Abstract
The metabolic syndrome (MetS) is an increasingly prevalent condition in people with schizophrenia. It remains highly prevalent in the general population in developed countries, but recently health promotion campaigns and greater awareness of the high associated mortality rates have resulted in improvements in the rates of cardiovascular risk factors. This is not the case for people with schizophrenia who continue to have more than twice the rates of MetS and significantly higher mortality rates than the general population. Various behavioural and pharmacological interventions have been used to improve conditions that are linked to MetS, mainly smoking and obesity. This review aims to provide an update of the latest knowledge about the behavioural, pharmacological and other interventions that might help to combat this life-threatening problem in people with schizophrenia.
Keywords
The metabolic syndrome in schizophrenia: introduction
Schizophrenia is a highly heritable condition which is associated with a dramatic reduction in lifespan. A meta-analysis of existing data revealed a substantial gap between the health of people with schizophrenia and the general population [Saha et al. 2007]. Mortality in schizophrenia is largely due to cardiovascular disease [Tandon et al. 2009]. Sudden cardiac death, often resulting from cardiac arrhythmias, is also an important cause of mortality [Koponen et al. 2008].
Schizophrenia has been associated with an increased risk of diabetes since the nineteenth century [Maudsley, 1979]. Henry Maudsley was one of the first psychiatrists to notice an association between diabetes and schizophrenia. This was prior to the development of antipsychotic treatments. Even today, a significant number of studies have demonstrated that antipsychotic-naïve patients have impaired glucose tolerance, increased insulin resistance and increased visceral fat distribution compared with normal controls [Thakore et al. 2002; Venkatasubramanian et al. 2007; Fernandez-Egea et al. 2009]. More importantly, other studies have shown increased glucose intolerance in the siblings of people with schizophrenia and an increased prevalence of type 2 diabetes in the parents of nonaffective psychosis subjects [Fernandez-Egea et al. 2008a, 2008b]. Recently, a Danish study found that having schizophrenia is associated with an at-risk allele for type II diabetes located in the TCF7L2 gene [Hansen et al. 2011]. These findings suggest that diabetes and schizophrenia may share familial risk factors or common genetic predisposition.
The metabolic syndrome (MetS; also known as syndrome X, syndrome of chronic cardiovascular disease and Reaven’s syndrome) is a constellation of different conditions, including abdominal obesity, insulin resistance, dyslipidaemia and elevated blood pressure (BP). All components of MetS (with obesity holding a central role in its development) have been recognized as independent risk factors for cardiovascular disease. Therefore, the presence of MetS is associated with other comorbidities such as the prothrombotic state, proinflammatory state, nonalcoholic fatty liver disease and reproductive disorders [Cornier et al. 2008].
It has been estimated that in the USA as many as 60% of people with schizophrenia meet the criteria for MetS, as opposed to 30% for the general population [Mendelson, 2008]. Numerous studies have shown that overweight and diabetes are in general increased in patients with schizophrenia, with a two- to fourfold increase in the risk of diabetes compared with the general population [Leucht et al. 2007].
As the presence of MetS is associated with an increased risk for cardiovascular disease and death, and patients with schizophrenia are increasingly predisposed to develop it, it is of paramount importance to develop and implement strategies which can tackle this problem in this particular group of patients. It is also imperative that the awareness of clinicians is increased regarding this insidious but also potentially lethal condition.
Rationale and objectives of our review
In this review we provide an update about how best to combat MetS in schizophrenia. We aim to explore any pharmacological, behavioural and combined interventions targeting physical health and improving cardiovascular risk factors in patients with schizophrenia.
We set the following objectives at the beginning of our review:
(1) To summarize the interventions currently available, their effectiveness and most importantly to ascertain which interventions appear to be the most effective and therefore should attract clinical interest.
(2) To discuss the importance of monitoring in the early detection of MetS.
Review methodology
Eligibility criteria
We included original articles published in English language up until 2011 that provided evidence on behavioural, pharmacological and combined interventions. We excluded studies of poor methodological or informative value, such as case reports or pre-experimental observational studies.
Information sources
Articles were retrieved from the ISI Web of KnowledgeSM platform (Thomson Reuters), a comprehensive database that incorporates the Web of Science (1970 to present) and MEDLINE (1950 to present) and also includes articles from PsychINFO and the Cochrane Review Database.
