Abstract
Women are at increased risk for cardiovascular disease (CVD) compared with men. While traditional risk factors for CVD seem to disproportionately affect women and contribute to this disparity, increased prevalence of CVD at midlife calls into question the contribution of menopause. Given the potential role that declining hormone levels play in this transition, menopause hormone therapies (MHT) have been proposed as a strategy for risk factor reduction. Unfortunately, trials have not consistently shown cardiovascular benefit with use, and several describe significant risks. Notably, the timing of hormone administration seems to play a role in its relative risks and benefits. At present, MHT is not recommended for primary or secondary prevention of CVD. For women who may benefit from the associated vasomotor, genitourinary, and/or bone health properties of MHT, CVD risks should be taken into account prior to administration. Further research is needed to assess routes, dosing, and formulations of MHT in order to elucidate appropriate timing for administration. Here, we aim to review both traditional and sex-specific risk factors contributing to increased CVD risk in women with a focus on menopause, understand cardiovascular effects of MHT through a review of several landmark clinical trials, summarize guidelines for appropriate MHT use, and discuss a comprehensive strategy for reducing CV risk in women.
Introduction
Despite recent improvements in sex and gender research in cardiovascular disease (CVD), disparities still exist. Ischemic heart disease (IHD) in particular remains a leading cause of death in women. 1 The reasons for this are multifactorial and include biological, social, environmental, and economic factors. 2 While men and women share many traditional risk factors for CVD, these alone do not explain the sex-specific increased risk of CVD in women. Additional female-specific risk factors, most notably menopause, contribute significantly. Menopause has been associated with an increased CVD risk in women aged ⩾55 years. 2 The transition to menopause has shown to be associated with negative alterations in the lipid profile, increased susceptibility to weight gain and metabolic syndrome, and both epicardial and paracardial fat deposition.3–6 It represents a vulnerable time for women and an opportunity for consideration of menopause hormone therapy (MHT). 4
In prior decades, MHT was thought to be effective in primary and secondary prevention of CVD. However, large trials demonstrating the risks of MHT have changed clinician and public perception of these strategies. As our understanding of MHT has evolved, so has our knowledge regarding the appropriate timing and patient characteristics for safe administration. Given the efficacy of MHT in reducing the unpleasant vasomotor and genitourinary symptoms associated with menopause, which can severely alter quality of life, understanding the safety and potential cardiovascular (CV) risks and benefits of MHT is important in optimizing the health of our female patients.
Traditional CV risk factors not equal in men and women
Well-established risk factors for CVD include diabetes mellitus (DM), tobacco use, hypertension (HTN), obesity, and lack of physical activity. These risk factors affect men and women differently and portend a higher risk of CVD in women. See Table 1 for a summary.
Traditional CV risk factors and their disproportionate effects on CVD in women compared with men.

CV, cardiovascular; CVD, cardiovascular disease; DM, diabetes mellitus; HTN, hypertension.
