Abstract
Over 90% of patients experience peritoneal adhesions after abdominal surgery, necessitating the use of anti-adhesion barriers. To effectively prevent tissue adhesions, strategies that combine physical and chemical blocking are essential. A self-healing hydrogel presents physical blocking by ensuring prolonged retention at the target site. To achieve this, dynamic covalent bonds (DCBs) between polyvinyl alcohol (PVA) and amino phenylboronic acid (APBA) are introduced. In this study, PVA and chondroitin sulfate type C (CS(C)) conjugated with APBA (CSAPBA) are employed to develop a dual-sprayable, self-healing, and anti-inflammatory hydrogel barrier. The hydrogel offers both physical barrier and anti-inflammatory benefits, facilitated by ROS scavenging effect of APBA and macrophage polarization effect of CS(C). As a result, in vivo rat studies show that dual-spraying CSAPBA and PVA forms a self-healing hydrogel that effectively prevents adhesions through both physical and chemical mechanisms. These results demonstrate that the PVA/CSAPBA hydrogel is a promising candidate for preventing post-surgical adhesions.
Introduction
Tissue adhesions are post-surgery problems indicated by abnormal fibrous band connections formed between organs or surrounding tissue. 1 Induced by surgery, natural healing processes generate reactive oxygen species (ROS) that polarize macrophages to the M1 phenotype. These M1 macrophages produce inflammatory cytokines such as TNF-α and IL-6, leading to inflammation that promotes tissue adhesion. 2 Every year in the USA, over 20 million people get invasive operations and 95% of surgeries trigger post-surgery adhesions regardless of surgical sites. 3 Especially in abdominal surgeries, post-operative adhesions may lead to complications including severe pain, organ malfunction, and intestinal obstructions. In fact, 15%–30% of patients may require an additional operation to remove the fibrous bands caused by these adhesions.1,3,4 The only available treatment after the outbreak is adhesiolysis. However, this surgical procedure can result in the recurrence of secondary adhesions or even more serious complications.5,6
To address this issue, effective prevention of peritoneal adhesions requires strategies that combine both physical and chemical blocking. In this study, a self-healing hydrogel provides optimal physical blocking by ensuring prolonged retention at the injury site. 7 This self-healing capability is achieved through the incorporation of dynamic covalent bonds (DCBs) between polyvinyl alcohol (PVA) and amino phenylboronic acid (APBA), which also provides anti-inflammatory properties to the hydrogel. PVA was chosen due to its cis-diol group for DCBs, and it’s in vivo safety and efficacy was demonstrated in previous studies.
In the past few decades, research discussions have primarily focused on clinical therapies for preventing abdominal tissue adhesions. Anti-adhesion barriers have been extensively studied as a means of intervention between opposing tissues during the healing process.8,9 There are two main types of anti-adhesion barriers: solution and solid-based. These barriers are typically composed of either polysaccharides or synthetic polymers.3,8,10 These barrier systems are widely used, but several issues have yet to be fully resolved. 11 For instance, polymer solutions can readily flow down and hardly be maintained on the intended target site.11,12 Solid barriers, such as films or preformed hydrogels, may not adhere properly to tissue surfaces and can easily detach due to their excessive stiffness and strength, making them unsuitable for remaining on wounds. Additionally, these barriers may not be able to fully cover irregular target areas. 3
Therefore, further studies were conducted to develop long-lasting anti-adhesion barriers that are easily applicable to solving the unmet clinical needs in this area. First, reversible DCBs are introduced into the hydrogel network to create a self-healing anti-adhesion barrier. Reversible cis-diol ester bonds between APBA and PVA may serve as a key factor for self-healing.12 –14 By enabling the hydrogel to return to its initial form when its figure is transformed by the motion or derived pressure from surrounding organs, the self-healing capability allows the hydrogel to remain localized at the defective site for an extended time.2,7,15 Moreover, the use of a hydrogel system endows the anti-adhesion barrier an elastic property, which further enhances its ability to remain in place for extended periods. The flexibility of the hydrogel also allows it to cover non-flat geometries, facilitating easier application. 14
To further enhance benefits such as easy and quick application, the multi-functional PVA/CSAPBA hydrogel was developed for a dual-spray-type anti-adhesion barrier. Unlike film-type barriers such as Seprafilm, which face challenges in prolonged maintenance and adequate coverage due to their stiffness, spray-type hydrogel can easily cover irregular surfaces with minimal fluid waste, further enhancing the simplicity and efficiency of the application process.3,16 –19 Each solution for optimal dual-spraying method was refined via concentration-dependent spraying force test on a coordinate plane. Obtaining a suitable viscosity to be sprayed, the hydrogel precursor solutions can be formulated to enable dual-spraying onto the surface, allowing for instant and in situ fabrication of the hydrogel complex.
In addition to physically separating tissues, biochemical blocking mechanism of the anti-adhesion barrier is essential for preventing peritoneal adhesions to address the inflammation response, which is a key mechanism in the formation of peritoneal adhesions. 20 According to the previous studies, the inflammation cycle induces peritoneal adhesion in the peritoneal cavity.20 –22 After a peritoneal injury, a significant number of inflammatory cells, including primarily lymphocytes, monocytes, and neutrophils, are exuded along with peritoneal fluid. Monocytes in the peritoneal fluid can differentiate into macrophages, which accumulate at the injury site and release inflammatory cytokines. 20 Macrophages are categorized into two main types: M1 and M2. M1 macrophages secrete pro-inflammatory factors, such as TNF-α and IL-6, and activate nitric oxide synthase (NOS), contributing to oxidative stress. In contrast, M2 macrophages exhibit anti-inflammatory properties during the later stages of inflammation, facilitating wound healing. 23 Initially, M1 macrophages dominate the damaged peritoneum, releasing pro-inflammatory factors such as TNF-α and IL-6, which bind to receptors on mesothelial cells, triggering the NF-κB signaling pathway and further amplifying the inflammatory response. Therefore, promoting the conversion of M1 macrophages to the M2 phenotype could be an effective strategy for preventing peritoneal adhesions.
Consequently, ROS scavenging and promoting macrophage polarization are the main target points of this study. Among the six major types of CS (type A/B/C/D/E/H), CS (C), which is sulfated at position 6, has been found to have anti-inflammatory effects and can help reduce inflammation at the surgical site.24 –26 CS(C) mitigates the inflammatory response in macrophages by strongly suppressing nitric oxide (NO) and signal cytokines of M1 macrophage such as IL-6 and TNF-α.24,26,27 In contrast, CS(A) is known to activate NF-κB in macrophages and induce TNF-α production.26,28 Compared to CS(A), the CS(C)-induced suppression of pro-inflammatory M1 macrophage was accompanied by an elevated IL-10 level, suggesting a potential switch toward anti-inflammatory M2 state. 27 In addition, APBA can scavenge ROS, which are known to be involved in inflammation and the development of post-operative adhesions.29,30
Therefore, APBA was chosen as a suitable material for DCBs that prevents tissue adhesion resulting from inflammation. CS(C), which promotes the polarization of pro-inflammatory M1 macrophages to anti-inflammatory M2 macrophages, was conjugated with APBA to enhance the anti-inflammatory effects of the hydrogel. This resulted in the formation of CSAPBA, which crosslinks with PVA via DCBs.
By incorporating this CSAPBA into the hydrogel compound, the complex can potentially provide strong anti-inflammatory effects, making it a promising solution for post-operative adhesions. This implies that the PVA/CSAPBA hydrogel can prevent tissue adhesion through both physical and chemical mechanisms.
The prepared hydrogel was tested by performing in vitro and in vivo assays, and the results demonstrated that the sprayable, anti-inflammatory hydrogel is effective in preventing adhesions, especially in a rat ischemic button model. Herein, we report an easy-applicable, multi-functional PVA/CSAPBA hydrogel that has the ability to be stable when sprayed onto anatomically complex surfaces and, at the same time, exhibits suitable mechanical and biochemical properties for preventing peritoneal adhesions. These features make the PVA/CSAPBA hydrogel a highly promising candidate for use as an anti-adhesion barrier for preventing abdominal adhesions (Scheme 1).