Search
We searched for articles using the terms: Title=(schizophrenia OR severe mental illness OR antipsychotic) AND Title=(exercise OR weight management OR weight reduction OR smoking cessation OR smoking reduction OR health intervention OR well being OR wellness OR caloric restriction OR diet OR nutrition), published up until 2011. Our initial search generated 320 hits. We completed our search by checking against previously published reviews and extracting additional articles (Table 1).
Published reviews on healthy living interventions in schizophrenia and severe mental illness.
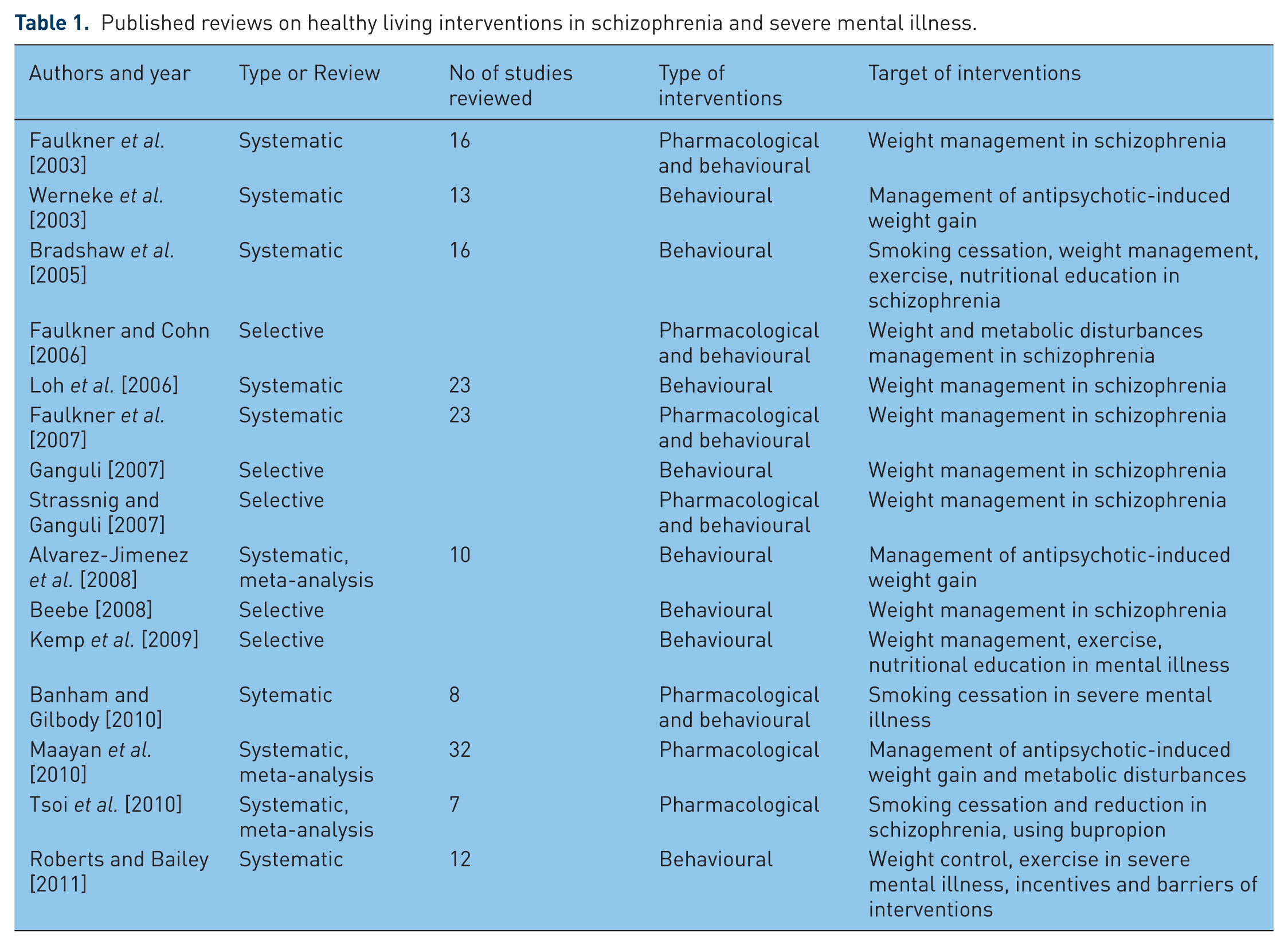
Study selection
Screening of articles was based on titles and abstract reading. Only articles fulfilling our eligibility criteria were included and full texts were subsequently obtained. We originally planned to include randomized controlled trials (RCTs) and other trials of high evidential quality. However, it soon became apparent that by doing so we would inevitably create an important source of bias. By excluding naturalistic studies, leaving us with significantly more pharmacological studies, as the majority of studies describing behavioural interventions are naturalistic, this would consequently disturb the balance we wished to maintain between the two kinds of interventions in our review. Furthermore, naturalistic studies, despite their less robust methodology, often convey valuable information, at a pragmatic level, and we did not want to appear dismissive of their contribution. So in addition to experimental studies (RCTs and non-RCTs) we included some naturalistic studies too.
