Abstract
This study aimed to examine the histological and mechanical effects of cryopreservation on human aortic tissues, focussing on storage duration and conditions. Assessments included smooth muscle cell integrity, elastic fibre preservation, and endothelial viability. Cryopreservation with dimethyl sulphoxide (DMSO) significantly reduced smooth muscle cell nuclei loss and maintained elastic fibre integrity. However, elastic fibre thickness increased after 12 months. Isolectin B4 staining showed reduced endothelial cell viability across all groups. No significant changes were observed in mucoid extracellular matrix accumulation or elastic fibre fragmentation. These findings suggest that cryopreservation with DMSO effectively maintains structural integrity for up to 12 months but requires refinement to address endothelial and biomechanical concerns. Cryopreserved aortic allografts demonstrated structural and functional performance when stored at low temperatures, confirming their viability for reconstructive surgeries. The study highlights the importance of the timely utilisation of cryopreserved grafts and optimising preservation techniques to advance surgical applications.
Introduction
The use of cryopreserved aortic allografts for airway reconstruction has emerged as a feasible option to produce viable substitutes for airway segments, including the trachea, carina, and bronchi. 1 In recent years, the thoracic surgery team at National Taiwan University Hospital has successfully applied cryopreserved aortic homografts in allogeneic transplantation to treat patients with tracheal injuries and stenosis. These clinical cases provide real-world validation of the feasibility and therapeutic potential of cryopreserved aortas for complex airway reconstruction.2,3 Previous studies have indicated that although short-term refrigeration may adversely affect the mechanical properties of tissues, freezing at extremely low temperatures (−80°C) helps maintain the structural integrity and biological properties of the allografts.4,5 This innovative approach, which employs stented aortic matrices, not only provides structural support but also facilitates tissue regeneration, making it a promising technique for airway reconstruction.1,6 The feasibility of bioengineered tracheal and bronchial reconstruction using stented cryopreserved aortic allografts has been demonstrated in preclinical studies and preliminary human trials.1,7 In a study on In Vivo Tissue Engineering of Human Airways, the authors showed the potential of constructing human airway tissues using tissue engineering techniques, further validating the clinical potential of cryopreserved aortic matrices. 8 This study highlighted the complexities of in vivo airway tissue engineering and its impact on long-term functional repair, laying the groundwork for future clinical applications.
The quality of cryopreserved aortas is crucial for determining their functional performance and long-term durability. Understanding the effect of cryopreservation on the mechanical properties and viability of aortic homografts is essential for optimising patient outcomes. Several studies have explored the composition, biomechanical properties, and viability of cryopreserved aortas, revealing factors that affect the survival of these grafts.5,9,10 Moreover, a previous study analysed the mechanical properties and structural changes that occur during the thawing process, providing valuable insights for enhancing preservation techniques and optimising the use of cryopreserved aortas in various clinical scenarios. 10 Prior studies further indicate that, despite preserving graft usability, decellularisation can impair long-term mechanical performance as a result of differential alterations in collagen and elastin fibres accompanied by vessel-wall thinning.11,12
The primary objective of this study was to assess the impact of storage time prior to cryopreservation on the mechanical properties and viability of aortic homografts. By comparing the properties of aortic homografts cryopreserved for different durations, we aimed to identify potential changes in their mechanical behaviour and overall quality.9,10 In addition, this study aimed to investigate factors that contribute to the development of dysfunction in cryopreserved aortic allografts, offering a comprehensive analysis of their long-term function and associated risk factors.9,13 The study findings will be useful in improving outcomes for high-risk populations undergoing aortic reconstruction procedures and enhancing the overall understanding of cryopreservation techniques for aortic tissues.
The present study is significant because it is the first study to collect human aortas on a large scale and conduct comprehensive long-term preservation experiments.
Furthermore, given the scarcity of donated human aortas, this study aimed to explore the potential use of porcine aortas for xenotransplantation, based on experience with porcine valve replacements for human heart valves. A review article demonstrated a good correspondence between the stiffness of porcine and human aortas, suggesting that porcine aortas could serve as a viable alternative in the future. 14 Therefore, this study aimed to analyse the mechanical features of porcine aortas and compare them with those of human aortas, potentially expanding the applications of cryopreserved aortic homografts in clinical practice.
Results
Histopathology cross-sectional investigation of human CAo grafts
Individual histopathological data for the assessment of various groups are provided in Supplemental appendices; the comparative results are summarised in Table 1. The Kruskal–Wallis test, Dunn’s multiple comparisons, and the Fisher exact test were used for statistical analysis. Group E, with samples preserved at −80°C, served as the control group to assess the effect of different preservation conditions on the integrity of aortic tissue.
Comparative histological results from human CAo graft groups and cross-sectional investigation.

