Abstract
Coronary artery disease (CAD) encompasses a spectrum of pathologies driven by atherosclerosis, trauma, inflammation, or other etiologies that compromise coronary morphology and function, ultimately leading to myocardial ischemia and infarction. While organ-on-a-chip (OOC) technology has emerged as a transformative tool for cardiovascular research, existing reviews have consistently marginalized coronary-specific pathophysiology, treating it merely as a subset of generic vascular biology. This review presents the first dedicated, critical analysis of microphysiological system (MPS) engineered explicitly as CAD-on-a-chip platform. We deliberately depart from generalized vascular models by exclusively evaluating systems designed to recapitulate the unique coronary-specific hallmarks: distinct geometric constraints, pro-inflammatory microenvironments, and dynamic hemodynamic shear stress profiles inherent to human coronary arteries. Following a concise introduction to OOC fabrication materials and techniques, we systematically present vessel-on-a-chip (VOC) models derived from diverse cellular sources. We then emphasize the biomedical applications of VOC in CAD field and analyze key CAD-specific pathological processes, including flow-mediated endothelial dysfunction, atherosclerotic plaque formation, plaque rupture-induced atherothrombosis, and coronary artery aneurysm. Finally, we critically discuss current limitations and outline future directions of OOC technology in CAD research. This review by focusing on the specific pathological features of CAD and the requirements for in vitro modeling, aim to establish a targeted knowledge framework to promote the clinical transformation of VOC technology in CAD diagnosis and treatment.
Introduction
CAD, manifesting as stenosis, plaque formation, calcification and thrombosis, requires prompt diagnosis and treatment due to its potential to cause severe outcomes, including myocardial ischemia and infarction.1–3 Uncontrollable factors (such as age, gender, and family history of heart disease) and controllable factors (like smoking, high blood pressure, high cholesterol, diabetes, obesity, lack of exercise, poor diet, chronic stress, sleep issues, and excessive drinking) are major causes and risk factors for CAD. In addition, the risk of CAD can also be raised by metabolic syndrome, obstructive sleep apnea, elevated homocysteine levels, autoimmune diseases, and chronic kidney disease. 4 While the mortality from cardiovascular disease (CVD) has declined significantly over the past few decades as economies have grown and health care systems have improved, the risks associated with CVD and aging remain a concern.5–7 Therefore, it is still necessary to establish different types of CAD models, which will provide a solid foundation for exploring the molecular mechanism of CVD, evaluating new drugs, and targeting therapies.
Conventional cardiovascular research has long depended on animal models and two-dimensional (2D) cell cultures.8,9 In vivo studies involving rodents, pigs and canines have successfully recapitulated key aspects of atherosclerosis, including plaque progression, thrombo-inflammatory responses, and reperfusion injury. These models have been instrumental in identifying critical pathways, such as those mediated by low-density lipoprotein (LDL) receptor deficiency or high-fat diet-induced metabolic dysregulation, as well as underlying disease pathogenesis.10–12 Moreover, animal systems provide an intact immune environment, systemic metabolic context, and clinically meaningful endpoints such as infarct size and survival. In parallel, conventional 2D cell cultures offer simplicity, cost-effectiveness, and ease of standardization, enabling high-throughput screening of numerous compounds under tightly regulated culture conditions (e.g. temperature, pH and nutrient supply).13–15 However, these established models present important limitations. Animal studies are constrained by interspecies differences, variable genetic backgrounds, and the inability to fully account for how chronic comorbidities modulate drug responses. Meanwhile, 2D cultures lack essential physiological features such as hemodynamic shear stress, three-dimensional (3D) extracellular matrix (ECM) environment, and multicellular communication. As a result, they fail to mimic the oscillatory shear profiles found at coronary bifurcations, the biomechanical interactions at the plaque-blood interface, or the spatiotemporal dynamics of thrombosis and plaque rupture in real-time.16,17 By reconstructing the patient-specific and perfusable 3D coronary microenvironment and supporting real-time imaging, OOC platforms hold great potential to bridge the translational gap in cardiovascular research. 18
Microfluidic OOC technology is an emerging bioinspired approach that leverages microfluidics and biomaterial engineering to construct highly simulated tissue or organ models on small-scale chips by recapitulating the microenvironment of human organs.19–21 The microstructures and ECM materials of the OOC system can support cell proliferation, differentiation, and function maintenance, and thus can be used to build bionic models with high relevance to tissues or organs in the body.22–24 Through the design of microstructures and microchannels, different levels of cell arrangement can be achieved, reflecting the key structure and function of real tissues. This capability provides a reliable experimental platform for the construction of disease models and the development of new therapeutic strategies.25,26 More significantly, because OOC technology has strong universality and scalability, customized services can be realized according to different tissue or organ needs, and even multi-organ chips.27–29 This flexibility makes OOC a promising tool with broad application prospects in biomedical fields such as basic research, drug development, and disease treatment.30,31 For CAD research, OOC technology offers a new platform for disease mechanism research and drug screening. This platform not only provides a high-fidelity physiological environment, but also significantly improves the efficiency and safety of drug screening, enabling personalized medicine. 32 Accordingly, OOC is gradually becoming an indispensable means for modern medicine research and clinical applications, which is expected to promote the research and treatment progress in the field of CVD.33,34
This review focuses on the application of OOC in CAD modeling and systematically summarizes the progress, challenges and future development of OOC in CAD research (Figure 1). In contrast to recent reviews that broadly address OOC applications across the cardiovascular system, often treating CAD as one among many vascular conditions, this work is specifically structured specifically around CAD-exclusive pathophysiology.35–37 Our review begins by deconstructing the design principles of current OOC platforms for CAD, offering a critical appraisal of benchmark materials, including polydimethylsiloxane (PDMS), hydrogel, paper, and glass capillary, with a focused discussion on their respective efficacy and limitations in replicating critical coronary-specific variables. We then provide a detailed catalog of cellular components and scrutinize the use of cell lines, primary cells, patient-specific induced pluripotent stem cells (iPSCs)-derived coronary cells, and ancillary stromal cells. After that, this review particularly emphasizes the capability of existing platforms to model four CAD-specific pathophenotypes: (i) flow-dependent endothelial dysfunction, (ii) atherosclerotic plaque formation, (iii) plaque rupture-driven atherothrombosis, and (iv) coronary artery aneurysm. Finally, we articulate a critical perspective on the profound gaps that persist specifically for coronary modeling, laying the foundation for the subsequent discussion on limitations and prospects.

Schematic diagram of the construction and application of VOC in CAD research, including VOCs prepared for CAD pathological model modeling from different cell sources and chip types.