Outcome
A total of 95 original studies were identified (Tables 2–4). Several researchers have tried to summarize the current evidence of MetS in patients with schizophrenia in numerous systematic or selective reviews. We identified a significant number of reviews that focus on behavioural and pharmacological interventions targeting metabolic disturbances in schizophrenia and severe mental illness. There are a number of reviews that focused on epidemiological studies, which also attempted to address the pathophysiological connections between MetS and schizophrenia. In addition, a group of reviews have focused particularly on studies of metabolic features associated with the use of second-generation antipsychotics. The latter two categories are not covered here as they are beyond the scope of this article.
Original articles on behavioural physical health interventions in schizophrenia.

BMI, body mass index; CBT, cognitive behavioural therapy; CLOZ, clozapine; DART, diabetes awareness rehabilitation training; FE, first episode; HAL, haloperidol; NR, nicotine replacement; OLZ, olanzapine; RCT, randomized controlled trial; RISP, risperidone; SCZ, schizophrenia; SCZA, schizoaffective disorder; SMI, serious mental illness; TAU: treatment as usual.
Original articles on pharmacological physical health interventions in schizophrenia.

AMT, amantadine; ATM, atomoxetine; BPR, bupropion; C&A, children and adolescents; CBT, cognitive behavioural therapy; CHLORPH, chlorphenetermine; CLOZ, clozapine; D-FNFL, D-fenfluramine; DXA-S, dextroamphetamine sulphate; FE, first episode; FGA, first-generation antipsychotics; FLV, fluvoxamine; FLX, fluoxetine; HAL, haloperidol; MTF, metformin; NR, nicotine replacement; NZT, nizatidine; OLZ, olanzapine; ORL, orlistat; PCB, placebo; PHENME, phenmetrazine; PHENYL, phenylpropanolamine; QUET, quetiapine; RBX, reboxetine; RCT, randomized controlled trial; RISP, risperidone; RSL, rosiglitazone; SBT, sibutramine; SCZ, schizophrenia; SCZA, schizoaffective disorder; SMI, serious mental illness; TAU, treatment as usual; TPR, topiramate; VRN, varenicline; ZIPR, ziprasidone.
Original articles on mixed physical health interventions in schizophrenia.

BPR, bupropion; BT, behavioural therapy; CBT, cognitive behavioural therapy; LFI, lifestyle intervention; MTF, metformin; OLZ, olanzapine; PCB, placebo; RCT, randomized controlled trial; SCZ, schizophrenia; SCZA, schizoaffective disorder; SGT, supportive group therapy; SMI, serious mental illness; TAU, treatment as usual.
Description and discussion of studies
Studies of behavioural interventions related to metabolic syndrome in severe mental illness
A total of 42 studies were identified in this category, testing interventions that targeted physical health and cardiovascular fitness, smoking and weight. Behavioural interventions are described in various terms, which may also be used interchangeably or share common concepts. Among the most commonly mentioned are the following:
(1) Wellbeing programmes, a holistic approach, which incorporates physical health, checks, physical exercise and dietary advice. They can target specific conditions (smoking, obesity) but also aim at the overall improvement of an individual’s quality of life, placing special emphasis on mental health. Their duration varies from a few weeks to several months and they are ‘tailor-made’ to respond to patients’ needs.
(2) Cognitive behavioural treatment (CBT), a broadly used psychological module, which aims primarily to modify erroneous beliefs and behaviours. CBT can have various applications, such as in smoking reduction.
(3) Nutritional education, which usually consists of specialist dietary input, focusing on calorie restriction and healthy diet.
