Abstract
Objective:
The objective was to analyze the efficacy of recombinant human growth hormone (rhGH) treatment in children born small for gestational age (SGA) without catch-up growth treated before the onset of puberty, with follow-up until adult height. The influence of demographic and auxological factors on the final response evaluated as adult height and height gain was assessed.
Patients and methods:
A prospective longitudinal observational study performed in a tertiary hospital, involving SGA patients, who started treatment with rhGH between October 2003 and April 2015. Potential response predictors were evaluated by multiple regression analysis and receiver operating characteristic curves.
Results:
Of the initial 96 patients included, 61 patients (28 boys and 33 girls) reached adult height. Adult height gain in standard deviation (SDS) was 0.99 (0.8) and 1.49 (0.94), respectively (p < 0.05). An adult height greater than −2 SDS was reached in 75% of the girls but only in 53% of the boys. The pubertal height gain was 22.6 (5.8) cm in boys and 18.8 (4.5) cm in girls. The multiple regression model obtained for total height gain explained 42% of the variability in this variable including sex, height gain during the first year, and the difference from target height at the start of treatment. A first-year height gain of 0.69 SDS was the optimal point for assessing a final height gain greater than 1.5 SDS with a specificity of 70% and a sensitivity of 71%.
Conclusion:
Most SGA patients achieve normalization of height above −2 SD, the percentage being higher in girls. According to our predictive model, height gain in the first year is the most important variable for predicting good response to treatment. During puberty, there is a loss of height SDS, probably due to a lower total pubertal gain with respect to the reference population, which is more marked in boys.
Introduction and objectives
The term small for gestational age (SGA) refers to newborns with a length or birth weight 2 or more standard deviations below the mean for their gestational age and sex,1,2 based on the reference population. These patients represent 3.1–5.5% of the population. 3
The causes of SGA are multifactorial and include maternal lifestyle, obstetric factors, placental dysfunction, and fetal genetic and epigenetic abnormalities. However, in a large proportion of SGA patients (about 40%), no clear cause can be established.
Persistent short stature is one of the most common complications of SGA births. These infants usually have a more marked catch-up growth during the first 6 months, and this is typically completed in the first 2 years of life (in some cases extending up to 4 years, especially in preterm infants). 4 However, based on previous studies, it is known that approximately 10% of these children do not show this catch-up growth between 2 and 3 years of life, eventually achieving a low adult height (AH).
Treatment with recombinant human growth hormone (rhGH) in SGA children without postnatal catch-up growth was approved in 2003 by the European Medicines Agency. 5 In Spain, according to data from the working group of the Spanish Society of Pediatric Endocrinology, SGA patients represent approximately 21% of all authorized treatments. 6 The recommended starting dose is 0.035 mg/kg per day, which can be increased to a maximum of 0.050 mg/kg per day, administered subcutaneously.
The effectiveness of rhGH treatment has been well documented,4,7 and multiple studies have compared the final height of SGA patients treated and not treated with rhGH. Nonetheless, because of the heterogeneous nature of SGA children, the response to treatment with rhGH can vary. Accordingly, multiple studies are currently aimed at determining the factors involved in the results obtained, and there is evidence that around 5–8% of patients are considered nonresponders. 8 However, given that insufficient time has elapsed since the approval of rhGH treatment, most of these studies do not include a large number of patients who have reached AH.
Height and age at onset of puberty, as well as the magnitude and duration of pubertal growth, are important determinants of AH, explaining 15–20% of this. 9 In SGA children, puberty generally begins within the normal range for their age, but perhaps relatively early for their low height at onset of puberty. Most published studies on puberty in SGA children consider height and age at the onset of puberty but not total pubertal gain (TPG) and its impact on AH.
