Abstract
Osteoporosis is a progressive skeletal disorder marked by an imbalance between bone resorption and formation, resulting in compromised microarchitecture and increased fracture risk. However, conventional pharmacological therapies have systemic side effects and limited targeting efficiency. Therefore, these limitations highlights the need for innovative strategies, and biomaterials have emerged as versatile tools, offering both structural support and the ability to modulate the osteoporotic bone microenvironment. This review outlines the key pathophysiological changes in osteoporosis including cellular dysregulation, ECM alteration, inflammation, and impaired vascularization underscoring the importance of restoring this niche for effective regeneration. A wide range of biomaterials, including natural/synthetic polymers, bioceramics, and metallic biomaterials and their alloys, are explored for their osteoconductive, osteoinductive, and mechanical features tailored to osteoporotic bone. This review also focuses on the functionalization approaches for the controlled delivery of drugs and growth factors (e.g. BMP-2, VEGF), and emerging gene/RNA therapies. The integration of biomaterials with stem cells and extracellular vesicles is discussed for enhancing osteogenesis, angiogenesis, and immunomodulation. Additionally, immuno-informed scaffold designs and bio-responsive materials responsive to pathological cues such as inflammation and oxidative stress are reviewed. Advanced technologies like three-dimensional printing and sensor-enabled scaffolds for real-time feedback are also addressed. Finally, the review considers translational barriers and highlights future directions combining material science, regenerative medicine, and personalized therapy for osteoporotic bone repair.

Introduction
Osteoporosis (OP) is a progressive skeletal disorder characterized by reduced bone mass, microarchitectural deterioration, and increased fracture risk. Diagnostic criteria generally follow World health organization (WHO) standards, with a T-score of ⩽ –2.5 indicating OP. Globally, the disease affects around 500 million individuals aged 50 years and over, with prevalence rates in the age group of 21.2% in women and 6.3% in men. In 2019, approximately 37 million osteoporotic fractures were reported worldwide. 1 OP is more prevalent in women (nearly 1 in 3) compared to men (1 in 5), with hip fractures predicted to rise by 240% in women and 310% in men by 2050 relative to 1990 levels. 2 In Europe, OP-related disability surpasses many chronic diseases, and in the U.S., annual direct costs reach almost USD19 billion. 3
The uncoupling of bone remodeling is central to OP pathophysiology, in which osteoclast (OC)-driven resorption overtakes osteoblast (OB)-mediated formation. 4 Under normal conditions, bone homeostasis depends on a delicate balance between these cell types, guided by osteocytes acting as mechanosensors and regulators. 5 In OP, this balance is disrupted as OCs accelerate resorption while OBs and mesenchymal stem cells (MSCs) fail to adequately compensate, often compounded by osteocyte dysfunction and signaling breakdown. 6
Beyond cellular imbalance, the bone microenvironment in OP undergoes significant deterioration, which affects the extracellular matrix (ECM), vasculature, cytokines, and mechanical stimuli. The ECM becomes sparse and mechanically weaker, angiogenesis slows, and inflammatory cytokines, oxidative stress, and microarchitectural disruption collectively create a hostile niche for bone regeneration.7–9 A damaged microenvironment not only prevents repair but may also actively promote bone fragility.
Current pharmacological treatments, such as bisphosphonates (BPs), selective estrogen receptor modulators (SERMs), and parathyroid hormone analogs, primarily focus on systemic suppression of resorption or stimulation of formation. While effective at reducing fracture risk, these approaches have limitations: systemic distribution can cause off-target effects, a lack of localized targeting can reduce efficacy at the bone site, and long-term use raises concerns regarding atypical fractures or osteonecrosis.10–12
In response to these challenges, biomaterials have emerged as a transformative platform in regenerative medicine and targeted therapies. By integrating structural support with site-specific drug or cell delivery, these materials can actively shape the microenvironment. Examples include injectable hydrogels, nanoparticles (NPs), 3D-printed scaffolds, and composite bone cements, all engineered to provide mechanical integrity and influence cellular behavior. 13 Recent advances in 3D bioprinting further highlight its ability to fabricate highly biomimetic tissue analogs with precise microarchitecture, expanding the potential of engineered scaffolds for regenerative applications. 14
Although a number of reviews have summarized biomaterial- and nanotechnology-based strategies for bone repair, most have either focused on specific delivery systems or broadly addressed bone regeneration without emphasizing the distinct challenges of osteoporotic bone. To clarify how the present review differs from prior works, a comparative summary (Table 1) has been incorporated, outlining the main focus and remaining gaps in recent reviews on biomaterials, NPs, and extracellular vesicle (EV)-based approaches. Unlike previous reviews, this article integrates these domains and emphasizes microenvironment-responsive biomaterial systems that address the pathological features of OP through mechanical reinforcement, biochemical modulation, and immunoregulatory mechanisms, while also considering translational aspects relevant to clinical application.
Summary of recent review articles relevant to osteoporotic bone microenvironment engineering, NP strategies, and EV-based therapeutics.

This table compares prior reviews in terms of their publication year, primary focus or scope, identified gaps or limitations, and the unique perspective provided by each article. Collectively, these reviews cover NP chemistries and targeting strategies, scaffold design, EV biology, regenerative platforms, and osteoporotic microenvironmental dysfunctions. The table highlights where current literature remains limited, such as insufficient discussion of scaffold mechanics, NP–EV integration, engineering pathways for EV delivery, and microenvironment-responsive biomaterial design; and how the present review addresses these gaps by providing a comprehensive, engineering-oriented roadmap for developing multifunctional biomaterials for OP treatment.
Scope and Literature Selection Strategy
This mini review examines the evolving role of biomaterials in precisely modulating the osteoporotic bone microenvironment. The versatility of these platforms is also reflected in other tissues, such as skin, where 3D bioprinting has enabled layered constructs that closely mimic native tissue organization. 15 This review provides a comprehensive analysis of advancements in regenerative biomaterials for the treatment of OP, focusing on approaches that actively modulate the bone microenvironment. Due to the high volume of research in this area, we established specific criteria to ensure a focused and impactful discussion. The review is primarily focused on polymeric, ceramic, and composite biomaterials used in the form of NPs, hydrogels, and porous scaffolds. We have also included emerging research on bio-absorbable metallic biomaterials to address the critical need for mechanical support in osteoporotic bone. Papers included for detailed discussion were selected based on one or more of the following criteria: they demonstrate a novel biomaterial design or functionalization strategy, report in vivo efficacy in a relevant animal model of OP (such as ovariectomy or glucocorticoid-induced models), or provide a definitive and impactful proof-of-concept for a specific therapeutic strategy, including gene delivery or immunomodulation. Studies focusing solely on inert metallic fixation, basic material synthesis without orthopedic application, or non-skeletal diseases were excluded. The studies cited throughout the text were chosen for their novelty, high relevance, or representativeness of a particular biomaterial class or therapeutic mechanism, serving as illustrative examples rather than an exhaustive list.
The osteoporotic bone microenvironment
Cellular imbalance and dysregulation
A hallmark of OP is the hyperactivation of OCs alongside OBs dysfunction, which can lead to excessive bone resorption. OC overactivation is driven primarily by heightened RANKL-to-OPG ratios, inflammatory cytokines (TNF-α, IL-1, IL-6), and oxidative stress, which collectively amplify osteoclastogenesis via NF-κB and MAPK signaling pathways,23,24 resulting in increased bone turnover but a net loss of bone mass.
