Abstract
Background:
Amiodarone treatment is effective against various types of arrhythmias but is associated with adverse effects affecting, among other organs, thyroid function. Amiodarone-induced thyroid dysfunction was not thoroughly evaluated in children as it was in adults, yet this affection may lead to irreversible neurodevelopmental complications. Our study aimed to define the incidence and risk factors of amiodarone-induced thyroid dysfunction in children.
Methods:
The study was designed as an observational study with a retrospective clinical series of 152 children treated by amiodarone in the Pediatric Cardiology Unit of our center from 1990 to 2019. All patients were divided into three groups according to their thyroid status: euthyroid, AIH (amiodarone-induced hypothyroidism) or AIT (amiodarone-induced thyrotoxicosis). Patients from these three groups were compared in terms of key clinical and therapeutic features.
Results:
Amiodarone-induced thyroid dysfunction was present in 23% of patients. AIT (5.3%) was three times less common than AIH (17.7%), and its occurrence increased with older age (p < 0.05), treatment dosage (p < 0.05), treatment duration (p < 0.05) and the number of loading doses administered (p < 0.05). There were no distinctive clinical features between euthyroid and AIH groups. A multivariable prediction model of AIT was built, with a yield of 66.7% as positive predictive value and 96.7% as negative predictive value.
Conclusion:
We observed that one in five children developed amiodarone-induced thyroid dysfunction. Special attention is required for older children with a high dosage and long-term therapy and who received a large number of loading doses, since these children are at risk to develop AIT, which is more delicate to manage than AIH.
Keywords
Introduction
Amiodarone is a class III antiarrhythmic agent according to the Vaughan Williams classification 1 that prolongs the duration of the action potential of cardiomyocytes by blocking ions channels. While this agent is preferred over other antiarrhythmics for the management of supraventricular and ventricular tachycardia because of its efficacy, 2 it triggers a wide array of adverse effects that affects, among other organs, thyroid.
Two properties of the amiodarone molecule explain its effects on the thyroid. First, each 200 mg pill of amiodarone releases 6 mg of free iodine into the circulation (compared with the 0.15 mg that is needed daily 2 ) and second, amiodarone bears a significant structural resemblance to the thyroid hormones triiodothyronine (T3) and thyroxine (T4). 3 Due to these properties, 20% of adults under therapy develop thyroid disorders, as amiodarone-induced hypothyroidism (AIH) or amiodarone-induced thyrotoxicosis (AIT). 4 AIH may induce the need for substitution therapy whereas AIT is more delicate to treat since it may favor tachyarrhythmias and lead to amiodarone discontinuation. 5
Although widely used in children with arrhythmia, amiodarone and its effects on the thyroid were not thoroughly evaluated in children as they were in adults, yet this treatment may be required in early infancy and lead to irreversible AIH- or AIT-induced neurodevelopmental complications when administered early in infancy. Since the mechanisms of arrhythmias in children may differ from those in adults, and because the metabolism of amiodarone may vary within various age groups, comparison of amiodarone effectiveness and side effects in children and adults may not be warranted. Furthermore, the indications of amiodarone therapy differ in adults, with ischemic heart disease, and in children, for whom these are mostly represented by arrhythmias occurring during the post-operative period of surgery for congenital heart defects.
Studies that evaluated the adverse effects of amiodarone on thyroid function in children showed conflicting results.6–9 Three retrospective studies analysed the effectiveness of amiodarone in children and all its adverse effects. They showed that AIH and AIT are less common in children than in adults: changes in thyroid hormone levels occurred in less than 10% of patients and these developed symptoms in less than 5% of cases.6–8 These studies also reported a higher prevalence of AIT than AIH. The first study, from Coumel and Fidelle, 6 retrospectively analyzed 135 children: two patients developed AIT and one AIH. Garson et al. 7 followed 39 children: two children developed biochemical evidence of AIT, but none developed symptoms. Guccione et al. 8 focused their interest on the long-term (up for >18 months) follow-up of amiodarone therapy of 95 children. The incidence of thyroid dysfunction was 9% and no patient was symptomatic.