Taking these aspects into account, our study had two objectives. The primary objective was to analyze the efficacy of rhGH treatment by studying the factors affecting the final response to treatment assessed by AH and height gain (HG). The secondary objective was to study the pubertal gain in SGA patients treated with rhGH before the onset of puberty, to compare these patients with those born appropriate for gestational age as well as with SGA patients not treated with rhGH, and to determine whether or not there are differences between the sexes that may have an impact on AH.
Patients and methods
Study design and patient selection
A single-center longitudinal prospective observational study was conducted. The study was carried out in a tertiary level hospital, involving SGA patients treated with rhGH from October 2003 to April 2015. All the participants met the inclusion criteria for treatment with rhGH in children born SGA established by the European Medicines Agency:
Length and/or weight less than −2 SDS at birth for gestational age, according to the tables of the 2010 Spanish Growth Study.1,2
No catch-up growth at 4 years of life.
At the start of treatment, height less than −2.5 SDS (according to the reference tables) and less than 1 SDS adjusted to the target height.
None of the patients had begun puberty at the start of treatment.
The exclusion criteria were the following:
Noncompliance with auxological inclusion criteria.
Patients with malformation syndromes or genetic alterations.
Patients with uncontrolled chronic or systemic disease.
Treatment suspended before termination for any cause.
The average dose used was 0.035 mg/kg/day.
In total, 96 patients in follow-up were included in the study. Of these, 61 patients who completed treatment and reached AH were included in our analysis (Figure 1). Thirty-five patients were excluded from the study. In 11 patients, treatment was discontinued for the following reasons: voluntary withdrawal (n = 2) (one case by parental decision and another by the patient’s own decision), professional decision due to lack of response to treatment (n = 4), and due to the appearance of adverse effects (n = 5), which are detailed below. Nonresponse was defined as a gain of less than 0.3 SDS after the first year of treatment, as established in other studies. 5 Nevertheless, some publications 10 consider patients to be nonresponders when the growth rate is less than 3 cm/year.

Patients included in the study.
Methods
Auxological data were expressed as age- and sex-adjusted SDS using the references for the Spanish population through AuxoLog® software (Pfizer Endocrine Care), based on data from the 2010 Spanish Growth Study.1,2 Height measurement was performed with a precision measuring meter (Holtain Stadiometer) by trained healthcare personnel. Pubertal staging was determined according to Tanner and Whitehouse. 11 Onset of puberty was defined by the presence of a testicular volume ⩾4 ml in boys and breast stage 2 (S2) in girls. Bone age was assessed based on the Greulich and Pyle atlas. 12
During the study, the following initial variables were recorded: date of birth, sex, gestational age, newborn weight (SDS) and length (SDS), parental height, target height (SDS), and mid-parental height (MPH). MPH was defined as the target height +2.1 cm in relation to the secular trend in growth. Height (cm) and weight (kg) were determined at the start of treatment. The follow-up variables (auxological and biochemical data) included were chronological age, height (cm), body weight (kg), pubertal stage, rhGH dose (mg/kg/day), IGF-1 (insulin-like growth factor 1) (ng/ml), insulinemia (µIU/mL), bone age, and growth velocity (cm/year). The pubertal variables considered were age of pubertal onset, height at pubertal onset (cm), bone age/chronological age ratio at pubertal onset, and TPG (cm). Growth to puberty was calculated as the difference in height at the onset of puberty and height at the onset of treatment in SDS.
AH was defined as the height attained when the growth rate was less than 2 cm/year. AH (SDS) was calculated using adult references (SDS for age 18 years). TPG, expressed in cm, is defined as AH minus height at the onset of puberty. HG, expressed in SDS, was calculated as AH minus height at the start of treatment. In addition, the variable distance to mid-parental height (DMPH) was determined as the distance from the target height with respect to height at the start of treatment in SDS.
Statistical analysis
R Commander Version 2.7-1 graphical user interface was used for the statistical analysis. Descriptive analysis was performed for all variables. Absolute and relative frequencies were presented for qualitative variables, and the main measures of central tendency and dispersion were presented for quantitative variables (mean ± SD).