Fabrication of the OOC for modeling CAD
Materials for microfluidic chips
Glass capillary
Owing to the excellent mechanical, optical and chemical stability, glass capillaries are among the most commonly used materials for constructing microfluidic devices. By regulating the assembly form of the glass capillary, various structured droplets, fibers or scaffolds can be conveniently generated. For instance, Ou et al. demonstrated the creation of uniform microfluidic channels tens of centimeters long using commercial glass pipettes through simple heating and stretching methods 38 (Figure 2(a)). Glass capillary microfluidic devices also perform exceptionally well in the diagnosis and detection of diseases. Given the established correlation between hyperglycemia and increased risk of coronary events, it is noteworthy that Govind et al. developed glass capillary blood glucose sensors utilizing radiofrequency complementary electric LC (CELC) resonators. Their work further demonstrated that this adaptable microfluidic architecture could be modified for the detection of myocardial injury proteins. This approach suggests the potential for an integrated coronary chip capable of assessing multiple risk factors, thereby providing a prototype for dual-mode, real-time metabolic-injury signal monitoring. 39 In this study, glass capillaries loaded with test samples were installed in the cut grooves on the central arm of the resonator. Compared with previous blood glucose concentration sensors, the recyclability of this microfluidic sensor reduces the cost of long-term use, and it also demonstrates excellent performance in terms of sensitivity and response time. Additionally, the glass capillary prevents the sensor from coming into close touch with the test sample, avoiding the risk of cross-contamination from any chance encounter (Figure 2(b)). In another study, Li et al. fabricated a capillary-based microfluidic chip that consists of a plastic shell and two functional capillaries (S-like and I-like) for rapid detection of acute myocardial infarction (AMI). 40 The inner surface of the capillary was modified with myocardial infarction signature antibodies, such as myoglobin (Myo), cardiac troponin I (cTnI) and creatine kinase-MB (CK-MB), following treatment with three-aminopropyltriethoxysilane and glutaraldehyde. The sample added to the capillary specifically binds to the capture antibody to form an antigen-antibody complex. This complex was then detected by fluorescently labeled secondary antibodies, and the content of biomarkers in the sample was quantitatively analyzed by the intensity of the fluorescence signal. The entire process was simple to operate, low in cost, time-efficient, and could be mass-produced, providing a reliable method for the rapid detection of biomarkers for CVDs (Figure 2(c)).

Microfluidic devices fabricated with glass capillary, PDMS and paper. (a) Scanning electron microscope (SEM) images of five cross-sections of a capillary microfluidic channel. (b) Image of the microfluidic sensor system. (c) Two capillary microfluidic chips with groove structures: S-like and I-like. (d) (i) Schematic illustration of the construction of TEBV-on-a-chip. (ii) Image of TEBV-on-a-chip. (iii) Image of TEBV under the perfusion experiment. (e) (i) Schematic of the PDMS chip. (ii) Cells cultured in microfluidic device with regular and pressurized mode. (iii) Cells stained with F-actin and VE-Cadherin in the static status and after applying FSS for 24 h (f) (i) Schematic of LFA-based μPAD. (ii) LFA-based μPAD layers and assembled chip. (g) (i) Photographs of the E-μPAD. (ii) Corresponding calibration curves measured by the E-μPADs using ABP samples spiked with cTnI, BNP-32, and D-Dimer, respectively.).
Glass capillary microfluidic platforms offer advantages such as low cost, recyclability, and ease of batch replication for constructing uniform channels. These systems have been utilized for non-invasive blood glucose monitoring and fluorescence quantification of acute myocardial infarction markers, serving as useful tools for rapid prototyping in cardiovascular diagnostics. However, most current methods are often single-function and lack integration. For example, while thermoforming enables channel fabrication, it often relies on manual operation, restricts channel geometry diversity, and complicates integration with active components such as valves or pumps. Similarly, radiofrequency sensing avoids fluid contamination but is limited to detecting shifts in electrical resonance frequency, lacking complementary optical or electrochemical validation. Furthermore, existing immunoassays for cardiac markers in glass capillaries typically require manual antibody immobilization, washing, and signal reading, steps prone to environmental interference and operator-dependent variability. These approaches also frequently overlook confounding factors such as plasma viscosity and hematocrit, which may influence fluorescence quenching. Consequently, without incorporating elastic membranes, automated liquid handling, multi-parameter sensing arrays, and internal calibration mechanisms, the reproducibility and clinical utility of glass capillary platforms will remain limited, impeding their translation from laboratory prototypes to standardized clinical products.
Polydimethylsiloxane
PDMS remains the workhorse of OOC platforms due to its optical clarity, biocompatibility and compatibility with rapid soft lithography. These properties enable the replication of diverse coronary anatomies-from focal stenoses to bifurcating aneurysms-within hours.41,42 Through soft lithography technology, PDMS can be created into various complex microstructures, including narrow, pleated, and multi-chambers, to mimic arterial structures in various clinical situations. 43 In addition, due to its excellent transparency, PDMS greatly enhances the convenience of observation by allowing optical imaging and real-time monitoring. 44 In a word, PDMS has become one of the main materials currently used in microfluidic devices owing to its excellent biocompatibility, transparency, and ease of processing. 45 Just as Wang et al. prepared a tissue-engineered blood vessel (TEBV) chip with a cylindrical channel in the middle using PDMS. 46 First, human aortic smooth muscle cells (HASMCs) were implanted into the chip for 1 week of culture, and then human umbilical vein endothelial cells (HUVECs) were continuously injected into the chip to form an endothelial lining. Cell culture medium was perfused with peristaltic pumps to simulate the dynamic movement and flow of blood vessels. The elasticity and flexibility of PDMS enable the TEBV to perform periodic contraction and expansion movements driven by pulsatile flow, thus better simulating the physiological state of blood vessels in vivo and ensuring the stability and reliability of the experiment (Figure 2(d)).
In the OOC field, silicon wafers are typically utilized to create precise features, such as electrodes, sensors, and microfluidic channels. 47 The production of silicon wafer-based microfluidic devices contains a series of precise craftsmanship, including photolithography, etching, thin film deposition and bonding. 48 In particular, PDMS is most commonly used to replicate the customized structure of silicon wafer-based microfluidic devices to construct OOC devices with good flexibility and high light transmittance. Taking advantage of the high elasticity of PDMS, Chu et al.’s team developed a PDMS chip to study the mechanical response of endothelial cells (ECs). 49 The master mold with microfluidic channels and air chambers was processed on a silicon wafer using traditional photolithography techniques, and then the PDMS microfluidic chip was created by replicating the mold. By periodically pressurizing and releasing the air chamber, the PDMS membrane deformed, thereby applying cyclic stretching (CS) forces on the cells. Simultaneously, continuous fluid flow was maintained in the microfluidic channels via a syringe pump, generating fluid shear stress (FSS). The combination of CS and FSS produces pulsatile flow, which effectively mimics the arterial environment and offers a novel tool for the research and treatment development of CVDs (Figure 2(e)).
Despite having many advantages, PDMS still exhibits well-documented limitations that become critical in long-term CAD experiments. The natural PDMS is intrinsically hydrophobic, which is not conducive to cell adhesion. The PDMS surface can be activated by plasma treatment and then coated with matrix proteins immediately to prevent the reversion of hydrophobicity. However, these processing methods still result in non-specific adsorption of lipophilic drugs and lipoproteins, leading to an underestimation of the drug dosage and an overestimation of lipid accumulation. Additionally, PDMS absorbs small molecules, such as hydrophobic drugs, resulting in a 100-fold decrease in its effective concentration, thereby confounding the dose-response analysis. 50 It is reported that using 0.1% bovine serum albumin (BSA) or additional hydrophilic passivation for a brief pre-rinsing process might be a way to alleviate these problems.51,52 Despite these issues, PDMS remains the most used material for chips.