Conversely, the prospective study of Costigan et al. 9 showed that the incidence of AIT and AIH in children and in adults was similar, and that AIH was more common than AIT. In this study, thyroid function of 15 children was systematically evaluated before and during treatment with amiodarone. Three patients developed AIH (clinical and biochemical evidence) and two needed a substitution therapy. One patient developed biological AIT but was asymptomatic.
Much more recently, Barett et al. 10 published the results of their retrospective study: in the 190 studied children, 31% developed AIH (among them, 17.3% were “subclinical AIH”, defined by thyroid-stimulating hormone (TSH) >5 and <10 mIU/l) and 2.1% developed AIT. These results suggest that amiodarone-induced thyroid dysfunction might be much more common than previous studies suggested.
Our work aimed to define the incidence of AIH and AIT during amiodarone therapy in children with arrhythmia and to specify the features of patients who developed thyroid dysfunction, with the purpose of developing a multivariable prediction model of this condition.
Material and methods
The THYRAMIO study was designed as a retrospective study with a clinical series of 232 children treated by amiodarone in the Pediatric Cardiology Unit of Cliniques universitaires Saint-Luc (tertiary health care center) from 1990 to 2019, with an average follow-up of 8 years. For each patient, the following data were collected: age, sex, SD scores for body mass index (BMI) using Belgian Flemish reference charts, 11 history of congenital heart disease, cardiac surgery, thyroid disease, indication of therapy, modality of treatment (cumulative amiodarone dose, duration of treatment and number of loading doses) and presence or absence of thyroid disease after amiodarone therapy (thyroid function, symptoms, treatment of thyroid dysfunction), confirmed by a medical diagnosis mentioned in the patient records.
All patients were divided into three groups along their thyroid status during the medical follow-up: euthyroid, AIH or AIT. We did not rely on raw blood test results to assess the thyroid function status but, rather, on the clinical diagnosis as reported in the medical records, because of some missing data about the reference values used for interpretation of laboratory results. Indeed, thyroid hormone assay units and reference values have changed between 1990 and 2019. When all units were converted in actual reference units [mU/l for TSH and pmol/l for free (F)T3 and free (F)T4], the extrapolated diagnosis differed from the diagnosis reported in the medical records. Biologically, euthyroidism was defined as normal thyroid function tests (serum TSH and FT4 within normal ranges). Hypothyroidism (in AIH) was defined when serum TSH levels were above normal range for age and sex, and when serum FT4 levels were either normal (compensated hypothyroidism) or below normal range (overt hypothyroidism). Hyperthyroidism (in AIT) was defined when serum TSH levels were suppressed (below normal range) and when serum FT4 and/or FT3 levels were above normal range or close to the upper limit of normal range. Our data collection referred only to type 2 AIT (i.e. excess of thyroid hormone secretion caused by an amiodarone-induced thyroiditis without underlying thyroid disease). Non-thyroid side effects attributed to amiodarone treatment were not evaluated.
Data analysis
Data were analyzed using of IBM® SPSS® Statistics 26, JMP® and Graphpad® softwares. Discrete variables are reported as numbers and proportion. Patients with normal thyroid function, AIH and AIT were compared using chi-square test. Continuous variables are reported as means with standard deviation (±SD) if normally distributed or as median with interquartile range (IQR) otherwise. Groups were compared using analysis of variance (ANOVA) with F-test if normally distributed or ANOVA with Kruskal–Wallis test if not normally distributed. Variables with a p-value < 0.10 were evaluated with univariate analyses. All significant variables in univariate analyses (p < 0.05) were entered into a multivariate logistic regression. Results are expressed as odds ratio with 95% confidence intervals. Receiver operating characteristic curve and confusion matrix were performed for the multivariate model.