: normal saline; p > 0.05 when compared with group E.
/**p < 0.05/0.01 when compared with group E.
Regarding age, there was no significant difference between the groups (p > 0.05), indicating that age did not affect the study outcomes.
Smooth muscle cell nuclei integrity
Smooth muscle cell integrity in the tunica media showed significant differences depending on the preservation method. Preservation at −80°C without dimethyl sulphoxide (DMSO; Group E) caused severe and widespread patchy disruption of smooth muscle nuclei. In contrast, samples preserved with DMSO for 3–12 months demonstrated significantly fewer alterations, reducing from moderate to almost negligible changes (p < 0.05). Besides, this protective effect of DMSO was observed in fresh samples stored at room temperature. However, samples stored at 4°C (Group G) exhibited similar disruptions compared to those preserved at −80°C, although the small sample size in Group G may have affected the results.
Regarding endothelial cell viability in cryopreserved human aorta samples, analysis was conducted at various time points, ranging from fresh samples to those preserved for up to 12 months. In both groups of older individuals, a distinct patchy loss of smooth muscle cells was observed in the aorta, becoming more pronounced as the cryopreservation period increased. In contrast, young males exhibited only sporadic loss of smooth muscle cells, mainly after 3 and 12 months of preservation. This indicates that younger individuals experienced a less severe impact of cryopreservation on smooth muscle cell viability compared to older individuals (Figure 1).
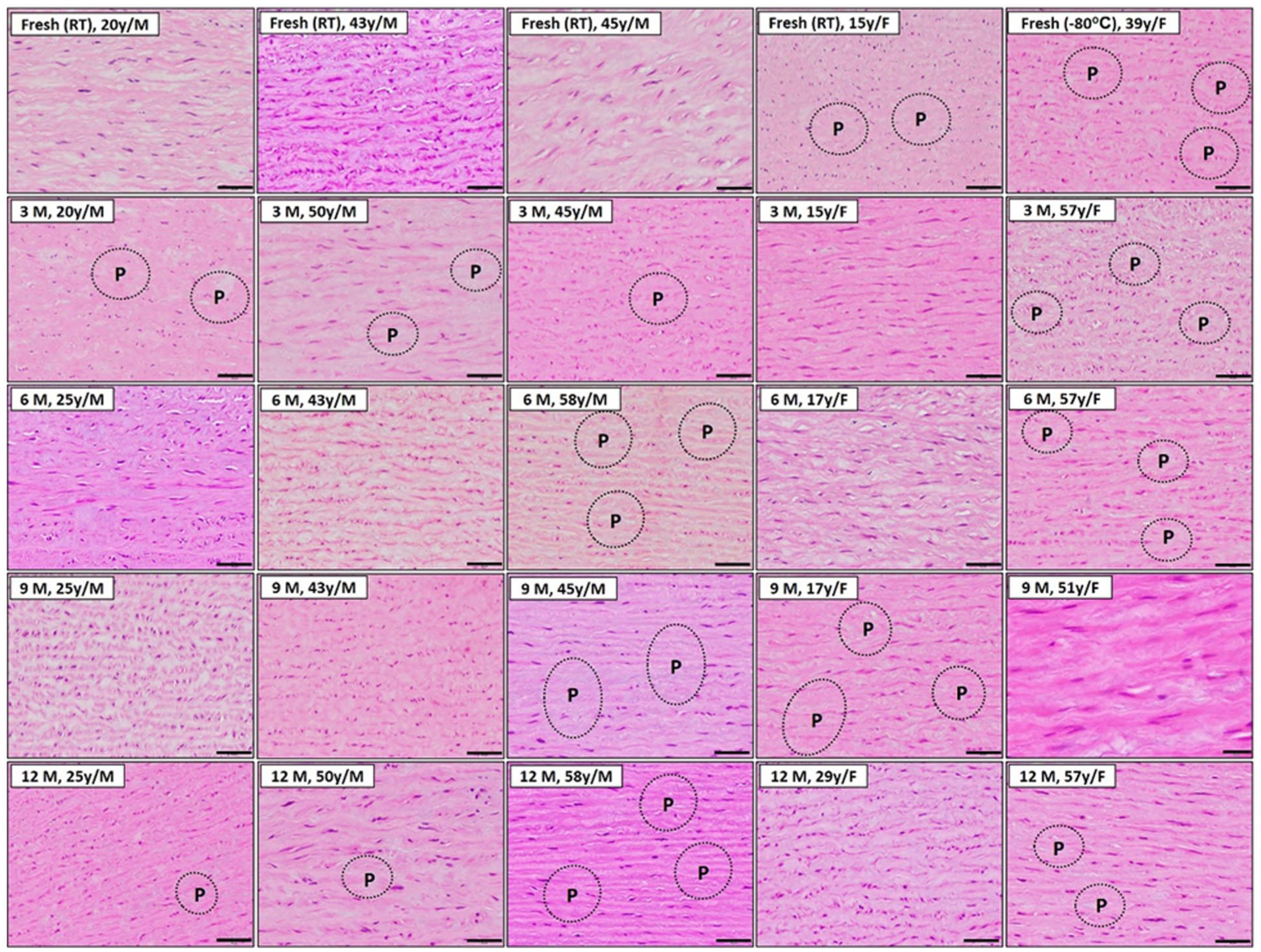
Histological sections of human aorta specimens stained with haematoxylin-eosin-saffron (scale bar = 50 µm). Cryopreserved human aorta samples were analysed for endothelial cell viability at different time points (fresh to twelve months). Both groups of older individuals exhibited significant patchy loss of smooth muscle cells in the aorta, which increased with the duration of cryopreservation. In contrast, young males displayed sporadic smooth muscle cell loss in the aorta after 3 and 12 months of preservation.
Endothelium integrity in tunica adventitia
In Group E, where samples were preserved at −80°C without DMSO, there was a rapid decline in the integrity of endothelial cells in the tunica adventitia, as shown by a marked decrease in Isolectin B4 labelling intensity. Even with the addition of DMSO, endothelial integrity continued to deteriorate in samples stored at 4°C and room temperature and in those preserved with DMSO for 3–12 months. However, these results were not statistically significant, and factors such as congenital conditions or post-mortem changes may have contributed to the decline in endothelial integrity.
Immunohistochemical (IHC) staining further demonstrated a significant reduction in aortic endothelial integrity after 1 year of preservation in both a 50-year-old male and a 57-year-old female (indicated by white arrowheads). In contrast, the aortic endothelial integrity of a 45-year-old male remained intact after undergoing three different types of preservation (Figure 2).

Histological sections of the human aorta stained with Isolectin B4 (scale bar = 50 µm). Immunohistochemical staining revealed a significant reduction in aortic endothelial integrity after 1 year of preservation in a 50-year-old male and a 57-year-old female (white arrowhead). In contrast, the endothelial integrity of the 45-year-old male aorta remained intact after undergoing three types of preservation.
Mucoid extracellular matrix accumulation (MEMA)
MEMA was assessed in two forms: translamellar (MEMA-T) and intralamellar (MEMA-I). MEMA-T, characterised by elastic fibre disruption, was observed sporadically across all groups, with no significant difference (p > 0.05). MEMA-I, which preserved elastic fibre integrity, showed no notable variations between groups, except for Group G, where the small sample size (n = 1) may have skewed the results. Overall, DMSO did not significantly affect MEMA accumulation, indicating that the preservation methods did not alter the formation of this abnormal extracellular matrix.
Elastic fibre fragmentation and loss
Elastic fibre fragmentation was observed across the groups; however, there were no significant differences between different preservation methods. Freshly harvested samples, as well as those preserved at −80°C, 4°C, or room temperature, showed no significant changes in elastic fibre fragmentation (p > 0.05). However, when samples were preserved in DMSO and stored at −80°C, the elastic fibres remained intact for 3–12 months.
Elastic fibre thickness and disorganisation
The thickness of elastic fibres varied depending on the preservation method. Samples stored at −80°C exhibited significantly thinner elastic fibres (2.978 ± 0.773 µm) compared to those stored at 4°C (3.618 ± 1.235 µm) and room temperature (4.742 ± 0.955 µm), indicating that low-temperature storage contributes to fibre thinning (p < 0.05). In contrast, DMSO-preserved samples showed a gradual increase in fibre thickness over time, with a significant increase observed in Group A after 12 months of preservation. However, elastic fibre disorganisation did not show significant differences across groups (p > 0.05), suggesting that preservation methods did not significantly affect the overall arrangement of the fibres.
In addition, degenerative elastic fibres, characterised by partial dilation and a dense purple appearance (marked by white arrowheads), increased in quantity as the preservation period lengthened. The most pronounced dilation of elastic fibres occurred after preservation at room temperature, followed by 9 months of preservation (Figure 3).