Parametric tests [Student’s t test or one-way analysis of variance (ANOVA)] were performed to study the differences between quantitative variables in different groups, such as sex. Pearson’s correlation coefficient and Spearman’s rho were used to examine the relationship between quantitative variables. Before the application of the hypothesis tests, the normality of the variables was checked with the Kolmogorov–Smirnov test (n ⩾ 30) and the applicability assumptions of each of the tests used. The significance level used throughout the study was p ⩽ 0.05.
To establish the influence of certain factors on treatment response, a multiple linear regression model was used with AH and total HG as dependent variables. The potential predictors of response were evaluated by receiver operating characteristic (ROC) curve analysis.
Ethical aspects
The research complied with all relevant national regulations and institutional policies and was in accordance with the principles of the Declaration of Helsinki. The study was approved by the Clinical Research Ethics Committee of the Regional University Hospital of Malaga. (Ref: 0788-N-19). Written informed consent was obtained from parents/guardians and children.
Results
A cohort of 61 SGA children (28 boys and 33 girls) who met the established inclusion criteria were prospectively followed up until reaching AH. The initial descriptive variables and comparison between sexes are presented in Table 1 and the developmental variables in Table 2.
Initial descriptive variables.

DMPH, distance to mid-parental height; SGA, small for gestational age.
Data expressed as mean (SD).
Developmental variables in the first year of treatment.

GV, growth velocity.
Data expressed as mean (SD).
Patients started rhGH therapy at a mean age of 6.4 (1.8) years in the boys and 6 (1.8) years in the girls, with a mean height (SDS) of −2.9 (0.6) in the boys and −3.2 (0.7) in the girls, reaching an AH of −1.9 (0.9) SDS in the boys and −1.7 (0.7) SDS in the girls.
From the start to the completion of rhGH therapy, a progressive increase in height was noted. After the first year of treatment, mean height had improved considerably in our patients, as can be seen in the data shown in Table 2. During this period, the greatest accumulated HG during the entire treatment of the patients occurred, in line with the findings described in the literature.
Puberty
Onset of puberty in the SGA boys was at the age of 11.9 (1) years and in the girls at 10.6 (1.3) years. HG during puberty was 22.62 (5.8) cm in the boys and 18.8 (4.5) cm in the girls. In the SGA girls, there were no statistically significant differences with respect to the reference population in terms of the age of onset of puberty or HG during puberty (pubertal growth spurt). For the boys, onset of puberty was at a normal age, but TPG was significantly lower with respect to the reference population (p < 0.01). The TPG in the Spanish population appropriate for gestational age (AGA) is 26.3 cm in boys, while in the case of girls is 20.3 cm. 1
The patients started puberty with a bone age in accordance with their chronological age, a bone age/chronological age ratio of 0.97 (0.12). In both sexes, growth from the start of treatment to the onset of puberty was similar, with an HG (SDS) during this period in the boys of +1.7 (1) and in the girls of +1.7 (0.8), with no significant differences. No patients in the study presented precocious puberty or rapidly progressive puberty. Data for the pubertal variables are provided (Table 3 and Figure 2).
Variables collected during puberty.

Data expressed as mean (SD).
