Abstract
Background:
Perioperative hyperglycemia is a risk factor for postoperative complications in the general population. However, it has not been clarified whether perioperative hyperglycemia increases postoperative complications in patients with type-2 diabetes mellitus (T2D). Therefore, we aimed to analyze the relationship between perioperative glycemic status and postoperative complications in non-intensive care unit (non-ICU) hospitalized patients with T2D.
Materials and Methods:
Medical records of 1217 patients with T2D who were admitted to the non-ICU in our hospital were analyzed retrospectively. Relationships between clinical characteristics including perioperative glycemic status and postoperative complications were assessed using univariate and multivariate analyses. Perioperative glycemic status was evaluated by calculating the mean, standard deviation (SD), and coefficient of variation (CV) of blood glucose (BG) measurements in preoperative and postoperative periods for three contiguous days before and after surgery, respectively. Postoperative complications were defined as infections, delayed wound healing, postoperative bleeding, and/or thrombosis.
Results:
Postoperative complications occurred in 139 patients (11.4%). These patients showed a lower BG immediately before surgery (P = 0.04) and a higher mean postoperative BG (P = 0.009) than those without postoperative complications. There were no differences in the other perioperative BG parameters including BG variability and the frequency of hypoglycemia. The multivariate analysis showed that BG immediately before surgery (adjusted odds ratio (95% confidence interval [CI]), 0.91 (0.85–0.98), P = 0.01) and mean postoperative BG (1.11 (1.05–1.18), P < 0.001) were independently associated with postoperative complications.
Conclusion:
Perioperative glycemic status, that is, a low BG immediately before surgery and a high mean postoperative BG, are associated with the increased incidence of postoperative complications in non-ICU patients with T2D.
Keywords
Introduction
Patients with diabetes are more likely to undergo surgery than those without diabetes, and they tend to have a higher risk of postoperative complications.1,2 However, the significance of perioperative hyperglycemia for the mortality and postoperative complications has not been verified in patients with diabetes. The Leuven 1 study, 3 a large-scale clinical trial for surgical intensive care unit (SICU) patients, showed the efficacy of glucose management on 1-month mortality; however, the subgroup analysis in patients with diabetes failed to show similar efficacy. Frisch et al. 2 performed a large observational study of the perioperative period in patients receiving noncardiac surgery and showed the efficacy of glucose management on hospital length of stay, complications, and mortality; although the subgroup analysis in patients with diabetes failed to show comparable efficacy.
Perioperative hyperglycemia is known to increase the risk of infection,3,4 stroke, 5 myocardial infarction, 5 and death3,5,6 in the general population. It is also considered to increase the risk of arterial and venous thrombosis because it increases platelet aggregation and the activation of coagulation factors.7–10 Furthermore, hyperglycemia per se increases the risk of delayed wound healing. 11 Therefore, the American Diabetes Association recommends that the target blood glucose (BG) range for the perioperative period in patients with diabetes should be 80–180 mg/dL. 12 The American College of Surgeons and the Surgical Infection Society recommend that all patients target a BG range of 110–150 mg/dL to lower the risk of surgical site infection (SSI) in the immediate preoperative period. 13 The Japan Diabetes Society recommends a fasting BG range of 100–140 mg/dL and a postprandial BG range of 160–200 mg/dL for patients with diabetes before surgery. 14 However, no evidence currently supports a recommended BG range for patients with diabetes undergoing surgery.
The aim of this study is to clarify the relationship between perioperative glycemic status and postoperative complications in patients with T2D. Here, we retrospectively analyzed the association between perioperative BG parameters and general postoperative complications in non-intensive care unit (non-ICU) patients with T2D who received intensive glucose management before surgery. In this study, the postoperative complications were defined as infections, delayed wound healing, bleeding, arterial thrombosis, and venous thrombosis because non-ICU patients with T2D were supposed to have lower possibilities of serious events, such as death and severe disabilities than ICU patients.