Paper
The working principle of microfluidic paper-based analytical devices (μPADs) is to create patterned hydrophobic boundaries on hydrophilic paper substrates, thereby forming microflow channels to control the delivery of liquids. For instance, hydrophobic materials (such as alkyl ketone alkenes) can be printed using an inkjet printer, photolithography, or wax printer to create the hydrophobic patterns. μPADs, as a low-cost, convenient, and environmentally friendly rapid test device in the form of test strips, have shown broad application prospects in clinical immediate detection.53–55 Ilacas et al.’s research group proposed a method for detecting glucose based on μPADs, which greatly improved the detection effect with the help of metal-organic frameworks (MOFs). 56 The authors designed two types of devices, a well-based chip and a lateral flow chromatography (LFA) platform. Both devices adopt wax printing technology to ensure the directional flow of solution and improve the stability and tightness of the chip through hot pressing treatment. The results showed that the well-based platform demonstrated a broad linear range in glucose detection, making it ideal for precise quantitative analysis in laboratory settings. In contrast, the LFA platform excels in rapid response, rendering it highly suitable for on-site rapid detection and home self-inspection (Figure 2(f)). For CVD diagnosis, Fu et al.’s team introduced an electrochemical μPAD (E-μPAD) for rapid and point-of-care (POC) testing of CVD protein markers in whole blood. 57 The chip included three electrochemical units, a plasma separation membrane and a buffer absorbent pad. Blood samples passed through the plasma separation membrane to reach the working electrode (WE), and then the target protein was detected by measuring the impedance change at the interface between WE and the solution under the action of the electron mediator solution. The experimental results showed that the detection limits (LOD) of E-μPAD were 4.6 pg·mL−1 (190 fM, cTnI), 1.2 pg·mL−1 (40 fM, bnp-32), and 146 pg·mL−1 (730 fM D-Dimer), respectively, all of which were lower than the clinical detection limits and comparable to commercial enzyme-linked immunosorbent assay (ELISA) kits. The entire process requires only 46 min, which is significantly shorter than the internationally recommended guideline of completing CVD testing within 60 min. The findings revealed that the E-μPAD chip offered significant benefits for the detection of CVD, including high sensitivity, rapid detection, user-friendly operation, low cost, and high portability. Collectively, these attributes position the E-μPAD chip as a highly promising candidate for on-site rapid diagnostics, thereby providing robust support for the early diagnosis and personalized treatment of CVD (Figure 2(g)).
While the above μPAD studies integrating wax printing, screen-printing, electrochemical impedance, and MOF catalysis successfully achieved simultaneous quantification of cTnI, BNP-32, and D-dimer with sub-picomolar sensitivity in 46 min at a cost under $0.3, presenting a streamlined “sample in-result out” approach for bedside screening of coronary events, these microfluidic platforms face a persistent disconnect between their performance capabilities and practical clinical implementation. For instance, although one blood glucose-MOF system exhibits a broad linear range, its colorimetric readout may be influenced by hematocrit and blood lipid interference, which has not been systematically evaluated. Similarly, while an E-μPAD platform achieves a detection limit superior to that of ELISA its plasma separation membrane exhibits a significant reduction in flow rate at hematocrit levels exceeding 55%, leading to extended detection times and increased coefficient of variation. Moreover, both systems necessitate open manual sample addition without capping microvalves, increasing the risk of leakage and signal diffusion, particularly in environments characterized by high humidity and dust. Critically, neither platform has been validated against central laboratory chemiluminescence assays to establish large-sample consistency, nor do they incorporate stratified diagnostic thresholds accounting for gender, age, or comorbidities. Without the integration of features such as automatic capping, built-in calibration strips, and real-time humidity and hematocrit correction modules, the clinical credibility of μPAD systems will remain limited, hindering their transition from proof-of-concept prototypes to guideline-level, real-time coronary diagnostic tools.
Hydrogel
Hydrogel is 3D network formed by the chemical or physical cross-linking of hydrophilic polymer chains, which possesses excellent biological properties such as soft texture, variable performance, good biocompatibility, and adjustable mechanical properties. The natural or synthetic polymers that make up hydrogels, the most common of which are gelatin, collagen, polyvinyl alcohol (PVA), alginate and cellulose, are highly suitable for the cells and tissues of organisms and can effectively promote the development and function of cells. By selecting appropriate hydrogel materials, researchers can create a microenvironment suitable for specific cell types, promoting cell proliferation, differentiation, and functional expression. 58 Nie et al. combined gelatin, gelatin methacrylate (GelMA) and alginate pairwise to obtain three different types of composite hydrogels. 59 Among them, the gelatin-GelMA composite hydrogel has a more excellent microstructure and transparency, which is more conducive to cell adhesion and diffusion, and maintains the normal morphology. In addition, the excellent biocompatibility and adjustable mechanical properties of hydrogels have enabled them to stand out in the field of in vitro cardiovascular modeling (Figure 3(a)). For instance, Gu et al.’s team utilized a 5% (w/v) GelMA solution mixed with HUVECs and a 5% (w/v) gelatin solution to create a perfusable VOC. 60 The two hydrogels were separately loaded into syringes and extruded through coaxial nozzles, with GelMA forming the outer layer and gelatin forming the inner layer. Upon UV curing, core-shell structured fibers were successfully generated. After 5 days of culture, a polycaprolactone (PCL) stent was introduced to provide internal support to the lumen, ensuring that the endothelialized blood vessel maintained its structural integrity and did not collapse. The CCK-8 assay revealed a marked increase in cellular activity over a 7-day period. Additionally, immunostaining for zonula occludens-1 (ZO-1, intercellular junction protein) and vinculin (cell-matrix adhesion protein) demonstrated the formation of a stable endothelial layer within the hydrogel fibers (Figure 3(b)). In conclusion, due to the outstanding biocompatibility, appropriate hardness and excellent permeability of hydrogels, they have been widely applied in the fields of tissue engineering and in vitro organ model research, especially in organoids and OOC systems.61,62

Microfluidic chips fabricated with hydrogel and 3D printing. (a) Comparison of the three hydrogel combinations. (i) Physical images for the demolded hydrogel sheets, bonded hydrogel-based chips, and ink perfusion of the hydrogel-based chips of the three composite hydrogels. (ii) Statistical analysis of the diameter changes between each step during the manufacturing process of the three composite hydrogels. (iii) Microstructures of the surface morphology of the three composite hydrogels under microscope. (iv) Images of cell attachment and spreading on the three composite hydrogels. (b) Fabrication of a perfusable VOC. (i) Fabrication process. (ii) Top view of the perfusion chip. (iii) Explosive view of the perfusion chip. (c) Schematic diagram of the procedure of fabricating endothelialized myocardium using the 3D bioprinting strategy. (d) Construction of a vascularized OOC. (i, ii) Transparent microfluidic chip prototypes with 3 mm tissue chamber. (iii, iv) Immunofluorescence staining image of CD-31 (red), nuclei (blue), and actin filaments (green). HDF lining the outer vessel walls as indicated by white arrows.
