Abstract
Background:
A questionnaire-based survey was conducted to evaluate attitudes towards a reusable self-injection system, SurePal™, among paediatric patients with growth disturbances who were prescribed treatment with somatropin (Omnitrope®) as part of routine clinical practice.
Methods:
This cross-sectional survey was incorporated into the multinational, multi-centre, noninterventional PAtients TReated with Omnitrope® (PATRO) Children study. Questions were mainly focused on five areas: the attractiveness of SurePal™; training received; use of the device; opinion of the low-drug wastage system; experience compared with previous devices used (among pretreated patients).
Results:
Final results from participants in France are reported. Completed questionnaires were returned by 409 participants. Most patients (55%) were male and 89% were recombinant human growth hormone (rhGH)-treatment naïve. Around 57% of children completed the questionnaire by themselves, while 43% had help from a family member/other person. The mean (standard deviation) age of all participants was 11.3 (3.6) years, and most patients were aged 10–12 years (n = 126) or 13–15 years (n = 117). Overall, 86% of patients reported that preparing SurePal™ for injection was easy/very easy. Similarly, 83% reported that performing injections with SurePal™ was easy/very easy. The attractiveness of SurePal™ was rated as good/excellent by the majority (85%) of patients; this proportion was similarly high (> 80%) across all age groups. The dose-memory function was rated as helpful/very helpful by 54% of patients. Of the 174 patients who reported using the low drug-waste feature, 90% found it to be helpful/very helpful. Among the 24 pretreated patients, 17 reported that SurePal™ was better/much better than their previous device.
Conclusions:
This questionnaire-based survey conducted in a large cohort of paediatric patients with growth disturbances from France confirms the ease of use of SurePal™ to support daily administration of Omnitrope® across all age groups. The demonstrated acceptability of the device may help to improve patient adherence to long-term daily treatment with rhGH.
Introduction
Poor adherence to recombinant human growth hormone (rhGH) therapy not only leads to reduced efficacy outcomes, such as failure of children to reach target height, but may also increase healthcare costs.1,2 A number of factors, affecting both the children and their families, can cause poor adherence. These include psychological/emotional problems, social/everyday problems and technical handling issues of the drug-delivery device. 3
Several device-related factors have been identified that may improve treatment adherence; these include simplicity, convenience and ease of use, together with the provision of appropriate education and training.4,5 Other factors identified as being important in the design of a rhGH-injection device intended for long-term use include reliability, lack of pain, safety during use/storage and the number of steps involved in the injection process.6,7
Seven rhGH products are available in France, and the associated injection devices for all products have undergone development and improvements to better meet the needs of patients. SurePal™ is a reusable self-injection system specifically designed to support the daily administration of somatropin (Omnitrope®) (Sandoz, Kundl, Austria). The device has been designed to be easy and convenient to use and to minimize drug wastage. 8 SurePal™ became available for patients to use in France in 2014. To better understand patients’ acceptability of the SurePal™ device, a questionnaire-based, cross-sectional, multicentre observational survey was conducted in France, Germany and the UK as part of the noninterventional PAtients TReated with Omnitrope® (PATRO) Children study.9,10 Interim survey data from France have been reported previously as part of a pooled analysis that also included data from participants in Germany and the UK. 10 Here, we report final results from French patients who participated in the questionnaire survey.
Methods
The study methodology has been reported previously. 9 In brief, the survey involved infants, children and adolescents who were already participating in the noninterventional PATRO Children study and therefore receiving treatment with Omnitrope®. All patients (or their caregivers) provided written additional informed consent to participate in the survey, and the survey was approved by the relevant ethics committees.
Eligible patients or their caregivers were provided with the questionnaire, an explanation of the survey and procedures, and timelines for returning the completed questionnaire. Completed questionnaires were to be returned at least 1 month after the start of use of SurePal™, and no later than 7 months after start of use. The questionnaire included questions on five main topics: training received; using SurePal™; attractiveness of the device; the low drug-wastage system; experience compared with other devices used previously (where applicable). Questions were generally scored on a 5-point scale, with -2 being the worst possible outcome (e.g. very hard or very poor) and 2 being the best possible outcome (e.g. very easy or excellent). Most questions also included an option to respond ‘I don’t know’.
Analyses were conducted for the French study population overall, by age group, and (for some questions) by who prepared the SurePal™ device and who performed the injections (myself [child] and not myself [child]).