Materials and methods
Study design
We conducted a retrospective study based on the medical records of patients with T2D who received intensive glucose management before surgery at Nippon Medical School Hospital (Tokyo, Japan) during the 7-year period from January 2012 to December 2018. The ethics committee of Nippon Medical School Hospital approved this study (No. R1-08-1174) and waived the requirement for informed consent because all patient data were anonymized. The opportunity to opt-out was guaranteed for the patients. We screened patients with T2D (N = 2271) who had been referred to our department for intensive glucose management before surgery and who underwent glucose monitoring four times a day before breakfast, lunch, and dinner (around 08:00, 12:00, and 18:00 under the condition of fasting), and 21:00 using a point-of-care testing device (One Touch Verio pro plus; Johnson and Johnson Co., New Brunswick, NJ). We excluded patients who were admitted to the ICU, used immunosuppressants, used corticosteroids, infected before surgery, underwent reoperation, ophthalmic surgery, and/or surgery under local anesthesia. However, only the initial surgery was included in our study when a patient underwent reoperation. We also excluded cases in which any of the BG levels were completely missing in the preoperative or postoperative period because we could not calculate mean BG. In other words, patients were eligible for this study if their BG levels were measured at least once using 4-point BG profiles each before and after surgery, even if their hospital stay was fairly short. A total of 1054 patients were excluded and thus the data were analyzed for 1217 patients (Figure 1).
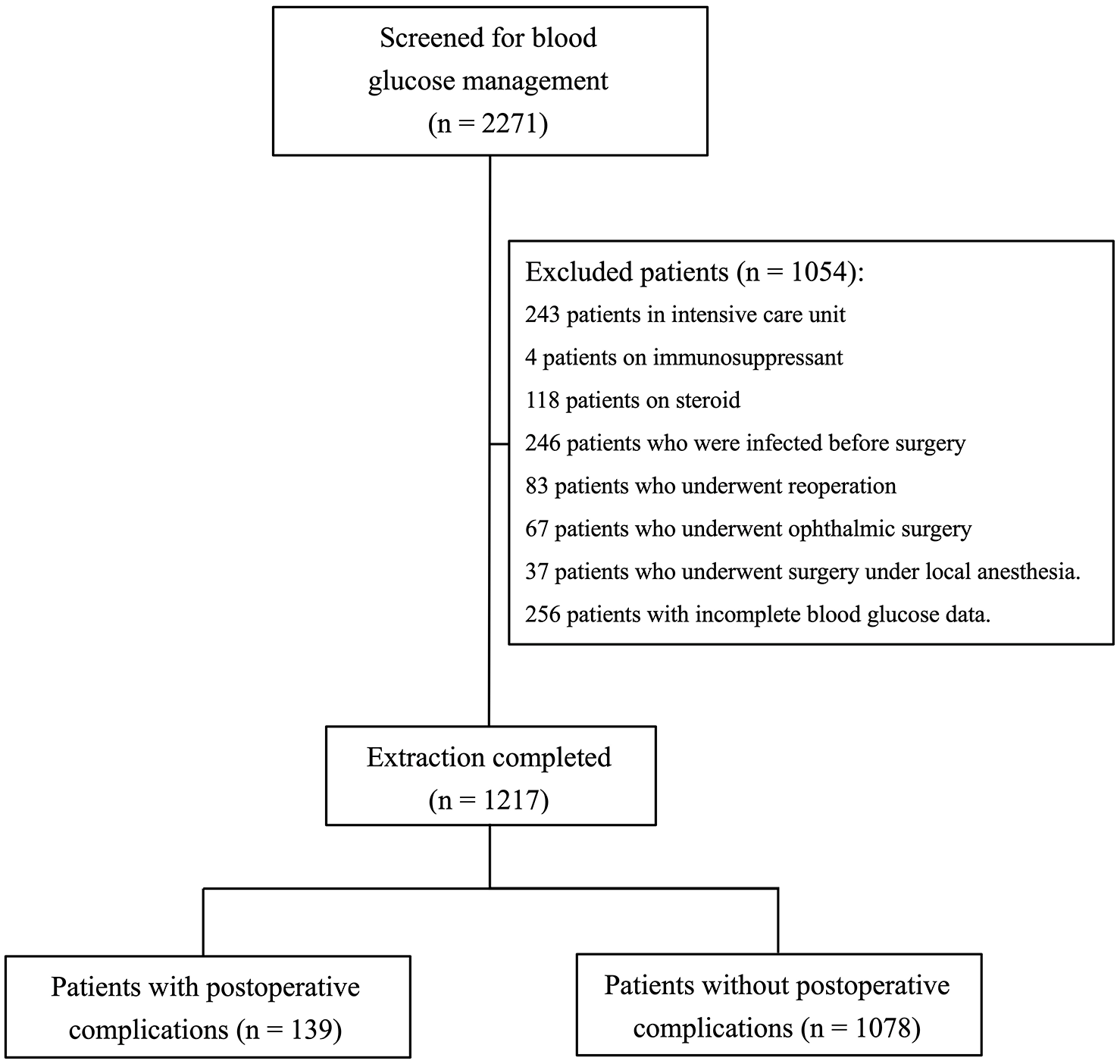
Participant flow through our study: in total, 2271 participants were screened, 1054 were excluded, and 1217 met the eligibility criteria and were enrolled in this retrospective study.