Histological images of the human aorta stained with Movat’s pentachrome (scale bar = 50 µm). In the detection of elastic fibres, the degenerative fibres, which partially dilated and appeared in dense purple (white arrowhead), increased with the preservation period. Notably, the elastic fibres were most dilated after preservation at room temperature, followed by 9 months of preservation.
Longitudinal investigation of histopathology
Longitudinal analysis of histological changes in aortic patches, as shown in Table 2, revealed no significant effect of long-term preservation on various parameters, including smooth muscle nuclei integrity, microvascular endothelium signal, MEMA-I and MEMA-T accumulation, elastic fibre fragmentation, lamellar media collapse, and elastic fibre disorganisation. The only notable finding was the increase in elastic fibre thickness after 12 months of preservation, which could have implications for the mechanical strength of the patches. This raises questions about the potential long-term effects of increased elastic fibre thickness on the structural integrity of aortic patches.
Comparative histological results from human CAo graft groups, longitudinal investigation, and the dangers of long-term preservation.

DMSO-related cytotoxicity
The potential cytotoxic effects of DMSO are presented in Table 3. There was no significant effect of 10% DMSO on critical histological parameters, such as smooth muscle nuclei preservation, microvascular endothelium integrity, MEMA formation, lamellar media integrity, and elastic fibre fragmentation or thickness. These findings suggest that the use of 10% DMSO is safe for preserving aortic patches over extended periods, without inducing detrimental histological changes.
Comparative histological results from human CAo graft groups, longitudinal investigation, and the dangers of DMSO.

Characterisation of porcine CAo graft
This study investigated the histopathological changes in porcine aortic tissue subjected to different preservation conditions, with a focus on the integrity of smooth muscle cells, microvascular endothelial cells, elastic fibres, and extracellular matrix components (Table 4).
Comparative histological results across preservation periods and methods in the porcine CAo graft groups.

p < 0.05 when compared with group E. **p < 0.01 when compared with group E.
Group A-D (Cryopreserved with 10% DMSO): Smooth muscle cells showed moderate to severe nuclei loss in groups preserved for 9–12 months, with Group D (3 months) showing the least damage. Elastic fibre fragmentation was most prominent in the 12-month preservation group, whereas endothelial integrity remained strong across all samples. Group E (−80°C without DMSO): Fresh samples showed minimal to no smooth muscle cell damage or elastic fibre loss, and the endothelial cell integrity was well preserved.
Loss of porcine smooth muscle cell nuclei
Smooth muscle cell nuclei loss varied significantly across the different preservation conditions and durations (Figure 4).