(4) Weight management, a term used to describe a combination of strategies targeting obesity or overweight, such as physical activity and modification of dietary habits.
(5) Psycho-education, usually describing information offered to patients regarding their medication and illness in a manner that can enhance medication adherence and promote relapse prevention.
Studies on physical health and cardiovascular fitness
Some naturalistic studies provided limited evidence that wellbeing support programmes, holistic approaches or exercise can generally improve physical health and cardiovascular fitness in patients with severe mental illness (SMI) or schizophrenia [Lukoff et al. 1986; Pelham et al. 1993; Fogarty and Happell, 2005; Eldridge et al. 2011]. An RCT comparing wellbeing training with standard care showed improved rates of self-reported heath status following a 12-month wellness training intervention [Chafetz et al. 2008]. Another recent RCT showed that a motivational intervention in patients with schizophrenia spectrum disorders significantly increased their attendance to a physical exercise programme [Beebe et al. 2011].
Studies on smoking cessation/reduction
An RCT showed that CBT added to nicotine replacement (NR) works better than NR alone for smoking reduction or discontinuation; however, abstinence was not achieved [Baker et al. 2006]. Another experimental study, employing an ABA crossover design (alternation of treatment ‘B’ and nontreatment ‘A’ phases within the same subjects) showed that monetary reinforcement can lead to both reduction and abstinence [Roll et al. 1998].
Studies on weight reduction/diet
Most of the studies on behavioural interventions targeted weight reduction, of which almost a third used an RCT design. This group of studies is utterly versatile, not only in terms of design but also with regards to the type of particular interventions tested (or combinations of these), duration of intervention or period of observation, number of participants and specification of intervention (weight reduction in general or in the context of antipsychotic medication). Although details of all these studies are included in the relevant tables, for the purposes of our discussion, we will not take into account studies with very small sample sizes (i.e. <10).
Wide availability of fruit and vegetables can improve diet but this effect was not sustained [McCreadie et al. 2005]. Calorie restriction, alone or combined with nutritional education and some behavioural or motivational strategies, appears to be effective in tackling weight gain [Sletten et al. 1967; Aquila and Emanuel, 2000; O’Keefe et al. 2003; Vreeland et al. 2003; Menza et al. 2004]. Diet and exercise can cause patients taking clozapine to lose weight [Wu et al. 2007], and individual nutritional education can attenuate weight gain in patients taking olanzapine, as shown by a recent RCT [Evans et al. 2005]. Despite these encouraging results, a 2-year retrospective chart review failed to show any weight reduction by calorie restriction only.
Comprehensive weight management programmes, including diet, exercise and counselling on lifestyle modifications, can also prove helpful in reducing weight, as shown by a naturalistic study and an RCT [Kwon et al. 2006; Poulin et al. 2007]. However, another naturalistic study showed no significant outcomes following a similar nurse-led programme in patients with schizophrenia spectrum disorders [Ohlsen et al. 2004].
Several other interventions, including behavioural components and psychoeducation, have shown various degrees of effectiveness in dealing with overweight obesity and improving antipsychotic-induced weight gain [Littrell et al. 2003; Brar et al. 2005; Kalarchian et al. 2005; Alvarez-Jimenez et al. 2006; Brown and Chan, 2006; McKibbin et al. 2006; Scocco et al. 2006; Pendlebury et al. 2007; Forsberg et al. 2008]. Some studies even demonstrated a relative advantage of behavioural techniques compared with diet or brief nutritional information [Harmatz and Lapuc, 1968; Khazaal et al. 2007]. Of note are two large naturalistic studies of structured wellbeing programmes (targeting both physical and mental health with emphasis on healthy lifestyle promotion) which showed weight reduction or improvements in lifestyle habits [Smith et al. 2007; Lindenmayer et al. 2009]. Smith and colleagues employed a multistep approach to provide a combination of assessment of physical health, lifestyle and medication side effects; feedback offered to clients; and referral to weight management/physical activity groups in a total of 956 outpatients with SMI lasting for up to 2 years. They noticed significant improvement in levels of physical activity, smoking, diet and self-esteem, though there were no changes in mean body mass index (BMI) or cardiovascular risk factors. Lindenmayer and colleagues described a 36-week inpatient programme for 275 chronically ill patients, offering a combination of psychoeducation, dietary advice and physical exercise and targeting primarily obesity and metabolic abnormalities. They found a significant decrease in BMI, especially in patients with diabetes.