Clinical measurements
Physical examinations (height and body weight) undergone on the first day of hospitalization, vital signs (systolic and diastolic blood pressure, pulse rate, and body temperature) checked on the same day, and results of a blood test performed after an overnight fasting on the second day of hospitalization were collected. On the blood test, fasting plasma glucose was measured using the glucose oxidase method (ADAMS Glucose GA-1170; Arkray, Kyoto, Japan). Glycated hemoglobin (HbA1c) was measured using high-performance liquid chromatography (ADAMS A1c HA-8160; Arkray) and is expressed as the percentage value of the National Glycohemoglobin Standardization Program according to the Japan Diabetes Society’s guideline. 15 Glycated albumin (GA) was measured using an enzymatic method that exploits albumin-specific proteinase and ketoamine oxidase (Lucica GA-L; Asahi Kasei Pharma, Tokyo, Japan) and is expressed as a percentage of total albumin. Serum and urinary C-peptide levels were measured using a chemiluminescent enzyme immunoassay (Fujirebio Inc., Tokyo, Japan). Serum total cholesterol (TC), high-density lipoprotein cholesterol, and triglyceride were measured enzymatically (Sekisui Medical, Tokyo, Japan). Low-density lipoprotein cholesterol (LDL-C) was calculated using the Friedewald formula. 16 Estimated glomerular filtration rate (eGFR) was calculated using the following formula: eGFR (mL/min/1.73 m2) = 194 × creatinine–1.094 × age–0.287 (for females, the total value was multiplied by 0.739). 17 Information on the duration of T2D and smoking habit (current, past, and never) at the time of admission was also collected.
Protocol for intensive glucose management
During the hospitalization period, dietary energy intake (kcal/day) was restricted to 27.5 kcal/kg of standard body weight (SBW) in non-obese subjects (body mass index (BMI, kg/m2) < 25) and to 25.0 kcal/kg of SBW in obese subjects (BMI ⩾ 25). SBW was calculated using the following formula because a BMI of 22 is regarded as the SBW: 18 SBW (kg) = (height (m))2 × 22 (BMI). The daily dietary energy intake was divided almost evenly among breakfast, lunch, and dinner meals. Each diet contained approximately 55% of energy as carbohydrate, 25% as fat, and 20% as protein. Patients using oral hypoglycemic agents before hospital admission had them withdrawn at the time of admission. The targets of perioperative glucose control were to maintain a fasting BG range of 100–140 mg/dL and a postprandial BG range of 160–200 mg/dL. To achieve the desired goals, the patients received basal-bolus insulin therapy using long- and/or rapid-acting insulin analogs. On the day of surgery, basal insulin was administered without changing the dosage and the bolus insulin was discontinued. We added one unit of regular insulin per 5.0–12.5 g of glucose in the infusion preparations, and measured BG immediately and at 2 and 4 hours after surgery. The administration of bolus insulin was resumed when the patient resumed oral diet after surgery. In case of unexpected hyperglycemia after surgery, we used continuous intravenous insulin infusion to control BG without using sliding scale insulin.