Simultaneously, OBs and their progenitor MSCs exhibit diminished recruitment and osteogenic potential.25,26 Aging and estrogen deficiency impair MSC differentiation by disrupting Wnt/β-catenin signaling and increasing mitochondrial dysfunction, which further compromises OB maturation and matrix production. 27
Another crucial element in bone homeostasis is osteocytes. These crucial mechanosensory cells orchestrate remodeling via sclerostin and RANKL but also become dysfunctional in OP. Meanwhile, osteocytes acquire a senescence-associated secretory phenotype (SASP), secreting pro-resorptive factors that tilt the balance toward resorption. Additionally, loss of osteocyte viability and mechanotransduction capacity impairs structural adaptation and compromises microdamage repair.28,29
ECM and biochemical changes
The osteoporotic bone microenvironment is characterized by a disrupted balance between bone formation and resorption, coupled with profound alterations in the composition and architecture of the ECM. The bone ECM comprises an organic phase predominantly type I collagen, proteoglycans, and non-collagenous proteins (such as osteocalcin, osteopontin, and bone sialoprotein) and an inorganic mineral phase mainly composed of hydroxyapatite (HA; Ca₁₀(PO₄)₆(OH)₂). Together, these components provide both mechanical strength and biochemical cues for cellular adhesion, differentiation, and remodeling. In OP, the ECM becomes disorganized, with reduced collagen crosslinking, altered mineralization density, and elevated matrix metalloproteinase (MMP) activity, leading to brittleness and reduced osteoconductivity. 30 Recent research also highlights how decellularized extracellular matrices, even those derived from pathological tissues, can serve as structurally and biologically meaningful scaffolds for regeneration. 31 The ECM in osteoporotic bone undergoes both qualitative and quantitative changes, including decreased collagen content, disrupted mineralization, and reduced cross-linking, which together lead to diminished stiffness and increased fracture risk. At the nanoscale, increased accumulation of advanced glycation end-products stiffens collagen fibrils but also makes them more brittle.32,33 Biochemically, OP is marked by chronic low-grade inflammation and oxidative stress. Excess reactive oxygen species (ROS), triggered by estrogen deficiency and aging, activate osteoclastogenesis and impair OB differentiation, primarily through MAPK and NF-κB pathways. ROS also dysregulate miRNAs, further skewing cell fate toward resorption. 34 Moreover, the osteoporotic microenvironment experiences vascular deterioration. Impaired angiogenesis results in fewer type-H capillaries, which are specialized vessels essential for coupling osteogenesis and angiogenesis, further diminishing nutrient, oxygen, and stem cell supply to bone surfaces. 35
Immunological Impact
The immune system plays a pivotal role in OP pathogenesis, with both innate and adaptive immune responses contributing to bone remodeling dysregulation. Macrophages, for example, act as both regulators and effectors in bone remodeling. Beyond secreting pro-inflammatory cytokines like TNF-α and IL-1β, which promote osteoclastogenesis, macrophages directly contribute to bone resorption by fusing into multinucleated OCs under the influence of RANKL and M-CSF. A study emphasized that activated M1 macrophages exacerbate bone loss by sustaining inflammation, while M2 macrophages, which secrete IL-10 and TGF-β, support OB differentiation and bone repair. 36
Another study showed that pro-inflammatory cytokines secreted by macrophages, dendritic cells, and Th17 cells (e.g. TNF-α, IL-1β, IL-6, and IL-17) enhance osteoclastogenesis via the RANK/RANKL signaling axis, thereby increasing bone resorption. 37 Conversely, regulatory T cells and B cells exert protective effects by producing anti-inflammatory cytokines and osteoprotegerin (OPG), a decoy receptor that inhibits RANKL-mediated OC activation. Furthermore, another study elaborated on how estrogen deficiency in postmenopausal women alters immune cell profiles, promoting OC activity and bone loss. 38 Additionally, chronic low-grade inflammation associated with aging and autoimmune diseases sustains a pro-osteoclastogenic environment. The immune system, comprising various cellular and biological components, therefore, plays a critical role with significant clinical implications. Consequently, effectively reversing these interconnected pathological changes demands localized, precision intervention capable of physically and biochemically reprograming the dysfunctional bone microenvironment.
Biomaterial-based strategies for the osteoporotic microenvironment
Fundamental design principles of bone biomaterials
To align with the principles of biomaterials science, various biomaterial designs (e.g. NP systems, porous scaffolds, injectable hydrogels, and topographically engineered surfaces) were evaluated based on their primary functional roles.
NPs provide biochemical modulation in osteoporotic bone by delivering OC inhibitors and osteogenic cues while offering intrinsic osteoconductive signaling, particularly in calcium-phosphate and HA systems. 39 Bone-affine functionalization improves targeting and reduces excessive resorption, while ion-releasing nanosystems enhance mineralization and MSC activity, establishing NPs as efficient regulators of osteoporotic turnover. 16 Porous scaffolds complement these effects by providing structural support for cell infiltration, angiogenesis, and matrix deposition. Hierarchically porous, ion-releasing designs show strong osteogenic and vascular integration and large-animal studies demonstrate substantial trabecular regeneration in osteoporotic defects. 40 Incorporation of bioactive factors further enhances MSC recruitment and differentiation. 41 Injectable hydrogels provide minimally invasive delivery of MSCs, exosomes, and anabolic cues, improving progenitor recruitment, focal adhesion signaling, and bone strength. Their controlled release of osteogenic and anti-resorptive signals supports angiogenesis and coordinated regeneration, with broad utility across all phases of repair.42,43 Nanotopographical cues strengthen focal adhesions and actomyosin organization, enabling osteogenesis even without growth factors, and exhibit strong synergy with ion-releasing or osteoinductive surface chemistries.44,45 Together, these strategies combine biochemical, structural, and mechanotransductive cues to orchestrate efficient and coordinated bone regeneration in osteoporotic defects (Figure 1).

Impact of biomaterial design on OP treatment: (a) NP delivery systems. Bare or exosome-coated NPs encapsulating growth factors are introduced into osteoporotic bone. These particles suppress OC activity while promoting differentiation of mesenchymal stem cells (MSCs), contributing to bone regeneration. (b) Scaffolds. Scaffolds with larger pore sizes support the attachment of pericytes, endothelial progenitor cells, and MSCs. This cellular integration facilitates angiogenesis and OB formation, restoring vascularization and bone structure. (c) Hydrogel encapsulation. Hydrogels loaded with MSCs and osteogenic growth factors enable delivery to osteoporotic sites. They also enhance MSC migration and stimulate osteogenesis, further aiding bone repair. (d) Topographically engineered biomaterials. Biomaterials with advanced surface topographies enhance MSC adhesion and guide their differentiation into OBs, thereby promoting more effective bone formation. Through these mechanisms, innovative biomaterial designs transform osteoporotic bone into healthier, structurally sound tissue (created with BioRender).