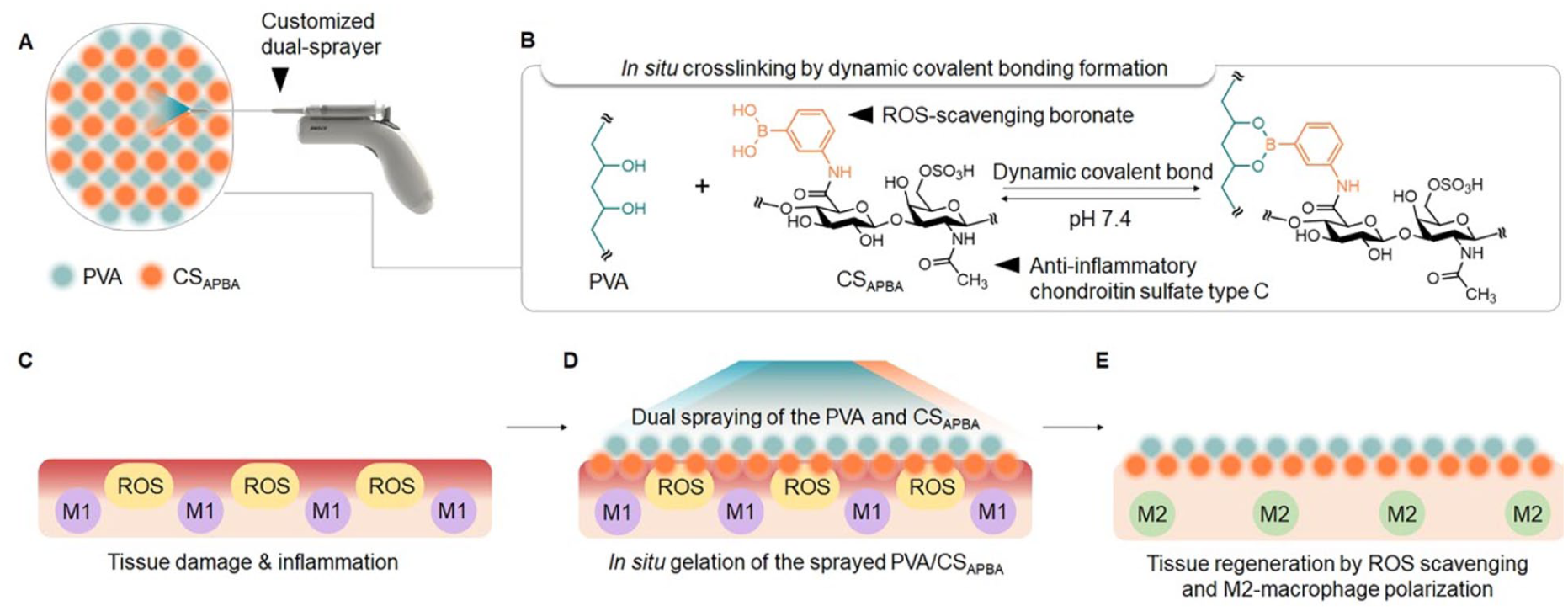
Schematic diagram of the PVA/CSAPBA hydrogel design and the dual-spray treatment: (a) illustration of the PVA/CSAPBA hydrogel treatment, highlighting in situ gelation through dual-spray application, (b) schematic illustration of the PVA/CSAPBA network formation via DCBs, and (c–e) schematic depiction of the PVA/CSAPBA application to the lesion site. The hydrogel prevents tissue adhesion both physically and chemically by providing a barrier between tissues and reducing inflammatory reactions.
Experimental section
Materials
Chondroitin Sulfate C sodium salt and N-Hydroxy succinimide (Sulfo-NHS) were provided by Biosynth (Switzerland). (1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride; EDC), BupH MES buffered saline packs, 2,7-dichlorodihydrofluoscein diacetate (H2DCFDA), Ethidium Homodimer-1 (EthD-1), Calcein AM, Arginase1 polyclonal antibody, Donkey anti-Rat IgG (H + L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor™ 594, Goat Anti-Rat IgG H&L (Alexa Fluor® 488), Goat Anti-Mouse IgG H&L (Alexa Fluor® 488) and Alexa Fluor™ 594 Phalloidin were provided by Thermo Fisher Scientific (USA). Chondroitin Sulfate A sodium salt, Poly (vinyl alcohol; 146–186 kDa), Calcium Hydroxide, Luminol sodium salt, Polystyrene latex beads, Hydrogen peroxide solution, and Lipopolysaccharides were provided by Sigma-Aldrich (USA). 3-Aminophenylboronic Acid (APBA) was provided by TCI chemicals (Japan). Phosphate buffered saline (PBS) was purchased in Dreamcell Co. (Korea). Ethylenediamine tetra acetic acid (EDTA) was provided by ISS (Insung science, Korea). Human/Mouse/Rat iNOS MAb (Clone 2D2-B2) was purchased from Novus Biologicals (USA). Omnipaque™ (iohexol) was obtained from GE healthCare (Korea). Non-absorbable black silk 4/0, Absorbable black silk 3/0 was purchased from Ailee CO. LTD (Korea). LEGEND MAX™ Rat TNF-α ELISA Kit, LEGEND MAX™ Rat IL-6 ELISA Kit, LEGEND MAX™ Mouse TNF-α ELISA Kit, and LEGEND MAX™ Mouse IL-6 ELISA Kit were provided by BioLegend (USA). Trichrome Stain Kit (Connective Tissue Stain) was obtained from Abcam Limited (UK).
Synthesis and characterization of CSAPBA
APBA was conjugated to CS(C) according to the previous published method 31 with some modifications. In brief, CS(C; 500 mg) was dispersed into a MES buffer (50 ml; pH 5.5). EDC (403 mg) and sulfo-NHS (456 mg) were added to the solution in sequence, and then the mixture was stirred for 20 min to activate the carboxylic acid moieties of CS. Subsequently, APBA (326 mg; 2:1 v/v;) was added slowly to the activated CS(C) solution. The mixture was stirred overnight at room temperature. The resulting solution was dialyzed against distilled water for three days after which it was lyophilized for 3 days to obtain CSAPBA. For the evaluation of APBA content in CSAPBA,1H-NMR (AvanceIII HD300 MHz; Bruker, German) analysis was performed. CSAPBA was dissolved in a D2O (1 w/v%) for 1H-NMR analysis.
Fabrication of PVA/CSAPBA hydrogel
PVA/CSAPBA hydrogels were prepared by first dissolving CSAPBA polymers in PBS (6 w/v%) with stirring or vortexing, PVA polymers was separately dissolved in PBS (5 w/v%) with stirring and heating (60°C). Both solutions were simultaneously dual-sprayed to create the PVA/CSAPBA hydrogel formulation.
Rheological studies
Rheological characterization was performed using an Advanced Rheometric Expansion System (ARES, Rheometric Scientific, UK). In brief, 200 µl of hydrogel precursor solutions with various polymer concentrations and component mass ratios were placed on a sample plate. The upper plate was then lowered to create a gap of 0.5 mm. The storage modulus (G’) and loss modulus (G’’) were recorded as functions of irradiation time at angular frequencies ranging from 1 to 100 rad/s and oscillatory strains (γ) ranging from 1% to 100%. Gelation was defined as the time point at which the G’ curves became higher than the G’’ curves. The viscosity (mPa•s, mPa•s−1) of each solution was also assessed at a shear rate of 100 to 103 across various concentrations (0%, 2%, 4%, 5%, 6%, 8%) with n = 3 each.
Sprayability test
Each solution was dyed by mixing non-polar colored ink into the solutions at a concentration of 1% v/v. In the mono-spray test, both PVA and CSAPBA solutions were dyed black. For the dual-spray test, the CSAPBA solution was colored red and the PVA solution was colored blue for easy classification. The solutions were loaded into a dual nozzle spray system and sprayed onto white A4 paper from a constant distance of 10 cm. The sprayed papers were collected and left to dry overnight at room temperature. The dried papers were then scanned, and the droplet distribution was assessed using ImageJ. The spray coverage (%) was quantified by analyzing the fraction of the whole image, employing the XY coordinate methods previously reported. 32
Inverted vial test
To test if the hydrogel formed by mixing two solutions (PVA and CSAPBA), each solution was prepared in various concentrations (CSAPBA at 3, 4, 5, and 6 w/v%; PVA at 3, 4, 5, and 6 w/v%). These solutions were cross-mixed in a glass vial, with 1 ml of each solution, and vortexed to form 16 different combinations of PVA/CSAPBA (n = 3 each). The different PVA/CSAPBA combinations were then inverted and left at room temperature for 10 min to observe gelation.
Gel block fusion test
The self-healing ability of the prepared 5% PVA/6% CSAPBA hydrogel was tested using a cutting and reattaching method. Initially, the hydrogel was cut into two sections, with one piece dyed red and the other dyed blue using India ink for easy observation. These sections were then tightly reattached at room temperature. Subsequently, the rejoined samples were allowed to self-heal for varying durations (0 –6 h) without any external stimulus, and they were covered to prevent water evaporation. After the self-healing treatment, the healed samples (0 h samples) were subjected to multiple stretching cycles using manual manipulation. This process was repeated in triplicate, assessing the integrity and resistance of the hydrogel after self-healing, thereby demonstrating its strength to bear tensile forces.
Pore size analysis of the PVA/CSAPBA hydrogel
The hydrogel was fabricated with an 8 mm diameter and lyophilized for 24 h. The morphology including pore size of PVA/CSAPBA hydrogel was observed by scanning electron microscope (mini-SEM, JCM-6000, JEOL Ltd., Korea).
