Abstract
Background:
Implantation is initiated when the blastocyst attaches to the endometrium during the peri-implantation period, and appropriate neovascularization is a prerequisite for the success of the subsequent process. The role of the renin–angiotensin system (RAS) in regulation of blood pressure and hydro-electrolyte balance has long been recognized, while its role in the peri-implantation endometrium remains unclear. This manuscript discusses endometrial RAS and its possible pathways in regulating endometrial angiogenesis and its influence on subsequent pregnancy outcomes.
Methods:
A comprehensive search of electronic databases was carried out to identify relevant published articles, and a literature review was then performed. Using immunohistochemistry, we also performed a pilot study to examine expression of angiotensin II receptors, including angiotensin II type 1 (AT1) receptor (AT1-R) and angiotensin II type 2 (AT2) receptor (AT2-R) in the human endometrium around the time of implantation.
Results:
The results of the pilot study showed expression of AT1-R and AT2-R in all endometrial compartments (luminal epithelium, glandular epithelium, stroma cells, and blood vessels), and altered expression was witnessed in women with recurrent miscarriage when compared with fertile control women from our preliminary result.
Conclusion:
Altered vasculature of the endometrium in the peri-implantation period is detrimental to implantation and may lead to recurrent miscarriage. Being an angiogenic mediators, endometrial RAS may play a role around the time of embryo implantation, affecting subsequent pregnancy outcomes.
Keywords
Introduction
The peri-implantation period, also known as the window of implantation (WOI), is a short, and the most crucial, period pertaining to embryo implantation. It involves close interaction between the receptive endometrium and a competent blastocyst; desynchronization between these two factors could result in implantation failure and further adverse pregnancy outcomes.1,2
It is known that appropriate endometrial neovascularization is a prerequisite for successful embryo implantation. Endometrial neovascularization consists of two distinct processes: vasculogenesis and angiogenesis. Vasculogenesis, which occurs mainly in fetal development, during which angioblasts form a primary vascular network, 3 consists of three steps: namely hemangioblasts and angioblasts induction regulated by fibroblast growth factor (FGF), primordial vessel gathering mediated by the endometrial vascular growth factor (VEGF) family, and the vasculogenesis-to-angiogenesis transition. 4 Unlike vasculogenesis, angiogenesis represents the formation of new vessels out of pre-existing ones in response to hypoxia. New vessel growth is characterized by four mechanisms: sprouting, intussusception, vessel elongation and incorporation. Sprouting involves several steps, starting with the activation of endothelial cells, which release proteases to break down the basement membrane and migrate towards angiogenic sites. 5 Intussusception is the process whereby endothelial cells grow inwards into the vessel lumen and form a network of interlocking vessels. 6 Elongation, also known as remodeling, happens where existing vessels restructure continuously in response to the metabolic demands of the surrounding cells. 7 No endothelial sprout is observed in human endometrium, where mainly intussusception and elongation are thought to be involved in endometrial angiogenesis. 8
The role of the renin–angiotensin system (RAS) in regulation of blood pressure and hydro-electrolyte balance has long been recognized. Expression of the RAS has been determined in various tissues, including human endometrium. 9 It is believed that the local RAS works either independently or in conjunction with the circulating RAS to function in a paracrine and autocrine manner. 10 The substrate of RAS, angiotensinogen, is released and cleaved in the circulation by renin to form angiotensin I, which is further cleaved into angiotensin II by angiotensin-converting enzyme (ACE). Angiotensin II is considered the main effector of the RAS by acting on specific membrane bound Ang II receptors, including angiotensin II type 1 (AT1) receptor (AT1-R) and angiotensin II type 2 (AT2) receptor (AT2-R).
Recurrent miscarriages (RM) could be defined as the loss of three or more consecutive pregnancies before 20 weeks of gestation, affecting approximately 1% couples who are trying to conceive worldwide. 11 It has been suggested that abnormal endometrial angiogenesis is associated with RM.12,13
Although there are various angiogenic factors and inhibitors in the human endometrium, the exact role that each of them plays in regulating angiogenesis remains unclear. To the best of our knowledge, the number of published studies examining RAS in the peri-implantation endometrium is rather limited. The aim of this manuscript was to provide a literature review on the role of the endometrial RAS around the time of implantation. We also performed a pilot study to compare expression of AT1-R and AT2-R in the endometrium of women with RM with that of fertile controls.