Histological sections of porcine aorta and cellular components cryopreserved at different time points (scale bar = 50 µm). Haematoxylin-eosin-saffron staining revealed no significant loss of smooth muscle cell nuclei across different preservation periods and methods.
These findings indicate that preservation at −80°C with 10% DMSO can effectively maintain smooth muscle cell integrity for up to 6 months, after which progressive degradation becomes evident, particularly beyond the 9-month threshold.
Microvascular endothelial cell integrity in porcine samples
The integrity of the endothelial cells in the tunica adventitia was assessed using Isolectin B4 staining and immunoreactive scores (IRS; Figure 5).
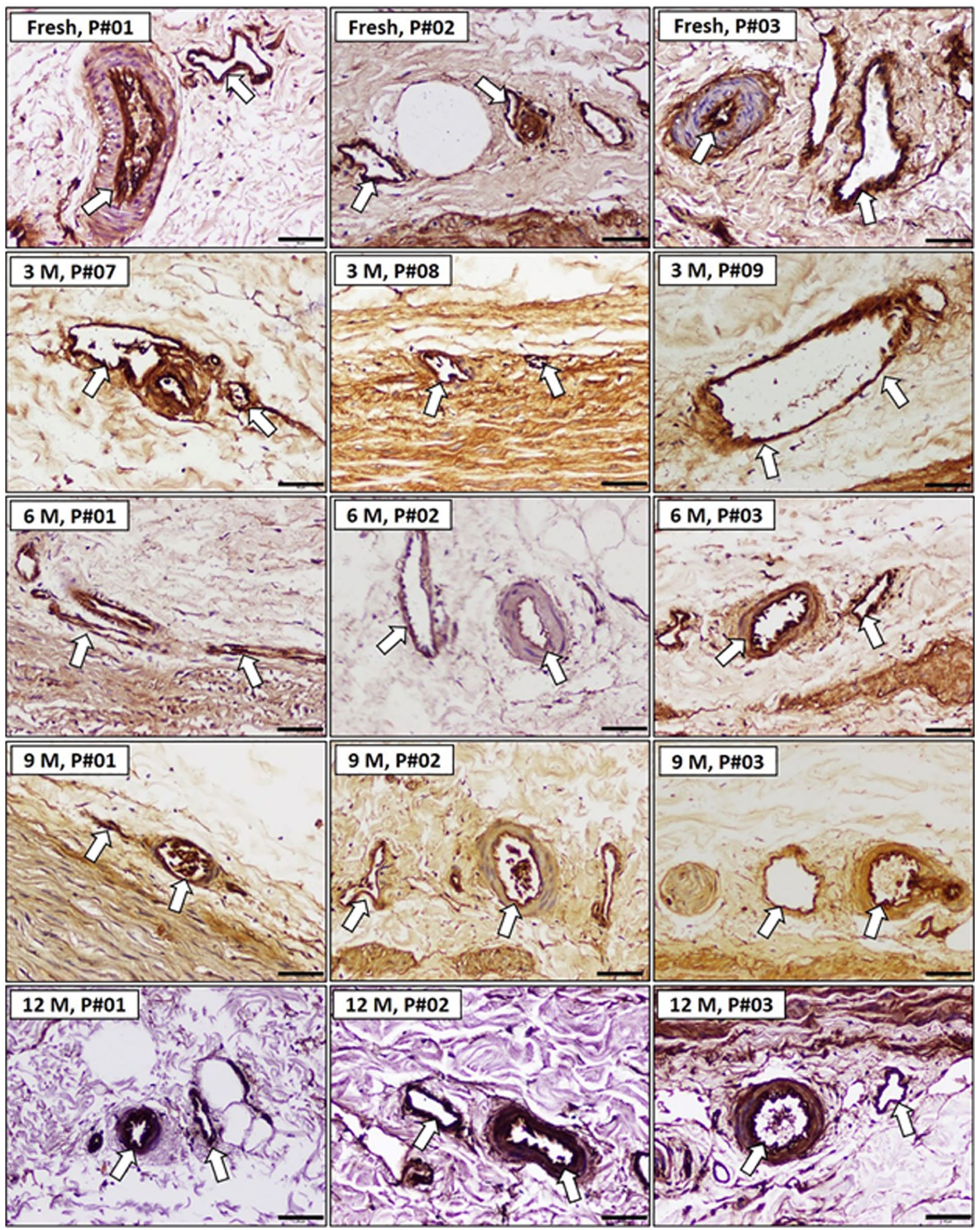
Histological sections of porcine aorta and cellular components cryopreserved at different time points (scale bar = 50 µm). Immunohistochemical staining indicated the preservation of endothelial integrity.
These results underscore the importance of DMSO in safeguarding endothelial cell integrity during cryopreservation. In contrast, preservation at −80°C without DMSO was associated with reduced endothelial health, highlighting the necessity of cryoprotectants for long-term tissue viability.
MEMA in porcine samples
The presence of MEMA-T and MEMA-I accumulation was evaluated to detect potential pathological alterations.
Overall, the results suggest that neither the cryopreservation methods nor storage duration contributed to MEMA and consequently compromised tissue functionality.
Elastic fibre fragmentation and loss in porcine samples
Elastic fibre integrity was evaluated as an important indicator of structural stability, with fragmentation signalling degradation (Figure 6).
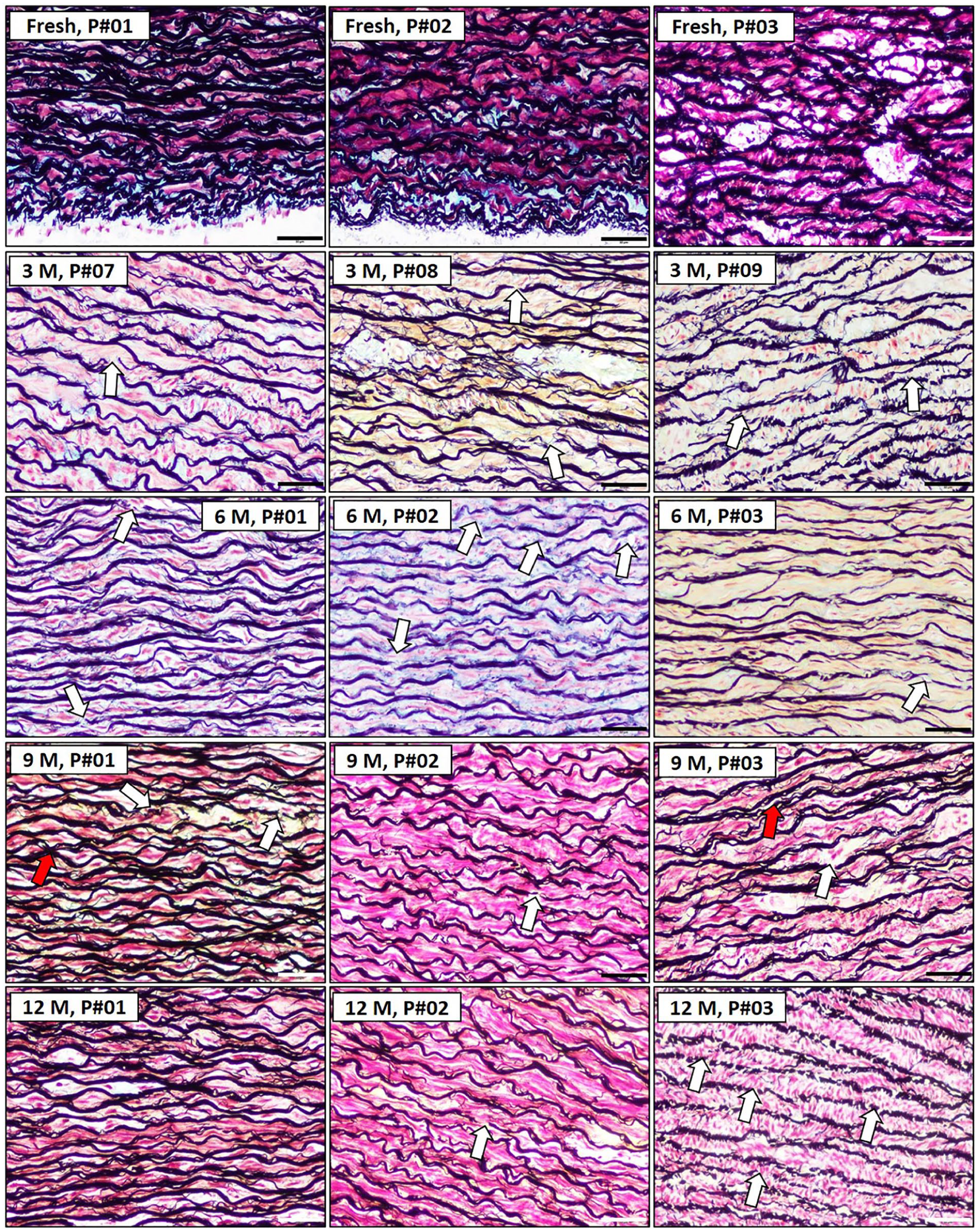
Histological sections of porcine aorta and cellular components cryopreserved at different time points (scale bar = 50 µm). Movat’s pentachrome staining identified fragmented (white arrowhead) and degenerated (red arrowhead) elastic fibres in porcine #01 and #03 after 6 and 9 months of preservation. Porcine #03 exhibited severe fragmented elastic debris surrounding the lamellar space.
These results indicate that elastic fibres begin to degrade after 9 months of storage at −80°C with DMSO. This degradation may compromise the mechanical properties of the tissue, especially for applications requiring the preservation of elasticity.
Lamellar media collapse and elastic fibre thinning in porcine samples
No significant lamellar media collapse was observed in any group, suggesting that none of the preservation methods caused structural collapse or thinning of the tissue.
Elastic fibre thickness and disorganisation in porcine samples
Elastic fibre thickness and disorganisation were assessed as markers of structural changes.
These results indicate that while elastic fibre thickening occurs over time, disorganisation becomes a concern after prolonged preservation, potentially affecting the mechanical performance of the tissue under stress.
Aorta tensile strength test
We performed independent sample and repeated measures variance analyses to investigate whether there were significant differences in the thickness, subfailure stress, max stress, elongation, low stretch modulus, and high stretch modulus of human and porcine arteries at different preservation times (0, 3, 6, 9, and 12 months; Figure 7). The average values of thickness, subfailure stress, max stress, elongation, low stretch modulus, and high stretch modulus for human and porcine arteries at different preservation times were presented using bar charts. In addition, Pearson correlation analysis was used to explore the linear relationships between max stress, elongation, high stretch modulus and thickness, subfailure stress, low stretch modulus, and age. The results are presented below.