Women are disproportionately affected by DM: mortality for diabetic women is an estimated 2.1 million
HTN is another well-established risk factor for CVD and the leading cause of CV mortality worldwide. 11 Women with HTN have a higher population-adjusted CV mortality when compared with men and are less likely to be treated to guideline-directed blood pressure goals. 12 Postmenopausal women are particularly susceptible to this – as estrogen levels decline, so do its protective vasodilatory effects and resulting benefits on blood pressure. 8
Obesity, another traditional CV risk factor, is more prevalent in women than in men. According to the National Health and Nutrition Examination Survey in 2013, among the 37.7% of adults aged 20 years or older who were classified as obese, 35% were men and 40.4% were women. 13 Obesity has independently shown to be associated with increased risk of CVD in women. The Framingham Heart Study, for example, found obesity to increase relative risk of CAD by 64% in women compared with 46% in men.8,12 Additionally, there is evidence to suggest inactivity levels are higher among women than in men, especially with older age. 14 Given observational data that higher levels of activity may be associated with lower rates of chronic diseases such as CVD, this may also be adding to the increased CV risk that women face. 14
CV risk factors unique to women
Female-specific biological factors, in addition to the unique clinical conditions encountered by women, pose additional CV risks. Adverse pregnancy outcomes, for example, have shown to have lasting implications for long-term CV risk and mortality, which persist despite adjustment for traditional CVD risk factors.15,16 Earlier occurrence of preeclampsia during pregnancy is associated with poorer outcomes overall, and its severity can be correlated with the severity of CVD later in life. Gestational DM, defined as a new diagnosis of DM beyond the first trimester of pregnancy, increases risk of type 2 DM later in life by 7-fold, and raises CVD risk significantly more than type 2 DM alone.17,18
Similarly, polycystic ovarian syndrome (PCOS) is an endocrine disorder that typically manifests as infertility and hyperandrogenism in reproductive aged women and also increases risk for CVD. It is the most common endocrine disorder in young women, affecting between 5% and 10% of women worldwide. 19 PCOS patients tend to have insulin secretory defects and insulin resistance, predisposing them to metabolic syndrome, which is known to be associated with CVD. 20 Presence of this diagnosis is associated with multiple comorbidities such as obesity and coronary artery disease. For example, the Coronary Artery Risk Development in Young Adults (CARDIA) study reported a 4-fold increase in coronary artery calcification in PCOS patients when compared with the general community. 21
Systemic autoimmune disorders represent another category of CVD risk factors that disproportionately affect women: females are two to three times more likely to develop rheumatoid arthritis (RA) than men, and 9 times more likely to develop systemic lupus erythematosus (SLE). 22 Systemic inflammation seen in these autoimmune disorders has been linked to atherosclerosis and CMD. Evidence suggests that more active autoimmune disease measured by the frequency of flares heralds a worse CV prognosis.23,24
The age at which a young woman experiences menarche may also play a role in predicting risk for CV events later in life. Interestingly, increased risk is observed in patients who began menstruating either “early” or “late”. 25 In a study of 648 women who underwent coronary angiography for suspected ischemia, the age of reported menarche had a J-shaped relationship to major adverse cardiac events (MACE). For example, the adjusted MACE hazard ratio for menarche at ⩽10 years (early) was 4.53 when compared with women with menarche at age 12 years (median age of menarche). Similarly, women who started menstruating at ⩾15 years (late) had a hazard ratio of 2.58 for MACE. This trend was also seen after adjustment for multiple CV risk factors, including estrogen exposure and inflammatory markers. 25 Similar findings were reported in the United Kingdom (UK) population-based Million Women Study. 26 These studies suggest that both early and late age at menarche predict higher risk for adverse CVD outcomes, though the mechanisms for this remain unclear.
On the other end of a woman’s reproductive life is menopause, which marks an important transition with regard to risk of CVD in women. Below, we describe the effects of menopause on CVD processes, discuss MHT and its role in CVD, and review current guidelines for appropriate use.