In vitro anti-oxidant assay
ROS levels were estimated using DCF-DA, a compound that enters cells and reacts with reactive oxygen species (ROS) to produce a green fluorescent compound called dichlorofluorescein (DCF). The estimation of ROS was measured using fluorescence microscopy (EVOS FL Auto 2, Thermo Fisher Scientific, USA).
RAW 264.7 cells were seeded onto 96-well plates at a density of 5 × 103 cells per well using culture medium (DMEM containing 10% FBS) and incubated for 12 h. The cells were then treated with a 500 μM H2O2. After a 1 h incubation period, different treatments including CS(C), CSAPBA, and 5% PVA/6% CSAPBA at a concentration of 0.1% w/v were administered to each group (n = 3 each). After 24 h incubation, cells were washed with PBS three times, and the DCFDA assay was then performed.
For the DCFDA assay, a stock solution of DCF-DA (10 mM) was prepared in methanol and then diluted with culture medium to a working concentration of 100 μM. Cells treated with the materials were washed with PBS and then treated with 100 μM DCF-DA for 30 min at 37°C. Subsequently, cells were lysed with alkaline solutions, and fluorescence intensity was measured using excitation at 485 nm and emission at 520 nm. Cells treated with only the 500 μM H2O2 solution served as a positive control. The number of ROS-induced fluorescent cells was quantified using GraphPad Prism, and the proportion of fluorescent cells was then calculated and presented graphically.
Immunocytochemistry
RAW 264.7 cells were seeded onto 96-well plates at a density of 3 × 103 cells per well using DMEM supplemented with 10% FBS, and incubated for 12 h. The cells were then treated with a LPS solution (100 ng/ml) for 24 h. Then the cells were treated with CS, CSAPBA, and PVA/CSAPBA at a concentration of 0.1% w/v for another 24 h (n = 4 each). After the specific time point, macrophages were incubated overnight at 37°C with primary antibodies anti-mouse iNOS and anti-mouse arginase1 followed by reaction with secondary antibodies Goat Anti-Mouse IgG H&L (Alexa Fluor® 488) and Alexa Fluor™ 594 Phalloidin. Nuclei were counterstained with DAPI, washed with PBS, and observed using a fluorescence microscope (EVOS FL Auto 2, Thermo Fisher Scientific, USA). For quantitative analysis, the percentage of M2 macrophages was determined by counting M2 macrophages among all RAW 264.7 cells.
In vitro cytokine measurement
The supernatants of oxidative stress-induced RAW 264.7 cells were collected from the immunocytochemistry assay, centrifuged at 3000 rpm, and the supernatant was aliquoted. The TNF-α and IL-6 concentrations in the RAW 264.7 cell supernatant were measured using specific murine ELISA kits (Biolegend, 430,907 (mouse TNF-α), 431,307 (mouse IL-6)) according to the manufacturer’s instructions. Samples were assayed (n = 4 for TNF-α, n = 3 for IL-6), and absorbance was read using the TECAN microplate reader (Tecan Group Ltd., Switzerland) at 450 nm.
Cell proliferation assay
RAW 264.7 cells were seeded in 96-well plates at a density of 3 × 103 cells per well, using DMEM containing 10% FBS. After a 24 h treatment of macrophage cells with LPS, the culture medium was replaced with the respective materials for each group (n = 4 each) which included control, LPS-only (100 ng/ml), 0.1% of CS(A), CS(C), CS(C)APBA, and 5% PVA/6% CS(C)APBA. Cell proliferation was evaluated using the Cell Counting Kit-8 (CCK-8; Dojindo, USA) according to the manufacturer’s instructions. Briefly, 10 µl of CCK-8 solution was added to each well and incubated for 2 h. Subsequently, the absorbance at OD 450 nm was measured using a microplate reader.
Macrophage aggregation assay
The macrophage aggregation assay was performed following published protocol with slight modifications. 33
Murine macrophage isolation and activation
All experimental procedures were approved by the Institutional Animal Care and Use Committee (IACUC) of the Seoul National University (IACUC number: SNU-231206-4-1). Experiments were conducted in accordance with guidelines established by the Committee.
After anesthetizing the C57BL/6 mouse (Female, 8 weeks, Nara Biotech, Korea), an incision was made in the outer skin of the peritoneum to expose the peritoneal wall. Ice-cold PBS containing 2 mM EDTA was then injected into the peritoneal cavity using a syringe. Following the injection, gentle stomach massage was performed to facilitate cell detachment within the peritoneal cavity. The peritoneal fluid was carefully collected with a syringe, taking care not to damage other organs. This process was repeated three times to collect a total of 8 ml of peritoneal fluid. If additional fluid was needed, the peritoneum was incised further to collect any remaining fluid, ensuring no blood was present in the collected fluid. The collected fluid was then centrifuged at 1500 rpm (4°C, 8 min). After centrifugation, the supernatant was discarded, and the cell pellet was resuspended in DMEM containing 10% FBS and seeded on coverslips placed in 100 mm dish. After a 3 h incubation at 37°C, approximately 90% of the adhered cells were large peritoneal macrophages.
Large peritoneal macrophage aggregation assay
Large peritoneal macrophages were detached from the dish bottom using Versene™ at 4°C. After transferring the detached cells to a warmed glass vial at 37°C, cells were treated with 100 μM CS(C), 100 μM CSAPBA (treatment groups), or 2 mM calcium (negative control) dissolved in PBS (n = 3 each), and continuously stirred at 400 rpm. After 10 min, cell aggregation was observed via microscopy.
In vitro cytocompatibility assay
The cytocompatibility of the 5% PVA/6% CSAPBA hydrogel was evaluated on NIH3T3 cells using the Live/Dead assay. For day 1 analysis, NIH3T3 cells were cultured in DMEM containing 10% FBS and 0.1% of the PVA/CSAPBA hydrogel (n = 4). After 24 h, cell culture medium was removed, and 1 ml of PBS containing calcein AM and ethidium homodimer was added to the NIH3T3 cells and incubated at 37°C for 30 min. Microscopic images of Live/Dead assay were obtained with a fluorescence microscope (EVOS FL Auto 2, Thermo Fisher Scientific, USA). For day 3 analysis, NIH3T3 cells were cultured in the same culture medium containing PVA/CSAPBA (n = 4) for 72 h, and the same process was repeated.
In vivo biocompatibility assay
All experimental procedures were approved by the Institutional Animal Care and Use Committee (IACUC) of the Seoul National University (IACUC number: SNU-231206-4-1). Experiments were conducted in accordance with guidelines established by the Committee.
The in vivo biocompatibility of 5% PVA/6% CSAPBA was assessed by quantifying subcutaneous luminol activity, following previously published methods.34,35
For in vivo detection of biomaterial-induced reactive oxygen species (ROS), 100 μl of PBS (negative control), 5% PVA, 6% CSAPBA, 5% PVA/6% CSAPBA, 4% PS (polystyrene beads), and 5% PVA/6% CSAPBA + 4% PS (n = 4 each) were injected separately using an 18-gage needle into six different subcutaneous sites on the dorsal side of C57BL/6 mice (Female, 8 weeks, Nara Biotech, Korea). To minimize background fluorescence interference, mice were treated with an AIN-93G diet. Prior to injections, mice were anesthetized with isoflurane inhalation.
After 3 days, mice were anesthetized with isoflurane and 200 mg • kg−1 of luminol sodium salt (5-amino-2,3-dihydro-1,4-phthalazinedione, sodium salt) resuspended in 100 μl of PBS was injected into the peritoneum. After 15 min, animals were imaged using an IVIS Spectrum 200 (PerkinElmer Inc., USA) with a 3 min exposure for bioluminescence or a 1 s exposure for fluorescence. Luminescence intensity was quantified using Living Image®, with a 5 mm diameter region of interest (ROI) serving as the quantification area. The signals were normalized against the background signal from rodent skin. This approach allowed visualization and quantification of ROS levels induced by the biomaterials.
In vivo retention and degradation assay
All experimental procedures were approved by the Institutional Animal Care and Use Committee (IACUC) of the Seoul National University (IACUC number: SNU-231206-4-1). Experiments were conducted in accordance with guidelines established by the Committee.
For the in vivo retention assay, animals were anesthetized with isoflurane and 500 μL of the 5% PVA/6% CSAPBA hydrogel (n = 3) was subcutaneously injected into the dorsal region of the BALBc/Nu mouse (Female, 8 weeks, Nara Biotech, Korea). Changes in the volume of the hydrogel were monitored using a Micro-CT Imaging System (Quantum GXII, Perkin Elmer, USA) from day of injection to day 12 post-injection.
Rat ischemic button model
All experimental procedures were approved by the Institutional Animal Care and Use Committee (IACUC) of the Seoul National University (IACUC number: SNU-231206-4-1). Experiments were conducted in accordance with guidelines established by the Committee. The ischemic button model was employed to replicate intraperitoneal adhesions. Sprague Dawley rats (Female, 8 weeks, DBL, Korea), were divided into three groups: (1) control, (2) Seprafilm (3) PVA/CSAPBA with n = 4 animals per group. The animals were anesthetized using isoflurane and were placed on a heating pad at 38℃. The abdominal fur was removed using an electric razor and hair removal cream. For disinfection, iodine and 70% ethyl alcohol were applied. To alleviate postoperative pain, carprofen (4 mg/kg; Rimadyl, Pfizer) was administered via subcutaneous injection before the surgery.