Results
Criteria for inclusion and exclusion of patients
In our THYRAMIO study, criteria for inclusion were to be treated with amiodarone for one or more doses for any indication (see below) in the Pediatric Cardiology Unit of our center, to be younger than 18 years and to have been screened for thyroid function (with medical interpretation of data mentioned in medical records), regardless of follow-up duration. Forty-three patients were excluded from the study because they were young adults (>18 years) at the onset of amiodarone therapy. Thirty-seven patients were excluded because of incomplete medical records (thyroid function not assessed or not reported). We included 152 patients in the study (Figure 1).
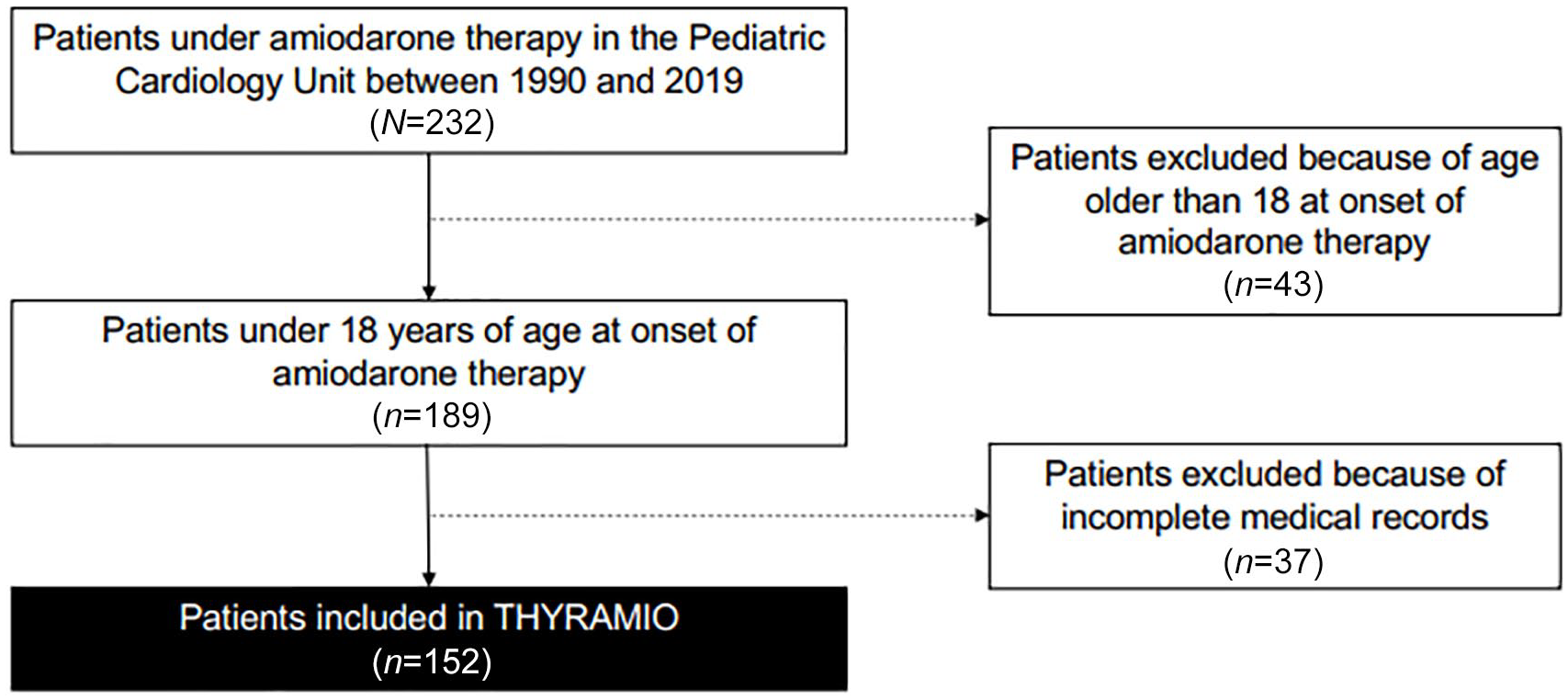
THYRAMIO study flow diagram.