Materials and methods
Literature search
A comprehensive search of electronic databases, including Web of Science, Google Scholar, MEDLINE, PubMed, and Scopus, was done to identify relevant published articles from inception to May 2019. A combination of the following text words was used: “renin angiotensin system”, “angiotensin II”, “angiopoietin-TIE”, “vascular endothelial growth factor”, “endometrium”, “implantation” and “angiogenesis”. Literature reviews, systematic reviews, and meta-analyses found in the search were also hand-searched and cross-referenced for relevant articles.
Ethical issues
We also carried out a pilot study, which was approved by Joint Chinese University of Hong Kong–New Territories East Cluster Clinical Research Ethics Committee (Approval Ref. No.: CREC-2018.365). All endometrial biopsies were collected with written informed consent of the participants.
Subjects and endometrial biopsy
A total of 20 women were included in this pilot study: ten with RM and ten fertile controls. All subjects had a daily urine dipstick test from day 9 of the menstrual cycle to identify the luteinizing hormone (LH) surge, and the endometrial biopsies were timed precisely on day 7 after the LH surge (LH+7), which is regarded as the peri-implantation period in a natural cycle.
Pipelle endometrial samplers (Prodimed, Neuilly-en-Thelle, France) were used to obtain the specimens, which were placed immediately into 10% neutral buffered formalin for fixation and then embedded in paraffin wax.
Immunohistochemistry
Immunohistochemistry was used to examine expression of AT-1 and AT-2 receptors. After embedding in paraffin wax, endometrial tissue was cut into 3.5 μm sections and mounted onto slides coated with 3-aminopropyl-triethoxysilane (Sigma-Aldrich, St. Louis, MO, USA), dewaxed in xylene, rehydrated through a descending alcohol series to phosphate-buffered saline (PBS) (pH 7.6) and quenched in 3% hydrogen peroxide in methanol for 20 min. After washing, unmasking of antigen was performed in sodium citrate buffer (pH 6.0) in a microwave oven. The paraffin-embedded human endometrial tissue sections were dewaxed in xylene, rehydrated through a descending ethanol series to Tris-buffered saline and antigen was unmasked in citrate buffer. The primary antibodies were rabbit anti-human angiotensin II receptor type-1 polyclonal antibody (AAR-011, Alomone Labs, Jerusalem, Israel) at a dilution of 1:100 and rabbit anti-human angiotensin II type 2 receptor antibody (ab19134, AbCam, UK) at a dilution of 1:100. After incubation, slides were washed in PBS Tween-20 and incubated in secondary antibody (goat anti-rabbit) for 1 h. Binding was visualized by incubation with peroxidase substrate 3.3-diaminobenzidene tetrahydrochloride (DAB; Dako, Copenhagen, Denmark). Slides were washed in distilled water and counterstained with 20% hematoxylin solution for 10 min and dehydrated through an alcohol series, cleared in xylene, and mounted in DPX medium (Sigma-Aldrich).
Scoring of staining results
This pilot study examined the localization of AT1-R and AT2-R in endometrial sections. The relative expression of these two receptors in various compartments of the endometrium was also examined and scored subjectively by the first author without knowledge of which group the specimens belonged to. The findings will be later confirmed by semi-quantitative histological evaluation using H-score, as described in a previous study. 14
Results
Literature on RAS and endometrium
After a comprehensive literature search, we identified a total of 66 relevant publications. The full articles of these 66 publications were retrieved for review, and a summary of the subject is presented in the Discussion.
Pilot results on the endometrial expression of AT1-R and AT2-R
In this pilot study, AT1-R and AT2-R appeared to be expressed in all endometrial compartments (luminal epithelium, glandular epithelium and stroma cells, and blood vessels) (Figure 1) to a variable degree (Table 1). In control subjects, both AT1-R and AT2-R expressed strongly in glandular epithelium, with relatively weak staining in the luminal epithelium. Specimen from women with RM appeared to have increased expression of AT1-R in glandular epithelium and stroma cells, and increased expression of AT2-R in glandular epithelium and blood vessels, compared with those in fertile controls from our preliminary results. Around the blood vessels, the staining of AT2-R appeared to be stronger than that of AT1-R in both the control and RM subjects.