Perioperative glycemic status parameters and outcome measurements
The primary outcome was postoperative complications which included infections (including SSI), delayed wound healing (including anastomotic leakage), bleeding, arterial thrombosis (i.e. ischemic stroke and myocardial infarction), and venous thrombosis (i.e. deep vein thrombosis and pulmonary embolism). We used the CDC-2017 criteria for diagnosing SSI. 19 Delayed wound healing was determined with reference to medical records. Except for SSIs, the follow-up period for postoperative complications was 30 days. Perioperative glycemic status was examined by calculating the mean, standard deviation (SD), and coefficient of variation (CV) in preoperative and postoperative periods for three contiguous days before and after surgery, respectively. If their hospital stay was short and 3 days of BG data were not available, the BG parameters were calculated based on that data for 1 or 2 days. The mean number of preoperative, postoperative, and perioperative BG measurements were 11.0 ± 1.9, 11.5 ± 1.5, and 26.4 ± 2.6 times, respectively. The BG data were a composite of BG measurements before breakfast, lunch, and dinner, and at bedtime for each period. Mean BG after surgery was calculated from the BG immediately, and at 2 and 4 hours after surgery. Mean perioperative BG was the average value of all perioperative BG, except for those measured during surgery. BG immediately before surgery was the last BG recorded before surgery; in detail, BG around 08:00 in the case of morning surgery or BG around 12:00 in the case of afternoon surgery. Since all the patients did not eat after 21:00 on the day before surgery, BG immediately before surgery was all measured in the overnight fasting condition. SD and CV were calculated from BG data over the hospitalization period to investigate the relationship between BG variability and postoperative complications. Hypoglycemia and severe hypoglycemia were defined as BG levels of ⩽70 mg/dL and <54 mg/dL, respectively, as recommended by the International Hypoglycaemia Study Group. 20
Statistical analysis
Continuous variables were expressed as the median and interquartile range, and categorical variables were expressed as the number and percentage. Continuous variables with a normal distribution were compared using the Student’s t-test, and those abnormally distributed were compared using the Mann–Whitney U-test. All dichotomous variables were compared using Pearson’s χ2 test or Fisher’s exact test. Multivariate regression models were used to determine statistically significant predictive factors for postoperative complications. The independent variables used for multivariate analysis were gender, statistically significant variables in the univariate analysis, and previously reported risk factors for postoperative complications, such as smoking, 21 obesity,22–24 age,24–27 operation duration,27,28 amount of intraoperative blood loss,29–32 length of hospital stay,28,33,34 and nutritional status.35,36 All analyses were two-sided, and a P value of <0.05 was considered statistically significant. We performed a complete case analysis which excluded missing values because these were missing at random and overall infrequent. A receiver operating characteristic (ROC) curve was generated, and the area under the curve (AUC) was calculated to determine the optimal BG required to reduce postoperative complications. All analyses were performed using JMP 13.2 software (SAS Institute Inc., Cary, North Carolina) and EZR software 37 (Saitama Medical Center, Jichi Medical University, Saitama, Japan), which is a graphical-user interface for R (The R Foundation for Statistical Computing, Vienna, Austria).
Results
Of the 1217 patients with T2D who underwent surgery, postoperative complications were reported for 139 (11.4%). The number of patients in each postoperative complication category is shown in Table 1. Urinary tract infections and delayed wound healing were the most frequent postoperative complications. The characteristic differences between the patients with and without postoperative complications are summarized in Table 2. Albumin, TC, and LDL-C were significantly lower in patients with postoperative complications, and pulse rate on the day of surgery was significantly higher than in patients who did not develop postoperative complications. Also, more patients with postoperative complications than without them had a history of ischemic heart disease.
Breakdown of postoperative complications.

SSI, surgical site infection.
Postoperative complications include infections (including SSI), delayed wound healing (including anastomotic leakage), bleeding, arterial thrombosis (i.e. ischemic stroke, myocardial infarction, and others), and venous thrombosis (i.e. deep vein thrombosis and pulmonary embolism). The CDC-2017 criteria 19 were used for diagnosing SSI. Delayed wound healing was determined with reference to medical records in this study. Except for SSIs, the follow-up period for postoperative complications was 30 days.
Baseline characteristics of patients.

BMI, body mass index; bpm, beats per minute; eGFR, estimated glomerular filtration rate; HDL, high-density lipoprotein; LDL, low-density lipoprotein.
Continuous variables are expressed as median (interquartile range).
Table 3 shows the differences in BG parameters and operative outcomes between patients with and without postoperative complications. BG immediately before surgery was significantly lower in patients with, as opposed to those without, postoperative complications. Conversely, mean postoperative BG was significantly higher in patients with versus without postoperative complications. There were no differences in the other BG parameters, including BG variability (SD and CV), and number of patients with hypoglycemia between the two groups. Operation duration and the amount of intraoperative blood loss were significantly longer and larger, respectively, in patients with postoperative complications compared with those without them. Mortality was not different between groups. In the multivariate analysis (R2 = 0.07; P < 0.001), which included gender, age, smoking, BMI, and significant variables from the univariate analysis, the following independent variables were statistically significant predictors of postoperative complications: history of ischemic heart disease, BG immediately before surgery, mean postoperative BG, and amount of intraoperative blood loss (Table 4). In addition, we performed univariate and multivariate analyses on each individual outcome, that is, infections, delayed wound healing, postoperative bleeding, or thrombosis (Supplementary Table 1–4). When we performed a multivariate analysis including gender, age, and significant variables in the univariate analysis for each outcome, only mean postoperative BG was a statistically significant predictor of postoperative bleeding among the BG parameters (Supplementary Table 3).