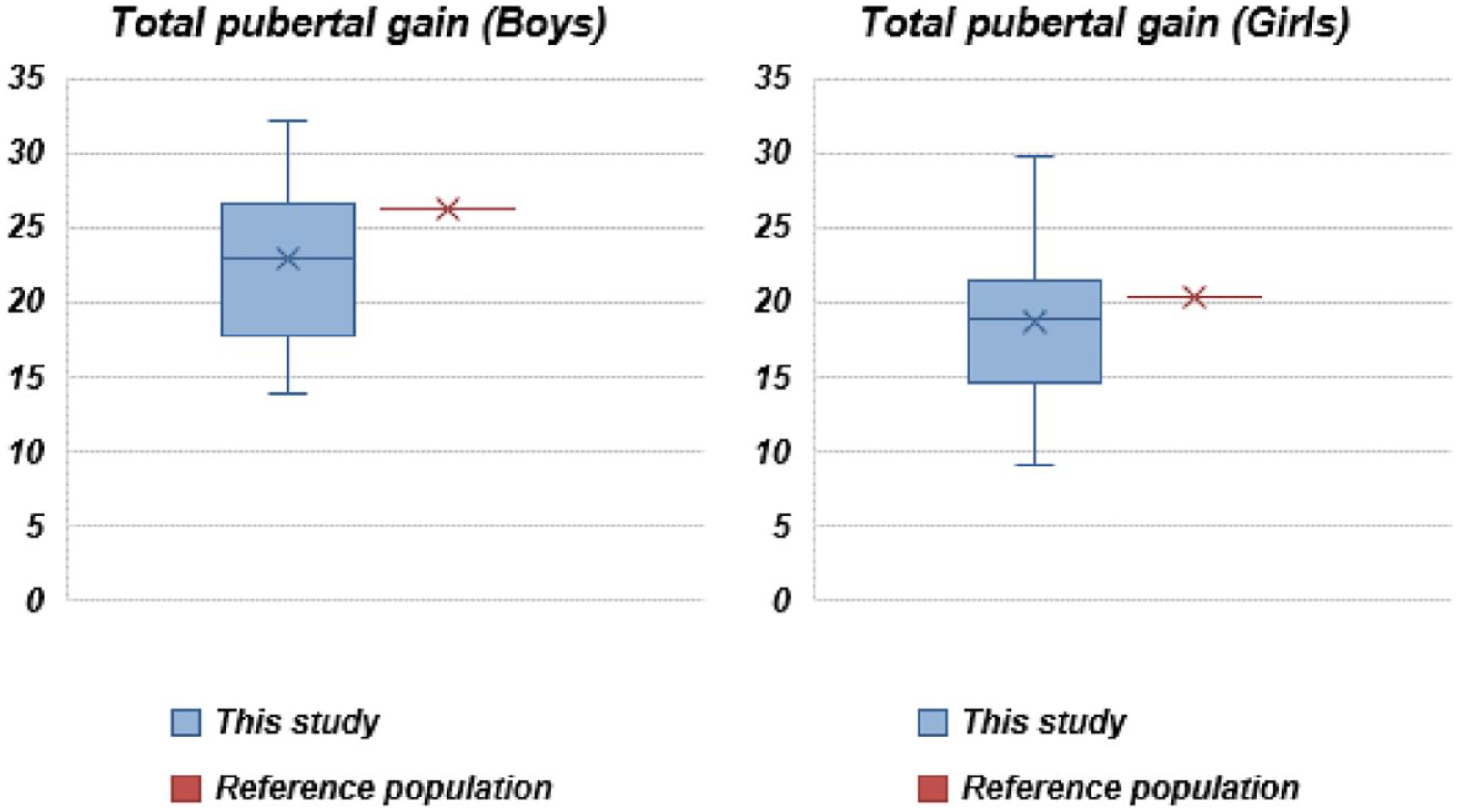
Comparison with the reference population according to Ferrández et al. 2
AH and HG
AH (SDS) was −1.94 (0.94) in boys and −1.72 (0.68) in girls. Seventy-five percent of the girls achieved an AH greater than −2 SDS, while only 53% of the boys were able to exceed this level. In total, 42.6% of the patients reached their target height range: 45.4% of the girls and 37.9% of the boys. There were no differences in AH between the sexes (p > 0.05).
In terms of total HG, we found a significant difference between the sexes, with the boys presenting an HG (SDS) of 0.99 (0.8) versus 1.49 (0.94) in the girls (p = 0.008).
Figures 3 and 4 illustrate the differences in AH between the boys and the girls. Despite a very large HG in SDS at the onset of puberty compared with at the start of treatment, after puberty, there was a reduction in height in SDS, which was more marked in boys.