The aforementioned studies collectively highlight the advantages of hydrogels in coronary and microvascular chips, emphasizing their “bio-friendly and mechanically adjustable” properties. For instance, a GelMA/gelatin/alginate composite matrix can be rapidly fabricated into a perfusable and stent-reinforceable core-shell fiber via coaxial extrusion-photocuring within minutes. This construct facilitates the formation of a continuous endothelial barrier, evidenced by ZO-1 and vinculin expression, within 7 days, thus offering a rapid prototyping platform for degradable vascular scaffolds and vascular OOCs. Despite these advances, existing models present inherent simplifications in both mechanical and biological aspects. Mechanically, the stiffness of commonly used hydrogels is substantially lower than that of the medial layer of native human coronary arteries. Moreover, GelMA undergoes gradual hydrolysis and softening during long-term culture, often resulting in lumen collapse and necessitating reinforcement with synthetic polymers such as polycaprolactone (PCL). While PCL scaffolds provide mechanical support, they disrupt physiological stress–cell feedback mechanisms. Biologically, the typical core-shell configuration supports only a monolayer of ECs (e.g. HUVECs), lacking essential vascular components such as smooth muscle cells (SMCs), macrophages, and lipid-rich cores. As a result, these models fail to recapitulate the multilayer mechanical instability seen in coronary aneurysms-for instance, the complex structure involving a collagen cap, calcification, and inflammatory infiltration. Further challenges include the mismatch among crosslinking density, degradation kinetics, and cellular remodeling. After 30 days in culture, significant matrix shrinkage (>15%) is commonly observed. The absence of real-time strain calibration and flow-rate compensation also leads to inaccuracies in computational fluid dynamics simulations due to ill-defined boundary conditions. Future progress in hydrogel VOCs, enabling them to transition from “rapid prototyping” to accurately predicting coronary aneurysm rupture or providing effective stent support, achieving a biologically and mechanically dual-fidelity platform contingent upon the integration of patient-specific elastic modulus matching, co-culture of iPSCs-derived diseased ECs, SMCs, and macrophages, and real-time creep correction.
Processing technologies of microfluidic chips
Soft lithography
Soft lithography, one of the most commonly used technique in micro-nano manufacturing and bioengineering, is closely related to the manufacturing of PDMS microfluidic chips. The core concept of soft lithography is to prepare microstructures using photosensitive materials and flexible materials (such as PDMS). 48 Firstly, a layer of photosensitive resin, also known as photoresist, is covered on the surface of the silicon wafer, and a chemical reaction occurs in a specific area through photolithography exposure. Secondly, the photoresist in the unexposed areas is removed by a developer to form the desired microstructure mold. Thirdly, thoroughly stir the liquid PDMS with the curing agent in the typical ratio of 10:1 or 20:1 and inject it into the mold. After removing the bubbles through a vacuum machine, place it in an oven to cure the PDMS. Finally, the PDMS is peeled off from the mold to obtain a microfluidic chip with specific microstructure. However, PDMS itself has a certain hydrophobicity and is not suitable for cell culture. Through technologies such as oxygen plasma treatment, hydrophilic coating, and chemical modification, the hydrophilicity of PDMS chips can be significantly enhanced, making them more suitable for fluid processing and cell culture. Meanwhile, by adding nanoparticles, improving curing conditions and adjusting the design, PDMS chips can be made harder, thereby having better stability and durability under long-term use and high pressure. In short, soft lithography technology with relatively simple operation, affordable price and excellent resolution has become one of the most widely used technologies in the production of microfluidic chips.
Soft lithography using PDMS chips has emerged as a widely adopted technique for fabricating coronary vascular models, owing to its well-established processes such as base-to-curing agent casting and oxygen plasma bonding. These methods enable cost-effective production with sub-micron accuracy. Recent enhancements including nanoparticle doping, hydrophilic coatings, and UV post-curing have significantly improved chip properties, increasing elastic modulus by three times and reducing the contact angle to 20°, thereby enhancing sealing performance and cellular compatibility under prolonged high-pressure perfusion conditions. Despite these advantages, this established technique faces three major limitations. First, the inherent viscoelasticity of PDMS differs by an order of magnitude from that of the coronary media, leading to a creep of up to 15% over 30 days. This creep causes continuous drift in wall thickness, channel height, and local shear force, with limited options for online calibration and feedback correction. Second, the adsorption of small hydrophobic molecules can result in a false rightward shift in dose-response curves. Moreover, hydrophilic treatment layers degrade and detach within 7 days at 37°C in serum, compromising reproducibility. Third, curing shrinkage and demolding stresses induce a barreling deformation in aspect ratio structures exceeding 500 µm in height. Such geometric distortions impede accurate replication of patient-specific coronary anatomy, particularly in regions with aneurysms or tortuous bifurcations. Without integrating strategies for real-time correction of elastic, geometric, and surface properties, such as employing thermoplastic elastomers, PDMS-glass hybrid systems, or real-time strain compensation, PDMS-based chips are likely to remain confined to rapid prototyping applications. They are presently inadequate for reliably predicting coronary aneurysm rupture or providing sufficient stent support evaluation, thereby restricting progress toward a dual-fidelity biomechanical platform for coronary research.
3D bioprinting
With the development of 3D printing technology, it is becoming increasingly feasible and convenient to manufacture microfluidic chips using 3D printing. The adoption of 3D printing technology can significantly simplify the processing of microfluidic chips and is also very flexible in the selection of printing materials. 3D bioprinting is an extension of 3D printing, which is based on additive manufacturing technology. It is a method of printing biomedical structures using living cells, biomolecules and biomaterials (bio-inks, such as hydrogels), by depositing biomaterials layer by layer to create 3D structures such as tissues and organs. Common 3D bioprinting technologies mainly include inkjet printing, extrusion printing and light-assisted printing, etc. Each printing method can print cell scaffolds and directly encapsulate cells within the scaffolds to construct tissue structures.63–65 In this regard, Zhang et al. fabricated 3D microfiber scaffolds using bioprinting technology for the construction of a heart-on-a-chip platform for drug screening and disease modeling. 66 In this study, alginate and GelMA were used as bioinks to prepare layer-by-layer stacked fiber scaffolds by extrusion printing. Then, cardiomyocytes and HUVECs were simultaneously injected into the microfiber scaffold to form myocardial tissue. The proposed myocardial model can monitor the electrophysiological activities and mechanical properties of myocardial cells in real time and shows good drug reactivity (Figure 3(c)). In addition, a vascularized OOC was constructed by printing angiogenic bioinks containing HUVECs and human dermal fibroblasts (HDF) through multiple printheads. 67 During the 14-day culture period, the formation and function of the vascular network were verified by immunofluorescence staining and confocal microscopy imaging, providing the possibility for large-scale production and application of vascularized OOC (Figure 3(d)). Overall, 3D bioprinting technology can continuously generate complex structures including ECM and cells, featuring a fast turnaround time and great flexibility in chip design. In view of this, the 3D printing OOC platform enables multiorgan-on-a-chip to create 3D tissues that are closer to the human body, becoming an effective means for understanding organ functions, disease modeling and drug screening.
3D bioprinting facilitates the direct fabrication of vascular networks and microfibrous structures by combining living cells with hydrogel-based bioinks. This approach enables concurrent evaluation of electrophysiological behavior and contractile function in engineered cardiac tissues within as few as 14 days, providing a rapid and versatile platform for preliminary drug screening in the context of coronary artery disease. Despite these capabilities, current bioprinting systems exhibit notable limitations related to biomechanics and temporal fidelity. For instance, extrusion-based printing of 250 μm fibers, while efficient, subject cells to high shear stress and UV cross-linking, leading to cell mortality rates exceeding 30%. Moreover, hydrogels such as GelMA and alginate possess mechanical properties significantly softer than those of native coronary medial layers, resulting in progressive creep and structural collapse during prolonged culture. This often necessitates supporting scaffolds that disrupt physiologically relevant stress-cell feedback loops. While multi-nozzle printing can create vascular networks with lumens, these constructs typically lack critical hallmarks of mature vasculature, including functional smooth muscle layers, lipid-rich cores, and infiltrating macrophages. Consequently, they fail to recapitulate the mechanical instabilities associated with coronary aneurysms, such as collagen cap formation, calcification, and inflammatory remodeling. The 14-day observation endpoint remains substantially shorter than the months to years required for clinical plaque progression and stabilization. In addition, key pathophysiological processes, including extracellular matrix degradation, calcium deposition, and donor-specific responses, are not monitored in real time, limiting the translational relevance of drug response assessments compared to in vivo outcomes. To transition from conceptual prototypes to predictive models for coronary aneurysms or long-term stent performance, future developments must concurrently address low-shear printing techniques, patient-specific elastic modulus matching, co-culture of multi-lineage disease-relevant cells, and real-time monitoring of mechanical properties and calcification.