Arterial mechanics quantified and summarised under tensile test.
Results of the ANOVA for human arteries preserved over different durations
Table 5 presents the results of the ANOVA for human arteries preserved over different periods. The findings indicate no significant differences across most variables, including thickness, subfailure stress, max stress, elongation, low stretch modulus, and high stretch modulus. For thickness, although the measurements fluctuated between preservation periods (e.g. 1.414 mm at 0 months and 1.274 mm at 9 months), the changes were not significant (p = 0.633). This suggests that preservation time did not considerably affect arterial wall thickness.
Results of ANOVA for human arteries preserved over different periods.

Similarly, subfailure stress and max stress showed variations across preservation periods; however, the changes were not significant (p = 0.779 and 0.205, respectively). The preservation duration did not have a notable effect on the ability of the arteries to withstand stress before failure or at maximum capacity.
Elongation, a measure of the arterial ability to stretch, also varied, with a lower elongation observed at 6 months (110.196%) than at 0 months (153.726%); however, this difference was marginally insignificant (p = 0.085). Meanwhile, low and high stretch modulus, which reflect arterial stiffness at different stress levels, did not demonstrate significant variations across different periods (p = 0.532 and 0.089, respectively).
Overall, these results suggest that human arteries preserved for up to 12 months showed no significant changes in mechanical properties, indicating that preservation time does not have a significant effect on the biomechanical integrity of arterial tissue (Figure 8).

Box plots demonstrating the mechanical and morphological properties of human aortic tissues following cryopreservation for 0, 3, 6, 9, and 12 months. Parameters include: (a) subfailure stress (MPa), (b) elongation (%), (c) low stretch modulus (MPa), (d) maximum stress (MPa), (e) wall thickness (mm), and (f) high stretch modulus (MPa).
Repeated measures variance analysis of porcine arteries
Table 6 presents the results of the repeated measures ANOVA for porcine arteries over various preservation periods. The analysis showed that low stretch modulus exhibited a significant difference across different periods (p = 0.035). Specifically, post-hoc analysis revealed that porcine arteries preserved for 0 and 3 months had significantly higher low stretch modulus values compared to those preserved for 9 months, indicating that arterial stiffness decreased as the preservation time increased. This suggests that porcine arteries become more flexible with prolonged preservation, as indicated by the reduction in low stretch modulus over time.
Repeated measures ANOVA for porcine arteries across different preservation periods.

p < 0.05. **p < 0.01. ***p < 0.001.
Although thickness (p = 0.071), subfailure stress (p = 0.138), max stress (p = 0.577), elongation (p = 0.379), and high stretch modulus (p = 0.692) showed variations across preservation periods, none of these differences were significant. For example, thickness increased slightly from 1.492 mm at 3 months to 1.877 mm at 9 months; however, the change was not significant. Similarly, max stress fluctuated between 1.002 MPa at 0 months and 1.158 MPa at 12 months, without reaching statistical significance.
Overall, the findings showed that preservation time had a significant effect on the flexibility of porcine arteries, as reflected in the low stretch modulus. However, other mechanical properties, such as thickness, max stress, and elongation, did not show significant changes over time, suggesting that these properties remain relatively stable during the preservation period (Figure 9).

Box plots illustrating the mechanical and morphological properties of porcine aortic tissues preserved for 0, 3, 6, 9, and 12 months. Measured parameters: (a) subfailure stress (MPa), (b) elongation (%), (c) low stretch modulus (MPa), (d) maximum stress (MPa), (e) wall thickness (mm), and (f) high stretch modulus (MPa).
Pearson correlation analysis for human arteries
Table 7 presents the Pearson correlation analysis for human arteries, focussing on the relationships between max stress, elongation, high stretch modulus, and other variables, such as thickness, subfailure stress, low stretch modulus, and age. The results indicated significant correlations across several variables.
Pearson correlation analysis for human arteries.

p < 0.01. ***p < 0.001.
Max stress showed a strong positive correlation with subfailure stress (r = 0.575, p < 0.001) and low stretch modulus (r = 0.392, p < 0.01), suggesting that arteries with higher subfailure stress and low stretch modulus can endure higher max stress. Conversely, max stress exhibited a negative correlation with thickness (r = -0.484, p < 0.001) and age (r = -0.439, p < 0.01). This implies that older arteries and those with greater thickness tend to have lower max stress capacity, potentially due to changes in arterial structure over time.
Elongation was negatively correlated with low stretch modulus (r = -0.487, p < 0.001) and age (r = -0.627, p < 0.001), indicating that as arteries become stiffer and individuals age, their ability to stretch decreases. This finding highlights the loss of elasticity in aging arteries.
Finally, high stretch modulus was positively correlated with low stretch modulus (r = 0.458, p < 0.01), indicating that arteries with higher stiffness at lower stress levels tend to exhibit higher stiffness at higher stress levels. Overall, these results emphasise the relationships between arterial stiffness, age, and mechanical properties, providing insights into how arterial performance changes with aging and varying structural conditions.
Pearson correlation analysis for porcine arteries
Pearson correlation analysis for porcine arteries, as shown in Table 8, highlighted several significant relationships between important variables. Max stress exhibited a significant negative correlation with thickness (r = -0.321, p < 0.05) and age (r = -0.345, p < 0.05), indicating that as the artery thickness and age increase, the maximum stress that the arteries can endure decreases. This suggests that older and thicker arteries may be more prone to reduced mechanical strength. Furthermore, max stress showed a positive correlation with subfailure stress (r = 0.397, p < 0.01) and low stretch modulus (r = 0.373, p < 0.01), implying that higher subfailure stress and low stretch modulus are associated with an increased ability of the artery to withstand maximum stress.
Pearson correlation analysis for porcine arteries.