Classification of biomaterials
Biomaterials for bone tissue engineering can either comprise natural polymers, synthetic polymers, or bioceramics. Each has distinct advantages for addressing osteoporotic microenvironments.46–48 Natural polymers, such as collagen, chitosan, alginate, and hyaluronic acid, are extremely biocompatible and mimic the ECM.49–51 Fibrin-based matrices have recently gained attention due to their intrinsic bioactivity and ability to promote vascularization, making them valuable candidates in tissue engineering applications. 52 A 2019 study demonstrated that collagen–HA scaffolds loaded with bone morphogenic protein-2 (BMP-2) and VEGF achieved complete healing of critical-size calvarial defects within 4 weeks, resulting in a 28-fold increase in new bone volume and a sevenfold increase in bone area compared to controls thanks to HA-mediated growth factor sequestration Figure 2. 53 Similarly, chitosan-alginate-HA composites have been shown to sustain BMP-2 release and support MSC-driven bone formation in vivo. 54

Evaluation of dual growth factor-loaded CHA scaffolds in a rodent calvarial defect model: (a) Experimental setup used to evaluate the in vivo bone regeneration potential of dual growth factor (GF)-loaded CHA scaffolds in a rodent critical-sized calvarial defect model. (b) Micro-computed tomography (CT)-based 3D reconstructions of rat calvarial defects at 4 weeks post-implantation with (a) GF-free CHA scaffolds and (b) dual growth factor (rhBMP-2 + rhVEGF)-loaded CHA scaffolds. The dual GF-loaded CHA scaffolds exhibited visibly enhanced bone regeneration across all specimens compared with the GF-free group, highlighting the osteoinductive and angiogenic synergy of the incorporated growth factors. (c) Histological evaluation of bone regeneration at 4 weeks post-implantation using hematoxylin and eosin (H&E) and Masson’s trichrome (MT) staining. (a) GF-free CHA scaffold group and (b) GF-loaded CHA scaffold group (rhBMP-2 + rhVEGF). Panels (i) and (iii) show low-magnification H&E and MT-stained sections, respectively, while panels (ii) and (iv) show high-magnification views of selected regions. In the GF-free group, scaffolds remained largely acellular with minimal new bone formation or integration. In contrast, the GF-loaded scaffold group showed greater scaffold integration, abundant cellular infiltration, and significant new bone and collagen matrix deposition. H&E stains nuclei purple and ECM pink, while MT stains mineralized bone/collagen blue and osteoid red. (Adapted with permission from © John Wiley and Sons).
Recent work on modified natural-polymer hydrogels has shown promising results in osteoporotic bone repair. For example, a rat study used a supramolecular gelatin hydrogel (GelCD) loaded with the secretome of human fetal MSCs (GelHFS) in ovariectomized femoral defects: the GelHFS group exhibited significantly higher bone volume and BMD as early as 2 weeks post-implantation compared to controls, and the construct showed enhanced stiffness and ultimate load after 8 weeks via recruitment of integrin β1-positive endogenous MSCs.
55
Separate work on injectable methacrylated gelatin (GelMA) hydrogels reinforced by
Synthetic polymers, notably poly (lactic-co-glycolic acid; PLGA), polycaprolactone (PCL), and polyethylene glycol (PEG), allow precise control over degradation rates and mechanical integrity. For example, aspirin-loaded PLGA coatings on titanium implants foster anti-inflammatory responses, suppress RANKL-driven osteoclastogenesis, and promote OB adhesion via NF-κB/MAPK pathway modulation, offering a dual osteogenic and anti-resorptive strategy.57,58 A 2024 study fabricated a spiral-structured scaffold composed of PCL/PLGA/β-TCP: the hybrid polymer blend improved mechanical stability over single-polymer scaffolds, and β-TCP enhanced osteogenic differentiation of human fetal OBs. 59 In parallel, a 2023 investigation with PLGA/β-TCP 3D-printed scaffolds plus low-intensity pulsed ultrasound and lipid microbubbles showed improved bone regeneration outcomes, highlighting how polymer-based devices can be combined with adjunctive therapies. 60 A systematic review of PCL scaffolds concluded that PCL combined with bioactive agents (HA, bioactive glass, growth factors) achieved higher cell viability and osteoinductive potential, though degradation rate and acidic by-products (especially for PLGA) must still be addressed. 61 For osteoporotic bone repair, synthetic polymers therefore serve as the structural backbone that can be further functionalized (e.g. with ceramics or drug release) to meet both mechanical and biological demands.
Bioceramics and composites, such as HA, β-tricalcium phosphate, and bioactive glass, replicate the mineral structure of bone while supporting osteogenesis. One study explored the development and characterization of hydroxyapatite-collagen (HC) scaffolds integrated with O-carboxymethyl chitosan microspheres for sequential release of VEGF and recombinant human BMP2 (rhBMP-2), aiming to enhance bone tissue engineering. The scaffolds were designed to mimic the natural bone healing process, where VEGF induces angiogenesis early, followed by rhBMP-2 promoting bone formation. The study demonstrated that the sequential release of these factors significantly improved bone regeneration, as shown by the in vitro and in vivo experiments. The HC scaffolds demonstrated excellent biocompatibility, structural integrity, and sustained release properties, with VEGF showing rapid early release and rhBMP-2 maintaining prolonged release. Histological observations confirmed enhanced neovascularization and collagen deposition in scaffolds loaded with both growth factors compared to single-factor or pure material scaffolds. This innovative approach highlights the potential of HC scaffolds as effective biomaterials for bone repair and regeneration. Bone tissue growth and neovascularization were minimal in HC scaffolds but were significantly boosted in hydroxyapatite single-factor composite scaffolds (HCB) and hydroxyapatite collagen two-factor composite scaffolds (HCBV). Moreover, HCBV scaffolds showed the strongest promotion of new blood vessels and collagen deposition due to the combined effects of VEGF and rhBMP-2, ultimately improving scaffold osteogenesis (Figure 3). 62