Cell source
The basic tissue components of blood vessels are endothelium, muscle tissue and connective tissue, which are the channels for blood circulation. Among them, ECs are located in the innermost layer of blood vessels, directly in contact with blood flow, and have important functions such as anticoagulation, anti-inflammation and regulation of vascular tension.68,69 Muscle tissue is located in the middle layer of blood vessels, mainly composed of SMCs, which function is to regulate the diameter of blood vessels through contraction and relaxation, thereby controlling blood pressure and blood flow velocity.70,71 Connective tissue provides support and protection for blood vessels, ensuring their integrity and stability. Consequently, ECs or ECs-SMCs co-culture are the main form of VOC. Here we introduce VOC based on different cell sources, and each type of cell has specific advantages and disadvantages.
Cell lines
Cell lines are groups of cells with unlimited proliferation ability obtained through long-term in vitro passage culture. Because of its benefits such as easy access, low cost, rapid proliferation and clear genetic background, OOC derived from cell lines has been employed extensively in drug screening and disease model construction. 72 Yadav et al.’s team constructed a perfusable 3D Xeno vascular chip (Xeno-vessel-chip) to simulate the co-culture environment of the porcine aortic endothelial cell line (MPN3) with human and monkey neutrophils. 73 MPN3 cells were seeded into microfluidic channels and perfused for 5 days to form a 3D xenogeneic vascular chip. Neutrophils isolated from the venous blood of healthy adults and macaques were then inoculated in the chip for co-culture. It was found that DNA-histone complexes significantly increased, indicating the formation of neutrophil extracellular traps (NETs, which are considered as a mechanism of xenograft rejection) (Figure 4(a)).

VOCs prepared from cell lines, primary cells and stem cells. (a) (i) Image of Xeno-vessel-chip. (ii) The orthogonal views of the MPN3 cell line lumen. Cells were stained with Calcein AM (green) and Hoechst (blue). (b) (i) Schematic view of the VFP-microchannel. (ii) 3D structure of the created microvessel. (iii) Chosen frames from the z-stack image. Primary HUVECs were stained with phalloidin and Hoechst. (c) (i) Schematic showing the design of the chip. (ii, iii) Representative fluorescent images showing multicellular components of the fully vascularized chip. (iv) Cross-section of the vascular channel. (Green: endothelial monolayer; blue: fibroblasts; red: nuclei). (d) (i) Schematic of the VOC protocol. (ii) Representative immunofluorescence images of microvascular network showing the hiPSCs-ECs (magenta; agglutinin) and mural cells (green; SM22). (iii) Quantification of normalized mean cell SM22 intensity, (iv) number of SM22 + cells and (v) normalized mean cell SM22 intensity of mural cells in contact with hiPSCs-ECs in hiPSCs-VSMCs, HBVPs and HBVSMCs.
While cell lines used in the OOC platforms offer benefits such as indefinite expandability and well-defined genetic backgrounds, enabling rapid, reproducible drug screening and studies on xenograft rejection, their tumor-derived origin and long-term culture adaptations, including phenotypic drift, chromosomal abnormalities, and altered signaling pathway activation, significantly constrain their physiological relevance. 74 For instance, ECs lines like MPN3 struggle to accurately replicate the physiological responses of normal coronary endothelium to shear stress, inflammatory mediators, or lipid exposure. Moreover, the lack of donor diversity, absence of stratification based on age and comorbidities, and the inability to capture spatiotemporal interactions with SMCs and macrophages within a homogeneous cell culture model collectively reduce the correlation between OOC-derived data and in vivo evaluations of plaque vulnerability or scaffold reendothelialization.
Primary cells
Primary cells are cells directly isolated from tissues or organs, and their biological characteristics are closer to the in vivo state, which can better simulate the physiological functions of organs. 75 Besides, primary cells have unique advantages in personalized medicine and disease mechanism research, such as using patient-derived primary cells to construct OOC for personalized drug screening and treatment plan formulation. 76 For example, Konopka et al. designed a VOC capable of effectively expanding ECs, in which the microfluidic device was generated by a method called Viscous Finger Patterning (VFP). 77 Then the effects of different contents of vascular endothelial growth factor (VEGF) on the growth status of primary HUVECs were studied. In the proposed VOC, HUVECs grew well and maintained the ability of barrier formation and angiogenesis, and the activity levels of related important genes (such as VE-cadherin and PECAM-1) were similar to those of ECs in vivo. Overall, the team successfully achieved effective expansion of ECs while maintaining their barrier function and angiogenesis capacity (Figure 4(b)). In another important study, Vo et al. proposed an innovative microfluidic chip that could successfully reconstruct a 3D-matrix (collagen and fibrin) embedded multicellular microvascular model with uniform shear force induction. 78 The results proved the morphological characteristics of the reconstructed microvessels, including lumen diameters similar to those of the microvessels in the body, uniform wall thickness, and the formation of a complex vascular network with branches and connection points. Finally, the barrier function, substance transfer ability and cell activity of the vascular model were verified (Figure 4(c)). In brief, primary cells are extremely useful in research with high biological authenticity and specificity, especially for simulating in vivo conditions.
Primary cell-based VOC models offer the advantage of “in vivo-like gene expression combined with realistic barrier function and angiogenic budding,” providing a high-fidelity in vitro platform for studying coronary microvascular regeneration, individualized VEGF dosing, and 3D collagen-fibrin network remodeling. However, their translational potential is constrained by three major bottlenecks related to “lifespan, source, and batch variability”: First, primary HUVECs exhibit aging and phenotypic drift after fewer than six passages, rendering them unsuitable for modeling slow pathological processes such as chronic atherosclerosis or in-stent restenosis, which evolve over months to years. 79 Second, although techniques such as VFP molding enable rapid expansion, donor-specific ECs can show two- to threefold variations in response to identical shear stress or VEGF stimuli. The absence of standardized isolation and quality control protocols further contributes to poor chip-to-chip reproducibility. 80 Lastly, primary cell sourcing remains dependent on umbilical cord or skin biopsies, which limits the diversity of donor backgrounds. This constraint hinders the representation of critical high-risk conditions such as diabetes and hyperlipidemia, thereby reducing physiological and clinical relevance. 81
Stem cells
Although primary cells can better simulate the real state of cells in vivo, it is difficult to obtain scalable primary cells from humans, especially cardiomyocytes, which greatly limits the use of primary cells. Stem cells are a type of primitive cell with the ability to self-renew and differentiate into various types of tissue cells, and they are also progenitor cells that form various tissues and organs in the human body. Recently, with the technological development of iPSCs, the technical solutions for constructing human organoid models using iPSCs have gradually matured. The cardiac organoids constructed thereby can “beat” and vascular organoids can not only achieve the physiological functions of the organs but also have hollow cavity structures similar to those of real organs and express similar markers. In the field of cardiovascular research, iPSCs can be derived into functional ECs and SMCs to construct physiologically related vascular models.82–84 Vila Cuenca et al. described a VOC that is generated from an entirely human-induced pluripotent stem cells (hiPSCs)-derived composition with ECs and SMCs. 81 They compared the discrepancy of hiPSCs-SMCs and human brain vascular smooth muscle cells (HBVSMCs) when co-cultured with hiPSCs-ECs. The results indicated that hiPSCs-ECs were able to generate stable microvessel networks with both hiPSC-SMCs and HBVSMCs. However, hiPSCs-SMCs showed lower SM22 (expression of the contractile marker) staining intensities and higher total cell numbers, indicating hiPSC-SMCs are less differentiated in the VOC (Figure 4(d)). Similarly, another study also proposed a hiPSCs-derived VOC, which includes a hydrogel chamber and two reservoirs. 85 Straight microfluidic channels were formed by removing the nylon filaments embedded in the hydrogel chamber. Stem cell-derived ECs (SC-ECs) were seeded into the microchannels of the chip and cultured for 2 days under static or bidirectional flow conditions. SC-ECs formed vascular structures with intact endothelial barrier function under flow conditions and also showed higher levels of NO (an important index for evaluating ECs’ function) synthesis, which is consistent with vascular function in vivo (Figure 5(a)).