Differences in perioperative glycemic control parameters and operative outcomes between patients with and without postoperative complications.

BG, blood glucose; CV, coefficient of variation; SD, standard deviation.
Continuous variables are expressed as median (interquartile range). Hypoglycemia is defined as BG level of 70 mg/dL or below. Severe hypoglycemia is defined as BG level below 54 mg/dL.
Univariate and multivariate analyses of independent variables associated with postoperative complications.
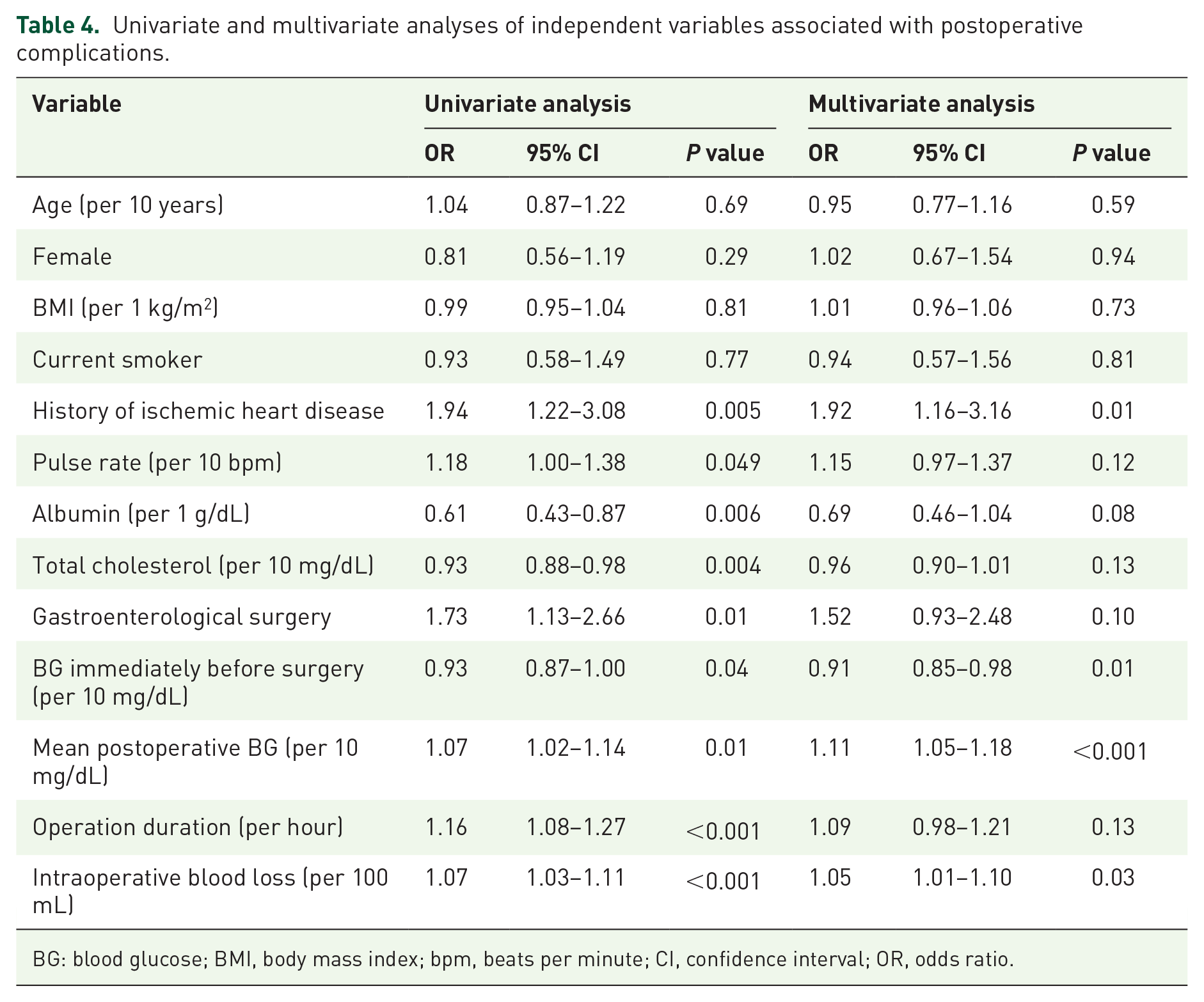
BG: blood glucose; BMI, body mass index; bpm, beats per minute; CI, confidence interval; OR, odds ratio.