Expression of AT1-R and AT2-R in the peri-implantation endometrium. Representative micrographs of immunohistochemical stain of (a) AT1-R in fertile control, (b) AT1-R in RM, (c) AT2-R in fertile control, (d) AT2-R in RM. Magnification ×200. Scale bar = 200 μm.
Comparison of AT1-R and AT2-R between fertile controls and women with RM in all compartments of endometrium during the peri-implantation period.

AT1-R, angiotensin type 1 receptor; AT2-R, angiotensin type 2 receptor; BV, blood vessels; CON, fertile controls; GE, glandular epithelium; LE, luminal epithelium; RM, women with recurrent miscarriage; ST, stroma.
Discussion
Endometrial angiogenesis
The endometrium is one unique site where periodical physiological angiogenesis happens.15,16 Rogers and colleagues have correlated new blood vessel growth with endometrial expressions of angiogenic growth factors. 17 They found that endothelial cell proliferation occurs throughout the menstrual cycle in human endometrium, reflecting continual vascular remodeling during menstrual cycles. 18 Endometrial angiogenesis happens in three distinct stages: to repair the vascular bed during menstruation, to allow rapid growth of endometrium during the proliferative stage, and to promote spiral arteriole coiling and growth during the secretory phase. 19
Another important function of endometrial angiogenesis is to prepare for embryo implantation and subsequent placentation. Although it is difficult to evaluate the profile of endometrial vascularity in early pregnancy, a good endometrial blood supply has been considered as an essential factor for successful implantation. During embryo implantation, the rapid growth of the uterus is supported by rich vessel formation to ensure supply of oxygen and nutrients to the uterus and the embryo.20,21 Studies have shown that endometrial vascularization is a good predictor for pregnancy outcome in both in vitro fertilization-embryo transfer (IVF-ET) cycles and frozen embryo transfer (FET) treatment cycles. 22 However, dysregulation of angiogenic factors and their inhibitors during the peri-implantation period may result in first-trimester miscarriage or defective placentation and increased risks of pregnancy disorders. 23 The process of angiogenesis is characterized by increasing vascular permeability and endothelial cell proliferation and migration. Angiogenesis is known to be regulated by various growth factors, among which the endometrial vascular growth factor (VEGF) family and the angiopoietin-TIE (Ang-Tie) system are the most investigated.
RAS and other angiogenic molecules in endometrium
It is suggested that AT1-R regulates vasoconstriction, while AT2-R plays a role in vasodilation in microvasculature. 24 In addition to the regulation of fluid and electrolyte balance and peripheral vascular resistance, angiotensin II has been shown to function as an important angiogenic growth factor in the regeneration of new blood vessels.25,26 One previous study found that angiotensin II was involved in 85% of the positive neovascularization in all implanted corneas, suggesting that angiotensin II not only activates preexisting collateral vascular pathways but also plays an active role in the angiogenic process. 27 In fact, it has been recognized for some time that angiotensin II is involved in mediating vascular permeability and stimulating angiogenesis in the uterus.28,29 The angiogenic process is initiated by the stimulation of growth factors, the most well-known being the VEGF family, consisting of six members: VEGF-A,-B,-C,-D,-E, placental growth factor (PIGF), and two VEGF receptors: VEGF receptor-1, Flt-1 (VEGFR-1) and KDR (VEGFR-2). KDR is widely considered as the central VEGF receptor in angiogenesis, while Flt-1 plays a supporting role. Previous studies have observed the expression in human endometrium of VEGF-A, VEGF-C, and PlGF, which are thought to play a critical role in implantation via promoting endometrial vascular permeability and dilation. 14 VEGF-A is the best studied of the VEGF family. It induces endothelial cell proliferation, migration, and differentiation, and it could also increase vascular permeability together with vascular integrity. VEGF-C is known to affect migration and proliferation of endothelial cells, acting as a growth factor for lymphatic vessels. PlGF is an important paracrine regulator of decidual angiogenesis and an autocrine mediator of trophoblast function. 30