VOCs fabricated with stem cells and additional cell sources. (a) (i) Cross-sectional view of the 3D-printed chip. (ii) Immunofluorescence images of SC-ECs (VE-cadherin) under bidirectional flow or static conditions. (b) (i) Schematic illustrations of the vascular remodeling induced by flow in vitro. (ii) Immunofluorescence images of HUVECs (CD146), pericytes (α-SMA), and nuclei (DAPI). (c) Images of microvascular networks derived from ECs-BMSCs and ECs-ASCs co-cultures and ECs alone. (d) Immunofluorescent images of CM (cTnT) and CF (VIM) markers in cardiac tissues fabricated using normal CMs or LQTS CMs and normal CFs or LQTS CFs (e) (i) Schematic overview of the VOC. (ii) Endothelial layer assessment, with healthy endothelial layer, macrophage embedded endothelial layer and TNF-α treated endothelial layer. (Nucleus: white; Actin: Red; Ox-LDL: Green). (iii) Deposition of fibrin with a monoculture of hiPSCs-ECs (Control) and co-cultured macrophages. (f) (i) Microfluidics device design. (ii) Micrographs show the monolayer of HUVECs and perivascular coverage of pericytes of the neovessels after static and flow culture.
Mesenchymal stem cells (MSCs) belong to adult stem cells and have the potential for self-renewal, proliferation and multi-directional differentiation, which can form specific types of tissue cells and have a wide range of applications.86,87 MSCs have the ability to develop toward pericytes during the angiogenesis process, which helps to support and stabilize ECs of the vascular wall. The co-culture of ECs and MSCs can promote the development and stability of blood vessels. 88 For instance, Sano et al. present a microfluidic device that comprises three parallel microchannels, with the middle channel used to fill the gel containing HUVECs and MSCs and the two side channels used to fill the culture medium. 89 Notably, MSCs differentiated into perivascular cells on day 5. With the extension of the culture time, the vascular diameter increased significantly, and the expressions of NO and MMP-9, which play a key role in vascular remodeling, also increased significantly. Additionally, bone marrow-derived MSCs (BMSCs) and adipose tissue-derived MSCs (ASCs) have been widely studied due to their ease of access and similar characteristics (Figure 5(b)). A microfluidic chip was proposed for comparing the angiogenesis ability of BMSCs and ASCs under dynamic culture conditions. 90 In this study, GFP-HUVECs were mixed with BMSCs or ASCs at a 5:1 ratio and grew together in a fibrin hydrogel to form a blood vessel network. The results indicated that BMSCs exhibited stronger angiogenic potential in supporting microvascular network formation and can form larger, more mature and fully perfusable microvascular networks. Additionally, BMSCs co-cultured with HUVECs exhibit better support around the cells, which may help them play a more effective role in the microvascular network (Figure 5(c)).
However, hiPSCs-based vascular modules still face several practical and often underappreciated challenges: (i) line-to-line variability in differentiation efficiency produces batches in which <30% of cells acquire a mature arterial phenotype, forcing repeated costly runs; (ii) long protocols (⩾3 weeks) involving recombinant cytokines and small-molecule cocktails push per-chip material costs above USD 600, an order of magnitude higher than primary-cell alternatives; (iii) immature fetal-like contractile protein profiles compromise shear-response fidelity in chronic CAD studies; and (iv) poor batch-to-batch reproducibility has led journals to recommend independent triplicate differentiations for every on-chip assay, substantially increasing both cost and labor.91–95 For example, studies have shown that hiPSCs-SMCs predominantly express the fetal isoform of β-myosin heavy chain (β-MHC), rather than the adult α-MHC isoform characteristic of mature coronary smooth muscle. This expression profile reflects an immature phenotypic state that adversely affects contractile behavior and functional maturation. 96 This immature molecular signature may compromise the contractile functionality and physiological relevance of the derived cardiomyocytes. Until these bottlenecks are solved, hiPSCs-derived VOCs are best reserved for short-term mechanistic screens rather than the multi-week perfusion models required for plaque evolution or drug-efficacy validation.
Additional stromal cells
Fibroblasts are involved in vascular growth, repair, stability, and disease occurrence in several significant ways. To improve the integrity of plaques and heal ruptured atherosclerotic plaques, SMCs can transdifferentiate into fibroblasts and release the extracellular matrix proteins, including collagen III and fibronectin. 97 In addition, fibroblasts assist in the formation and development of blood vessels by releasing substances that promote blood vessel growth and alter the surrounding supporting structures. An example mentioned above is that a VOC based on hydrogel was presented for dynamic co-culture of human lung fibroblasts (hLFs) and human lung microvascular endothelial cells (hLMVEnCs). 78 The experimental results proved that hLFs not only provided structural support and adhesion sites for hLMVEnCs but also affected the function and vascular barrier integrity of hLMVEnCs by regulating matrix stiffness and secreting growth factors (Figure 3(c)). Another case in point is that a cardiac model was constructed by embedding a mixture of hiPSCs-derived cardiomyocytes (hiPSCs-CMs), cardiac fibroblasts (CFs), and HUVECs using decellularized extracellular matrix hydrogel. 32 They evaluated the toxicity of various drugs and successfully simulated diseases such as long QT syndrome (LQTS) and cardiac fibrosis. In addition, when the prepared cardiac tissue was transplanted into the hearts of rats with myocardial infarction, it was found that cardiac function was significantly improved, fibrosis in the myocardial infarction area was reduced, and vascular density was increased (Figure 5(d)).
Macrophages are innate immune cells involved in preventing infections, wound healing, and vascular inflammation. However, they are also associated with chronic diseases, such as invading the endothelial layer in early atherosclerosis and promoting vascular inflammation. 98 Under pathological conditions, vascular ECs dysfunction triggers the coagulation cascade reaction and inflammatory response process. Among them, the contact between ECs, platelets and white blood cells, as well as the regulation of cells by numerous soluble factors, closely link the two processes, jointly constituting the “thromboinflammation” process.99,100 Understanding thromboinflammation is of great significance for the treatment and secondary prevention of CVDs. Middelkamp et al.’ group developed a VOC model that enables the co-culture of hiPSCs-ECs and macrophages, providing a new platform for studying thromboinflammation. 101 In this study, whole blood was perfused through the channel at a flow rate of 6 μL·min−1 (shear rate approximately 35 s−1), which was consistent with the shear rate of human veins. The experiment found that under the co-culture conditions of macrophages, fibrin deposition significantly increased, and the size of thrombi enlarged, indicating that macrophages significantly promoted the formation of thrombi. The underlying mechanism may involve the inflammatory factors released by macrophages, which can alter ECs function and subsequently trigger the coagulation cascade. Overall, these findings underscore the pivotal role of macrophages in vascular inflammation and thrombosis, thereby offering a robust in vitro model for elucidating the mechanisms of thromboinflammation (Figure 5(e)).