The ischemic button model was created following the previous published method. 36 The technique involves making a midline incision and creating four ischemic buttons on each side of the abdominal wall (eight buttons per animal), positioned 1 cm lateral to the midline incision and spaced 1 cm apart. Each button was formed by grasping a small 5 mm portion of peritoneal wall and ligating the base of the tissue with a non-absorbable suture (Ailee black silk 4/0). After tying the suture, it was wrapped around the entire base of the tissue and tied again to create the ischemic button. This procedure was repeated for all eight buttons. Immediately after button formation, materials corresponding to each group (control, Seprafilm, PVA/CSAPBA) were treated on the entire peritoneal wall, which contains ischemic buttons. PVA/CSAPBA was applied by using a dual-spray applicator. Prior to the dual-spray application, the sprayer nozzles were sonicated three times in distilled water and 70% alcohol, then dried in an oven to ensure sanitization. Subsequently, the abdomen and the skin were closed along the midline using absorbable sutures (3-0 Polyglactin 910 Vicryl® Ethicon). Following the procedure, the animals were placed on a heating pad (38℃) to aid in postoperative recovery.
A week after the surgery, rats were sacrificed for the evaluation of peritoneal adhesion severity. Serum and peritoneal wall samples were harvested for anti-inflammation assessment. The quantitative analysis of adhesion formation was scored by calculating the amount of peritoneal adhesions formed to the number of remaining buttons among eight buttons for each animal. Some buttons may slip away because of postoperative mobilization. The picture of each peritoneum was taken, and standard scoring system (Launder score) was utilized for the evaluation. 37
In vivo cytokine measurement
Rat serum samples were collected from peritoneal adhesion models on day 7. After resting at RT for 30 min, the serum was centrifuged at 1000–2000 xg at 4°C using a serum separation gel tube, and the supernatant was aliquoted. TNF-α and IL-6 concentrations in rat serum (n = 4) were measured using specific murine ELISA kits (Biolegend, 438,207 (rat TNF-α), and 437,107 (rat IL-6)) according to the manufacturer’s instructions. Samples were assayed in quadruplicates, and absorbance was read using the TECAN microplate reader (Tecan Group Ltd., Switzerland) at 450 nm.
Histology
To assess the degree of adhesions in the control group, Seprafilm-treated rats, and PVA/CSAPBA hydrogel-treated rats, all rats were observed 1 week after administration of each treatment. After sacrificing rats, the affected areas (peritoneal wall, liver, etc.) were harvested and fixed immediately in 4% paraformaldehyde. Following paraffin embedding and sectioning, the tissues were stained with Masson’s Trichrome (MTC) Stain using the Trichrome Stain Kit (Abcam, ab150686) for histopathological analysis. The experiment was repeated in triplicate.
Immunohistochemistry
Eight-micrometer-thick sections of paraffin-embedded tissue were heated in a 60°C oven for 10 min and deparaffinized in stages. After deparaffinization, the sections were treated with Tris-EDTA buffer (pH 9.0) at 95°C for 10 min to restore epitopes. Following cooling to room temperature, the sections were washed three times with PBS. Subsequently, the tissues were treated with a permeabilization solution (a mixture of 1% BSA and 0.3% Triton X) for 10 min. After three more washes with PBS, the sections were incubated with 10% normal goat serum (Kormed, Korea) for 1 h at room temperature. Following another three PBS washes, the tissues were treated with primary antibodies: Human/Mouse/Rat iNOS MAb (dilution 1:100) and Human/Mouse/Rat Arginase1 polyclonal antibody (dilution 1:100) for 1 h at room temperature or overnight at 4°C. After three PBS washes, the sections were treated with the appropriate secondary antibody (dilution 1:500) for 1 h in a dark environment, followed by three final washes in PBS. Finally, the sections were mounted on slides using a mounting medium with DAPI staining (Fluoroshield). All processes were repeated in triplicate. Microscopy images were captured at 20X magnification using the Eclipse Ti2 (Nikon Imaging Corporation, Korea). These images were then processed with ImageJ software to measure the immunoreactive intensity. The percentage of fluorescence-positive areas relative to the total area was calculated. For quantitative analysis, the ratio of M2 to M1 macrophages was determined by counting both M2 and M1 macrophages and dividing the number of M2 cells by the number of M1 cells.
Statistical analysis
All results are presented as mean ± standard deviation (s.d.). Comparisons between two groups were conducted using a two-tailed Student’s t-test. For comparisons across multiple groups, one-way analysis of variance (ANOVA) was utilized. Statistical significance was defined as *p < 0.05, ** p < 0.01, and *** p < 0.005.
Results
Synthesis and characterization of CSAPBA
APBA was conjugated to CS through the formation of EDC/NHS-mediated amide bonds between the primary amino group of APBA and the carboxylic acid group of CS (Figure 1(a)). The synthesis of CSAPBA was confirmed by 1H NMR spectroscopy. The 1H NMR spectrum of CSAPBA (Figure 1(b)) revealed distinct signals corresponding to the protons of the N-acetyl group of CS (a: 1.8 ppm) and the aromatic protons of APBA (b: 7.3–7.4 ppm; c: 7.6–7.7 ppm). The degree of substitution (DS) of CSAPBA was determined using this 1H NMR spectroscopy, revealing a substitution level of 14.81%. 38

Synthesis and characterization of CSAPBA: (a) schematic illustration of CSAPBA synthesis via amide crosslink and (b) the 1H-NMR result of CSAPBA.
Viscosity and spray test of the hydrogel precursors
In this study, PVA/CSAPBA hydrogel was applied using a dual-spray applicator to enable in situ gelation and facile application. Following the approach of Song et al., Individual PVA and CSAPBA solutions were loaded into a customized dual-sprayer and sprayed simultaneously but separately, integrating during flight and delivering to the target site in gel form. 39 For dual-spraying, both PVA and CSAPBA solutions are required to be simultaneously dual-sprayed, leading to in-situ gelation. This spray method would allow for even application onto lesions with uneven surfaces with minimal fluid waste.
The low viscosity of each solution makes them suitable for spraying onto lesions, providing great spread ability compared to stiff film types, which are commonly used in clinical application. Therefore, it is essential to conduct viscosity assessments for each solution to optimize the concentration of the solutions for the dual-spray method.
The viscosity of varying concentrations of PVA (Figure 2(a) and (b)) and CSAPBA solutions was measured (Figure 2(c) and (d)). The viscosity of each solution at the maximum shear rate (103) was specifically depicted in Figure 2(b) and (d). As shown in Figure 2(b), the average viscosity of PVA at various concentrations and the maximum shear rate was 6.77, 46.62, 87.34, 240.23, and 905.86 mPa·s for 2%, 4%, 5%, 6%, and 8% PVA solutions, respectively. In Figure 2(d), the average viscosity of CSAPBA at various concentrations and the maximum shear rate was 4.36, 2.57, 4.03, and 7.87 mPa·s for 2%, 4%, 6%, and 8% CSAPBA solutions, respectively. Upon comparing the viscosity of the two solutions at the maximum shear rate, it was observed that PVA exhibited a substantial viscosity alteration with changing concentration, unlike CSAPBA solutions.

Optimization of the PVA/CSAPBA hydrogel formulation for spraying. Viscosity measurement of (a) PVA and (c) CSAPBA. Graphical representation of viscosity at the maximum shear rate (103 S−1), (b) PVA and (d) CSAPBA, (e) sprayability of PVA solution, (f) graphical illustration of sprayed PVA solution on a coordinate plane, (g) quantitative analysis of the coverage area of sprayed PVA, (h) sprayability of CSAPBA solution, (i) graphical illustration of sprayed CSAPBA solution on a coordinate plane, and (j) quantitative analysis of the coverage area of sprayed CSAPBA.
After analyzing the viscosity of each solution at different concentrations, a spray test was conducted. Each solution was sprayed onto a piece of white A4 paper from a distance of 10 cm. The sprayed papers were dried and subsequently scanned for visualization on an XY coordinate plane. The coverage of each solution was analyzed by calculating the occupied area. In the case of PVA, there was a notable correlation between concentration and coverage, with a decrease in coverage with an increase in PVA concentration (Figure 2(e)–(g)). In Figure 2(g), the average spray coverage area for PVA solutions was 26.99%, 17.53%, 8.61%, and 8.17% at concentrations of 3%, 5%, 7%, and 9%, respectively. Conversely, for CSAPBA, except for 2%, there was no significant difference in the coverage area with an increase in concentration (Figure 2(h)–(j)). In Figure 2(j), the average spray coverage area for CSAPBA solutions was 36.85%, 25.98%, 30.36%, and 30.13% at concentrations of 2%, 4%, 6%, and 8%, respectively. Therefore, the optimal concentration for achieving dual-spray capability should be determined based on the optimized concentration of PVA. Referring to Figure 2(b) and (e), it was established that the optimal concentration of PVA should be under 7% for optimal spraying. The CSAPBA exhibited a similar spray coverage area across different concentrations, except for the 2% group.