Patient characteristics
Between 1990 and 2019, 152 eligible children (median age: 0.27 years; IQR: 0.03–2.32) underwent amiodarone therapy in our center (Table 1). The youngest child received a first dose of amiodarone shortly after birth and the oldest child at 17 years and 4 months of age. Fifty-five percent of patients suffered from congenital heart defects. The main indication of amiodarone therapy was de novo (i.e. not post-surgery) supraventricular tachycardia (64%). Twenty-seven percent of patients received amiodarone for arrhythmias occurring post-operatively (all but one in the context of congenital heart defect, the remaining patient underwent surgery for an endocarditis). Other reported indications were ventricular tachycardia (5.3%) or combined supraventricular and ventricular tachycardias (2%). Four cases (4/152, 2.6%) were classified in the “Other” category: two patients with a dilated cardiomyopathy and a high risk of arrhythmia, one with a restrictive cardiomyopathy and one who presented a syncope triggered by an undefined tachyarrhythmia.
Baseline characteristics and treatment features of the study cohort.

AIH, amiodarone-induced hypothyroidism; AIT, amiodarone-induced thyrotoxicosis; BMI, body mass index; IQR, interquartile range; SDS, standard deviation score; VST, supraventricular tachycardia; VT, ventricular tachycardia.
The median administered cumulative dose of amiodarone was 28.45 g/m2 (IQR: 14.59–63.25), yet most (42%) patients received a unique loading dose. Chronic treatments lasted 124.5 days (IQR: 57.75–304.00) on median average.
Prevalence of thyroid dysfunction
In our study, 35 patients (35/152; 23%) presented thyroid dysfunction after amiodarone therapy. Twenty-seven patients developed AIH (27/152; 17.7%) and eight patients developed AIT (8/152; 5.3%). One hundred and seventeen patients (117/152; 77%) remained euthyroid. Thyroid hormone assays were graphically displayed when concordant with the clinical diagnosis reported in the medical records (see Material and Methods) (Figure 2). The median duration between the initiation of amiodarone therapy and thyroid dysfunction was 22 days (IQR: 6–383 days): 14 days for AIH (IQR: 5–32) and 2 years for AIT (IQR: 302–958). Four patients had thyroid dysfunction before the onset of treatment. Only three patients (3/27; 11%) with AIH experienced symptoms (two cases of constipation and one case of suspected pubertal delay). Seventeen cases (17/27; 63%) required L-thyroxine replacement therapy (mean dose: 3.7 µg/kg; min 0.8 µg/kg, max 11.3 µg/kg). Three patients (3/8; 38%) with AIT presented a combination of the following symptoms: fatigue, difficulty concentrating, shivers and weight loss, dyspnea, palpitations, flushing, tremors of the hands, insomnia, tremors, palpitations, dysphagia and cervical discomfort. One of those three patients was 16 years old and benefited from propylthiouracil therapy. Two other cases required cessation of amiodarone treatment. Ten cases (10/27; 37%) of AIH were documented by thyroid ultrasound. Abnormal results occurred in only one case, with mildly heterogenous thyroid. Thyroid ultrasound was performed for three cases (3/8; 37%) of AIT and showed either mildly heterogenous thyroid or slight increase in the thyroid volume. Thyroid isotope (I123) scan was performed for one case of AIT and showed normal results. The average clinical follow-up (between the initiation of amiodarone therapy and the last clinic visit) was 8 years (IQR: 3.0–12.8 years).