VEGF, recognized as one of the earliest genes activated in the preimplantation embryo, could be produced by both decidual cells and the invading trophoblast.31,32 A significant increase in VEGF and its receptors are seen during the peri-implantation period.30,33 Abnormal expression of VEGF receptors may be a cause of lethality during embryogenesis. Studies have shown that trophoblastic knobs fuse with uterine epithelial cells, invade the sub-epithelial vessels, and become part of the vessel wall in mice during days 7 and 8 of pregnancy. 34 Therefore, it appears that VEGF could serve as a signal between the embryo and endometrial vascular structures. Similar findings have been found in another study, which analyzed directional VEGF secretion in polarized human endometrial epithelial cell cultures. 35
Another key system collaborating with the VEGF family to initiate angiogenesis in endometrium is the Ang-Tie signaling system. The Ang-Tie family is a binary system maintaining quiescence while responding to angiogenic stimuli. The human angiopoietin family consists of two receptors, Tie-1 and Tie-2, and three ligands, Ang-1, Ang-2, and Ang-4. Both Ang-1 and Ang-2 bind to Tie2, but only Ang-1 can activate Tie2 by inducing its autophosphorylation, 36 while Ang-2 acts as a competitive Ang-1 antagonist to inhibit Ang-1/Tie2 signaling in a context-dependent manner. Although Ang-4 has not been as well studied yet, it is thought to act like Ang-1. 37 Ang-Tie has a profound effect on blood vessel growth and maturation during angiogenesis.38–40 The angiopoietin family is thought to regulate angiogenesis by mediating perivascular cell migration and the formation of basement membranes. In the presence of angiogenic stimulators, sprouting endothelial cells can release Ang-2 and enhance mural cell detachment, vascular permeability, and endothelial cell sprouting. Ang-1, antagonist of Ang-2, is a natural inhibitor of vascular permeability, protecting against plasma leakage by tightening preexisting vessels. 41 Ang-1 is chemotactic for human endothelial cells, mediating the dialogue between vascular smooth muscle and the endothelium, thus playing a role in vessel stabilization. 42 On the other hand, Ang-2 mainly mediates vascular dilation, and disrupting vessel integrity.43,44
During the peri-implantation period in marmosets, Ang-1, Ang-2, and Tie-2 are found to participate in endometrial preparation for implantation, remodeling maternal vascularization, and trophoblast invasion. 45 In human endometrium, previous studies have shown expression of Ang-1, Ang-2, and Tie-2 in both epithelial and stromal components.42,46 Another study illustrated altered angiogenesis status in the peri-implantation endometrium after gonadotrophin-stimulation by investigating the expression of Ang-1 and Ang-2 together with VEGF-A. 47
VEGF acts in the early stages of angiogenesis by promoting the formation of primitive vessel structures prone to leakage while the Ang-Tie family contributes mainly to forming endothelial tight junctions and recruiting pericytes. 48 In response to VEGF, vascular permeability increases, which allows extravasation of plasma proteins to form a provisional scaffold for migrating endothelial cells. 49 Micro-vessels mediated by Ang-1 would degenerate if VEGF was absent, whereas, by maintaining the level of VEGF, proliferation of endothelial cells is promoted. Ang-1 could, in turn, induce sprouting of VEGF by phosphorylating tyrosine in Tie-2. 50 In addition, the effect of Ang-2 on angiogenesis, such as dropping off pericyte and altering extracellular matrix, depends largely on VEGF. 51 It is found that Ang-2 stimulates the formation of new blood vessels in the presence of abundant VEGF. However, Ang-2 expression is negatively associated with vascular structure regression when the concentration of VEGF is low. 52 Thus, co-expression of Ang-1, Ang-2, and VEGF can cause angiogenesis.