Optimization and characterization of the PVA/CSAPBA hydrogel
The inverted vial test was performed to optimize the concentration of each solution for gelation to obtain the PVA/CSAPBA hydrogel (Figure 3(a)). Specifically, groups with PVA - CSAPBA concentrations of 5%–6%, 6%–5%, and 6%–6% demonstrated firm gelation (Figure 3(b)).

Optimization of the dual-sprayability of the PVA/CSAPBA hydrogel: (a) optimization of PVA and CSAPBA concentrations for gelation using inverted vial tests, (b) graphical depiction of the optimized PVA and CSAPBA concentrations required for gelation of PVA/CSAPBA hydrogel, (c and d) storage modulus (g) and loss modulus (g) of PVA/CSAPBA hydrogel at various PVA concentrations and fixed CSAPBA concentration (6%) measured at different angular frequencies and oscillatory strains respectively. Dual-sprayability of PVA/CSAPBA hydrogel: (e) sprayability of different concentrations of PVA and CSAPBA to confirm in situ gelation, (f) graphical illustration on a coordinate plane and (g) quantitative analysis of the coverage area. Based on the results of (e–g) the formulation consisting of 5% PVA and 6% CSAPBA has been identified as the optimal option, (h) gel block fusion test and (i) stretching test of the 5% PVA/6% CSAPBA hydrogel.
To further validate gelation behavior at different concentrations, G’ (storage modulus) and G” (loss modulus) values were examined under various angular frequency and oscillatory strain conditions (Figure 3(c) and (d)). For instances of firm gelation, the G’ value, representing the elastic modulus, should surpass the G” value, which represents the viscous modulus. 40 For instance, at an angular frequency of 10 rad/s, the PVA 5% – CSAPBA 6% hydrogel exhibited a G’ value of 73.4967 and a G’” value of 50.4681. At a 10% oscillatory strain, the same hydrogel showed a G’ value of 113.579 and a G’’ value of 100.986. This rheological analysis aids in understanding the mechanical properties and stability of the hydrogel at different concentrations and conditions.
Furthermore, for optimal ease of spraying, it is preferable to use a lower concentration of PVA solution. Therefore, the optimal concentration of PVA should be under 7% (Figure 2(e)). Conversely, in the case of CSAPBA, the viscosity of each solution was considerably lower than that of PVA, and the concentration-dependent variation was not as pronounced in the 4%–8% groups (Figure 2(j)). Hence, the concentration optimization for CSAPBA should be determined based on the optimized concentration of PVA, which is determined to be 5%.
In the dual-spray test, the PVA 5% - CSAPBA 6% hydrogel demonstrated optimal gelation (Figure 3(e)). The spray coverage area is depicted in the XY coordinate system and analyzed quantitatively (Figure 3(f) and (g)). The average coverage area for each solution (3%, 4%, 5%, and 6% PVA, with a fixed CSAPBA concentration of 6%) was 24.803%, 17.962%, 11.983%, and 5.862%, respectively (Figure 3(g)). The lower the concentration of PVA, the wider the spray coverage area. For instance, within the PVA 6% - CSAPBA 6% hydrogel group, the PVA exhibited atomization due to its excessively high concentration (Figure 3(e)). However, for gelation to occur, the PVA concentration must be above 5%. Therefore, the concentration of each solution was fine-tuned, resulting in the optimized ratio of PVA 5%- CSAPBA 6%.
This combination ensures effective sprayability and desirable coverage during the dual spray application. The dual-spray in situ gelation technique facilitated an easy application of the PVA/CSAPBA hydrogel to the lesion site. The gelation process was completed in under 30 s, indicating rapid gelation.
The self-healing properties facilitated by the dynamic covalent bond between PVA and APBA conjugated to CS were evaluated by performing a self-healing test (Figure 3(h) and (i)). Initially, the PVA/CSAPBA hydrogel was cut into two blocks, and then the blocks were reattached, immediately initiating the self-healing process. Over time, the self-healed area, indicated by the purple color, increased, demonstrating the material’s ability to repair itself (Figure 3(h)). To further demonstrate its excellent healing capability, the hydrogel was immediately subjected to stretching following reattachment (Figure 3(i)). After multiple stretches, the volume of the purple hydrogel increased, highlighting its robust self-healing behavior. This implies that the material undergoes reversible changes in viscosity in response to applied stress, demonstrating it as a promising material for anti-adhesion barriers where self-repair and adjustable viscosity are desirable features.
Furthermore, the porosity of the developed system was analyzed. The SEM results showed that the pore size of the PVA/CSAPBA was found to be six times larger than the PVA-only hydrogel (Supplemental Figures 1A-G). The average pore size of the PVA-only hydrogel was 19.5 μm, whereas the PVA/CSAPBA hydrogel had an average pore size of 133.25 μm (Supplemental Figure 1G). The larger pores exhibited by PVA/CSAPBA hydrogel when used as an anti-adhesion barrier would favor the close adhesion of the patch to the peritoneal wall. 41 Furthermore, this substantial increase in pore size may offer potential advantages for supporting cell integration and, consequently, could be beneficial for tissue regeneration applications. 42 Therefore, this asymmetrical porous microstructure may effectively promote anti-adhesion and pro-healing of the injured peritoneal wall.
ROS scavenging effect of APBA
After optimizing the sprayable viscosity and gelation concentration, the biological effects of each material were analyzed. Boronic acid has been reported to scavenge ROS,30,43 based on this the effect of APBA treatment in reducing the ROS level induced by an oxidative stress trigger in the macrophage cell line RAW 264.7 was analyzed. For this purpose, hydrogen peroxide (H2O2) was included as an oxidative stress trigger (Figure 4(a)). The RAW 264.7 cells were incubated with different treatment groups (control, H2O2-only, PVA, CSAPBA, and 5% PVA/ 6% CSAPBA). The incubated cells were then treated with H2O2 and the ROS level was evaluated using the ROS probe 2’,7’-dichlorodihydrofluorescein diacetate (H2DCFDA).

ROS-scavenging effect of APBA: (a) graphical illustration of the DCFDA assay, (b) microscopic image of DCFDC + RAW 264.7 cells (green cells) after addition of different treatment groups, and (c) quantitative analysis to assess the percentage of DCFDA+ cells indicating ROS generation.
As shown in Figure 4(b), the decrease in fluorescence intensity was observed in APBA-treated cells indicating a reduction in ROS levels. The percentage of DCFDA+ cells for each group was as follows: 26.02% (control), 81.89% (H2O2), 83% (PVA), 76.332% (CS), 4.094% (CSAPBA), and 3.486% (PVA/CSAPBA). Considering that all groups containing APBA exhibited ROS reduction, these results collectively provide evidence of the ROS-scavenging effect of the PVA/CSAPBA hydrogel, likely owing to the presence of APBA (Figure 4(c)). Therefore, the PVA/CSAPBA hydrogel was shown to prevent ROS-mediated inflammation.
In vitro and ex vivo anti-inflammatory and anti-aggregation properties of CS(C) in the PVA/CS(C)APBA hydrogel
Chondroitin sulfate type C (CS(C)) has been reported to induce polarization of macrophages from M1 to M2 (Figure 5(a)), thereby exerting an anti-inflammatory effect in contrast to other forms of chondroitin sulfate, such as CS type A (CS(A)).24,44

The in vitro anti-inflammatory activity of CS (C): (a) visual representation showing the macrophage polarization from the pro-inflammatory M1 phenotype to the anti-inflammatory M2 phenotype induced by CS(C), (b) overview of the immunocytochemistry procedure employed in the study, (c) immunocytochemistry results demonstrating macrophage polarization from M1-like state (red cells) to the M2-like state (green cells), (d) quantitative assessment of the percentage of M2-like macrophages, (e and f) in vitro analysis of signal cytokines for M1 macrophages (TNF-α, IL-6) tested by ELISA, and (g) the in vitro anti-inflammatory effectiveness of CS(C) demonstrated through macrophage viability.
To investigate the in vitro impact of CS(C) on macrophage polarization, RAW264.7 macrophages were initially exposed to LPS to induce inflammation (Figure 5(b)). Subsequently, these macrophages were cultured with various materials, corresponding to groups such as control, LPS, CS(A), CS(C), CS(C)APBA, PVA/CS(C)APBA. Following the treatment, the polarization of macrophages was assessed using iNOS and Arginase 1, which serve as markers for M1 and M2 phenotypes, respectively.