Evolution of height (SDS) at different time points of rhGH treatment.

Evolution of height (SDS) at different time points of rhGH treatment.
Response to treatment measured in HG correlated positively and significantly with DMPH in the boys (r = 0.041) (p = 0.03) and in the girls (r = 0.54) (p = 0.0001). In addition, a large HG during the first year correlated with total HG again in both sexes: boys (r = 0.43) (p = 0.01) and girls (r = 0.62) (p = 0.001).
Concerning AH, we found that in the boys, there was a positive correlation with length at birth (r = 0.4) (p = 0.035) and height at the start of treatment (r = 0.479) (p = 0.01), and in the girls, there was a positive correlation with height at the onset of puberty (r = 0.4) (p = 0.01).
An analysis of the sensitivity and specificity of HG in the first year as a predictor of good response (Figures 5 and 6) shows that, assuming an HG greater than 1 SDS as a good response to treatment, a gain of 0.69 SDS after the first year of treatment has a specificity of 90% and a sensitivity of 52%. However, considering an HG greater than 1.5 SDS as a good response, we find that a gain of 0.59 SDS after the first year has a specificity of 70% and a sensitivity of 71%. Similarly, a gain of 0.78 SDS after the first year has a specificity of 87% and a sensitivity of 59% for a final gain greater than 1.5 SDS. On examination by sex (Figures 7 and 8), we observe that a first-year gain of 0.61 SDS in the boys has a sensitivity of 71% and a specificity of 72% for a gain greater than 1.5 SDS, and a first-year gain in the girls of 0.64 SDS has a sensitivity of 62% and a specificity of 65% for a final HG greater than 1.5 SDS.

ROC curve predictor > height gain 1 SDS.

ROC curve predictor > height gain 1.5 SDS.

ROC curve predictor > height gain 1.5 SDS (girls).

ROC curve predictor > height gain 1.5 SDS (boys).
Pubertal gain
With respect to pubertal gain, age at start of rhGH was inversely correlated in both the girls (r = −0.37) (p = 0.03) and the boys but without reaching statistical significance (r = −0.17) (p = 0.38). A negative correlation was also observed between age of pubertal onset and TPG in both sexes: boys (r = −0.4) (p = 0.04) and girls (r = −0.6) (p = 0.01).
The results of the statistical analysis are shown in Table 4.
Statistical results of the linear correlation.

DMPH, distance to mid-parental height; GH, growth hormone; NB, newborn; TPG, total pubertal gain.
Those values in bold correspond to the results that have statistical significance.
Insulinemia
Insulinemia at the start of treatment in the boys was 3.9 (3.2) uU/L, while in the girls, it was 5.2 (5.1) uU/L. After 1 year of treatment, there was a stable trend in general, maintaining normal values in both sexes. However, coinciding with puberty, we observed a slight increase in the girls to 5.8 (3.8) uU/L and an increase in the boys to 10.08 (9.6) uU/L. No significant differences were found.
Multiple linear regression model
The variables identified through univariate analysis (p < 0.05) were those included in the multiple regression analysis. In the model reported in detail in Table 5, we identify the factors that explain 42% of the variability in HG expressed in SDS of SGA children treated with rhGH before the onset of puberty. For the prediction of AH gain in SDS, the final model included three variables. The predictors with a positive effect were HG at 1 year and DMPH, whereas male sex had a negative effect.
Predictive model of adult height gain.

DMPH, distance to mid-parental height.
The gain of 1 SDS in the first year after the start of treatment increased the mean AH gain by 0.70 SDS (p = 0.0001) controlling for DMPH and sex. The difference of 1 SDS with the target height at the start of treatment increased the mean AH gain by 0.34 SDS (p = 0.09). The boys showed a lower mean AH gain of −0.26 SDS (p = 0.15).