p < 0.05. **p < 0.01. ***p < 0.001
For elongation, a significant positive correlation with thickness (r = 0.358, p < 0.05) suggests that thicker arteries have a greater ability to stretch. However, a negative correlation with low stretch modulus (r = -0.309, p < 0.05) implies that arteries with higher low stretch modulus tend to elongate less.
Finally, high stretch modulus was significantly negatively correlated with thickness (r = -0.407, p < 0.01) and age (r = -0.465, p < 0.001), indicating that older and thicker arteries have reduced stiffness under higher stress. These findings highlight the complex mechanical interactions between arterial properties and their ability to withstand stress and deformation.
Summary of the comparative analysis of human and porcine arteries
Table 9 presents the analysis investigating the differences between human and porcine arteries across various preservation periods (0, 3, 6, 9, and 12 months) using independent sample t-tests. The key variables examined include thickness, subfailure stress, max stress, elongation, low stretch modulus, and high stretch modulus (Figure 10).
1. Elongation and High Stretch Modulus at 6, 9, and 12 Months:
○ At 6 months, the analysis revealed significant differences in elongation (t(13.508) = -4.761, p < .001) and high stretch modulus (t(18) = 2.867, p < .05) between human and porcine arteries. Specifically, porcine arteries exhibited significantly higher elongation than did human arteries, indicating greater flexibility. Conversely, human arteries showed significantly higher high stretch modulus, suggesting they were stiffer under higher stress conditions.
○ At 9 months, significant differences were observed in thickness (t(16) = -3.124, p < 0.01) and high stretch modulus (t(9.926) = 2.238, p < 0.05). Porcine arteries were significantly thicker than human arteries, whereas human arteries had a higher high stretch modulus.
○ At 12 months, the trend continued, with significant differences in elongation (t(18) = -3.444, p < 0.01) and high stretch modulus (t(18) = 2.453, p < 0.05). Porcine arteries demonstrated greater elongation, whereas human arteries remained stiffer.
2. Thickness:
○ Thickness of human and porcine arteries did not show significant differences at 0 and 3 months. However, at 9 months, porcine arteries were significantly thicker than human arteries (p < 0.01). This suggests that preservation time may affect arterial thickness differently in humans and pigs.
3. Subfailure Stress and Max Stress:
○ Across all preservation periods, subfailure stress and max stress did not exhibit significant differences between human and porcine arteries. Both species maintained similar capacities to withstand stress before failure and at maximum levels, indicating that these properties remained relatively stable regardless of species and preservation time.
4. Low Stretch Modulus:
○ Although low stretch modulus generally showed no significant differences between human and porcine arteries, a notable finding was observed at 6 months, where porcine arteries exhibited a marginally lower low stretch modulus than did human arteries (p = 0.069). This suggests that at this time point, porcine arteries may be slightly more flexible under lower stress conditions.
Differences between human and porcine arteries across various preservation periods using independent sample t-tests.

p < 0.01. ***p < 0.001.