Results
Completed questionnaires were returned by 409 of 547 (74.8%) French patients invited to participate. Full data and complete responses were not available for 20 of the 409 patients. Around 57% of children completed the questionnaire by themselves, while 43% had help from a family member or another person.
Study population
The majority of patients (55%) were male and most participants (89%) were rhGH treatment naïve. Of the 24 rhGH-pretreated patients, three had previously been treated with Omnitrope®. At the time of completing the questionnaire, the mean ± standard deviation (SD) duration of SurePal™ use was 125.9 ± 142.8 days for the overall study population, 123.2 ± 141.6 days for rhGH treatment-naïve patients and 128.0 ± 111.1 days for pretreated patients. Most patients overall had growth hormone deficiency (43%) or were children born small for gestational age (42%) (Table 1). The mean age ± SD of all participants was 11.3 ± 3.6 years, with most patients in the age groups 10–12 years (n = 126) or 13–15 years (n = 117) when completing the questionnaire.
Key characteristics of study participants.

Missing data are not shown. SD, standard deviation.
Training
Overall, most patients were trained in the use of SurePal™ by a hospital nurse (46.5%), followed by a doctor (23.2%), another person (20.0%) (person not specified) and a homecare nurse (9.5%). Over three-quarters of patients (78%) found that learning to use SurePal™ was very easy or easy; this proportion was more than 70% across all age groups (Figure 1). Only 19 of the 409 patients found the device very hard or hard to learn to use, and these were all rhGH-naïve patients.

Ease of learning to use SurePal™.
Preparing SurePal™ and performing injections
Half of children prepared their SurePal™ for injection themselves, while 47% and 2% had a family member or nurse, respectively, prepare their device. When children (or caregivers) were asked about the ease of preparing SurePal™ for injection, 86% overall reported this to be easy or very easy (Figure 2a). Across all age groups, more than 80% reported that preparing SurePal™ for injection was easy/very easy (Figure 2a). The proportion who found preparing SurePal™ for injection was easy or very easy was higher in the ‘myself (child)’ than in the ‘not myself (child)’ group (Figure 2b and 2c); this applied overall and across all age groups. Among patients who prepared injections for themselves, the proportion who reported this as easy/very easy was more than 85% across all age groups (Figure 2c).
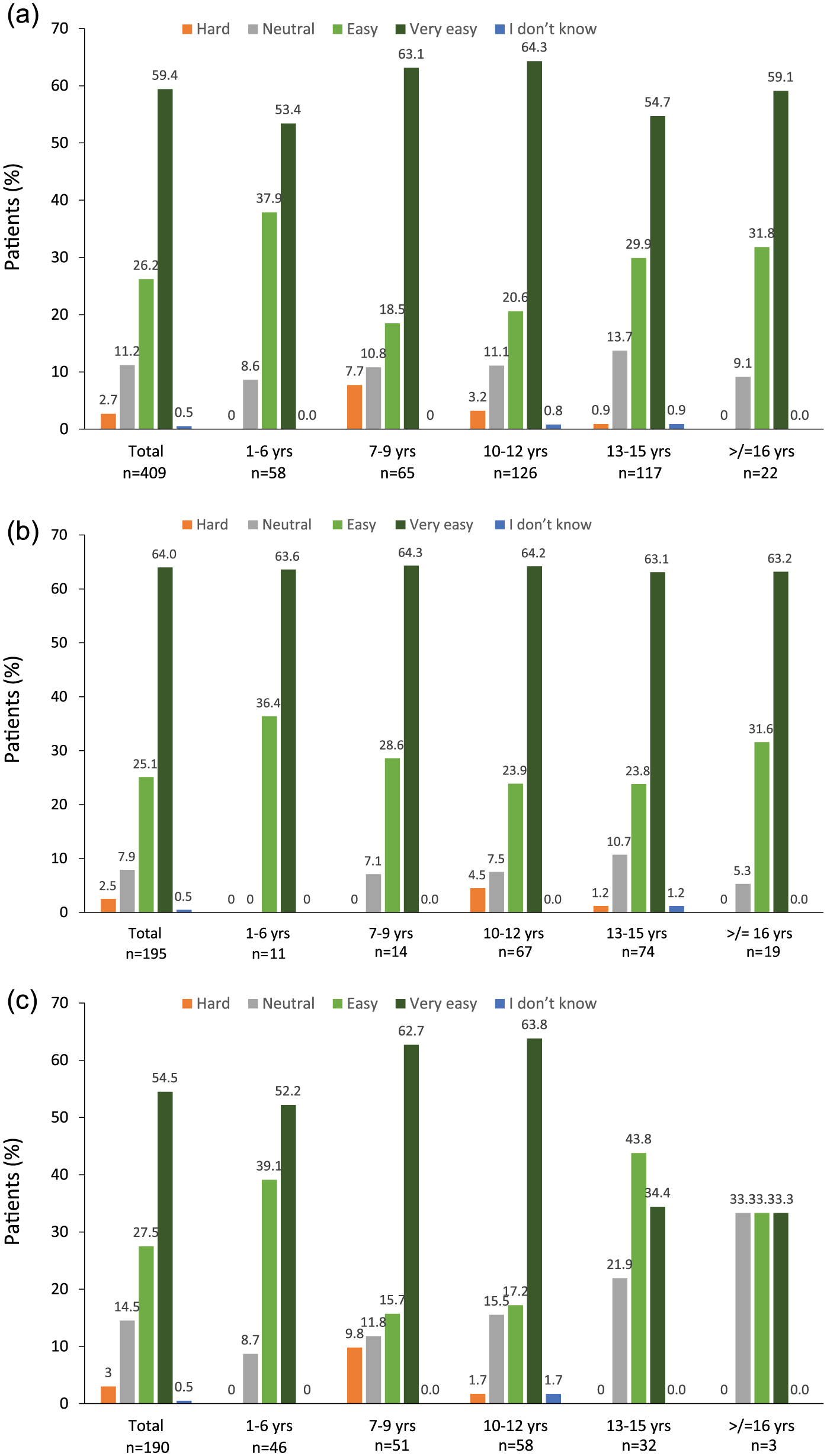
Ease of preparing SurePal™.