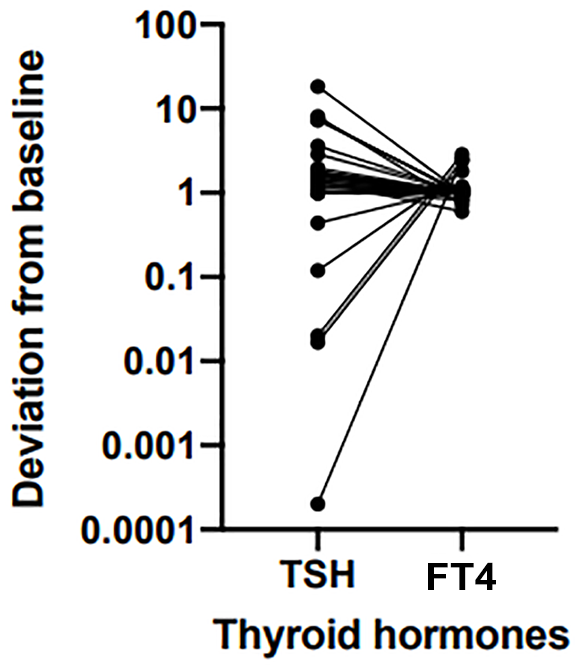
Thyroid function tests in patients with thyroid dysfunction.
Comparison between euthyroid patients, AIH and AIT
Four features of the amiodarone treatment showed a positive correlation with the occurrence of AIT: age (p < 0.01), cumulative dose (p < 0.01), treatment duration (p < 0.01) and number of loading doses (p < 0.01). Comparison between the three patient groups is described in Table 2 and Figure 3 (a–d). Relatively to the euthyroid and the AIH groups, patients within the AIT group were older (respectively 6.6 ± 2.0 years versus 1.0 ± 0.4 versus 2.62 ± 0.8 years), received a higher cumulative dose (285.4 g/m2 versus 24.5 g/m2 versus 48.1 g/m2), underwent a longer treatment (980 days versus 109 days versus 114 days) and received a larger number of loading doses than other groups (6 versus 1 versus 1.5). Characteristics of gender, BMI, treatment indication, congenital heart disease and previous thyroid disease did not differ in the three groups. Also, there were no distinctive clinical features among euthyroid and AIH groups.
Characteristics and treatment features: comparison between euthyroid, AIH and AIT groups.

Categorical variables were analyzed using chi-square test. Continuous variables were analyzed using analysis of variance if normally distributed [reported as mean with standard deviation (±SD)] or Kruskal–Wallis test if not [reported as median with interquartile range (IQR)].
AIH, amiodarone-induced hypothyroidism; AIT, amiodarone-induced thyrotoxicosis; BMI, body mass index; SDS, standard deviation score; VST, supraventricular tachycardia; VT, ventricular tachycardia.