Studies of pharmacological interventions related to metabolic syndrome in severe mental illness
A total of 44 studies were identified in this category, testing interventions that target smoking and weight. The majority of studies adopted a robust RCT design.
Studies on smoking cessation/reduction
Bupropion
Four RCTs tested bupropion (BPR) versus placebo, on its own or as an adjunct treatment to CBT or NR. They favoured BPR for either abstinence or smoking reduction; however, outcomes are not always sustainable [George et al. 2002, 2008; Fatemi et al. 2005; Evins et al. 2007].
Nicotine replacement
One RCT showed NR to be effective in smoking reduction [Dalack and Meador-Woodruff, 1999].
Varenicline
One RCT showed improved abstinence in patients receiving varenicline; however, it involved a very small sample of nine patients in total [Weiner et al. 2011].
Studies on weight reduction
A large number of RCTs tested various agents for weight reduction, usually in the context of antipsychotic medication. In a 2010 systematic review and meta-analysis of 32 RCTs of pharmacological interventions to attenuate antipsychotic-related weight gain, Maayan and colleagues ranked a number of medications according to their efficacy in reducing weight (from the most efficacious to the least): metformin, d-fenfluramine, sibutramine, topiramate, reboxetine, amantadine, nizatidine, orlistat, metformin plus sibutramine, famotidine, dextroamphetamine, fluoxetine, rosiglitazone [Maayan et al. 2010]. Specific findings per agent are briefly described below.
Dextroamphetamine sulphate
This medication did not modify appetite [Modell and Hussar, 1965].
Amantadine
Amantadine attenuated olanzapine-induced weight gain [Correa et al. 1987; Floris et al. 2001; Deberdt et al. 2005; Graham et al. 2005].
Atomoxetine
Atomoxetine was not effective in reducing weight gain associated with olanzapine or clozapine treatment [Ball et al. 2011].
Chlorphenetermine, phenmetrazine
When compared with each other, neither drug significantly reduced weight [Sletten et al. 1967].
D-Fenfluramine
This medication attenuated neuroleptic-induced obesity [Goodall et al. 1988].
Fluvoxamine
Contradictory findings were reported from two RCTs assessing fluvoxamine’s efficacy in reducing clozapine-induced weight gain [Hinze-Selch et al. 2000; Lu et al. 2004].
Fluoxetine
Fluoxetine did not attenuate olanzapine-induced weight gain [Poyurovsky et al. 2002; Bustillo et al. 2003].
Famotidine
Famotidine did not attenuate olanzapine-induced weight gain [Poyurovsky et al. 2004].
Metformin
Most studies point towards metformin being efficacious in attenuating olanzapine-, clozapine-, risperidone- and quetiapine-induced weight gain [Morrison et al. 2002; Klein et al. 2006; Baptista et al. 2007; Arman et al. 2008; Wu et al. 2008a; Carrizo et al. 2009]. Only a few studies provided contradictory findings, also when metformin was combined with sibutramine [Baptista et al. 2001, 2006, 2008].
Nizatidine
Contradictory findings were reported from three RCTs about the efficacy of nizatidine in attenuating olanzapine-induced weight gain [Atmaca et al. 2003; Cavazzoni et al. 2003]. Although it appears to attenuate quetiapine-induced weight gain [Atmaca et al. 2004].
Orlistat
Orlistat appears to be more efficacious for olanzapine- or clozapine-induced weight gain in men [Joffe et al. 2008; Tchoukhine et al. 2011].
Phenylpropanolamine
This medication did not attenuate clozapine-induced weight gain [Borovicka et al. 2002].
Reboxetine
Reboxetine attenuated olanzapine-induced weight gain [Poyurovsky et al. 2003, 2007].
Rosiglitazone
This medication did not attenuate olanzapine-induced weight gain or improve glucose metabolism [Baptista et al. 2009; Henderson et al. 2009].
Sibutramine
Sibutramine attenuated olanzapine-induced weight gain but not clozapine-induced weight gain [Henderson et al. 2005, 2007].
Topiramate
This medication attenuated olanzapine-induced weight gain [Ko et al. 2005; Nickel et al. 2005; Kim et al. 2006].
Switching antipsychotic agents
A naturalistic study showed that switching from risperidone or olanzapine to ziprasidone led to weight reduction, however this effect was not observed when switching from first-generation antipsychotics to ziprasidone [Weiden et al. 2003].