It was observed that the M1 and M2 phenotypes of macrophages exhibit significantly different cell morphologies, consistent with previous research. 45 The M0 macrophages exhibited a small, round shape with a few short protrusions. Exposure to LPS for 24 h transformed these macrophages (M0) into large, irregular, flat-shaped cells with longer pseudopodia (M1). The introduction of CS(C) induced the polarization of M1 macrophages toward the M2 phenotype, which is characterized by cell elongation (Figure 5(c)). Cell elongation was defined as having a long axis length of more than twice that of the short axis.
Following that, the phenotypic distribution of M2-like macrophages in each group was examined.
The immunofluorescence staining revealed increased expression of iNOS (red fluorescence), a marker for M1 macrophages, in the groups treated with LPS. However, upon the inclusion of CS(C), there was a reduction in the expression of iNOS and an augmentation in the expression of the M2 marker, arginase 1 (green fluorescence), as illustrated in Figure 5(c). Since all cells are macrophages (RAW 264.7), the percentage of M2-like macrophages was evaluated (Figure 5(d)). Non-M2 cells indicate either M0 or M1-like phenotypes. Each group showed the following average percentage of M2-like cells: 1.841% (control), 0.0365% (LPS), 0.0169% (CS(A)), 100% (CS(C)), 100% (CS(C)APBA), and 100% (PVA/CS(C)APBA). The groups treated with CS(C) exhibited a significantly higher rate of M2-like polarization compared to other groups. These results provide evidence that PVA/CSAPBA hydrogel containing CS(C) actively promotes the polarization of macrophages into the M2-like phenotype.
Furthermore, the secretion of signal cytokines of pro-inflammatory M1 macrophages (TNF-α, IL-6) from the treated RAW 264.7 cells was measured by ELISA in different groups with or without the treatment of PVA/CSAPBA hydrogel (Figure 5(e) and (f)). The average expression levels of TNF-α for each group (Control, LPS, CS(A), CS(C), CS(C)APBA) were 50.221, 1224.748, 1230.306, 383.262, and 235.607 pg•ml−1, respectively (Figure 5(e)). In Figure 5(f), the average expression levels of IL-6 for each group (Control, LPS, CS(A), CS(C), CS(C)APBA) were 62, 178.959, 160.251, 26.403, 44.586, and 36.659 pg•ml−1. The average expression levels of TNF-α and IL-6 in the PVA/CSAPBA group were lowered by approximately 4-fold and 5-fold compared to the LPS group, respectively (Figure 5(e) and (f)). In conclusion, compared to the control, LPS, CS(A) groups, both inflammatory cytokines were expressed at lower levels in the PVA/CSAPBA group. A statistically significant difference was observed between the control, LPS, CS(A) groups and the CS(C)-containing groups, indicating that PVA/CSAPBA effectively downregulated inflammation.
Moreover, it is established that inflammation can adversely affect cell viability. 46 To validate the anti-inflammatory effect of CS(C), the viability and proliferation capability of RAW 264.7 cells were assessed using the CCK-8 assay. Following a 24 h treatment of macrophage cells with LPS to induce ROS, the culture medium was replaced with each respective material-incubated medium corresponding to the control (cells alone), LPS-only, CS(A), CS(C), CS(C)APBA, and PVA/CS(C)APBA groups. The findings from the CCK-8 proliferation assay demonstrated a decrease in absorbance at OD 450 nm after treating macrophage cells for 24 and 48 h with LPS, indicating a significant inhibition of cell proliferation (Figure 5(g)). Notably, cell proliferation was restored when the cells were treated with CS(C). The results of the CCK-8 assay further demonstrated that PVA/CSAPBA hydrogel can effectively augment macrophage survival rates compared to the LPS-only or CS(A) treated groups. The groups containing CS(C; CS(C), CS(C)APBA, and PVA/CS(C)APBA) improved the macrophage survival rate, likely due to the reduction of the macrophage inflammatory response.
Following the in vitro assessments, an ex vivo macrophage experiment was performed to further validate the additional function of CS(C). Abdomen injury triggers the aggregation of macrophages within the mammalian body cavities, which can lead to the formation of peritoneal adhesions due to collagen deposition (Supplemental Figure 2A). 33 Following injury, macrophages quickly aggregate at the wound site or on the surface of nearby abdominal organs forming bridges between the injured area and intra-abdominal organs like the omentum or small intestine. Within 3 days, these macrophage aggregates become covered with a mesothelial lining. After 7 days, collagen begins to be deposited within these macrophage aggregates. Both the mesothelial lining coverage and collagen deposition are considered key indicators of peritoneal adhesion formation. 33
In line with the observation that negatively charged materials have the potential to decrease macrophage aggregation, 33 an investigation was conducted to assess whether negatively charged CS(C) could reduce macrophage aggregation. The macrophage aggregation assay involved the use of murine large peritoneal macrophages (LPM) obtained from C5BL/6 mice (Supplemental Figure 2B). Following incubation, these murine LPMs were treated with various materials, including Ca2+ (positively charged) and EDTA (negatively charged; Supplemental Figure 2C). The CS(C) and CSAPBA groups also included Ca2+ to promote macrophage aggregation. Quantitatively analyzed in Supplemental Figure 2D, the Ca 2 ⁺ group showed an average aggregated macrophage percentage of 58.3275%. In contrast, the EDTA group showed an average aggregated macrophage percentage of 7.788%, while the CS(C) and CSAPBA groups showed 16.215% and 13.793%, respectively. The results reveal a substantial reduction in macrophage aggregation in the group containing CS(C). The EDTA-treated group exhibited the most significant reduction in aggregation compared to all CS(C)-containing groups, which could be due to the highly negative charge of EDTA. 47 Therefore, it may imply superior performance in reducing aggregation compared to other groups, as supported by our previous findings on the influence of negatively charged materials on macrophage aggregation. 39
Taken together, these evaluations suggest that the PVA/CSAPBA hydrogel has the potential to play a role in preventing macrophage aggregation, thereby mitigating tissue adhesion.
In vitro and in vivo biocompatibility of the PVA/CSAPBA hydrogel
After demonstrating the biological efficacy of the hydrogel in vitro, a cytotoxicity assay was conducted in vitro. Microscope images of live (green) and dead (red) NIH3T3 cells was obtained by Live/Dead assay, stained with calcein AM and ethidium homodimer-1 (Figure 6(a)). The viability of cells after being cultured in culture medium (control) and culture medium containing PVA/CSAPBA hydrogel for 1 and 3 days was visualized and analyzed (Figure 6(b) and (c)). In Figure 6(c), all groups on day 1 showed 100% cell viability. On day 3, the control group showed 99.555% of cell viability, and the PVA/CSAPBA group showed 99.778% of cell viability. Consequently, there was no significant difference in the viability between the control and hydrogel-treated groups. This suggests that the hydrogel formulation does not compromise cells.

In vitro and in vivo toxicity tests of PVA/CSAPBA hydrogel: (a) graphical summary of Live/Dead assay in vitro. NIH3T3 cells treated with PVA/CSAPBA hydrogel observed on day 1 and 3, (b) microscopic image of live (green) and dead (red) cells, (c) quantitative assessment of cell viability percentage, (d) graphical representation illustrating the experimental procedure and outcome data of luminol inflammation test in vivo, (e) IVIS result of bioluminescence and (f) Quantitative analysis from IVIS results.
To evaluate biocompatibility in vivo, the release of ROS produced by inflammatory cells in response to biomaterials was measured using a luminol assay. 48 A bioluminescence animal imaging method (IVIS) was employed to observe ROS released in response to subcutaneously implanted materials in live mice. In the presence of peroxide, ROS oxidizes luminol (5-amino-2,3-dihydro-1,4-phthalazinedione, sodium salt), generating an excited product known as 3-aminophthalate that emits light at 425 nm (bioluminescence). 49 For the luminol assay, various formulations were administered to mice via dorsal subcutaneous injection 3 days prior to imaging. PBS served as the control group, and polystyrene (PS) was chosen as a potent inflammation-inducing material, representing the positive control group. 35 Additionally, a PS + PVA/CSAPBA hydrogel group was included to assess whether the hydrogel could mitigate the strong inflammation induced by PS. In vivo luminescent imaging of mouse models with each material was conducted 20 min after intraperitoneal administration of luminol. The outcome revealed that PVA, CSAPBA, and PVA/CSAPBA hydrogel did not induce inflammation (Figure 6(e) and (f)). However, the PVA/CSAPBA hydrogel did not alleviate the high level of inflammation induced by PS but it is observed that each component used in the hydrogel formulation did not induce inflammation, indicating its biocompatible nature.