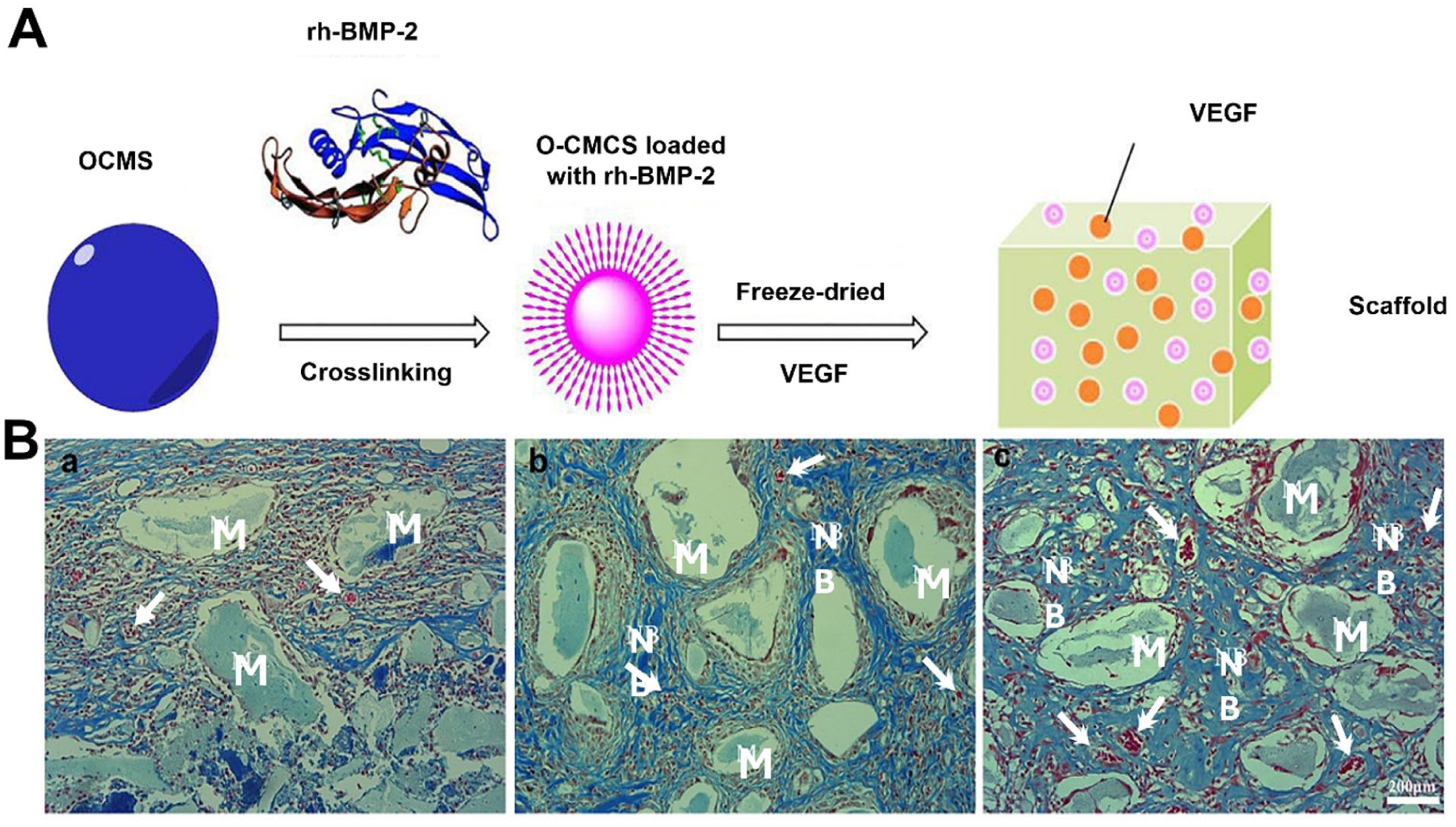
Bioactive scaffold fabrication and evaluation of osteogenesis and angiogenesis: (a) Schematic illustration of the preparation process for the bioactive scaffold. Oxidized carboxymethyl chitosan (O-CMCS) was first crosslinked with rhBMP-2 to form O-CMCS microspheres loaded with rhBMP-2. The microspheres were then incorporated into a scaffold matrix containing VEGF through freeze-drying, resulting in a composite scaffold designed to promote both osteogenesis and angiogenesis. (b) Masson’s trichrome staining of HC (a), HCB (b), and HCBV (c) scaffolds showing bone tissue ingrowth and neovascularization. “M” denotes scaffold material, “NB” indicates new bone formation, and the arrows point out newly formed blood vessels. Minimal tissue ingrowth and vascularization were observed in HC scaffolds, while HCB and HCBV scaffolds exhibited extensive collagen deposition, neovascularization, and new bone formation. HCBV scaffolds, containing both rhBMP-2 and VEGF, showed the most robust angiogenic and osteogenic responses. (Adapted with permission from © Elsevier).
While the focus in bone tissue engineering frequently centers on polymeric, ceramic, and composite scaffolds, metallic biomaterials represent a foundational class essential for clinical success, particularly in applications requiring high mechanical integrity, such as long-bone fracture fixation, joint replacement, and spinal fusion devices. 56 Titanium (Ti) and its alloys (e.g. Ti-6Al-4V) are the prevailing choice due to their outstanding biocompatibility, low density, and high corrosion resistance, properties largely attributed to the formation of a stable, inert oxide layer on their surface. 63 Despite these advantages, the elastic modulus of traditional Ti alloys (110–140 GPa) is significantly higher than that of cortical bone (11–30 GPa), a disparity that results in stress shielding the reduction of mechanical load on the surrounding bone, which can lead to bone resorption and eventual implant failure. 64 To mitigate stress shielding and enhance osseointegration, significant efforts are dedicated to surface modification techniques and the development of new alloy systems. For instance, creating porous metallic structures, such as those using Tantalum (Ta), can reduce the overall stiffness of the implant and promote rapid bone ingrowth via interconnecting pores. 65 Furthermore, the emergent field of biodegradable metals, primarily magnesium (Mg) and zinc (Zn) alloys offers a revolutionary alternative. 66 These materials are designed to degrade in vivo at a controlled rate, gradually transferring mechanical load back to the regenerating bone and avoiding the need for a second surgery to remove the implant. However, the primary challenge remains in optimizing their degradation kinetics to match the rate of bone healing, managing the rapid production of hydrogen gas and controlling the release of metal ions to prevent localized cytotoxicity.67,68 Ongoing research in this area utilizes advanced manufacturing (such as additive manufacturing) and surface engineering (e.g. bioactive coatings of HA or calcium phosphate) to precisely tailor the mechanical, biological, and degradation profiles of metallic implants for use in complex orthopedic scenarios.69,70
Collectively, these findings highlight that although natural, synthetic, and ceramic-based biomaterials each contribute valuable attributes to osteoporotic bone regeneration, their comparative performance differs markedly. Natural polymers (collagen, chitosan, alginate) mimic extracellular-matrix biochemistry and support excellent cell adhesion and early osteogenic signaling; however, they lack mechanical robustness and exhibit variable degradation that restricts load-bearing use. Synthetic polymers (PLGA, PCL, PEG) provide tunable strength and predictable degradation profiles but require surface functionalization or bioactive fillers to overcome limited bioactivity, and acidic degradation products may hinder osteogenesis if not buffered. Bioceramics (HA, β-TCP, bioactive glass) and polymer–ceramic composites supply superior osteoconductivity and stiffness but can be brittle and difficult to fabricate at large scale. Overall, natural polymers = biological performance but limited mechanics; synthetics = mechanical control but weak bioactivity; ceramics/composites = balanced properties yet brittle or complex to process. For future OP-oriented designs, multifunctional hybrid systems that integrate polymeric flexibility, ceramic bioactivity, and targeted molecular delivery represent the most promising direction.
Osteoconductivity, osteoinductivity, and biomechanics
Effective scaffolds must be osteoconductive (supporting cell attachment and matrix growth), osteoinductive (stimulating undifferentiated cells toward bone lineage), and biomechanically suitable for osteoporotic bone. 71 Collagen-HA scaffolds not only encourage MSC adhesion and alkaline phosphatase activity but are also delivery vehicles for BMP-2, enhancing their osteoinductive properties. Freeze-casting techniques have yielded HA scaffolds with ~50% porosity and compressive strength up to ~145 MPa. This allows them to withstand partial loads while maintaining pore interconnectivity, which is crucial for vascularization and cell migration. 72 Design parameters like pore size (100–300 µm) and interconnectivity are critical to the prevention of hypoxic zones, while supporting nutrient transport and promoting angiogenesis in compromised osteoporotic bone. 73
Mechanistic regulation of cellular pathways by biomaterial cues
The need for localized influence has made the development of therapeutic biomaterials a central focus, as the interaction between biomaterials and cells involves a complex mechanochemical dialog that regulates intracellular signaling cascades. Surface chemistry, topography, stiffness, and ion release can selectively activate or suppress pathways such as FAK/ERK, MAPK, and NF-κB, thereby determining osteogenic or inflammatory outcomes, as described in Table 2.