Studies of mixed interventions related to metabolic syndrome in severe mental illness
A limited number of studies attempted to test the efficacy of combinations of behavioural and pharmacological interventions or even compare different kinds of interventions. Only nine studies were identified in this category. Two naturalistic studies showed that behavioural techniques (group therapy, motivational enhancement, positive reinforcement and anxiety reduction) can help maintain abstinence from smoking when combined with NR [Ziedonis and George, 1997; Addington et al. 1998]. When group therapy was added to NR, in order to reduce smoking, there was no difference between modules specially designed for patients with schizophrenia and those aimed at the general public. However, it seems that combining NR with atypical antipsychotic agents provided better outcomes [George et al. 2000]. BPR appeared quite a promising intervention when combined with either group therapy or CBT to reduce smoking [Evins et al. 2001, 2005; Weiner et al. 2001, 2007]. Switching from olanzapine to risperidone significantly reduced prevalence rates of MetS. However, adding behavioural treatment to risperidone did not add to this effect [Meyer et al. 2005]. Finally, an RCT comparing metformin and a lifestyle intervention (LFI) in weight reduction found a combination of the two interventions to be more effective than either intervention alone. Metformin was superior to LFI [Wu et al. 2008b].
Findings from reviews of healthy-living interventions in schizophrenia and severe mental illness
Behavioural interventions
In 2003, Werneke and colleagues systematically reviewed 13 studies on behavioural management of antipsychotic-induced weight gain; none of these studies were RCTs [Werneke et al. 2003]. They found that behavioural approaches, including diet, exercise and modification of treatment were possibly effective, considering limited evidence and methodological flaws.
In 2003, Bradshaw and colleagues published a systematic review of 16 studies of healthy living interventions in schizophrenia, including smoking cessation, weight management, exercise and nutritional education [Bradshaw et al. 2005]. They acknowledged the poor quality of the majority of studies; however, they noted that most studies showed positive outcomes.
In 2006, Loh and colleagues published another systematic review of 23 articles on interventions for weight management in schizophrenia [Loh et al. 2006]. Despite the fact that most of the literature was not methodologically sound, some controlled studies suggested that behavioural interventions could prevent weight gain and in some cases promote weight loss.
In 2007, Ganguli published a selective review of weight loss therapy in schizophrenia, confirming the above findings and also suggesting that weight can be controlled on a long-term basis [Ganguli, 2007].
In a 2008 systematic review and meta-analysis of 10 RCTs of nonpharmacological management of antipsychotic-induced weight gain, Alvarez-Jimenez and colleagues showed that individual, group, cognitive-behavioural or nutritional counselling interventions were effective in reducing or attenuating weight gain and maintaining treatment effects over time [Alvarez-Jimenez et al. 2008].
In a 2008 selective review of obesity in schizophrenia, assuming a nurses’ perspective, Beebe noted that measures such as diet teaching (adapted to the cognitive capacities of patients) and exercise could be effective in dealing with obesity in schizophrenia [Beebe, 2008].
In another 2009 selective review of weight management, exercise and nutritional education in mental illness, Kemp and colleagues found that most studies reported modest success during the period of intervention. However, this effect is not generally sustainable [Kemp et al. 2009]. They commented that even limited success could significantly reduce the likelihood of development of physical comorbidities.
Finally, in 2011 Roberts and Bailey presented a systematic review of 12 quantitative and qualitative studies of LFIs in SMI, including weight control and exercise [Roberts and Bailey, 2011]. They also identified possible barriers (illness symptoms, treatment effects, lack of support, negative staff attitude) and incentives (symptom reduction, peer and staff support, knowledge, personal attitudes) to these interventions.
Mixed behavioural and pharmacological interventions
Faulkner and colleagues reviewed the evidence for both pharmacological and behavioural interventions for weight management in schizophrenia by publishing two systematic reviews of 16 studies and 23 RCTs respectively and one selective review published between 2003 and 2007 [Faulkner et al. 2003, 2007; Faulkner and Cohn, 2006]. They concluded that, although difficult, the prevention of weight gain and promotion of weight loss is not impossible and can be achieved with a combination of medication and LFIs.