In order to propose a target perioperative BG range in patients with T2D, we constructed ROC curves for the prediction of postoperative complications using BG immediately before surgery and mean postoperative BG (Figure 2). When the BG cut-off level was set at 118 mg/dL immediately before surgery, it was optimally predictive of postoperative complications, and the sensitivity and specificity were 68.2% and 41.2%, respectively. The AUC for postoperative complications was 0.553 (95% confidence interval (CI): 0.501–0.606). When the mean postoperative BG cut-off level was set at 150 mg/dL, it was optimally predictive of postoperative complications, and the sensitivity and specificity was 39.3% and 73.4%, respectively. The AUC for postoperative complications was 0.568 (95% CI, 0.517–0.619).

Receiver operating characteristic (ROC) curves for the prediction of postoperative complications using blood glucose (BG) immediately before surgery at 118 mg/dL (a) and mean postoperative BG at 150 mg/dL (b). ROC curves were generated to optimally predict postoperative BG complications using immediately before surgery and mean postoperative BG.
Discussion
Most previous studies have shown no association between hyperglycemia and postoperative complications, including mortality, in patients with diabetes.2,3,38 In fact, these previous studies recruited patients who were admitted to the ICU or cardiovascular surgery department. In addition, postoperative complications reported in the previous studies were limited to serious events such as death and severe disabilities.3,38 However, this study accessed the association between perioperative BG parameters and general postoperative complications including infections, delayed wound healing, bleeding, arterial thrombosis, and venous thrombosis because non-ICU patients with T2D were supposed to have lower possibilities of serious events than ICU patients. We demonstrated here that a decrease in BG immediately before surgery and an increase in mean postoperative BG were associated with the increased incidence of general postoperative complications.
In this study, we showed that the risk of postoperative complications was associated with a low BG immediately before surgery. King et al. 39 reported that mean BG in the first 24 hours after surgery of <80 mg/dL and >150 mg/dL increased rate of infection in patients with diabetes undergoing noncardiac surgery. Another study of total knee arthroplasty in non-diabetic patients showed that hypoglycemia increased a risk of re-operation by increasing postoperative infections. 40 These studies suggest that not only perioperative hyperglycemia but also hypoglycemia could be a risk factor for postoperative infection. Furthermore, hypoglycemia can stimulate a secretion of glucocorticoid which impairs leukocyte adhesion to the vascular endothelium and migration to infected or damaged tissues, 41 likely resulting in postoperative infection and delayed wound healing. In addition, hypoglycemia stimulates a sympathetic response by catecholamine and following activation of plasminogen activator inhibitor-1 (PAI-1) will promote thrombosis.42–44 We therefore consider that the insulin dose should be adjusted to achieve moderate glucose control for avoiding hypoglycemia particularly during the preoperative period.
In contrast, since the risk of postoperative complications was associated with an increase in mean postoperative BG, strict glucose control should be considered after surgery. The concept can be supported by the finding in the DIGAMI study 45 (a multi-center, open-label, randomized trial comparing the effects of two BG targets on 1-year mortality in diabetic patients with acute myocardial infarction) that intensive treatment group with mean BG 173 mg/dL reduced relative mortality by 29% as compared with control group with mean BG 211 mg/dL. Potential mechanisms underlying the association between postoperative hyperglycemia and postoperative complications could be considered as follows. First, previous studies have pointed that hyperglycemia impairs polymorphonuclear neutrophil cell function by modulating endothelial adherence, chemotaxis, phagocytosis, and bacterial killing function.46,47 Hyperglycemia also affects cytokine response by slowing production of inflammatory cytokines and/or by reducing production of local proinflammatory cytokines.48,49 Such effects of hyperglycemia on the immune system could increase susceptibility to postoperative infection. Second, hyperglycemia increases reactive oxygen species production from the mitochondrial electron transport chain through the protein kinase C-dependent activation of NAD(P)H oxidase.50–52 The increased oxidative stress could induce microcirculatory disturbances in the wound, likely resulting in delayed wound healing. Third, hyperglycemia increases activities of blood coagulation factors, that is, factors VIIa and VIII, the thrombin–anti-thrombin complex, von Willebrand factor, and tissue factor pathway inhibitor, and decreases activity of PAI-1.40,53–55 Following platelet aggregation, blood coagulation could increase the risk of arterial and venous thrombosis.