Summary of key biomaterial properties and their associated mechanotransductive or immunomodulatory signaling pathways influencing MSC osteogenesis or macrophage polarization.

Matrix stiffness, nanotopography, ion release, and surface chemistry regulate integrin-dependent (FAK/ERK, Akt/GSK-3β/β-catenin), MAPK-based, and osteoimmunomodulatory (NF-κB, BMP/Smad) signaling to modulate osteogenic differentiation and immune-mediated bone regeneration.
Modifying surface and architecture for microenvironmental modulation
Surface and architectural refinements enhance how scaffolds influence the osteoporotic niche. Nanoscale topographies, such as nanograin HA layers or collagen-coated titanium implants, enhance integrin binding and promote osteogenic differentiation via the FAK/ERK pathways. 80 Functional coatings containing bioactive molecules such as RGD peptides, BMPs, or VEGF further enhance MSC differentiation and vascularization. For instance, the dual delivery of BMP-2 and VEGF from HC scaffolds simultaneously stimulated angiogenesis and bone formation in a mandibular defect model via the MEK/ERK and PI3K/Akt pathways. 81
By combining matrix-mimicking natural polymers, structurally tunable synthetics, and osteoconductive ceramics with advanced surface nano topographies, porosity control, and bioactive cues, it was possible to synthesize scaffolds that go beyond passive support to actively modulate the osteoporotic microenvironment, restoring balance in cellular activity, vascular supply, and mechanical integrity to facilitate effective bone regeneration.
Beyond serving as mechanical and structural templates, scaffolds also provide an excellent platform for localized therapeutic delivery. By incorporating NPs, growth factors, or gene carriers within their matrices, these biomaterials can transform from passive frameworks into active drug reservoirs that modulate the osteoporotic microenvironment.
Biomaterials as therapeutic delivery platform
Building upon the scaffold-based approaches described above, the next section focuses on how biomaterials can be engineered as delivery vehicles for pharmacological and genetic therapies. These systems, ranging from nanoparticles to composite scaffolds, are designed to release bioactive agents in a controlled or stimulus-responsive manner, thus bridging the gap between structural regeneration and targeted molecular therapy.
Controlled release systems
A key strategy in biomaterial-based OP therapy is the controlled release of therapeutics from scaffolds or nanocarriers. Anti-resorptive agents, such as BPs (e.g. alendronate, risedronate), are commonly incorporated into biodegradable carriers to enhance bone targeting and reduce systemic side effects. For instance, calcium-risedronate NPs (Ca-RISNPs), which are formed via calcium ion precipitation, enabled sustained drug delivery with high loading efficiency. These NPs dissociate selectively in acidic endosomal conditions and demonstrate enhanced bone penetration and therapeutic potential in disease models.16,82 Similarly, PLGA-based NPs coated with alendronate exhibited strong HA binding and improved uptake by MC3T3 osteoblastic cells, suggesting their viability of these NPs as targeted bone delivery systems. 83
Building on these advances, a recent study proposed a novel therapeutic strategy employing stepwise administration of BP-conjugated lipid NPs (BP-LNPs) encapsulating valproic acid (VPA) and tauroursodeoxycholic acid (TUDCA). This approach not only ensured targeted delivery but also enhanced stemness and induced osteogenic transdifferentiation of fibroblasts, resulting in superior bone regeneration in an ovariectomy-induced OP mouse model. Notably, it offers a safe, clinically translatable alternative to conventional genetic reprograming methods. 84 Concurrently, recent work highlighted several emerging NP-based systems that address limitations of conventional OP therapies, such as poor bioavailability and off-target effects. These include silica NPs functionalized with pentapeptides to promote OB differentiation, titanium nanotubes loaded with raloxifene to stimulate bone formation, and quercetin-loaded solid lipid NPs that strengthened bone mineral density while reducing OC activity. Collectively, these innovations highlight the potential of nanocarrier platforms to enable targeted bone delivery, controlled release, and enhanced therapeutic efficacy. 18
Targeted and responsive delivery platforms
Advanced stimuli-responsive systems enable precise drug release in pathological microenvironments. Indeed, pH-sensitive linkages (e.g. hydrazone, acetal, imine bonds) and redox-sensitive disulfide bridges are frequently used in carrier designs that respond to the acidity or oxidative stress within bone lesions. 17
Bone-targeting moieties, such as alendronate or zoledronate, can be conjugated to nanocarrier surfaces to specifically direct therapeutic payloads to bone tissue. For example, PLGA-PEG NPs conjugated with alendronate achieved a sixfold increase in bone accumulation, while zoledronate-modified PLGA-PEG NPs exhibited a 50-fold increase in bone localization compared to unconjugated controls. 85 These BP-functionalized carriers improve not only drug bioavailability in osseous tissue but also reduce off-target toxicity and enable integration with imaging modalities. 86
Emerging delivery systems are moving beyond simple bone-affinity ligands to smart, microenvironment-responsive platforms tuned for osteoporotic bone. For example, one study developed amorphous calcium carbonate NPs coated with a glutamic-acid hexapeptide that neutralizes local acidity and selectively releases Oroxylin A in OC-rich resorption zones, effectively reversing bone loss in ovariectomized mice. 87 In parallel, another study reported milk-derived EVs functionalized with a bone-targeting peptide (DSS₆), loaded with the SIRT1 agonist SRT2104 and a Mn-based contrast agent, which significantly increased bone accumulation, improved BMD, and enabled MRI-guided therapy in osteoporotic models. 88 These advances signal a shift toward platforms that combine ligand-mediated osteotropism, triggered release (pH, enzymatic, ROS cues), therapeutic payloads (drugs, ions, nucleic acids) and imaging or monitoring functionality, offering precise spatiotemporal control of treatment within the pathologic bone niche.
Emerging genes and RNA delivery platforms
In addition to small molecules and proteins, other emerging strategies to modulate OB and OC function at the genetic level include RNA therapies delivered via biomaterials. siRNA (RANK, SOST)-loaded mesoporous silica NPs with polymer caps have achieved up to 90% gene knockdown in vitro, potentially inhibiting osteoclastogenic signals or reprograming MSCs. 89 Lipid NP systems, often functionalized with bone-targeting BPs, have successfully delivered miRNA modulators (e.g. antagomir-148a) to reduce OC activity in osteoporotic mice (Figure 4). 90 Moreover, alendronate-functionalized liposomal NPs delivering the Stromal cell-derived factor-1 (SDF-1) gene have been used to recruit MSCs to bone surfaces, facilitating regeneration in osteoporotic mouse models. 91 Other emerging platforms also include BMP-9 gene delivery via ionizable lipid NPs, demonstrating efficient transfection and showing promise as gene therapies for OP (Figure 5). 92

Tissue-specific distribution of antagomir-148a in vivo: (a) Representative biophotonic images showing the tissue distribution of FAM-labeled antagomir-148a in ovariectomized (OVX) mice following administration of different formulations: free FAM-antagomir-148a, in vivo jetPEI–FAM-antagomir-148a, liposome–FAM-antagomir-148a, and (D-Asp8)-modified liposome–FAM-antagomir-148a. Fluorescence signals were assessed in harvested tissues, including the heart, liver, spleen, lung, kidney, and proximal tibia group. (b) Quantitative fluorescence analysis of FAM-antagomir-148a accumulation in each tissue was performed using a microplate reader. Separate groups of OVX mice received each formulation, while fluorescence intensity was measured in the heart, liver, spleen, lung, kidney, and bone tissue. *indicates the statistical significance (P<0.05). (Adapted with permission from Elsevier).