Adverse effects
The adverse effects possibly related to the treatment were very few and mild, and the treatment was therefore very well tolerated by the patients. Two patients had alterations in carbohydrate metabolism that reverted after discontinuation of treatment. One patient developed Perthes disease at 11 years of age, which led to the discontinuation of treatment. One patient experienced accelerated bone aging. Treatment was therefore discontinued early to avoid possible repercussions on final height. It should be noted that during treatment, one patient developed a giant cell tumor of the mandible that led to discontinuation of treatment. However, we found no relationship between this finding and rhGH treatment.
Discussion
This prospective follow-up study shows the final result of AH in SGA patients treated with rhGH from the prepubertal period, confirming, as is already known, that treatment with rhGH in these patients is effective. The study emphasizes the existence of predictive values for treatment efficacy, as well as the influence of puberty, a critical stage of the growth period during the pediatric age. Among the most important findings of our study, we found that HG in the first year is the most important variable to predict a good response to treatment.
During puberty, there is a loss of SDS in height, due to a lower TPG with respect to the reference population, which is more marked in the boy.
In our study, we detected a shorter AH in boys than in girls; however, we found no significant differences between the sexes. The published studies that differentiate AH by sex have also shown no differences, obtaining results similar to ours, which can be seen in Supplemental Table 66,13–16 and Table 7.17–20 Ranke et al. 17 found an AH of −1.8 SDS (boys) and −1.9 SDS (girls) with no significant differences between sexes, as observed in our study. The data series that differs most from ours is the one of Sánchez Zahonero 15 in which most of the patients who reached AH did not achieve normalization. Nevertheless, they did attain a height SDS that was quite close to that of the shorter parent. The fact that no study has found statistically significant differences is probably due to a small number of patients in relation to the short time that has elapsed since the approval of the treatment. In successive studies, the follow-up that we are carrying out on a greater number of patients will allow us to find more definitive conclusions.
Although we found no differences between the sexes in AH, we did find differences in HG, which is one of the most important aspects of our study. In our multiple regression model, we identified that only three factors explained 42% of the variability in HG: sex, HG in the first year, and DMPH. It was to be expected that HG in the first year would be a determining factor in our predictive model, as this is one of the most described predictors of good response to treatment in the literature. 16 Our ROC curve analysis provided additional prognostic information, since a gain in the first year of 0.6 SDS would allow us to practically ensure normalization of height, reaching AH > −2 SDS. However, as our analysis only explains 42% of the variability and there are other dependent variables, it would not be enough to determine treatment discontinuation in those patients who are not good responders, and further data are needed to be able to advise termination of rhGH treatment.
DMPH was positively associated with HG, which is consistent with other studies.17,21 Specifically in the study carried out with data form KIGS database 20 in which DMPH at the start of rhGH is the most important predictor variable. MPH in both the boys and the girls in our study was within the limits of normal height in the Spanish adult population, with no differences by sex, as in the rest of the recently published national series (national table). Because the girls had a significantly lower height at the start of treatment, they therefore presented a higher DMPH than the boys, which may further influence the difference in HG that we observed in the girls with respect to the boys. However, other studies, such as that of Beisti, 16 found a smaller difference in genetic height as a factor associated with AH gain. The difference between the sexes observed in our study in terms of HG is not described in the literature; in fact, in the Belgian study, 18 a lower AH and HG was observed in girls compared with boys. This last study showed a positive correlation between AH and height at the beginning of treatment, a result similar to that obtained in our sample.
The age at onset of puberty was within the normal range compared with the reference population, as described in most of the literature.19,22 Our data are similar to those of the Spanish reference population, and although we did not find differences with respect to this population, a recent meta-analysis 23 does show that SGA children present an earlier onset of puberty that is more marked in girls.
