-I levels in patients with PWS. This is a complex topic that would require an appropriately designed prospective study to investigate robustly. As PATRO Children was a non-interventional study, physicians were not mandated to collect IGF-I measurements at specific timepoints, nor in association with AEs, precluding a reliable analysis of this relationship. The potential relationship between AEs and IGF-I levels should be investigated in future studies in patients with PWS, as outcomes could help to guide clinical management of these patients.
The PATRO Children study has some limitations that are common to all observational studies. First, there is a risk of selection bias due to enrollment of patients only from selected clinics, and information bias due to missing or erroneous data collection, as data are collected according to routine clinical practice. As a result, some assessments in the current analysis are based on a relatively small amount of data. Second, calculation and justification of the sample size for this analysis were not carried out. The sample size included in the analysis was relatively small; therefore, our findings may need to be verified in a larger population. Furthermore, the median treatment duration was relatively short (approximately 4.7 years), which may limit the interpretation of some data, such as side effects of treatment that may take several years to develop.
As patients were enrolled from multiple centers, there may be some differences in the management of patients with PWS at a country and center level; any potential differences were not identified or considered in our analysis. Finally, as there was often a 6–12-month period between patient visits, and limited consultation time during routine visits in some cases, there is a possibility that some AEs were under-reported.
Conclusion
These data demonstrate that Omnitrope treatment is well tolerated and effective in this population of pediatric patients with PWS treated in real-world clinical practice. Our data extend the evidence-base for the benefits of rhGH when used in this patient population. Well-known safety issues in patients with PWS (such as respiratory and sleep disorders, scoliosis, and diabetes) should continue to be closely monitored during rhGH treatment.
Footnotes
Acknowledgements
Medical writing support was provided by Eve Blumson, PhD, of Apollo, OPEN Health Communications, and funded by Sandoz/Hexal AG, in accordance with Good Publication Practice (GPP) guidelines (![]() ). Support with submission was provided by Aven Bang of OPEN Health Communications, and funded by Sandoz/Hexal AG. All authors have authorized the submission of their manuscript via a third party and approved all statements and declarations. Statistical input was provided by Masiur Rahman of Novartis Pharmaceuticals, Wallingford, CT, USA. The authors would like to thank all patients and investigators who participated in the PATRO Children study.
). Support with submission was provided by Aven Bang of OPEN Health Communications, and funded by Sandoz/Hexal AG. All authors have authorized the submission of their manuscript via a third party and approved all statements and declarations. Statistical input was provided by Masiur Rahman of Novartis Pharmaceuticals, Wallingford, CT, USA. The authors would like to thank all patients and investigators who participated in the PATRO Children study.