In a similar selective review published in 2007, Strassnig and colleagues found that antiobesity drugs, behavioural approaches, and in some cases bariatric surgery may all lead to significant weight loss in patients with obesity and schizophrenia [Strassnig and Ganguli, 2007]. The authors emphasized the need for rigorous studies to determine whether weight loss achieved in short-term interventions is maintained.
Finally, in 2010 Banham and Gilbody published a systematic review of eight RCTs of both pharmacological and behavioural interventions for smoking cessation in SMI [Banham and Gilbody, 2010]. They concluded that treating tobacco dependence is effective in patients with SMI, and the same treatments are effective in the general population and those with mental illness. They also found that treating tobacco dependence in patients with stable psychiatric conditions does not worsen their mental state.
Pharmacological interventions
In 2010, Maayan and colleagues published a systematic review and meta-analysis of 32 RCTs of pharmacological interventions to attenuate antipsychotic-related weight gain and metabolic abnormalities [Maayan et al. 2010]. Across a total of 1482 subjects, 15 different medications were tested: amantadine, dextroamphetamine, d-fenfluramine, famotidine, fluoxetine, fluvoxamine, metformin, hizatidine, orlistat, phenylpropanolamine, reboxetine, rosiglitazone, sibutramine, topiramate, and the combination of metformin and sibutramine. Compared with placebo, metformin showed the greatest weight loss, followed by d-fenfluramine, sibutramine, topiramate and reboxetine. Weight loss was found to be significant with metformin after weight gain had occurred, but not when started concomitantly with antipsychotics.
In a 2010 systematic meta-analysis, Tsoi and colleagues examined the efficacy of BPR for smoking cessation and reduction in schizophrenia, comparing data from seven RCTs. These authors found that biochemically verified self-reported smoking cessation rates (measuring expired carbon monoxide levels) after BPR were significantly higher than placebo at the end of the treatment. They concluded that BPR increases rates of abstinence in smokers with schizophrenia, without jeopardizing their mental state [Tsoi et al. 2010].
Discussion
Can metabolic syndrome be prevented? The role of clinicians and monitoring
Prevention (when feasible) is better than cure, and in the case of MetS, this can be achieved by relatively inexpensive means. The fact that MetS can quickly develop as a response to antipsychotic medication renders the role of clinicians paramount in its early detection, and the only way to do this is using a rigorous monitoring plan. The level of monitoring in many cases is far from being satisfactory, especially in the community. A large audit of 48 assertive outreach teams in the UK, including 1966 patients, revealed that more than 60% of this population had no evidence of screening for BP, obesity, blood glucose and lipid profile [Barnes et al. 2008]. However, those numbers significantly improved 1 year after implementing a blend of approaches to influence the behaviour of mental health professionals. Various research teams have suggested different approaches to the monitoring challenge.
The Belgian Consensus Group
This group recommended the following monitoring for basic features of MetS [De Nayer et al. 2005]:
(1) Weight and waist circumference (WC): weekly in hospital care, monthly in ambulatory care.
(2) Fasting plasma glucose (FPG): monthly in patients with a family history of diabetes/obesity or who are overweight/have obesity or impaired fasting glucose; at 6 and 12 weeks then every 3 months in patients without risk factors.
(3) Fasting plasma lipids (FPLs): every 3 months for the first year of treatment, then monthly.
(4) BP: every 3 months.
The British Association of Psychopharmacology
This group recommended a thorough evaluation of risks of developing MetS in all patients receiving antipsychotic medication, followed by individual tailoring of pharmacological treatment to minimize metabolic risk and education/advice [Barnett et al. 2007]. Their suggested monitoring includes:
(1) Personal family history: at baseline.
(2) Height/weight and BMI: baseline, 4 weeks, 8 weeks, 12 weeks, 6 months then annually.
(3) BP: at baseline, 12 weeks then every 6 months.
(4) FPG: at baseline, 4 weeks, 8 weeks, 12 weeks then every 6 months.
(5) FPL: at baseline, 12 weeks then every 6 months.
The European Psychiatric Association, supported by the European Association for the Study of Diabetes and the European Society of Cardiology
These organizations recommended a comprehensive four-step monitoring and management algorithm [De Hert et al. 2009]:
(1) Step 1 (history including previous diseases, family history, smoking, exercise, dietary habits): at baseline, 12 months, then annually.
(2) Step 2 (BP, weight, WC, BMI): at baseline, 6 weeks, 12 weeks, 12 months then annually.