Mechanistic illustration of NP delivery and therapeutic gene modulation. The figure schematically outlines the multi-step process for delivering therapeutic cargoes (such as siRNA, miRNA, or gene plasmids) via a NP (e.g. LNP or MSNP). The NP is internalized by the target cell via endocytosis, followed by endosomal escape to ensure the cargo is released into the cytosol rather than being degraded. Once released, the cargo utilizes its molecular machinery to initiate a chain of events that results in the specific silencing of the target gene or the expression of a therapeutic protein. MSNP: mesoporous silica nanoparticle; LNP: lipid nanoparticle. Created by BioRender.
Biomaterial for osteoporotic bone defects
Bone defect healing in osteoporotic conditions remains a major challenge due to diminished osteogenic potential, excessive osteoclastic activity, and poor angiogenesis. To address these barriers, recent advances in biomaterial engineering have focused on localized and sustained delivery systems capable of directly modulating the osteoporotic microenvironment. Among the most promising strategies are ion-releasing and bioresponsive scaffolds, which integrate mechanical stability with biochemical signaling. Injectable Mg-based hydrogels have demonstrated controlled hydrogen release under oxidative conditions, effectively scavenging ROS, promoting M2 macrophage polarization, and nearly doubling bone-volume fraction in osteoporotic defect models compared with untreated controls. 93 Likewise, biodegradable Zn–Mg alloy scaffolds exhibiting adaptive degradation behavior have shown bidirectional regulation of bone homeostasis by enhancing OB differentiation while suppressing OC activity, resulting in faster remodeling and higher bone mineral density in osteoporotic femoral defects. 94 These results highlight that ion-releasing scaffolds act as active modulators of the redox and immune landscape, aligning local inflammatory and osteogenic processes to restore balanced bone regeneration.
Expanding upon these approaches, bioactive molecule-releasing scaffolds have been developed to deliver osteoinductive or anti-resorptive agents in a controlled, site-specific manner. Collagen-based matrices loaded with BPs have achieved more than a twofold increase in defect fill and notable improvements in trabecular microarchitecture in osteoporotic animal models. 95 Similarly, carbonate apatite scaffolds functionalized with anti-sclerostin antibodies have restored bone remodeling dynamics by facilitating osteocyte differentiation and reducing osteoclastic activity. 96 Multifunctional polymeric composites combining parathyroid hormone and simvastatin within GelMA/PLA scaffolds have further enhanced osteogenic marker expression (ALP, BMP-2, COL-1, and OCN) and promoted robust bone formation in osteoporotic cranial defects. 97 Together, these developments trace a clear evolution, from ion-mediated immunomodulation to multi-drug, microenvironment-responsive delivery systems transforming biomaterials from passive defect fillers into dynamic therapeutic platforms that orchestrate osteogenesis, angiogenesis, and immune regulation for functional restoration of osteoporotic bone.
Biomaterials combined biological therapeutics
MSC-Loaded scaffolds
MSC-loaded scaffolds have emerged as powerful platforms for bone regeneration due to their ability to enhance osteogenic differentiation and paracrine signaling. MSCs seeded onto biomimetic scaffolds not only differentiate into OBs but also modulate local environments through secreted factors that support angiogenesis and immune modulation. Moreover, studies have consistently reported that MSC-loaded hydrogels, nanofibrous meshes, and 3D-printed porous scaffolds increase bone volume and quality in preclinical models. 98 Preconditioning MSCs via BMP-2 priming or genetic modification further amplifies these benefits. For example, BMP-2-pretreated MSCs on nanofibrous bacterial cellulose scaffolds exhibited early and enhanced matrix mineralization and bone maturation. 99
Recent studies have advanced MSC-loaded scaffold strategies by integrating angiogenic stimulation, biochemical modulation, and natural compound loading to synergistically enhance osteogenesis. A study fabricated a 3D-printed porous titanium alloy scaffold combined with adipose-derived MSCs (ADSCs) and platelet-rich-plasma (PRP) gel, demonstrating in a rabbit femoral defect model that the construct significantly enhanced vascular network formation and new bone deposition compared to scaffolds alone. 100 Similarly, another study reported that a kaempferol-loaded bioactive glass-based scaffold seeded with BMMSCs achieved an approximately 200% increase in new bone area (NB%) after 12 weeks relative to non-cell-loaded controls in a rat calvarial defect model. 101 Complementary reviews, such as Saberian et al., summarize how scaffold systems combined with stem cells benefit further from preconditioning and growth-factor gradients, including stromal cell-derived factor-1 (SDF-1), connective tissue growth factor (CTGF), and transforming growth factor β3 (TGF-β3) that improve MSC recruitment, survival, and differentiation in bone regeneration settings. 102 Collectively, these findings highlight that coupling MSCs with pro-angiogenic cues, natural osteoactive compounds, and targeted cytokine signaling can markedly amplify scaffold-mediated bone repair outcomes.
Extracellular vesicle-functionalized biomaterials
To address challenges associated with cell therapies, such as immune reactions and poor cell survival rates, the focus has recently shifted to MSC-derived EVs, which carry bioactive cargoes such as miRNAs and proteins, that promote osteogenesis, angiogenesis, and immune regulation. 21 A recent review synthesized advances in EV-functionalized scaffolds that support bone repair through improved osteogenesis and angiogenesis. For instance, EV-integrated bioactive scaffolds enhanced vascularized bone regeneration compared to controls. 103 Additionally, macrophage-derived EVs (particularly from the M2 phenotype) further demonstrated immunomodulatory potential: EVs derived from M2 macrophages (M2 EVs) increased MSC osteoinductive gene expression both in vitro and in rat calvarial models, compared to EVs derived from M1 macrophages (M1 EVs), which impaired healing. 104 In a 2024 study, Al-Sharabi et al. isolated osteogenic MSC-derived EVs (Osteo-EVs) enriched in collagen I, osteopontin and alkaline phosphatase, and immobilized them on collagen scaffolds, which markedly enhanced new bone formation in rat calvarial defects compared with unmodified EVs or scaffold alone. 105 Mechanistically, MSC-EVs promote M2 macrophage polarization via miR-1246 and miR-451a and stimulate angiogenesis through miR-21/miR-27b, while delivering Runx2/osterix-related proteins that trigger osteogenic differentiation 106 When incorporated into hydrogels or ceramic–polymer scaffolds, EVs show improved retention, prolonged signaling, and superior vascularized bone repair relative to direct EV injection. These findings highlight EV-integrated scaffolds as promising acellular therapeutics capable of coordinated osteogenesis, angiogenesis, and immune modulation in osteoporotic bone.