Menopause and CVD risk
As early as 1976, Framingham investigators reported a 2.6-fold higher incidence of CV events in age-matched postmenopausal women when compared with premenopausal women. 27 These same data also found that, on average, women develop CVD 7–10 years later in life compared with men. 28 Observational studies since then have also consistently shown that early age at menopause is positively associated with IHD.29–31 As a result, it has been hypothesized that the pronounced risk of IHD in older women is related to the mid-life withdrawal of endogenous sex steroids associated with the menopausal transition. 4
This hypothesis is supported by several observations. First, menopause has shown to be associated with more atherogenic shifts in the lipid profile. After menopause, women are noted to have higher total cholesterol, triglyceride, and low-density lipoprotein cholesterol (LDL-C) levels in addition to reduced high-density lipoprotein cholesterol (HDL-C) levels. 3 In the Study of Women Across the Nation, LDL-C and apolipoprotein B in particular, when compared with other common CV risk factors, were the only two factors to change in association with menopause and not just age. 32 Second, studies have revealed that the weight gain experienced in midlife, specifically increased fat mass, loss of skeletal mass, and increased waist circumference, cannot be explained by increasing age alone but by ovarian aging associated with the last menstrual period. 6 The menopause transition has shown to contribute to the development of metabolic syndrome by impairing glucose metabolism in addition to causing negative effects on blood pressure, weight gain, and central abdominal obesity. Third, menopause is associated with greater percentage of both epicardial and paracardial adipose tissue, two emerging risk factors for IHD.4,5
Systemic effects of estrogen
Estrogens regulate a variety of systemic factors – they can alter serum lipid concentrations, coagulation and fibrinolytic systems, antioxidant systems, and the production of vasoactive molecules such as nitric oxide and prostaglandins. 33 Estrogens can also have direct effects on vascular cells and tissues, enacted through both “genomic” and “non-genomic” pathways. The genomic pathway employs ligand-activated estrogen receptors located in the cell nucleus that are responsible for regulating gene transcription and expression in response to hormone binding. Non-genomic effects are mediated by estrogen receptors localized to cell membrane signaling domains. While the genomic mechanism is thought to be responsible for estrogen’s longer-term effects, the non-genomic pathway can explain estrogen’s more rapid effects, such as arterial vasodilation in response to estrogen administration. This mechanism is thought to play a role in inhibiting progression of atherosclerosis. 33
Estrogen’s effects on the vasculature can depend partly on the extent to which atherosclerosis has been established in a given vessel. Estrogen’s potential anti-atherosclerotic effects may be less robust in diseased arteries. Not only has existing atherosclerosis shown to result in diminished estrogen receptor expression, but the effects of estrogen itself may have different consequences depending on the state of the vessel and its endothelium. Studies in several animal subjects have shown that anti-atherosclerotic effects of exogenous estrogens are most apparent when minimal underlying atherosclerosis is present at the time of therapy initiation.34,35
Menopause hormone therapy
Based on the above, it is probable that decline in endogenous sex hormone levels play a part in the increased CV risk women face with age. However, only a few longitudinal studies have gone so far as to study associations between circulating sex hormone, sex hormone-binding globulin levels, and CVD, with varying results.36,37 Moreover, as both menopause and biological aging are interrelated in time, the extent to which one contributes to age-related CV risk over the other is difficult to delineate. However, the question of a possible CV benefit from MHT has been promoted for decades, as noted in several observational studies such as the Nurse’s Health Study, which found postmenopausal hormone use to decrease risk for major coronary events in women without previous CVD. 38 Since the 1990s, several placebo-controlled trials studying various MHT have taken place, looking at both primary and secondary prevention outcomes.
The 1998 Heart and Estrogen/Progestin Replacement Study (HERS) was the first published secondary prevention trial to evaluate hormone therapy in women with known coronary disease.
39
Participants were randomly assigned to receive a combination of conjugated estrogens (CE) and medroxyprogesterone acetate (MPA). The primary outcome was nonfatal MI and IHD death with no significant decreases in rates of these events in women assigned to the hormone group compared with those assigned to the placebo group after 4.1 years of follow up.
The Women’s Health Initiative (WHI) also studied CE plus MPA in postmenopausal women aged 50–79, and was actually stopped early due to a 24% increased risk of coronary heart disease in this group compared with placebo. 42 This group also had increased risk of stroke, pulmonary embolism, and breast cancer. These effects were most apparent at 1 year, and with the exception of breast cancer risk, dissipated after stopping intervention. 43 When results were examined by time since menopause, risk of IHD was neutral for women <10 years from menopause and increased for women >20 years from menopause.