Pubertal growth in girls was similar to that found in other published series (Supplemental Tables 6 and 7). However, in boys, we saw a decrease in pubertal gain relative to the reference population taking into account a TPG of 22.62 (5.8) cm in our cohort versus 26.2 (4.4) cm in the normal population according to Ferrández et al. 2 Comparing these data with those described in SGA children without rhGH treatment, we observed the same difference in TPG: the data published by Vicens-Calvet et al. 24 reported a TPG of 25.3 cm in boys and 19.6 cm in girls, and the study by Lazar et al. 25 found a TPG of 26.1 (3.6) cm in boys and 18.1 (3.4) cm in girls.
We also observed that our male patients had a progressive decrease in growth, which results in a smaller pubertal gain. This progressive decrease is usually attributed to an early onset of puberty or an exponential increase in bone age, but our patients started puberty at a normal age with a bone age in accordance with their chronological age. Therefore, none of these hypotheses is sufficient to explain this decrease in gain. Recent series, such as that of Campos-Martorel et al., 14 have also found that SGA patients treated with GH have a lower TPG than the reference population, although their decrease in TPG is less than that of our analysis.
One hypothesis to explain the loss of HG during puberty in both boys and girls is a need to increase the dose of rhGH during this period. Some studies26,27 showed that an increase in the dose during the pubertal period resulted in a considerable increase in the final height. The use of gonadotropin-releasing hormone analog in patients with an extremely short stature at the onset of puberty could be beneficial, as they present a higher TPG than in those who were only treated with rhGH.19,28,29
Some studies,30,31 however, argue that the rhGH dose does not play an important role in TPG and that age and height at the onset of puberty, bone age, and DMPH are more important, showing a strong positive correlation between AH and height at the onset of puberty.
In other studies, 19 patients who started treatment after the onset of puberty had greater pubertal gain than those who started rhGH treatment in the prepubertal years, coinciding with what we have seen in our univariate analysis in which pubertal gain was inversely related to age at the start of rhGH treatment.
The safety of rhGH treatment has been well documented in SGA patients10,32,33 as is the case in our study in which, in most cases, rhGH-related side effects were mild and reversible and did not require any type of intervention.
The main limitations of our study are the absence of a matched control group, which would be unethical since rhGH is indicated for this group of patients. Few of the patients have reached their final AH, which limits our ability to draw conclusions. In addition, the method of calculating AH in our patients using references from adult patients (height in SDS for patients aged 18 years) may underestimate AH, as some patients may have grown a few centimeters after the last visit available in the registry, despite having taken into account the last growth velocity assessment in the calculation.
Conclusion
Our predictive model suggests that the first-year gain is the most important factor in determining treatment response, together with DMPH, which could provide data on the assessment of treatment response without completing treatment to the final height. More studies are needed that support these models that allow us to identify early responders and nonresponders to a long and costly treatment.
Based on the results obtained in our work, we can conclude that a lower total HG in boys appears to be associated with a decrease in TPG compared with the reference population, since up to this period, there are no significant differences in HG between both sexes. The mechanism underlying the unique pubertal growth pattern of children born SGA treated with rhGH remains unclear. None of the hypotheses proposed in the published studies has been able to explain the observed differences according to sex. This latter hypothesis should be confirmed in future studies to explain the influence of this loss of HG during puberty in boys, as well as its influence on AH.
Supplemental Material
sj-docx-1-tae-10.1177_20420188221083534 – Supplemental material for Factors influencing height gain in children born small for gestational age treated with recombinant growth hormone: what extent is puberty involved?
Supplemental material, sj-docx-1-tae-10.1177_20420188221083534 for Factors influencing height gain in children born small for gestational age treated with recombinant growth hormone: what extent is puberty involved? by Ramón Arroyo Ruiz, Aránzazu Ballester Pérez, Isabel Leiva-Gea, MªJosé Martínez-Aedo and Juan Pedro López-Siguero in Therapeutic Advances in Endocrinology and Metabolism
Footnotes
Acknowledgements
The authors would like to thank Maria Repice for help with the English language version of this article. Both the first and second authors have had the same work implication in this original article.
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