Immunomodulatory scaffolds
Endogenous immune responses are critical for bone healing, with macrophage polarization (M1 → M2) playing a pivotal role. 107 Biomaterials can be engineered to modulate macrophage phenotype and thereby enhance repair. Immunomodulatory scaffolds deliberately engage the osteoimmune axis to shift a pro-resorptive, inflammatory microenvironment toward a pro-regenerative one. By modulating the activity of macrophages, T cells, and B cells, these scaffolds influence osteoclastogenesis and osteoblast function through key mediators such as RANKL/OPG, TNF α, IL-1β, IL-6, and IL-17, thereby directly affecting bone remodeling. This immunomodulation is achieved through multiple strategies: biochemical cues, such as incorporation or controlled release of IL-10, TGF-β, or NF-κB inhibitors; physical cues, including surface chemistry, stiffness, nanotopography, and pore architecture that bias macrophage polarization toward M2-like, pro-repair states; and delivery of bioactive cargo, such as MSC-derived EVs or targeted drugs, which reprogram resident immune and progenitor cells. 108 In addition, scaffold materials can act as ROS scavengers or controlled-release depots to blunt chronic inflammation and senescence-associated secretory phenotypes (SASP), preserving tissue homeostasis. 109 Mechanistically, successful immunomodulatory scaffolds attenuate NF-κB and MAPK signaling while enhancing STAT3/SMAD and PI3K/Akt pathways in macrophages and MSCs, promoting IL-10 and TGF-β production, angiogenic factor release, and downstream Runx2/β-catenin–mediated osteogenic programs. 110 Collectively, these multifaceted strategies enable scaffolds to create a local microenvironment conducive to bone regeneration and functional repair.
Smart and bioresponsive biomaterials
Smart biomaterials that dynamically respond to pathological cues in osteoporotic bone, such as inflammation, acidity, or oxidative stress, are emerging as powerful therapeutic platforms. ROS-responsive hydrogels, for example, scavenge excessive ROS, alleviate inflammation, and inhibit OC activity, thereby enhancing bone regeneration. Recent studies utilized hydrogels that degraded in response to ROS, releasing therapeutic agents that reduced inflammation and promoted osteogenesis.111–113 Similarly, antioxidant scaffolds, often loaded with nanozyme particles, have demonstrated suppression of oxidative stress and improvement in osteogenic differentiation and vascularization in vivo. 114 Additionally, pH-sensitive scaffolds are engineered to exploit the acidic microenvironment of bone resorption zones. For example, mesostructured glass–HA composites release antibiotics like levofloxacin more rapidly under acidic conditions (~pH 5.5–6.7), which can help treat infection-mediated OP while sustaining bone-forming cues. 115 Advances in 3D printing techniques have led to scaffolds with spatiotemporal control over drug release. A milestone study by Zhu et al. introduced a polydopamine-modified PLLA scaffold carrying antibacterial eugenol and angiogenic DMOG (dimethyloxalylglycine)-loaded layered double hydroxides. This scaffold initially released antibacterial agents sequentially, before transmitting sustained osteogenic/angiogenic signals through DMOG, facilitating vascularized bone healing in vivo. 116 The incorporation of real-time monitoring sensors into biomaterials is an advanced yet promising area of research. Indeed, fiberoptic sensors, such as Raman-based probes embedded within calcium phosphate scaffolds, enable in vivo tracking of mineralization and tissue growth during healing. 117 In addition to Raman probes, innovative sensor approaches have been successfully integrated into orthopedic biomaterials for real-time, in vivo monitoring of bone regeneration. For instance, Schilling et al. developed electrospun fiber scaffolds incorporating a two-photon-excitable phosphorescent probe (PtP-C343), enabling high-resolution, real-time measurement of local oxygen tension during cranial bone defect healing via two-photon phosphorescence lifetime microscopy. 118 Another important strategy involves embedding near-infrared (NIR) fluorescent copolymer scaffolds (ZW-PCLG) paired with labeled stem cells to noninvasively track both scaffold degradation and cell localization in a rat calvarial defect model using NIR imaging. 119 Additionally, Tan et al. demonstrated a passive wireless magnetoelastic sensor embedded in bone-plate constructs that wirelessly measures strain evolution during healing in a goat tibia model, offering battery-free, longitudinal tracking of fracture stabilization. 120 Research on fiber Bragg grating sensors within 3D-printed constructs has demonstrated real-time monitoring of mechanical integrity, which could potentially be used in personalized osteoporotic care. 121
Clinical translation and challenges
The successful clinical translation of biomaterial-based therapies remains challenging, despite promising preclinical results in osteoporotic animal models. Many innovative scaffolds combining cells, growth factors, or smart materials achieve remarkable bone regeneration in rodent or ovine OP models, such as strontium-doped Ca-Si bioceramics enhancing bone and vascular regeneration in the ovariectomized model. 122 Mesoporous bioactive glass–PCL composites also demonstrated robust integration and neovascularization when implanted in osteoporotic sheep. 44 However, translating these findings into human therapy will have to overcome several hurdles.
Clinical trials and commercial products
Current clinical management of osteoporotic fractures combines pharmacological therapy with surgical reconstruction using biomaterials that restore bone integrity. While antiresorptive and anabolic drugs reduce systemic bone loss, local repair depends on osteoconductive bone substitutes, especially calcium phosphate–based materials such as HA and β-TCP, which have been used clinically for over five decades. Commercial β-TCP products like chronOS® (DePuy Synthes) and Vitoss® (Stryker) provide porous, resorbable scaffolds that promote osteointegration and remodeling. Clinical use of chronOS Inject has shown 93% fracture union with no implant-related complications in tibial defects. 123 Other clinically approved substitutes include bioactive glass (BonAlive® S53P4) and composite collagen-HA scaffolds (e.g. Collagraft®, Healos®), which improve handling, angiogenesis, and osteoconductivity in osteoporotic bone. 124 Calcium-based cements such as OsteoSet® and Stimulan® are also clinically used as injectable fillers and drug carriers in fragility fractures. 125 Collectively, these long-established biomaterials remain the clinical gold standard for osteoporotic bone defect repair due to their safety, regulatory approval, and extensive human data.
Technical and manufacturing hurdles
The “4Fs” framework (Form, Function, Fixation, and Formation) outlines essential scaffold requirements for clinical use. Meeting all these requirements simultaneously remains technically demanding. Moreover, production challenges persist in achieving reproducibility and scale-up under cGMP conditions, along with maintaining batch consistency and sterile handling. 126
Long-term biocompatibility and safety
Although short-term studies suggest safety, long-term outcomes, such as scaffold degradation profiles, immune responses, and the potential for heterotopic ossification, require extended follow-up. Non-invasive imaging modalities (e.g. micro-CT, magnetic resonance imaging (MRI), quantitative histology, and mechanical testing are essential), yet they are often lacking in clinical protocols. 127
Ethical and cost considerations
Patient-specific or cell-based construct therapies tend to be more expensive than conventional implants. Additionally, ethical concerns arise around cell sources and genetic modifications. Moreover, health economic evaluations and cost-effectiveness analyses are essential for real-world adoption but are sporadically performed in early-phase studies.
Future perspectives
The future of biomaterials in OP therapy lies at the intersection of 3D bioprinting, personalized medicine, and artificial intelligence (AI), reshaping scaffold design into patient-specific regenerative platforms. Recent advances highlight the 3D printing of bioactive materials such as polyetheretherketone (PEEK) into custom bone-plate scaffolds, with nanoscale HA coatings that greatly accelerate bone regeneration in rabbit femur models, heralding the possibility of personalized, load-bearing implants. 128 In addition to shape customization, the integration of bioactive coatings mimicking native bone surfaces has been shown to enhance MSC adhesion and osteogenesis.