These observations, in conjunction with observed systemic effects of estrogen described above, led to the development of the timing hypothesis: the idea that beneficial effects of MHT occur only when therapy is initiated early in the menopausal period, prior to the development of advanced atherosclerotic disease. This was theorized to be based on underlying characteristics of damaged vessel wall and presumed negatively altered vascular remodeling in atherosclerotic vessels. The Kronos Early Estrogen Prevention Study (KEEPS) evaluated the effectiveness of a combined estrogen/progestin in preventing progression of carotid intima-media thickness (CIMT) or coronary artery calcium (CAC) in women who are within 36 months of their final menstrual period. 44 In healthy women, within 36 months of menopause, therapy did not prove to slow the progression of CIMT or affect CAC. Treatment did improve mood, sleep, vasomotor symptoms, and bone density without an increase in adverse events, suggesting that there may be noncardiac benefits and safety in using MHT in recently postmenopausal women. 44
ELITE (Early
The SMART (Selective Estrogens, Menopause, and Response to Therapy) trials have gone a step further in evaluating effects of conjugated estrogens/bazidoxifene [a third generation selective estrogen receptor modulator (SERM)] on postmenopausal women. 46 At 12 months, this combination was associated with significant improvements in total cholesterol, LDL-C, and HDL-C levels when compared with placebo. Triglyceride levels, however, were significantly increased at both doses. 46
In contrast to the HERS and WHI, the KEEPS, ELITE, and SMART trials support the timing hypothesis and suggest no significant harm with possible CVD benefits when initiating MHT at the time of menopause. More recently, however, a 2017 Cochrane review of MHT including 22 published studies, mostly in menopausal women over 60, found that women taking continuous combination MHT had increased risk of IHD, VTE, stroke, breast cancer, biliary disease, and death from lung cancer. 47 Estrogen-only MHT was associated with decreased risk of breast cancer and bone fracture with no increased risk of IHD. Women with known CVD on combined MHT had an increased risk of DVT. 47 These results influenced current guidelines heavily, as we review below.
Benefits of MHT and current guidelines
MHT is effective in reducing vasomotor and genitourinary symptoms associated with menopause, promoting bone health, and, in many cases, improving quality of life. A Cochrane review including 24 randomized controlled trials studying MHT administration for vasomotor symptoms demonstrated a reduction in weekly hot flashes by 75% and an 87% decrease in severity of hot flashes, demonstrating it to be an effective therapy for this difficult-to-manage symptom of menopause, which on its own is associated with increased risk of CVD.48,49 MHT is currently approved by the United States (US) Food and Drug Administration (FDA) for treatment of vasomotor and genitourinary symptoms, prevention of osteoporosis, and as estrogen replacement in the setting of surgical menopause, hypogonadism, or premature ovarian insufficiency. 50 Options for MHT include conjugated equine or synthetic estrogens, micronized 17β-estradiol, or ethinyl estradiol. Progestogens are indicated for endometrial protection in patients with a uterus, and include MPA, norethindrone acetate, and progesterone. 51 Additionally, bazedoxifene, a SERM, can be combined with CE to form a tissue-selective estrogen complex, providing endometrial protection without the use of a progestogen. 52
Currently, MHT is not recommended for primary or secondary prevention of CVD and is not recommended for women with a high CVD risk.51,53,54 In women who may benefit from the vasomotor, genitourinary, and bone health properties of MHT, CV risk should be assessed and optimized prior to initiation. A 2020

CV risk factors women at the time of menopause stratified according to suitability for MHT use.