Effective anti-adhesion barriers should remain in place to separate injured tissue surfaces from other tissues for a period of 5–7 days as this duration is necessary for re-mesothelialization to occur. 50 Additionally, collagen deposition due to macrophage aggregation may occur within 7 days. 33
To confirm the durability of the PVA/CSAPBA hydrogel, 500 μL of the hydrogel was injected into the dorsal region of mice, and changes in the size of the hydrogel were monitored (Supplemental Figure 3A). The hydrogel figure remained visible up to day 7, and a subsequent decrease in the volume of the hydrogel was observed from the day of implantation to day 12 post-injection (Supplemental Figures 3B-F). The analysis of volume changes involved measuring the area and maximum height of the hydrogel in vivo. In Supplemental Figure H, the average PVA/CSAPBA hydrogel area and maximum height was found to be 620.586 mm2; 9.583 mm on day 0, which reduces to 483.56 mm2; 4.604 mm on day 3, 168.513 mm2; 2.877 mm on day 7 respectively. The PVA/CSAPBA hydrogel demonstrated retention for more than 7 days and completely disappeared by day 12 which is a critical period for tissue adhesion prevention. This suggests that the PVA/CSAPBA hydrogel holds promising potential for effective anti-adhesion functionality during this crucial initial 7-day timeframe. Additionally, the hydrogel degrades within 2 weeks, indicating its biodegradability after completing its role as a barrier in preventing peritoneal adhesion.
In vivo efficacy of the PVA/CSAPBA hydrogel
The efficacy of PVA/CSAPBA hydrogels in preventing adhesions after abdominal surgery was investigated using a rat peritoneal ischemic button model (Figure 7(a)). For imaging purpose, PVA was dyed blue and CSAPBA was dyed red. Following dual spraying, a purple hydrogel was formed by in situ gelation (Figure 7(b)). The ischemic button model has been well proven to be more proficient in generating a robust tissue adhesion compared to other standard abdominal adhesion models, and reliably induces severe peritoneal adhesions through inflammation and tissue damage. 51 A midline incision was made, and four ischemic buttons were created on each side of the peritoneal wall, totaling eight ischemic buttons per animal. Ischemic buttons were generated using a stick tie suture technique to induce a region of ischemia along the peritoneal wall (Figure 7(c)). Immediately after inducing ischemic buttons, rats were treated with either (i) dual-sprayed PVA/CSAPBA hydrogel (1 ml for each peritoneal wall), (ii) a commercially available adhesion barrier, Seprafilm (4 cm2), or (iii) received no treatment before closing the midline incision. Seprafilm is a commercially available film-type physical barrier that prevents adhesion formation between opposing tissues and was used as a positive control. Seprafilm is an FDA-approved adhesion barrier, and its efficacy has been clearly demonstrated for the prevention of abdominal and pelvic adhesions. 52 These treatments were directly applied over the ischemic buttons created on the peritoneal surface. In the case of PVA/CSAPBA hydrogel, each solution was dual-sprayed for the treatment, respectively (without dyeing, except for Figure 7(b) and the rightmost upper panels of 7C, where the hydrogel was dyed for imaging purposes). The hydrogel was gelled in situ and could completely cover the peritoneal wall in less than 30 s. Seprafilm was also applied directly to the lesion for treatment (Figure 7(c)). A week later, rats were sacrificed to assess the anti-adhesive efficacy of our PVA/CSAPBA hydrogel adhesion barrier. A midline incision was made to visualize the extent of adhesion formation before conducting blunt dissection. Sharp or blunt dissection techniques were applied to the tissue adhesions to evaluate the in vivo effectiveness of the adhesion barrier. Serum from rats was collected via cardiac puncture for evaluation of inflammatory cytokine levels. Adhesion scores were assigned using a double-blind clinical scoring system on a scale from 0 to 5 (Figure 7(d)). Subsequently, adhesions were dissected, and the peritoneal wall was preserved for histological analysis.

Prevention of peritoneal adhesions in rat ischemic button model using PVA/CSAPBA: (a) an illustrative image showing the dual-spraying of PVA/CSAPBA hydrogel onto the ischemic buttons. Four ischemic buttons were created on each side of the rat’s peritoneal wall, totaling eight buttons per rat, (b) photograph of dual-sprayed PVA/CSAPBA hydrogel. The blue PVA solution and red CSAPBA solution were dual-sprayed, resulting in the formation of a purple hydrogel, (dyed for imaging purpose), (c) representative image showing ischemic buttons on the peritoneal wall treated with different groups on the day of application and on day 7. White arrows in the images indicate adhesions, (d) a standard clinical scoring system for the assessment of peritoneal adhesions, and (e) plot of adhesion scores observed for different groups. Each dot represents the average adhesion score per each rat.
In the untreated control group, the adhesions were tightly adhered to the ischemic buttons (Figure 7(c)), resulting in an adhesion score of 4.625 ± 0.25 (Figure 7(e)). These adhesions required the use of scissors since they could not be removed with blunt dissection. The Seprafilm-treated group had an adhesion score of 3.5 ± 1.5, which was not statistically different from that of the control group (Figure 7(e)). Rats treated with the PVA/CSAPBA hydrogel exhibited the lowest adhesion score (1.723 ± 0.152; Figure 7(e)), and most adhesions that formed could be easily removed from the ischemic button using blunt dissection with forceps. These results demonstrate that the PVA/CSAPBA hydrogel adhesion barrier significantly reduced the incidence and severity of adhesions following abdominal surgery, allowing for more facile dissection compared to the control and commercial Seprafilm group. These observations support the idea that the anti-inflammatory PVA/CSAPBA hydrogel may contribute to its effective adhesion barrier function by physically blocking the injured tissue surface and potentially shutting down early inflammatory cascades. Additionally, we hypothesize that the self-healing properties of the hydrogel may prevent dislodging from the site of application, thereby reducing the risk of adhesive failure caused by tissue movement.
In vivo anti-inflammatory and anti-adhesion properties of the PVA/CSAPBA hydrogel
The localization of two subtypes of macrophages in ischemic buttons was visualized via double-immunofluorescence staining of iNOS (M1 macrophage marker) combined with arginase 1 (M2 macrophage marker; Figure 8(a)). The iNOS is expressed in M1 macrophages, while arginase 1 is expressed in M2 macrophages. 53 Unlike the immunocytochemistry results in Figure 5, not all cells were macrophages in the immunohistochemistry analysis. Other cell types may have been included, so the ratio of M2-like to M1-like macrophages was calculated.

Anti-inflammatory effect of PVA/CSAPBA hydrogel in vivo. Ischemic buttons and serum samples were collected from rats following the in vivo ischemic button experiment, for the analysis of anti-inflammatory effect of PVA/CSAPBA hydrogel by immunohistochemistry and ELISA: (a) microscopic photo of stained macrophage in each group (control, Seprafilm, PVA/CSAPBA). Red cells indicate M1-like macrophages, green cells indicate M2-like macrophages, (b) quantitative analysis of M2-like/M1-like ratio, (c and d) in vivo analysis of pro-inflammatory cytokines from M1-like macrophages: (c) TNF-α and D) IL-6) tested by ELISA, (e–g) representative images of MTC staining for each ischemic button, showing the adhesion severity in (e) control, (f) Seprafilm and (g) PVA/CSAPBA treated groups.
The average M2-like macrophage to M1-like macrophage ratio in the control group and Seprafilm group was 0.0233 and 0.0209, respectively. In contrast, the average M2-like/M1-like ratio in the PVA/CSAPBA group was 5.139. Compared to the control and Seprafilm groups, the M2/M1 ratio in the PVA/CSAPBA group increased by approximately 220-fold. The results showed a low level of M1-like macrophages and five times higher M2-like macrophages detected in the PVA/CSAPBA hydrogel-treated group (Figure 8(b)). This result suggests that the PVA/CSAPBA hydrogel polarized pro-inflammatory M1-like macrophages to anti-inflammatory M2-like macrophages. This shift indicates that PVA/CSAPBA possesses sufficient anti-inflammatory properties to potentially prevent tissue adhesion.
Furthermore, the expression levels of pro-inflammatory cytokines of M1 macrophages in rat serum were also assessed by ELISA (Figure 8(c) and (d)). In Figure 8(c), the average expression levels of TNF-α for each group (Control, Seprafilm, CSAPBA) were 146.6, 119.18, and 49.44 pg•ml−1, respectively. In Figure 8(d), the average expression levels of IL-6 for each group (Control, Seprafilm, CSAPBA) were 106.083, 37.299, and 29.365 pg•ml−1, respectively. Compared to the control group, the average expression levels of TNF-α and IL-6 in the PVA/CSAPBA hydrogel treated group were lowered by approximately 3 and 4-fold, respectively. In conclusion, compared to the control groups, both inflammatory cytokines were expressed at lower levels in the PVA/CSAPBA group. There was a statistically significant difference observed between the commercial product and the PVA/CSAPBA hydrogel group, indicating that the developed hydrogel could effectively downregulate inflammation.
Masson’s trichrome (MTC) staining was used for histological analysis of harvested ischemic buttons (Figure 8(e)–(g)). This staining method was employed to visualize collagen deposition and fibroblast activity. In the control group, observed 7 days after surgery, fibrinous adhesion was evident between the ischemic button and liver, accompanied by collagen deposition (Figure 8(e)). However, in the PVA/CSAPBA treated group, the created ischemic button was clearly recognizable and the collagen deposition appeared scattered, indicating its anti-adhesive effect (Figure 8(g)). In the Seprafilm group, fibrous bands formed, resulting in adhesions that were less severe than those in the control group but significantly more severe than in the PVA/CSAPBA group (Figure 8(f)).