Injections were performed by 44% of children themselves, by a family member in 52% of patients, and by a nurse for 2% of participants. Self-administration was performed by approximately 70% of patients in the 13–15 years age group and by more than 80% in the 16 years or older age group. Overall, 83% reported that performing injections with SurePal™ was easy or very easy (Figure 3a). Across all age groups, more than 79% reported that performing injections with SurePal™ was easy/very easy (Figure 3a). The proportion who found that performing injections with SurePal™ was easy or very easy was similar in the ‘myself (child)’ and in the ‘not myself (child)’ group (Figure 3b and c). Among patients who performed injections themselves, the proportion who reported this as easy/very easy was more than 80% across all age groups except for the 7–9 years age group (69%) (Figure 3c).

Performing injections with SurePal™.
Attractiveness and use of SurePal™
Overall, 83% reported that using SurePal™ in general was easy or very easy (Figure 4); this proportion was more than 80% across all age groups. The attractiveness of SurePal™ was rated as good or excellent by the majority (85%) of patients (Figure 5); this proportion was similarly high (> 80%) across all age groups. Only one patient reported the attractiveness of SurePal™ as poor.

Using SurePal™.

Attractiveness of SurePal™.
Participants were also asked questions on specific features and handling tasks of SurePal™. The dose-memory function of SurePal™ was rated as very helpful or helpful by 54% of patients. Use of the low drug-waste feature was reported by 174 patients; of these, 90% found it to be helpful or very helpful. Putting in a new preassembled cartridge, attaching the needle and turning the dose knob were rated as easy or very easy by 79%, 91% and 78% of patients, respectively. Similarly, taking SurePal™ apart after an injection was rated as easy or very easy by 83% of respondents.
Experience compared with previous devices used
Among the 24 pretreated patients, 17 reported that SurePal™ was better or much better than their previous device; two patients reported that it was worse. Among this same group, 14 patients felt that SurePal™ made their rhGH-treatment plan easier to follow compared with their previously used device.
Discussion
The need for long-term daily subcutaneous injections has stimulated research into better methods of administration in an attempt to improve adherence to rhGH therapy. SurePal™ is a reusable self-injection system that has been developed to support daily administration of Omnitrope®. The vast majority of participants from France found SurePal™ easy to learn to use, and also found it easy to prepare the device and perform injections. Setting the injection dose and taking the device apart after an injection were rated as very easy or easy by most participants (78% and 83%, respectively). Furthermore, overall usability was rated as easy or very easy by 83% of patients.