Optimizing CVD risk in women prior to initiating MHT
Prior to consideration of MHT, it should be emphasized that all women need aggressive lifetime CVD risk factor identification and reduction. 57 To this end, guidelines exist specifically for prevention of CVD in women – an effort that has grown to classify women systematically according to their risk and implement recommendations at all levels. 53 General lifestyle recommendations include advising women to stop cigarette smoking, participate in at least 150 min/week of moderate exercise, consume a diet rich in fruits and vegetables with whole-grain foods and fish at least twice a week, and maintain a body mass index (BMI) <25 or waist size <35 inches. Interventions for major medical risk factors such as HTN, DM, and dyslipidemia have also been delineated. Women are advised to maintain an optimal blood pressure of <120/80 mmHg and use appropriate pharmacotherapy when above 140/90 mmHg. With regard to lipids, women are recommended to have a goal LDL-C of <100 mg/dl, HDL-C > 50 mg/dl, triglycerides <150 mg/dl, and non-HDL-C < 130 mg/dl. LDL-C lowering drug therapy is recommended in conjunction with lifestyle modification to achieve an LDL-C < 100 mg/dl in most women and <70 mg/dl in very high-risk women. In women with DM, lifestyle and pharmacotherapy is recommended to achieve a hemoglobin A1c < 7%. 53
While MHT is not recommended for primary or secondary prevention of CVD, pre-existing CVD risk should be assessed in all women considering these therapies prior to initiation. The 2013 ACC/AHA Pooled Cohort Risk Equation for ASCVD (atherosclerotic cardiovascular disease) is one tool that has been recommended. This tool estimates 10-year primary risk of ASCVD in patients without pre-existing CVD who are between 40 and 79 years of age. It takes into account race, lipids, as well as history of HTN, DM, and tobacco use. A score of under 5% is considered low risk, 5–7.4% moderate risk, and 7.5% or greater high risk. 58 Those considered high risk with this calculator may seriously consider avoiding MHT while those who are low risk may be appropriate candidates for these therapies. 50
Women with moderate-risk scores may benefit from more thorough assessment using ASCVD “risk-enhancing factors” to guide decision making. Risk enhancing factors as defined in the 2019 ACC/AHA guideline on the primary prevention of CVD include family history of premature ASCVD, primary hypercholesterolemia, metabolic syndrome, chronic kidney disease, chronic inflammatory conditions, history of premature menopause, history of pregnancy associated conditions such as preeclampsia, high risk race/ethnicity, persistently elevated triglycerides, high lipoprotein (a), high apolipoprotein B, and ankle-brachial index <0.9. 