Comparative box plots of biomechanical and morphological parameters between human and porcine aortic tissues at each preservation time point (0, 3, 6, 9, and 12 months). Panels show: (a) subfailure stress, (b) elongation, (c) low stretch modulus, (d) maximum stress, (e) wall thickness, and (f) high stretch modulus.
Discussion
This study provides valuable insights into the histological changes in human and porcine aortic tissues under different preservation methods, particularly focussing on long-term cryopreservation with 10% DMSO. The results showed that cryopreservation effectively preserved tissue integrity, particularly smooth muscle cell nuclei and elastic fibres, compared to preservation at −80°C without DMSO. These findings agree with those of previous studies, indicating that cryopreservation reduces ice crystal formation and mechanical damage to tissues.15,16
Effects on smooth muscle cells
An important finding of this study is the progressive loss of smooth muscle cell integrity over time in porcine samples preserved at −80°C with 10% DMSO. 17 Samples stored at −80°C without DMSO showed extensive smooth muscle cell loss, whereas those preserved with DMSO experienced significantly less degradation, especially after 9 months of storage.18,19 This suggests that DMSO reduces intracellular ice formation and osmotic imbalance, which are important causes of cell death during freezing. 20 Smooth muscle cells are crucial for maintaining the contractile and structural integrity of the aortic wall, and preserving them is critical for preventing conditions such as aneurysms or dissections. 21 This study supports the recommendation to use aortic tissues preserved at −80°C with DMSO within 12 months for optimal surgical outcomes.
Elastic fibre integrity and thickening
A novel finding of this study is the progressive thickening of elastic fibres during preservation at −80°C with DMSO. 22 Elastic fibres are essential for maintaining the elasticity and tensile strength of the aortic wall. 18 Although the clinical implications of elastic fibre thickening are unclear, this change could alter the mechanical properties of the tissue, making the aorta stiffer, and less compliant under physiological stress. 23 In addition, significant disorganisation of elastic fibres was observed after 9 months of preservation.19,20 The thickening and disorganisation of elastic fibres suggest the need for further investigation of the biomechanical effects of these changes and their impact on long-term graft viability. 19
Endothelial integrity
The study showed that endothelial integrity, as indicated by Isolectin B4 staining, declined more significantly in tissues stored at −80°C without DMSO. 24 Although DMSO helped preserve endothelial cells better in preservation at −80°C, a decline in endothelial signal was observed after 12 months. 18 Endothelial cells play a critical role in vascular homeostasis, and their loss could increase the risk of thrombosis and graft failure. These findings suggest that DMSO provides better preservation of endothelial cells than storage at −80°C without DMSO; improvements in preservation techniques are needed to maintain endothelial integrity over longer periods.
MEMA and elastic fibre fragmentation
The study evaluated MEMA and elastic fibre fragmentation, both indicators of tissue degradation. The findings showed no significant changes in MEMA across preservation groups, suggesting that the preservation methods did not exacerbate this pathological alteration. Similarly, significant elastic fibre fragmentation was not observed, indicating that the overall structure of elastic fibres remained intact throughout the preservation period. These findings suggest that long-term cryopreservation does not induce harmful changes in the extracellular matrix or cause fragmentation of elastic fibres, both of which are critical for maintaining the mechanical strength of the aortic wall.
Lamellar media collapse and thinning
No significant lamellar media collapse was observed across the different preservation groups, including those stored at −80°C with DMSO. However, instances of elastic fibre thinning were observed, particularly in samples stored for longer periods. 22 Thinning of elastic fibres can compromise the tensile strength of the tissue, making it less resistant to mechanical stress. Future studies should investigate the molecular mechanisms underlying this thinning and explore strategies for mitigation.
Impact of cryopreservation on mechanical properties
A primary objective of this study was to assess how cryopreservation affects the mechanical properties of aortic homografts over time. 25 Our results showed that preservation at −80°C with the addition of 10% DMSO led to a significant retention of mechanical properties, including tensile strength, elasticity, and overall structural integrity, in contrast to tissues stored at −80°C without cryoprotectants. The application of cryoprotectants such as DMSO reduced ice crystal formation during freezing, which helped maintain the cellular and extracellular matrix structure of the graft. This is consistent with previous findings, which suggest that tissues preserved with DMSO showed fewer structural changes due to osmotic stress and intracellular ice formation, two of the major contributors to mechanical degradation during cryopreservation.
An interesting observation in this study was the progressive loss of tensile strength in the aortic grafts over extended storage periods. This was especially evident after 9 months, where a notable decline in mechanical performance was observed in tissues stored preservation at −80°C with DMSO, whereas those stored at −80°C exhibited even more rapid degradation. Clinically, this indicates that although cryopreservation with cryoprotectants preserves mechanical integrity better than freezing at −80°C, there remains a time-dependent decline in tissue quality. Therefore, grafts that are cryopreserved for more than 9 months need to be evaluated carefully before use in clinical procedures to ensure they maintain the necessary mechanical properties to withstand physiological stress during implantation.
Conclusion
This study aimed to investigate the effects of different preservation periods, storage conditions (room temperature, 4°C and −80°C), and the use of DMSO as a cryoprotectant on cadaveric aortic tissues. This is the first study globally to undertake a large-scale collection of human aortas, conducting long-term, comprehensive analysis and incorporating comparative results from animal experiments to provide broader insights. Cross-sectional and longitudinal methods were used to assess how these variables affect tissue integrity and function. Microscopic examination revealed widespread pyknosis, indicating irreversible cellular death across nearly all samples, regardless of DMSO usage or storage conditions. Moreover, the study faced challenges due to congenital conditions such as atherosclerosis, plaque formation, and calcification, which occasionally resulted in inconsistent data.
Despite these challenges, DMSO use demonstrated positive outcomes, particularly in significantly reducing smooth muscle cell nuclei loss and enhancing microvascular endothelial signalling. However, over time, especially after 12 months of preservation, there was a notable thickening of elastic fibres, suggesting potential structural changes that could affect biomechanical properties. This trend was corroborated by supplementary analyses. Electron microscopy and tensile strength tests showed that tensile strength decreased by 40% after 12 h of exposure to DMSO and by 23% after 24 h of exposure, indicating that although DMSO reduces ice crystal formation during freezing, it may also alter the mechanical properties of tissues over time.
Clinically, the findings suggest that cryopreservation with 10% DMSO is an effective method for maintaining the structural integrity of aortic tissues for up to 12 months. This presents a promising option for preserving tissues used in thoracic and vascular surgeries, especially given the limited availability of donors. However, the thickening of elastic fibres and decline in endothelial integrity with prolonged storage raise concerns. The study recommends that tissues preserved under these conditions be used within 1 year to mitigate the risk of undetected degradation. Long-term storage without regular evaluation could lead to compromised graft quality, increasing the risk of surgical failure.
Future research should focus on understanding the molecular mechanisms underlying elastic fibre thickening and explore alternative cryoprotectants that might better preserve endothelial cell function and tissue elasticity. Given that elastic fibres, composed of microfibrils and elastin, form a critical network that allows tissues like the aorta to stretch and contract under physiological stress, it is essential to further study their interactions with DMSO to optimise cryopreservation techniques.
The gradual degradation of elastic fibres poses a potential risk during surgical interventions, especially in high-pressure areas such as the aorta. If unnoticed, this deterioration could result in mechanical failure or graft breakdown. Therefore, aortic patches harvested from cadavers should be cryopreserved at −80°C with DMSO and used within 1 year to prevent such risks.
Although cryopreservation with DMSO effectively maintained tissue integrity for up to 12 months, the observed structural changes in elastic fibres and endothelial cells indicate that further improvements in preservation techniques are necessary. By refining cryopreservation methods and exploring new cryoprotectants, the longevity and performance of preserved tissues in clinical applications can be enhanced, leading to better outcomes in thoracic and airway reconstruction surgeries.
Methods
Aorta harvesting and preparation of cryopreserved aortic graft
When the thoracic surgery and transplantation team at our hospital accepts an organ donation, the team conducts organ harvesting and procure the thoracic aorta segment from the patient. This thoracic segment of the aorta spans from the aortic arch to the descending portion of the aorta that passes through the diaphragm. The harvested aorta is immersed in 0°C physiological saline, transported using three layers of sterile packaging, and immediately cryopreserved at the tissue bank of the hospital. The aorta must be prepared within 72 h of harvesting.
The preparation of the aortic grafts is based on established vascular tissue storage methods in previous studies, which have been validated and used to establish procedures for the harvesting, tissue processing, and cryopreservation of aortas from porcine and human donors (Supplemental Material). The collected aortic tissues are stored in a refrigerator at −80°C and further mechanical tests conducted after 3, 6, 9, and 12 months of storage.
Length of aorta and schedule for preliminary testing
Harvested aortas had a typical length of 13–15 cm, with a 6-cm straight segment without collateral branches or curves retained. Standard procedures for aortic cryopreservation were conducted. When a matching patient required tracheal reconstruction, the graft was used for transplantation. The remaining curved section of the harvested aorta was trimmed of connective tissues and divided into three 3-cm sections. All segments of the aorta were immersed in preservation solution and frozen using standard procedures. These segments were stored for 3, 6, 9, or 12 months before thawing. Once thawed, the specimens could be used by:
The pathology department to detect cell and tissue morphology damage due to long-term cryopreservation.
The laboratory department for bacterial culture to observe if long-term cryopreservation causes specimen contamination.
The medical engineering team to conduct stress and tension tests to determine if the original physical characteristics of the aorta are retained after long-term cryopreservation.
Aorta tensile strength test
Aorta specimens were cut in the longitudinal or circumferential direction into 2 cm × 1 cm segments and then into type I segments, resulting in the concentration of forces in the middle of the specimens during stretching. Prior to the tensile test, the width and thickness of the middle depression were recorded; thereafter, a hydraulic clamp was used to clamp the top and bottom wider parts of the segment with a clamp force of 10 psi. The original length of the stretched region was recorded after reaching a specimen force of zero. Finally, the specimen was stretched at a speed of 10 mm/min until breakage.
After the test, the force applied and the amount of stretching were divided by the cross-sectional area and original length to create a stress-strain curve. The following specimen characteristics were determined: (a) low stretch modulus, (b) sub-failure stress, (c) high stretch modulus, (d) yield stress, and (e) tensile stress.
The mechanical characteristics of the original and cryopreserved aortas were compared to determine the retention of mechanical characteristics and function after cryopreservation of the aorta specimens.
Histological assessment
To further evaluate the effect of cryopreservation on tissue structure, thawed aorta samples underwent histological analysis. The following staining methods were used:
a. Haematoxylin and Eosin (H&E) Staining:
Used to observe the general tissue structure, including cell morphology and overall tissue architecture.
b. Movat’s Pentachrome Staining:
Used to differentiate and visualise various tissue components, such as connective tissues, muscle, and collagen.
c. IHC Staining:
Used to detect specific antigens within the tissue, providing insight into protein expression and localisation.
Samples were fixed, dehydrated, embedded in paraffin, sectioned, and stained using the above methods to observe tissue structure, cell viability, and collagen fibre arrangement.
Histological assessment of cadaver aorta in different preservation methods and different storage periods
We investigated human cadaver aortic tissues from various age groups and storage periods, with or without preservation at −80°C with DMSO. A total of 41 samples from both sexes were collected and examined. The analysis included tensile testing and histological assessment, utilising H&E, Isolectin B4, and Movat’s pentachrome staining to elucidate the effects of long-term storage. The basic study designation is listed in Table 10.
Forty-one samples of human cadaver aortic tissue from various age groups and storage periods were collected.