In order to improve treatment adherence among children who require rhGH treatment, it is important that delivery devices are simple to learn to use, simple to use and well accepted by children of all ages as well as their parents/caregivers.5,11 In the current survey, the proportions of participants who rated SurePal™ as easy to learn to use (> 70%), easy to prepare for use (> 80%), easy to perform injections with (> 79%) and overall easy to use (> 80%) was high across all age groups of children. This is an important consideration, particularly among teens, for whom the highest rates of nonadherence have been reported. 12 Similarly, the proportion of participants who rated SurePal™ as easy to prepare for use (> 80%) and easy to perform injections with (> 80%) was high among both children who prepared/performed their own injections and those who did not prepare/perform their own injections.
Ease of use is a factor likely to determine choice of device and may also be associated with successful adherence with treatment.7,8 In comparative studies, devices considered by patients to be easier to use than comparator devices were associated with reduced handling errors, fear of injection (needle anxiety/needle phobia) and pain upon needle insertion, and were thus preferred. 13 SurePal™ has various features that may have contributed to patients rating the device as easy to use. These include an auto-priming feature, cartridges that are preassembled and ready-to-use, a sliding injection button that requires minimum force to perform an injection, and a dose-memory function that enables the correct dose to be preset and locked into the device. 8 SurePal™ appeared to compare well with previous devices used as 17 out of 24 pretreated patients reported that SurePal™ was better or much better than their previous device. However, it should be noted that the number of patients in the French cohort with experience of other rhGH-injection devices was relatively small.
In the current study, 43% of participants reported that they had used the low drug-waste feature, and of these, 90% found it to be helpful or very helpful. SurePal™ is specifically designed to minimize drug wastage. If a cartridge in the device does not contain enough drug to inject a full dose, the device automatically administers the correct amount of additional drug once a new cartridge is inserted, with no need for priming or adjusting the dose setting. This feature makes it more convenient to administer a second injection, compared with some other rhGH-injection devices. 8
A previous study assessed the usability and ease of use of SurePal™ among adults and children/adolescents in Germany and the USA. 8 In this study, participants performed injections into a pad. A very high proportion (92%) of participants rated the injection procedure as very easy or easy. In addition, 99% were able to disassemble the pen device successfully. 8 Findings reported here, along with previously reported combined data from France, Germany and the UK, 10 provide real-world clinical practice data that add to the findings reported by Rapaport and colleagues. 8
Over recent years, rhGH-injection devices have evolved to better help meet the needs of patients and improve adherence and potentially outcomes. These advances may need to be combined with other strategies to increase patient motivation to adhere to therapy, such as education about the injection device and injection techniques, and ongoing support from healthcare professionals, including comprehensive education about their condition, medication and expected outcomes. 13
The Injection Pen Assessment Questionnaire (IPAQ) was previously developed to evaluate perceptions of injection devices used to administer rhGH in children. The reliability and validity of IPAQ have been confirmed for the evaluation of usability and preference for rhGH treatment-injection devices. 14 The current SurePal™ survey used the same 5-point measurement scale as was included in IPAQ. Furthermore, the ease of use evaluation in the SurePal™ survey included the same measures of usability as IPAQ (preparing the device, setting the dose, injecting the medicine and disposal of the cartridge/needle). These similarities confirm the validity of the results from the current survey.
Potential limitations of the study include its open-label and uncontrolled design. In addition, the survey did not provide any information on whether the ease of use reported for SurePal™ translated into improved compliance with rhGH treatment. Also, the relatively small number of patients in the rhGH-pretreated subgroup precludes firm conclusions being made based on pretreatment status. Nevertheless, the survey questions were self-completed by participating children/their caregivers, with no influence or interference from healthcare professionals.
Conclusion
This questionnaire-based, cross-sectional survey conducted in a large cohort of paediatric patients with growth disturbances from France confirms the ease of use of SurePal™ to support daily administration of Omnitrope® across all age groups. The demonstrated acceptability of the device may help improve patient adherence to long-term daily administration of rhGH treatment.
Footnotes
Acknowledgements
The authors thank the patients and their caregivers who participated in the study, and all participating investigators and their specialist nurses.
Funding
This study was funded by Sandoz. Medical writing assistance was provided by Tony Reardon of Spirit Medical Communications Ltd, funded by Sandoz S.A.S., France.
Conflict of interest statement
RC has received speaker fees from Sandoz, Lilly, Merck, Pfizer and Novo Nordisk. The authors report no other conflicts of interest in this work.