59 The NAMS has created an app, MenoPro, which utilizes an algorithm accounting for severity of menopausal symptoms along with family history of breast cancer and CVD in conjunction with the ASCVD risk calculator to propose appropriate hormonal and non-hormonal therapeutic options for each individual. 60 It seeks to streamline this multi-faceted decision-making process for both clinicians and patients alike. Figure 2 illustrates the steps involved in ideal ASCVD risk assessment for women at menopause.
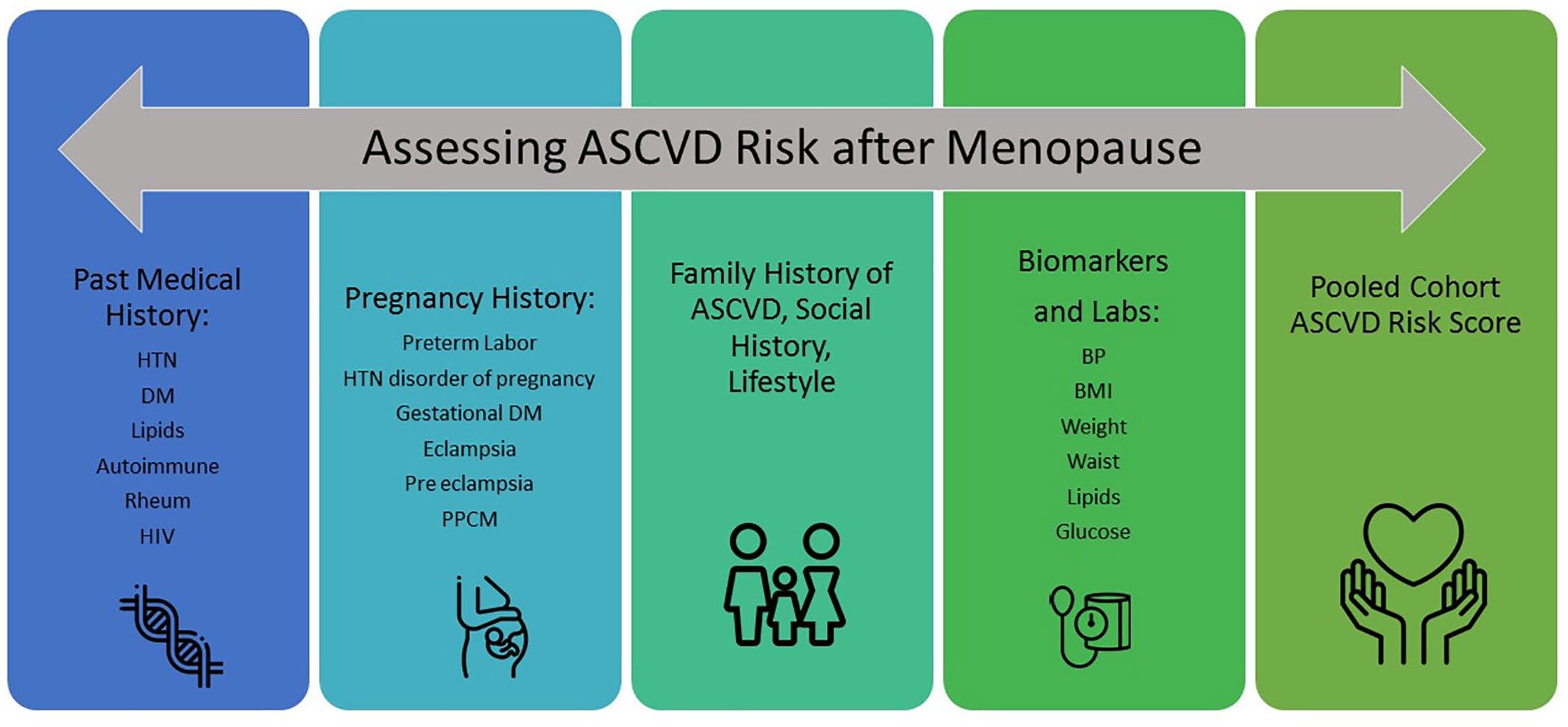
Ideal steps for assessing ASCVD risk in women at menopause.
Conclusion
CV risk in women increases at the time of menopause, likely related to a combination of aging and the menopausal transition. Proper CVD risk assessment is imperative for improving long term CVD outcomes, guiding risk reduction therapy, and determining safety of MHT if needed. A thorough history of adverse pregnancy outcomes and complications is required in all perimenopausal women. Assessment of family history of CVD in addition to any personal history of autoimmune or rheumatologic disorders and human immunodeficiency virus (HIV) is essential. All traditional risk factors should be assessed as well as current lipid cholesterol levels, blood glucose levels, and blood pressure. All women should receive education on ideal body weight, a plant-based heart healthy diet, and optimal exercise routines.
MHT is not recommended for primary or secondary prevention of CVD or in women with known ASCVD or high risk for CVD events. In select populations of women who may benefit from its vasomotor, genitourinary, and bone health effects, women at low CVD risk may be prescribed MHT safely until age 65. The principles underlying safe use of MHT include using low dose MHT for the shortest possible duration and employing transdermal, SERM, and topical formulations where appropriate, often as an initial strategy. 50 Lastly, it is imperative that both patients and providers are aware of the appropriate indications for MHT and individual risks, especially from a CV standpoint. Several calculators are available to further risk-stratify these patients and enable safe shared-decision making, such as the ASCVD pooled-cohort risk calculator and the NAMS MenoPro app. 60 Quality of life for postmenopausal women is an important consideration, and in addition to early risk assessment and CVD risk reduction, decisions regarding MHT should be made individually and reassessed over time.
Footnotes
Author contribution(s)
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