Pericytes are multipotent cells located on the walls of capillaries, closely connected to hiPSCs-ECs, and together form the vascular structure. They play an important role in the formation, maintenance, and functional regulation of blood vessels. Pericytes can sense the signals of new blood vessel growth and release growth factors (like VEGF) to encourage this process. In addition, pericytes possess stem cell characteristics and can differentiate into SMCs, glial cells, and even neurons under specific conditions. 102 Dijk’s research group designed a VOC containing HUVECs, pericytes and ECM for studying the interaction between HUVECs and pericytes. 103 After 3 days of static culture, HUVECs formed a complete endothelial monolayer in the microfluidic channel, while pericytes gradually migrated toward neovascularization and provided support. At a flow rate of 40 μL·min−1, the endothelial monolayer remained intact and the coverage of pericytes remained stable. Dextran leakage was significantly reduced with the support of pericytes, suggesting that pericytes are essential for maintaining endothelial barrier function. In addition, the distribution of pericytes around new blood vessels increased over time, especially in the areas close to HUVECs (Figure 5(f)).
Taken together, these studies establish a mesenchymal-immune-vascular triple coronary microenvironment in vitro. Within this system, fibroblasts help maintain plaque integrity through matrix remodeling and mechanical support. Macrophages, by releasing inflammatory factors, significantly increase fibrin deposition and thrombus volume. Pericytes, in turn, stabilize nascent vascular networks through VEGF signaling and direct endothelial coverage, reducing vascular leakage and offering a multicellular platform for investigating coronary artery repair, thromboinflammation, and collateral vessel formation. Nevertheless, several important limitations remain. Fibroblasts are often derived from limited donor sources, lacking diversity in age, diabetic status, or dyslipidemia background, which limits their ability to mimic patient-specific variations in collagen organization and crosslinking. Similarly, macrophage models, whether derived from cell lines or hiPSCs, often fail to recapitulate the dynamic spectrum of M1/M2 polarization or the lipid-loading heterogeneity observed in human plaques, leading to discrepancies between in vitro thrombus measurements and clinical plaque vulnerability. Furthermore, pericyte functionality has primarily been assessed in static, short-term cultures of 3–5 days, without extended monitoring under physiologically relevant pulsatile shear. Real-time dynamics of pericyte coverage and barrier function during matrix degradation have yet to be quantitatively correlated with microenvironmental changes. Consequently, these highly biologically faithful OOC models remain largely confined to providing mechanistic snapshots. The absence of long-term, dynamically regulated culture conditions limits their utility as scalable platforms for predicting patient-specific coronary repair processes or evaluating novel antithrombotic therapies.
Application of OOC in CAD
Recent advances in OOC technology have enabled the sophisticated vascular platform to recreate the highly curved, pulsating and cyclically strained microenvironment of human coronary artery in a coronary artery-specific manner. CAD progression is characterized by a cascade of pathophysiological alterations, including endothelial dysfunction, inflammatory activation, atherosclerotic plaque formation and rupture, atherothrombosis, and vascular remodeling. 104 Endothelial dysfunction, representing the initial pathophysiological manifestation, precipitates a series of deleterious events including impaired vasodilation, enhanced platelet adhesion and aggregation, and initiation of pro-inflammatory cascades. 105 The development and subsequent rupture of atherosclerotic plaque constitute the pivotal pathological hallmark of advanced CAD. The plaque disruption triggers thrombus formation, potentially resulting in vascular occlusion and consequent myocardial ischemia. These interconnected pathological processes ultimately culminate in coronary artery luminal stenosis and functional impairment, predisposing to catastrophic cardiovascular events such as acute coronary syndrome. 106 The underlying mechanisms involve complex interplay between biochemical mediators (including LDL, inflammatory factors, and coagulation factors) and cellular components. This section systematically examines the application of microfluidic platforms in modeling in both healthy and diseased coronary artery conditions.
Flow-dependent endothelial dysfunction
Endothelial dysfunction represents a pivotal early pathological event in CAD. Hemodynamic shear stress, defined as the tangential force exerted by blood flow on vascular ECs, serves as a critical regulator of endothelial biology. Laminar shear stress (≈3 dyn·cm−2 = 0.3 Pa; τ = μ × γ̇; γ̇ = 4Q/πr³ for a circular lumen of radius r, where μ is dynamic viscosity and Q is volumetric flow rate) exhibits atheroprotective properties, while disturbed or low shear stress (<3 dyn·cm−2 = 0.3 Pa; typically < 100 s⁻¹ shear rate) is strongly associated with endothelial dysfunction and atherogenesis. 107 Ko et al. systematically investigated the anisotropic mechanical properties of the endothelial layer under physiological fluid shear stress using microfluidic devices and biological atomic force microscopy (Bio-AFM). 108 This configuration lacks the pressure pulsatility and cyclic radial strain intrinsic to coronary arteries; consequently, it cannot reproduce the multi-axial mechanical coupling experienced by coronary ECs during the cardiac cycle. The microfluidic devices measured lateral elasticity, while Bio-AFM quantified vertical elasticity. The chip consists of a top cell culture layer, a middle PDMS elastic membrane, and a bottom electrofluidic circuit layer. Experimental findings demonstrated that HUVECs are arranged along the flow direction under the action of fluid shear stress, but there is no obvious direction under the condition of no shear stress. In addition, the decrease in sensor sensitivity after cell culture with shear stress is more serious than that without shear stress, indicating that the transverse elasticity of ECs is higher in the flow direction. Comparative studies under static versus perfused conditions revealed significant flow-direction-dependent variations in transverse elasticity, with maximal stiffness observed parallel to flow direction. This mechanical anisotropy correlated with shear-induced cytoskeletal reorganization, demonstrating the profound mechanobiological regulation of endothelial function (Figure 6(a)).

Application of OOC in endothelial dysfunction. (a) (i) Schematic and experimental photos of a microfluidic device. (ii) The phase contrast and fluorescence images (blue: nuclei; green: VE-cadherin; red: F-actin) of the HUVECs without and with fluid shear stress in the two different cell culture chambers, respectively. (b) (i) Photograph of an artificial artery chip. (ii) 3D fluorescence images and photographs (insets) of artificial artery chips with straight and annular channels. (iii) 3D confocal fluorescence image of HUVECs. (iv) Temporal changes in pressure in the soft microchannel. The pressure changes simulate hypertension, normal, and hypotension blood pressures, respectively. (v) HUVECs cultured in fluid shear force and (vi) combined fluid shear force and cycle stretching. Filamentous actin (F-actin) and nuclei were stained with red and blue fluorescence, respectively. (c) (i) Schematic depicting the structure of the device. (ii) Representative mages of HUVECs fluorescence upon addition of dextran red for different conditions. (iii) Graph shows average concentration of chemokines in conditioned media collected from HUVECs exposed to PBMCs.