There is considerable evidence of a direct and close relationship between RAS and VEGF. In an electrical stimulation study, angiogenesis was proven to be regulated by both angiotensin II and VEGF. 53 It is reported that VEGF may increase local angiotensin II by inducing ACE, and that the inhibition of ACE reduces VEGF expression. 54 Moreover, studies have shown that angiotensin II stimulates the expression of angiogenic factors VEGF and angiopoietins in endothelial cells.55,56 Angiotensin II can stimulate expression of VEGF and its receptor, which eventually contributes to tissue remodeling and angiogenesis. 57 Similar studies have shown that VEGF expression is regulated by the AT1 receptor in respond to angiotensin II in a dose-dependent manner. 58 All these findings suggest that RAS may be involved in mediation of VEGF as well as in angiogenesis.
Although VEGF and angiopoietins may not be the only downstream angiogenic mediators of RAS, they are likely to be RAS-dependent in some way. 55 One study suggested that angiotensin II exerts its function at retinal neovascularization by stimulating Ang-2 expression through AT1 receptor-mediated pathways. 59 It is reported that angiotensin II could selectively induce Ang2 and VEGF expression via heparin binding epidermal growth factor (EGF)-like growth factor (HB-EGF)-mediated EGF receptor. In addition, Ang-2 could trigger angiotensin II mediated angiogenesis by enhancing angiogenic activity of VEGF. 60
Although the VEGF family, Ang-Tie system, and RAS are different types of vascular regulatory molecules for endometrial angiogenesis, how they correlate and interact with each other is far from being fully understood. Considering that there is rather limited literature on endometrial RAS, further detailed studies are needed to better understand the involvement of RAS in human endometrial neovascularization.
RAS signaling pathway and its mediated angiogenesis
Being the main effector hormone, angiotensin II mediates various physiological effects mostly by binding to AT1-R and AT2-R .The well-accepted mechanism whereby angiotensin II regulates vasoconstriction is via classic G protein dependent pathways. AT1 activates G-protein-coupled second messengers, including phospholipase C (PLC), phospholipase A2 (PLA2), and phospholipase D (PLD). 61 Activation of PLC will lead to production of inositol triphosphate (IP3) and diacylglycerol (DAG). IP3 could open a calcium channel by binding to its receptors. Numerous Ca2+ could then act on calmodulin and trigger myosin light-chain kinase (KLCK), which could phosphorylate the myosin light chain and enhance the interaction between actin and myosin, leading to contraction of smooth muscle cells and migration of cells. 62 At the same time, myosin light chain phosphatase (MLCP) can counter the defect of MLCK, thus inducing sustained contraction. 63 DAG, another product of PLC, contributes to increasing the pH during cell contraction by phosphorylating the Na+/H+ pump. It is also involved in the Ras/Raf/MEK/ERK pathway. 64 PLA2 exerts its effect mainly in maintaining a balance between vasoconstriction and vasodilation. Arachidonic acid (AA) and its metabolites are produced after PLA2 is phosphorylated and activated by angiotensin II. AA not only acts on maintaining vascular tone but also on NAD(P)H oxidation of vascular smooth muscle cells (VSMCs). 65 AA metabolites hydroxyeicosatetraenoic acids have a pro-hypertensive effect on angiotensin II-regulated VSMC vasoconstriction by facilitating the entry of Ca2+ into cells. 