Some reports have shown an association between glycemic variability and mortality in hospitalized patients.56–59 Moreover, there was a similar report in patients who were undergoing surgery. 59 In this study, the indices of BG variability, that is, SD and CV, were not associated with postoperative complications. In fact, most of the previous reports were performed within ICU patients. It is likely to be considered that BG variability might not be associated with the non-severe postoperative complications. In addition, all the participants in this study received insulin therapy, and insulin doses were adjusted carefully by diabetologists to achieve good BG control without hypoglycemia. These can be the reasons why no significant differences in SD and CV were observed between T2D patients with postoperative complications and those without in this study.
In addition to the glycemic indices, the amount of intraoperative blood loss was significantly associated with the risk of postoperative complications in the multivariate analysis. Also, participants who had a history of ischemic heart disease were at higher risk for postoperative complications. This is likely because participants who had a history of ischemic heart disease tended to receive a treatment with antiplatelet agents (68.5% vs 10.6%, respectively; P < 0.001) and were at a high risk of intraoperative bleeding. Furthermore, the discontinuation of antiplatelet agents during the perioperative period could increase the risk of thrombosis. In general, patients with T2D and ischemic heart disease tend to have peripheral tissue ischemia which can cause delayed wound healing owing to advanced atherosclerosis. Of note, neither the type of surgery nor the operation duration was associated with the risk of postoperative complications. We consider that the increasing trend of endoscopic surgeries might affect the outcome because endoscopic surgeries tend to prolong operative duration but to be less invasive with fewer postoperative complications compared with open surgery.60–63 In this study, we could not include the factor of surgical procedures, that is, either endoscopic or open surgeries, in the analysis due to the following reasons. Endoscopic surgeries are not performed in the field of orthopedic, otolaryngology, plastic, breast, neurological, and cardiovascular surgery. For comparing endoscopic and open surgeries, we should restrict the field to thoracic and abdominal surgeries. However, since patients who are admitted to the non-ICU tend to receive less-invasive surgeries, most of the thoracic and abdominal surgeries were performed in endoscopic procedures. Therefore, a further study including ICU patients would be necessary to clarify the influence of surgical procedures on the relationships between perioperative glycemic status and postoperative complications.
There are several limitations associated with this study. First, although the sample size was reasonably large, this was a case–control study without randomization. In addition, as a single-center study, the findings cannot be generalized to all patients with T2D. Second, sample selection bias likely occurred because patients with T2D who were not referred to our department before operation were not included in this study. Third, it is possible that we underestimated postoperative complications, given the follow-up period was as short as 30 days in cases without SSI. Fourth, we did not evaluate the effect of perioperative artificial nutritional support on postoperative complications. Finally, the BG data used for analysis was limited to 3 days after surgery. Therefore, the relationship between BG after surgery and postoperative complications could not be evaluated over a longer term.
In conclusion, this study demonstrated that perioperative glycemic status, that is, a low BG immediately before surgery and a high postoperative BG, was independently associated with the increased incidence of postoperative complications in non-ICU patients with T2D. Accordingly, even for diabetic patients who receive less-invasive surgeries, tight glycemic control might be necessary in the postoperative period. Future interventional studies are warranted to provide an optimal range of perioperative BG levels for reducing the risk of postoperative complications in patients with diabetes.
Supplemental Material
sj-docx-1-tae-10.1177_20420188221099349 – Supplemental material for Perioperative glycemic status is linked to postoperative complications in non-intensive care unit patients with type-2 diabetes: a retrospective study
Supplemental material, sj-docx-1-tae-10.1177_20420188221099349 for Perioperative glycemic status is linked to postoperative complications in non-intensive care unit patients with type-2 diabetes: a retrospective study by Takeshi Oba, Mototsugu Nagao, Shunsuke Kobayashi, Yuji Yamaguchi, Tomoko Nagamine, Kyoko Tanimura-Inagaki, Izumi Fukuda and Hitoshi Sugihara in Therapeutic Advances in Endocrinology and Metabolism
Footnotes
Acknowledgements
All the authors of this manuscript contributed significantly to this work.
Author contribution(s)
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