RT: Room temperature.
In addition, we investigated porcine cadaver aortic tissues across different age groups and storage periods, with or without −80°C preservation. Twenty-seven samples were harvested and examined, including tensile testing and histological assessment to elucidate the effect of long-term storage using H&E, Isolectin B4, and Movat’s pentachrome staining. The basic study designation is listed in Table 11.
Twenty-seven samples of porcine cadaver aortic tissues from different age groups and storage periods, with or without preservation.

RT: Room temperature.
Tissue preservation and histological preparation
The submitted tissues were immersed in 10% neutral buffered formalin at room temperature for three consecutive days, followed by tissue trimming, gradient EtOH dehydration, xylene replacement, paraffin infiltration, and wax embedding. Thereafter, the tissue blocks were sectioned into 4–6 μm slices using a microtome. Afterward, the wax-embedded sections were subjected to H&E, Movat’s pentachrome, and Isolectin B4 staining to assess the following:
Smooth muscle cell nuclei loss
MEMA with elastic fibre thickness measurement in tunica media
Microvascular endothelial cell integrity in tunica adventitia (externa)
The experiments were performed in a single-blind manner by a board-certified toxic pathologist (JSTP registered membership number: 6272872007).
For H&E staining, the sections were rehydrated and immersed in Mayer’s haematoxylin for 3 min, followed by tap water and PBS irrigation prior to Eosin Y incubation.
For Movat’s pentachrome staining, the rehydrated sections were immersed in freshly prepared elastic fibre stain solution for 20 min and, thereafter, discoloured with 2% ferric chloride. The sections were dipped in 5% sodium thiosulphate solution and 3% acetic acid solution for 1 and 2 min, respectively. This was followed by staining with Alcian blue solution and Biebrich Scarlet/Acid Fusion solution, and counter-staining with Metanil yellow solution.
For Isolectin B4 staining, the rehydrated sections were digested with protease K for 5 min at room temperature to retrieve the antigen. Thereafter, the sections were quenched in BLOXALL® to block endogenous peroxidase. Isolectin B4 with biotin conjugation was applied for 30 min at room temperature, and the avidin-biotin complex method was used to reveal the positive signal via the DAB chromogen. The stained sections were cleared with xylene and sealed with cover slides using synthetic resin.
Statistical analysis
Data were presented as means ± standard deviation (mean ± SD). One-way ANOVA and t-tests were used to compare the mechanical properties and histological results between different groups. Statistical significance was set at p < 0.05. All statistical analyses were conducted using SPSS or R software.
Study approval
This study was conducted in accordance with the principles of the Declaration of Helsinki and approved by the Ethics Committee of National Taiwan University Hospital (Approval No. 202205070RINB). Informed consent was obtained from all participants or their legal representatives prior to study initiation. All animal experiments were approved by the Institutional Animal Care and Use Committee of the National Taiwan University College of Medicine and College of Public Health (IACUC No. 20220063).
Supplemental Material
sj-docx-1-tej-10.1177_20417314251397592 – Supplemental material for Comprehensive evaluation of cryopreserved aortic homografts: Long-term mechanical and histological preservation across human and porcine models
Supplemental material, sj-docx-1-tej-10.1177_20417314251397592 for Comprehensive evaluation of cryopreserved aortic homografts: Long-term mechanical and histological preservation across human and porcine models by Hsien-Chi Liao, Kuan-Chuan Tsou, Wan-Ting Hung, Chao-Wen Lu, Ying-Syuan Wu, Wei-Ching Su, Yu-Ten Ju, Hsao-Hsun Hsu, Jin-Shing Chen and Tai-Horng Young in Journal of Tissue Engineering
Footnotes
Acknowledgements
We would like to acknowledge the service provided by the Center for Cell Therapy at the National Taiwan University Hospital. We express our sincere gratitude to the National Taiwan University Animal Resource Center for their invaluable support and services. We thank Ms. Kuei-Ying Kao, Ms. Lu Yun, and Ms. Shu-Wei Wu for their help during the experiment. Special thanks go to Professor Yu-Ten Ju and his laboratory colleagues for their support. We also appreciate the staff of the Department of Medical Research, National Taiwan University Hospital, for their essential technical support during the study.
Glossary
Cryopreservation The process of preserving cells or tissues at very low temperatures (typically −80°C) to maintain structural and functional viability.
DMSO (Dimethyl Sulphoxide) A cryoprotective agent used to reduce ice crystal formation and cellular damage during freezing.
Tunica media The middle layer of the arterial wall, primarily composed of smooth muscle cells and elastic fibres.
Isolectin B4 A plant-derived lectin used in histochemistry to stain microvascular endothelial cells.
MEMA (Mucoid Extracellular Matrix Accumulation) An abnormal accumulation of glycosaminoglycan-rich matrix in the aortic wall, often associated with degeneration.
IRS score (Immunoreactive Score) A semi-quantitative method for evaluating staining intensity and distribution in immunohistochemistry.
Tensile strength The maximum stress a material can withstand while being stretched before breaking.
Stretch modulus A measure of a material’s stiffness or resistance to deformation under tensile stress.
Elastic fibre disorganisation The loss of regular structure and alignment of elastic fibres, often indicating tissue degeneration.
Lamellar media The layered structure of the tunica media in the arterial wall.
Thawing The process of returning cryopreserved tissues to physiological temperature prior to analysis or implantation.
Ethical considerations
This study complied with the Declaration of Helsinki and was approved by the Institutional Review Board of the National Taiwan University Hospital (IRB No. 202205070RINB). Written informed consent was obtained from all participants or their legal representatives. All animal procedures were approved by the Institutional Animal Care and Use Committee at the National Taiwan University College of Medicine and College of Public Health (IACUC No. 20220063).
Consent to participate
Human aortic tissues were procured with written informed donor consent (organ and tissue donation forms).
Author contributions
Hsien-Chi Liao: Investigation, Validation, Formal analysis, Visualisation, Writing – original draft, Writing – review & editing. Kuan-Chuan Tsou: Investigation, Data curation. Wan-Ting Hung: Investigation, Data curation. Chao-Wen Lu: Investigation, Data curation. Ying-Syuan Wu: Investigation, Visualisation, Formal analysis. Wei-Ching Su: Investigation, Data curation. Yu-Ten Ju: Methodology, Resources, Supervision. Hsao-Hsun Hsu: Conceptualisation, Methodology, Supervision. Jin-Shing Chen: Conceptualisation, Methodology, Supervision. Tai-Horng Young: Conceptualisation, Methodology, Supervision. All authors reviewed and approved the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Science and Technology, Taiwan (109-2321-E-002-044) and National Taiwan University Hospital (MD001-4, MD001-5, MD001-15).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available on request from the corresponding author, Tai-Horng Young, upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