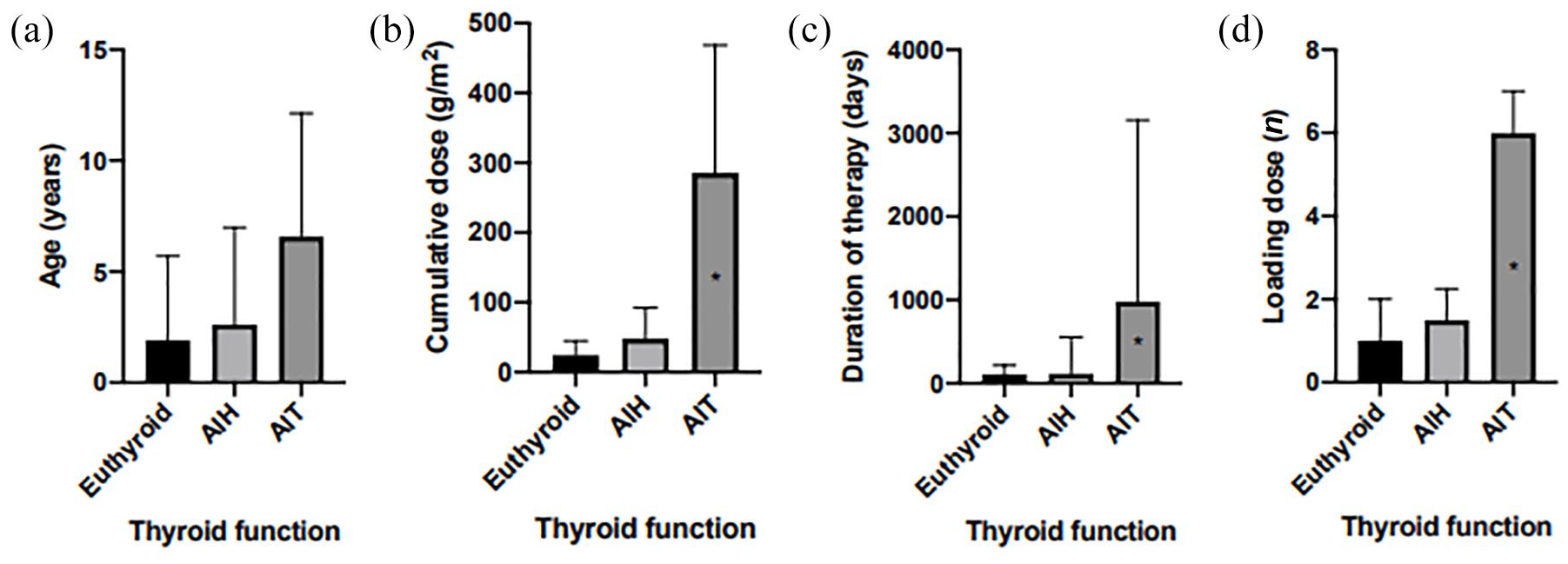
Impact of age, cumulative dose, duration of therapy and loading doses on thyroid function.
A multivariate regression model was built based on these four treatment features (i.e. cumulative dose, treatment duration, number of loading doses and age; Table 3). It provides an area under curve of 97.6%. Our equation was as follows: (P (AIT))/[1−p (AIT)] = e^(−7.142 + 0.191 × age + 0.027 × cumulative dose + 0.371 × loading dose - 0.002 × treatment duration). This equation was extracted from multivariate logistic regression based on variables which were significant in univariate analyses (p < 0.05) (i.e. cumulative dose, treatment duration, number of loading doses and age). It assesses the probability to develop an AIT after amiodarone therapy. The Euler number is represented by “e”, the age is expressed in years, the cumulative dose is expressed in g/m2, the loading dose is expressed by the number of times a loading dose was administered and the duration of therapy is expressed in days. The equation will give a predicted probability of the binary outcome AIT that can be turned into a prediction by setting a threshold (typically 0.5). If the predicted probability is higher than threshold, the predicted outcome is AIT; if it is smaller, the predicted outcome is no AIT (i.e. euthyroid or AIH). In our clinical series, this equation predicted the development of AIT in our clinical series with a positive predictive value of 66.7% and a negative predictive value of 96.7% for a threshold probability of 0.5.
Predictors for amiodarone-induced thyrotoxicosis within a univariate model.

Model was constructed using nominal logistic regression.
CI, confidence interval; OR, odds ratio.
Discussion
In our study of 152 cases, we observed that the prevalence of amiodarone-induced thyroid dysfunction was 23%, which was in line with previous pediatric and adult studies.4,9 AIT in children was three times less common than AIH, which differs from observations in adults, 4 yet we showed that AIT is more common with older age. We also found that AIT increases with treatment dosage, treatment duration and the number of loading doses administered.