(3) Step 3 (FPG, FPL, ECG): at baseline, 6 weeks, 12 weeks, 12 months then annually.
(4) Step 4 (advice on smoking cessation; food choices; physical activity): at baseline, 6 weeks, 12 weeks, 12 months then annually.
The Maudsley Prescribing Guidelines
These are detailed guidelines on antipsychotic profiles with regards to hypertension, weight gain, diabetes/impaired glucose tolerance and dyslipidaemia [Taylor et al. 2009]. The authors recommended a comprehensive schedule of monitoring covering a variety of physiological parameters for patients receiving antipsychotic medications:
(1) Urea and electrolytes (U&Es): at baseline then annually.
(2) Full blood count (FBC): at baseline then annually.
(3) FPL: at baseline, 3 months then annually.
(4) FPG: at baseline, 4–6 months then annually.
(5) Weight, BMI: at baseline, every 3 months for the first year then annually.
(6) Electrocardiogram (ECG): at baseline, after dose increases.
(7) BP: at baseline, frequently during dose titration.
(8) Prolactin: at baseline, 6 months then annually.
(9) Liver function tests (LFTs): at baseline then annually.
(10) Creatinine phosphokinase: at baseline, then if neuroleptic malignant syndrome is suspected.
What we need to emphasize here is that physical monitoring has to be treated as the responsibility of not only physicians and general practitioners but also treating psychiatrists. It is very important that a ‘metabolic monitoring toolkit’ consisting of screening for BMI, FPG, FPL and BP is incorporated into the regular follow up of patients, along with the standard psychiatric evaluation [Meyer and Stahl, 2009]. Even if adherence to the above-mentioned guidelines (and any others used locally) often proves problematic, the concept of physical checking patients with schizophrenia, especially those receiving antipsychotic medication, needs to be deeply embedded in the routine practice of psychiatrists.
Is metabolic syndrome in schizophrenia a manageable condition?
A variety of behavioural interventions were considered with regards to cardiovascular fitness, smoking and weight gain in patients with schizophrenia. Most studies in this area employed either a naturalistic or experimental design of poor methodology (non-RCT) and were unable to prove one intervention to be superior to another. Among the interventions studied were wellbeing programmes, CBT, nutritional education and diet, weight management and exercise programmes, and various other combinations. Almost all interventions appeared to have some benefit for patients, either towards improving their physical health or their health perception and views.
Pharmacological interventions mainly target smoking behaviour and antipsychotic-induced weight gain, and were supported by more robust studies, mostly of RCT design. BPR and NR appear to work in smoking cessation and reduction; however outcomes were hardly sustainable. Among various agents tested for antipsychotic-related weight gain, metformin, d-fenfluramine, sibutramine and topiramate seem to be the most effective in attenuating this side effect.
Few studies focused on mixed behavioural and pharmacological interventions. The results appear to be quite inconsistent and limited in this area, with some studies favouring pharmacological interventions over behavioural ones, while others show better outcomes by combining both kinds of interventions.
Monitoring, monitoring, monitoring
The cornerstone of early detection and effective management of MetS in schizophrenia is comprehensive monitoring, and a variety of guidelines provide structured schedules for this. Despite the introduction of guidelines for metabolic screening in schizophrenia, metabolic monitoring in routine clinical practice is still unusual. In their impressive meta-analysis of 48 studies, Mitchell and colleagues reviewed changes in monitoring of patients receiving antipsychotics before and after the implementation of relevant guidelines [Mitchell et al. 2012]. They concluded that although guidelines can increase monitoring, most patients still do not receive adequate tests.
Apart from the basic features of MetS (BMI, FPG, FPL, BP), other tests such as ECG and routine blood tests (U&Es, LFTs, FBC, prolactin levels) can complement the laboratory and physical checks of patients with schizophrenia, especially those in receipt of antipsychotic medication. A medical and family history should also be included in this monitoring, and in most cases it is useful to accompany the whole process with regular advice on healthy living. The frequency of monitoring can vary and be adapted to the individual needs of patients. However, it is more important that this process is incorporated in regular psychiatric follow up. The findings that certain ethnic groups, female sex, family history and type of medication all increase the risk of developing MetS can be used by practitioners to identify and target certain individuals who are likely to be at greater risk of life-threatening cardiovascular disease should they develop schizophrenia.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The author declares that there is no conflict of interest.