66 AT1 can also elicit PLD, resulting in the hydrolysis of phosphatidylcholine (PC) into choline and phosphatidic acid (PA). PA is converted rapidly to DAG, resulting in protein kinase C (PKC) activation and sustained muscle contraction. The DAG-PKC pathway is known to be implicated in vasoconstriction, angiogenesis, and endothelial permeability. In summary, alteration of PLC/PLD pathways may be involved in the pathogenesis of hypertension. 67 Possible pathways of angiotensin II-mediated angiogenesis are summarized in Figure 2. As a potent mediator of oxidative stress and oxidant signaling, angiotensin II stimulates the generation of reactive oxygen species (ROS) via membrane NAD(P)H oxidases in VSMCs after binding to AT1-R. 68 The production of ROS can regulate hypertension and vessel inflammation by increasing the release of cytokines and leukocyte adhesion molecules, inducing the recruitment of monocytes to endothelial damage. Moreover, ROS are involved in many angiotensin II-mediated effects, including production of hypoxia inducible factor-1α (HIF-1α) in vascular cells, 69 activation of p38MAPK, transcription factor nuclear factor kappa B (NF-kB), and nitric oxide (NO) in VSMCs. 70 Therefore, interactions between angiotensin II and ROS are critical in vascular physiology and pathology in terms of regulating vascular structure and function. HIF-1α is a well-known regulator of VEGF, and can regulate angiopoietin and VEGF by modulating ROS-dependent heparin binding-epidermal growth factor receptor (EGF-R). 62 Ang-Tie could also trigger the production of ROS through NADPH oxidase. 71 It has been verified in a rat model that VEGF and Ang-1 elicit the angiogenic process by sustaining phosphorylation of MAPKs and Akt pathway. 50
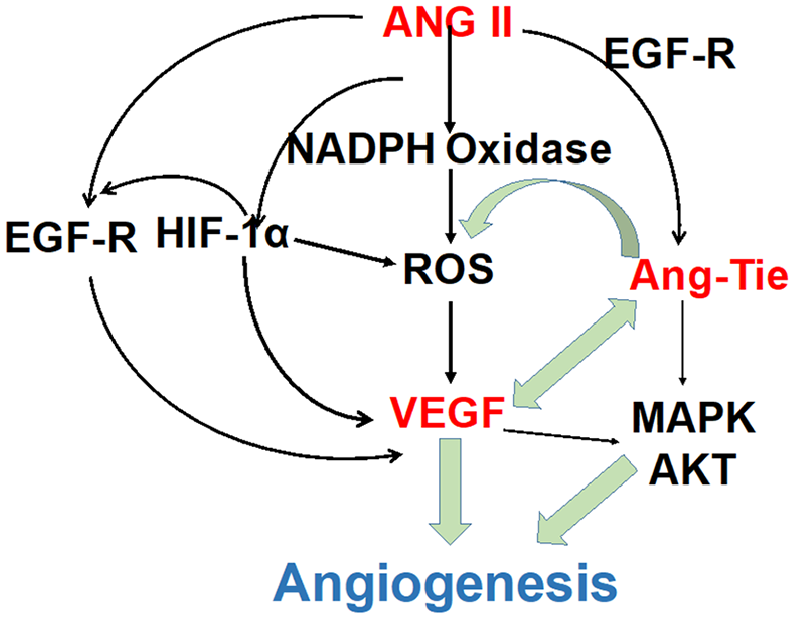
The possible pathways of Ang II-mediated angiogenesis. ANG II stimulates the generation of ROS through membrane NAD(P)H oxidases in VSMCs after binding to AT1-R. ROS are involved in many Ang II mediated effects, including production of HIF-1α in vascular cells, activation of p38MAPK, and transcription factor NF-kB. Interactions between Ang II and ROS are critical in vascular physiology and pathology in terms of regulating vascular structure and functions. Ang-Tie could also trigger the production of ROS through NADPH oxidase.
Potential prognostic value of RAS for pregnancy outcome
In the 1980s, Barker and colleagues found a strong relationship between decreasing birthweight and increasing death from coronary heart disease. 72 This hypothesis, often called the “Barker hypothesis”, states that adverse events, particularly during the intrauterine period, can result in increased disease risk later on. Peri-conceptional events are also found to have a long-term effect. A report from Sydney showed that one-third of mothers giving birth to small-for-gestational-age babies had been diagnosed with an eating disorder before the beginning of pregnancy. 73 Even very early perturbations in the maternal endometrium during the peri-implantation period may play a significant role in pregnancy outcome, and could also subsequently lead to detrimental consequences in adulthood. 74 Therefore, we believe that the endometrial environment, including angiogenesis in the peri-implantation period, could have an influence on the outcome of a subsequent pregnancy.
As shown in our preliminary work, a significantly higher expression was observed in women with RM compared with fertile controls. Although the result was insufficient to enable us to draw the conclusion that AT1-R and AT2-R serve as a potential predictors of implantation outcome, it did provide a significant clue as to the role of endometrial RAS. Further studies are needed to investigate whether endometrial RAS is involved in adverse pregnancy outcomes and could act as a prediction marker.