If AIT increases with high dosage, long-term treatment and loading doses, we can formulate the hypothesis that AIT increases with the drug blood level. Most AIT are indeed of type 2, that is, caused by a destructive inflammatory thyroiditis (type 1 is less common, especially in children, and is caused by latent disease).5,12 Amiodarone has pro-apoptotic effects and direct cytotoxic effects on thyroid follicular cells, corroborated by the iodine load released by the drug. 5 Histopathological studies showed disruption of thyroid structure including follicular damage, a reduced number of mitochondria, an increased number of lysosomes and dilatation of endoplasmic reticulum. 5 In our hypothesis, the higher the drug blood level is, the most severe the cytotoxicity will be and most frequently AIT will develop.
The impact of age in development of AIT can also be explained by the drug level. Studies suggested that amiodarone metabolization is faster in children than in adults, with the following arguments: there is a trend towards an increase in mean serum concentration with age; 13 the half-life of amiodarone was calculated in children to be between 6.9 and 11.4 days (compared with 100 days in adults); 13 children have less fat mass than adults, yet amiodarone concentrates in the adipose tissue 10–30 times more than in plasma, 6 and amiodarone treatment efficacy after cessation lasts less than a few weeks, as opposed to several months in adults. 12 Therefore, the drug concentration increases with age and so may the cytotoxicity. As a result, AIT is more common with age.
Our predictive model has a poor positive predictive value but a strong negative predictive value. It could still be useful for clinicians since AIT may be difficult to treat: it may even reveal counterproductivity by stimulating arrhythmias 14 and lead to amiodarone discontinuation. If our model yields a non-zero probability, practitioners should pay additional attention to the thyroid follow-up during amiodarone therapy. It is notable that the model is applicable to our clinical series but requires validation in new patients.
Our study is a retrospective study of patient records followed in the pediatric cardiology department at CUSL in Brussels. The study methodology may lead to some imprecisions. First of all, the single-site setting represents a recruitment bias. This is nevertheless limited by the significant referencing of CUSL (tertiary center) in Belgium and within the Louvain Health Network. Also, the retrospective study design produces limitations in the temporality of thyroid function testing, as such: for 87% of patients, there was no bioassay of thyroid parameters before the start of treatment with amiodarone. Finally, the iodine status of the patients was not evaluated, apart from it impacts both the development of dysthyroidism and also the type of dysthyroidism (TIA or AIH) developed.
To improve the external validity of our results and to develop a stronger prediction model, we recognize the obvious need to prospectively collect clinical data, at regular intervals, and to assess thyroid function before amiodarone initiation. Underlying thyroid diseases as Hashimoto thyroiditis or Graves’ disease are rare but not insignificant (respectively 1.2% of prevalence and 0.8/100,000 of incidence), and it is important to distinguish them from amiodarone-induced thyroid dysfunctions. 15 The important issue of the thyroid dysfunction reversibility after amiodarone cessation also needs to be assessed in further studies.
In conclusion, our study established the incidence of amiodarone-induced thyroid dysfunction in a large cohort of children and adolescents and described the characteristics of patients with normal thyroid function, AIH and AIT. In addition, we proposed a predictive model to assess the probability to develop AIT based on age, dosage, duration and number of loading doses of amiodarone. A prospective study is now required to validate our results.
Footnotes
Acknowledgements
The authors would like to thank Jean-Pierre Montenez and Keltoum Roussaky for proofreading of the manuscript.
Author contributions
S.M., S.M. and P.A.L. researched data, analyzed data and wrote the manuscript; L.D. and A.R. analyzed data, and reviewed/edited the manuscript.
Availability of data
The datasets generated during and/or analyzed during the current study are not publicly available but are available from the corresponding author on reasonable request.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethics statement
The research was conducted ethically in accordance with the World Medical Association Declaration of Helsinki. The local ethical committee (Comité d’Ethique Hospitalo-Facultaire Saint-Luc-UCLouvain) approved the study protocol (reference: 2018/14MAR/112). The study was designed as a retrospective study and therefore the ethics committee waived the need for informed consent.