Discussion
Conventional anti-adhesion barriers, such as solution-type drugs which have lower viscosity than hydrogels, are rapidly cleared from the peritoneal cavity. 54 Traditional film types tend to be too rigid to adequately adhere to the uneven target site for extended period. In contrast, the dual-spraying strategy combined with the self-healing properties of our hydrogel system provides a promising solution by allowing the material to be easily sprayed onto the target site, effectively covering wide and uneven areas while recovering from physical stress and maintaining its functionality.
This hydrogel offers advantages over film-type barriers like Seprafilm, which are commonly used in commercial applications. Seprafilm is a rigid film that may not conform well to target lesions due to its stiffness. In contrast, the dual-sprayable PVA/CSAPBA hydrogel shows self-healing properties and enhanced retention with targeting capability in the designated area. 55
Previous studies indicate that effective anti-adhesion barriers should be retained for more than 7 days.49,55 This duration is important because inflammatory mechanisms related to peritoneal adhesions typically occur within 3 days.33,56,57 Demonstrated by micro-CT analysis, the hydrogel remains in vivo for more than 7 days, fulfilling the necessary retention period for effective anti-adhesion efficacy.
In addition to its self-healing properties, another appealing factor of the hydrogel is its anti-inflammatory effects. Organic moieties such as phenol groups, sulfur, and boronic acid act as ROS scavenging agents. 43 APBA in the hydrogel scavenges ROS through electron transfer or proton donation. 30 CS(C) effectively promotes the polarization of M1-like macrophages to the M2-like phenotype, which exhibits elongated morphology, as previously observed in other studies. 58 As a result, the PVA/CSAPBA hydrogel significantly reduces inflammatory cytokine expression levels.
On the other hand, the PVA/CSAPBA hydrogel also exhibits an additional biochemical effect of reducing peritoneal adhesion by inhibiting macrophage aggregation. Negatively charged molecules like CS or hyaluronic acid bind to receptors on macrophages, inhibiting the ability of macrophage to bridge themselves as secondary tethers to make cell aggregates. This interaction may reduce macrophage aggregation at the injury site. 33 The negatively charged CS effectively reduces the recruitment of peritoneal macrophages, demonstrating its anti-aggregation properties that could prevent peritoneal adhesion by inhibiting macrophage-mediated collagen deposition.
The successful conjugation of APBA to CS was confirmed using 1H-NMR spectroscopy. Although the conjugation rate optimization is not presented here, we determined the ideal CS ratio (1:2) through NMR analysis, with no significant improvements observed at higher ratios.
Subsequent optimization of the hydrogel involved fine-tuning the PVA solution concentration to less than 6% to facilitate spraying and prevent problems such as solution splitting, with above 5% PVA being ideal for achieving sufficient gelation. The CSAPBA concentration was fixed at 6%, as confirmed through the inverted vial test and rheological measurements. Notably, at concentrations of 5% PVA and 6% CSAPBA, the storage modulus (G’) exceeded the loss modulus (G’’) across various angular frequencies, indicating successful gelation. Based on these findings, we finalized the concentrations at 5% PVA and 6% CSAPBA for optimal hydrogel coverage performance.
The hydrogel exhibits biocompatibility both in vitro and in vivo settings. Notably, as shown by the luminol assay, none of the hydrogel components induce significant inflammation in vivo. It demonstrates the biocompatibility of the hydrogel in vivo, as well as in vitro. Additionally, PS with PVA/CSAPBA was injected to assess whether the hydrogel could alleviate strong inflammation induced by PS. However, the hydrogel did not alleviate ROS levels in this context. This is likely due to the extremely high inflammation triggered by PS, which overwhelmed the mitigating capabilities of the PVA/CSAPBA hydrogel. Nevertheless, the hydrogel showed distinct anti-inflammatory effects in additional in vivo assays.
Here, we have presented a novel approach to managing post-surgical adhesions, with a particular emphasis on peritoneal adhesions. Our findings demonstrate that the PVA/CSAPBA hydrogel exhibits mechanical characteristics that are well-adapted to the dynamic environment of the peritoneum. It intervenes between the damaged tissue and the opposing tissue, acting as a barrier for approximately 7 days to prevent peritoneal adhesion. Additionally, it displays anti-inflammatory properties conducive to preventing adhesion. This study also provides a handy method to deliver the hydrogel onto the target lesion by dual-spraying. As a result, this work provides an efficient strategy for the design of a multi-functional barrier against postoperative tissue adhesion. The uniqueness of this study lies in the application of the self-healing properties between APBA and PVA as an anti-adhesion barrier. Furthermore, unlike previous studies, CS(C) was conjugated to APBA to enhance its anti-inflammatory properties, thereby enabling dual prevention of abdominal adhesions both physically and chemically.
Moreover, the dual-spray delivery requires only a compact applicator (nozzle), facilitating integration with catheter systems. This adaptability renders the hydrogel method suitable for minimally invasive endoscopic interventions as well as open surgeries. Consequently, catheter-based administration may additionally mitigate postoperative tissue adhesion by minimizing surgical trauma. Future studies will further investigate catheter-optimized dual-spray system to enhance clinical translatability
Conclusion
This study demonstrates that the dual-sprayable, multi-functional PVA/CSAPBA hydrogel possesses self-healing and anti-inflammatory properties, effectively preventing the formation of post-operative peritoneal adhesions. The hydrogel exhibits the necessary rheological properties for self-healing, allowing for extended retention on target lesions. It maintains sustained local retention for more than 7 days in vivo and forms a viscoelastic physical barrier between target tissues. Due to the presence of APBA and CS(C), the hydrogel also exhibits anti-inflammatory properties, which are crucial for preventing fibrous band formation. It effectively reduces ROS levels and promotes the polarization of M1-like macrophages to M2-like macrophages, thereby leading to a decrease in inflammatory cytokine levels. The application of the PVA/CSAPBA hydrogel overcomes the limitations of traditional adhesion barriers by combining multiple strategies. Easily applied through dual-spraying, the hydrogel reduces inflammation and effectively prevents peritoneal adhesions, establishing it as a promising solution for handling clinical challenges associated with post-surgical peritoneal adhesions.
Supplemental Material
sj-png-1-tej-10.1177_20417314261427227 – Supplemental material for Dual-sprayable, multi-functional hydrogel for effective prevention of peritoneal adhesions
Supplemental material, sj-png-1-tej-10.1177_20417314261427227 for Dual-sprayable, multi-functional hydrogel for effective prevention of peritoneal adhesions by Hyunjung Kim, Wonmoon Song, M. Nivedhitha Sundaram, Changyub Lee, Xiang Li, Kyoung-Ha So, Juan M. Melero-Martin and Nathaniel S. Hwang in Journal of Tissue Engineering
Supplemental Material
sj-png-2-tej-10.1177_20417314261427227 – Supplemental material for Dual-sprayable, multi-functional hydrogel for effective prevention of peritoneal adhesions
Supplemental material, sj-png-2-tej-10.1177_20417314261427227 for Dual-sprayable, multi-functional hydrogel for effective prevention of peritoneal adhesions by Hyunjung Kim, Wonmoon Song, M. Nivedhitha Sundaram, Changyub Lee, Xiang Li, Kyoung-Ha So, Juan M. Melero-Martin and Nathaniel S. Hwang in Journal of Tissue Engineering
Supplemental Material
sj-png-3-tej-10.1177_20417314261427227 – Supplemental material for Dual-sprayable, multi-functional hydrogel for effective prevention of peritoneal adhesions
Supplemental material, sj-png-3-tej-10.1177_20417314261427227 for Dual-sprayable, multi-functional hydrogel for effective prevention of peritoneal adhesions by Hyunjung Kim, Wonmoon Song, M. Nivedhitha Sundaram, Changyub Lee, Xiang Li, Kyoung-Ha So, Juan M. Melero-Martin and Nathaniel S. Hwang in Journal of Tissue Engineering
Footnotes
Acknowledgements
The authors thank Seh Ri Oh and Gwang-Bum Im for their excellent support and feedback during this study.
Author’s Note
Nathaniel S. Hwang is now affiliated with Bio-MAX/N-Bio Institute, Seoul National University, Republic of Korea.
Ethical considerations
This study does not involve human participants; therefore, informed consent is not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Research Foundation (NRF) funded by the Korean government (MSIT; RS-2023-NR077182, RS-2024-00411768). This work was supported by the Technology Innovation Program (20016553) funded by the Ministry of Trade Industry & Energy (MOTIE, Korea). This work was supported by R&D project from the Ministry of Food and Drug Safety (MFDS) of the Republic of Korea (RS-2024-00350502). This work was additionally supported by the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare of the Republic of Korea (RS-2025-02213873). The Institute of Engineering Research at Seoul National University provided research facilities.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