AI-driven and machine learning–augmented 3D bioprinting systems are now used to tune scaffold porosity, mechanical strength, and structural fidelity. One notable study by Daryabor et al. implemented a regression-based ML framework using empirical filament width data to predict and control scaffold geometry, thereby enhancing print accuracy and reproducibility in extrusion-based bioprinting.129,130 Another study reported on the development of a time-dependent optimization model for polymer scaffold porosity that maximizes stiffness throughout bone healing, a computational approach validated to reduce failure risk in biodegradable constructs. 131 These works confirm that AI-guided parameter adjustments in scaffold fabrication can deliver mechanically optimized, patient-specific constructs with reliable performance, marking a significant step toward truly customized, precision bone-regenerative scaffolds.
The next frontier involves multifunctional hybrid scaffolds that integrate drugs, EVs, smart sensors, and responsive elements. For example, combining antibacterials, growth factors, or exosome releases into staged or stimuli-sensitive layers can mimic temporal healing processes. AI-driven bioprinting of such complex architectures promises programmable local therapy with minimal invasiveness.
Looking further into the future, patient-specific regenerative platforms will reshape osteoporotic treatment. Using CT/MR data, AI-optimized CAD models will enable personalized bioprinting of scaffolds infused with the MSCs or EVs from the patient. Incorporating sensors into printed constructs, such as Raman-based probes, will allow real-time monitoring of healing, offering immediate feedback on effectiveness and enabling adaptive therapy adjustments (Figure 6). 132

Schematic illustration of a personalized bone regeneration workflow integrating 3D bioprinting and real-time monitoring technologies. The process begins with combining a patient-specific regenerative platform comprising autologous or compatible therapeutic cells (e.g. MSCs, OB, immune cells) and a biofunctional 3D ink optimized for bone tissue engineering. This composite is used in 3D bioprinting to fabricate a custom-made bone scaffold tailored to the individual anatomical and biological requirements of each patient. Embedded sensors within the scaffold enable real-time, in situ monitoring of the local microenvironment, such as mechanical strain, pH, or oxygen levels, during the healing process. These smart constructs support the implementation of personalized medicine approaches by providing dynamic feedback and facilitating timely clinical decisions, ultimately enhancing the precision and efficacy of bone regeneration.
In summary, the union of 3D bioprinting, AI personalization, and multifunctional design is paving the way for next-generation biomaterial scaffolds that are not just implants, but actual living, adaptive systems capable of actively regenerating osteoporotic bone in patients. The synergy of materials science, biology, and clinical need drives a vision of precision regenerative therapy that responds intelligently and dynamically to the unique healing journey of each patient.
Conclusion
OP represents a multifactorial disorder involving intricate disruptions in cellular communication, immune regulation, and microenvironmental homeostasis. Traditional pharmacological approaches alone are insufficient to restore structural and functional bone integrity. The rapid evolution of biomaterial science spanning natural, synthetic, and composite scaffolds; nanocarrier-based delivery systems; and multifunctional, stimuli-responsive matrices has introduced a paradigm shift from passive bone fillers to active, biointeractive systems. Key advances summarized in this review include (i) the development of nanostructured and surface-engineered scaffolds capable of directing osteogenesis and angiogenesis; (ii) localized, controlled drug and gene delivery strategies to rebalance OB-OC activity; (iii) integration of stem cells and extracellular vesicles to modulate immune and regenerative signaling; and (iv) the emergence of AI-driven 3D bioprinting and in situ monitoring platforms that enable personalized bone regeneration. In summary, as illustrated in Figure 7, these diverse biomaterial strategies converge to actively reprogram the osteoporotic niche, utilizing specific signaling pathways, such as integrin-mediated mechanotransduction and immunomodulatory polarization to drive the transition from a pathological state to functional bone restoration.

Schematic representation of biomaterial-mediated mechanisms for osteoporotic niche restoration. The transition from a pathological state to a restored niche is achieved through specific signaling pathways triggered by biomaterial interventions. The osteoporotic niche (left) is characterized by chronic inflammation, elevated Reactive Oxygen Species (ROS), loss of Type H vessels, ECM degradation, and upregulated RANKL activity. Biomaterial interventions (middle) target these dysfunctions through three primary mechanisms: (1) Porous Scaffolds provide physical cues that activate Integrin/mechanotransduction, stimulating FAK/ERK signaling to promote OB differentiation. (2) Bioactive Hydrogels facilitate ROS scavenging, driving an immunomodulatory M1 → M2 macrophage shift that resolves inflammation. (3) NPs and EVs ensure intracellular delivery of therapeutic cargoes, resulting in gene silencing (e.g. siRNA targeting RANK) or anabolic signaling (e.g. BMP2/VEGF) that restores angiogenesis and mineralization. Collectively, these mechanistic pathways re-establish bone homeostasis and structural integrity (niche restoration). (Created with BioRender).
Despite these promising developments, several challenges remain. The translation of preclinical success to clinical practice is hindered by issues of large-scale manufacturing, long-term biosafety, reproducibility, and standardization of regulatory pathways. Furthermore, understanding the temporal coordination between immune, vascular, and osteogenic cues in the osteoporotic niche remains incomplete. Future work should focus on creating smart, adaptive biomaterials that integrate real-time biosensing, data-driven design, and personalized therapeutic modulation. Interdisciplinary collaboration among materials scientists, biologists, and clinicians will be crucial to achieving precision regenerative therapy for osteoporotic bones. In essence, biomaterials are evolving from structural support into intelligent, patient-specific therapeutic systems, heralding a new era of integrative and responsive bone regeneration.
Footnotes
Abbreviations
AI: Artificial intelligence
β-TCP: β-tricalcium phosphate
BMP 2: Bone morphogenic protein-2
BP: Bisphosphonate
Ca RISNPs: Calcium risedronate nanoparticles
ECM: Extracellular matrix
EV: Extracellular vesicle
HA: Hydroxyapatite
HC: Hydroxyapatite collagen
IL-1: Interleukin-1
IL-6: Interleukin-6
MAPK: Mitogen-activated protein kinase
M1 EVs: Extracellular vesicle derived from macrophage 1
M2 EVs: Extracellular vesicle derived from macrophage 2
MSCs: Mesenchymal stem cells
NF-κB: Nuclear factor kappa-light-chain-enhancer of activated B cells
OB: Osteoblast
OC: Osteoclast
OPG: Osteoprotegerin
OP: Osteoporosis
PCL: Polycaprolactone
PEG: Polyethylene glycol
PEEK: Polyetheretherketone
PLGA: Poly(lactic-co-glycolic acid)
RANKL: Receptor activator of nuclear factor kappa-B ligand
rhBMP-2: Recombinant human bone morphogenetic protein-2
RNA: Ribonucleic acid
ROS: Reactive oxygen species
SASP: Senescence-associated secretory phenotype
SDF-1: Stromal cell-derived factor-1
SERM: Selective estrogen receptor modulators
siRNA: small interfering ribonucleic acid
TNF-α: Tumor necrosis factor-alpha
TUDCA: Tauroursodeoxycholic acid
VPA: Valproic acid
VEGF: Vascular endothelial growth factor
WHO: World health organization
Ethical Considerations
Not applicable
Consent to Participate
Not applicable
Author Contributions
The review topic was conceived and designed by P.D.A., Y.A., and S.L. The manuscript was drafted by P.D.A., and N.P., and it was revised and critically edited by Y.A., and S.L.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT; NRF-2022R1A2C3004850 and RS-2023-00208529) and Korean Fund for Regenerative Medicine funded by Ministry of Science and ICT, and Ministry of Health and Welfare (RS-2025-02072977, Republic of Korea).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