Disturbances in the uterine vascular system will cause an inadequate supply of nutrients and substrates, which is detrimental to normal intrauterine development. Some common adverse pregnancy outcomes, including pre-eclampsia and intrauterine growth restriction (IUGR), are thought to be associated with dysfunctional endometrial spiral arterioles remodeling. 75 In the first trimester of normal pregnancy, the endothelium, internal elastic lamina, and muscular coat of the spiral arteries are broken down in the placental bed after invasion of the cytotrophoblast.76,77 In the early second trimester, a second wave of cytotrophoblast invasion occurs, and the transformation of spiral arteries in myometrial segments happens. In pre-eclampsia and IUGR, it is estimated that only about 50–65% of the decidual spiral vessels undergo normal physiological changes, 78 and the rest do not achieve successful conversion of myometrial components. 79 As a result, placental blood flow is significantly decreased due to the impairment of primary invasion. The condition may become even worse owing to the increasing need of conceptus with advancing gestation. Pre-eclampsia is one of the leading causes of perinatal mortality, and is thought to be related to disturbed angiogenesis in the feto–maternal interface and defective placentation. 80 The RAS has long been considered to be related to preeclampsia. Back in 1961, all the components of the RAS, including renin, angiotensinogen, ACE, angiotensin I, angiotensin II and AT1 receptors were identified in human placental tissues, maternal decidua, and fetal placental tissues. 81 Angiotensin II can stimulate placental vasorelaxants, including NO and parathyroid hormone-related protein (PTHrP), which can either attenuate vasoconstrictors or cause concentration-dependent vasodilator effects in the feto-placental circulation to increase the basal perfusion pressure in the human placental cotyledon.82,83 Angiotensin II is reported to be a vital vasoconstrictor in feto-placental circulation, 84 and the receptor sites for angiotensin II have also been found in placenta. 85 A close relationship between aberrant expression of AT1-R and the occurrence of pre-eclampsia was also observed, and the inadequate placental perfusion is likely to be caused by a reduction of AT1.86–88
In animal studies, hypoxaemia in fetuses will activate the RAS and redistribution of blood towards fetal heart and brain. Vasoconstriction induced by placenta angiotensin II may exacerbate the disorder,89,90 and a similar effect is witnessed by infusing angiotensin II. 91 It has also been found that there are structural changes in placental vasculature in IUGR, and these changes are closely related to umbilical artery abnormality observed from Doppler flow velocity waveform. 92 Angiotensin II, being a trophic factor of vascular smooth muscle, could contribute directly to this remodeling process. 93 The “fetal origins” hypothesis is that the disproportionate growth in fetus could account for later coronary heart disease. 94 Epidemiological studies have revealed that IUGR in early fetal life is associated with hypertension development in later adulthood. 95 Given that numerous studies have investigated the relationship between RAS and hypertension, it would be very meaningful to investigate whether changes of RAS in the peri-implantation endometrium correlate with IUGR during subsequent pregnancy, or even hypertension during the later adult life of that fetus.
Conclusion
Optimal development of endometrial vasculature during implantation is a prerequisite for normal pregnancy. Our preliminary study found altered expression of AT1-R and AT2-R in endometrium in women suffering from RM. Further studies based on semi-quantitative H-scoring in a sufficiently large sample size are needed to confirm these findings and to explore the role of the RAS in angiogenesis and how it works with the VEGF family and Ang-Tie system. The potential value of endometrial RAS in predicting early and late pregnancy outcomes is also worth further investigation.
Footnotes
Acknowledgements
The authors would like to thank Terence Lao from The Chinese University of Hong Kong and Sotirios Saravelos from the In Vitro Fertilisation (IVF) Unit, Hammersmith Hospital, London, for his suggestions on preparation of the manuscript.
Author contributions
Conceptualization: RQ, TCL, and XC; writing—original draft preparation: RQ; writing—review and editing: TCL and XC; supervision: TCL; funding acquisition: XC.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Hong Kong Medical and Research Fund, grant number 06170186.