As a benchmark for coronary relevance, Chen et al. developed a pressure-responsive microfluidic vascular model that enables simultaneous modulation of pulsatile pressure, shear force and cyclic stretching through deformable circular channels. 109 Although exact pressure magnitudes and strain values were not quantified in that study, the design conceptually integrates the triad of mechanical cues, pulsatility, stretch and shear, that distinguishes coronary from peripheral vessels. This innovative model incorporates two elastic membranes between the upper and lower channels of the chip, creating a deformable soft channel that mimics the biomechanical properties of vascular systems under various physiological conditions. In their comprehensive study, the researchers investigated the mechanical behavior of soft microchannels under varying pressure conditions, focusing on maximum deflection and compliance. The experimental results demonstrated that the maximum deflection of the soft microchannels increases with pressure and stabilizes at higher pressures. Furthermore, the compliance of the soft microchannels was found to increase with pressure, highlighting their sensitivity to pressure changes and their potential for use in modeling dynamic vascular responses. Additionally, he system successfully modeled a range of pathological conditions, including normal blood pressure, hypertension, and hypotension. In the stenosis region, the flow becomes distorted, with shear forces distributed unevenly, and a recirculation zone forms downstream of the stenosis. These flow characteristics closely resemble those observed in atherosclerotic lesions in vivo, validating the model’s ability to replicate complex vascular hemodynamics under various pressure conditions (Figure 6(b)).
Expanding on these microfluidic vascular models, Rengarajan et al. used a 3D vascular MPS utilizing a trilayer PDMS configuration, with a central PDMS rod creating the luminal space between two functional layers. 110 HUVECs were perfused through the tubular architecture and allowed to adhere. Following 48-h exposure to a cytokine cocktail (TNF-α/VEGF-A/IL-6, 10 ng·mL−1 each), the endothelial barrier demonstrated significantly increased permeability to 70 kDa dextran (1.7-fold increase). Parallel co-culture experiments with peripheral blood mononuclear cells (PBMCs) revealed that activated PBMCs substantially disrupted calcium signaling dynamics while secreting elevated levels of proinflammatory cytokines compared to resting PBMCs (Figure 6(c)).
In summary, the VOCs developed by Ko, Chen, and Rengarajan, among others, successfully integrated key physiological stimuli, including shear stress, luminal pressure, and inflammatory signaling, within a single microfluidic system (Table 1). These integrated models enable real-time mechanical loading, quantitative evaluation of endothelial barrier permeability, and high-resolution imaging of cytoskeletal anisotropy, thereby offering a high spatiotemporal resolution platform for dissecting the interplay between vascular mechanics and biological function. To progress toward more authentic models of human coronary physiology, future devices should adopt pressure-responsive deformable channels, introduce heart-rate-frequency pulsatility, incorporate patient-specific curvature and bifurcation angles, and sustain multi-cellular coronary-lineage cultures under multi-axial mechanical coupling. Furthermore, sustaining multicellular cultures of coronary-relevant cell types under multi-axial mechanical coupling will be essential. Such enhancements will help bridge early mechanical perturbations to the development of advanced atherosclerotic lesions, paving the way for individualized vascular mechanical models’ genuine predictive capability.
Endothelial mechano-inflammatory-on-a-chip models.

Atherosclerotic plaque formation
Atherosclerotic plaques are defined by a pathological triad of lipid accumulation, SMCs, and inflammatory cells. The initiation of plaque formation begins with the development of lipid-laden streaks within the arterial intima, driven by the subendothelial retention of modified lipoproteins. 111 Subsequent inflammatory activation promotes macrophage differentiation into foam cells, establishing the foundation for lesion progression. Proinflammatory mediators further induce SMCs migration from the medial layer into the intima, where they proliferate and synthesize extracellular matrix components, ultimately contributing to plaque architecture. Over times, plaques are stabilized by fibrous caps composed primarily of collagen and SMCs, ultimately calcification may occur. Plaque stability critically depends on fibrous cap thickness and calcification patterns, with thin-cap fibroatheroma (TCFAs) predisposing to rupture, thrombin generation, and acute coronary syndromes through exposure of thrombogenic necrotic cores. 112 To model early-stage atherogenesis under physiological flow conditions, Zhang et al. engineered multilayer arterial human TEBVs. 113 The luminal structure was formed by injecting a collagen gel solution containing type I collagen, 10 × DMEM, 1 m NaOH, and human neonatal dermal fibroblasts (hNDFs) into semicircular channels, followed by gelation at 37°C for 30 min. Sequential layering incorporated human coronary artery SMCs embedded in collagen for the medial layer, while endothelial colony-forming cells (ECFCs) were perfused to establish a confluent luminal monolayer. Functional validation confirmed preserved vasoreactivity, with TEBVs exhibiting phenylephrine-induced vasoconstriction and acetylcholine-mediated vasodilation. Histological analysis confirmed a stratified architecture: an endothelialized lumen, a densely populated SMC-rich medial layer, and an outer fibroblast-derived supportive matrix (Figure 7(a)).

Application of OOC in atherosclerotic plaque formation and atherothrombosis. (a) (i) Schematic diagram of the perfusion system. (ii) Cross-sectional view of three-layer TEBV: endothelial layer (CD31-red), SMCs layer (cell tracker-yellow), fibroblast layer (αSMA-green). (iii) Vasoactivity of two- and three-layer TEBVs after 1-week perfusion. (b) Confocal micrographs of the preserved endothelial monolayer formed by GFP-labeled HUVECs surrounded by preserved-labeled AoSMCs cocultured with foam cells. (c) (i) Illustration of an early-atherosclerosis intimal-lumen model in a microfluidic device. (ii) Confirmation of SVEC-10 cells monolayer formation on the top channel. Plasma membrane (green), nucleus (blue) staining. (iii) Monocyte transmigration and foam cell formation in a microfluidic atherosclerosis intimal-lumen model. (d) Confocal fluorescence images from microfluidic chambers without (up) or with HUVECs (low) at 10 min of plasma perfusion. (e) (i) Fluorescence microscopy images showing the distribution of HUVECs (in purple) and SMCs (in yellow) within the constructed arterial models. (ii) Overlay CLSM images of nucleus and Dil-ox-LDL after 24 and 48 h incubation, respectively. (f) (i) Scheme of a perfusable 3D in vitro artery-mimicking multichannel system. (ii) vWF immunofluorescence staining micrographs of a multichannel device with four different degrees of stenosis channels in the static or perfusion environment.
Maringandi et al. developed plaque modeling through a 3D tubular microfluidic system co-culturing GFP-labeled HASMCs, BFP-tagged foam cells, and HUVECs. 114 Foam cells were generated by differentiating THP-1 monocytes into macrophages (phorbol ester stimulation) followed by 24-h exposure to Dil-oxLDL. Cellular interactions were studied under physiologically relevant shear stress, revealing subendothelial foam cell accumulation and early medial layer disorganization (Figure 7(b)). Similarly, Akther et al. developed a dual-channel intima-lumen chip to investigate monocyte transmigration. 115 The upper lumen channel, lined with TNF-α/LDL-treated SVEC-10 ECs, and the lower subendothelial channel, containing mouse aortic smooth muscle cells (MOVAS SMCs) in a collagen matrix, were subjected to arterial shear rates. Real-time imaging demonstrated significant increases in monocyte adhesion, transendothelial migration, and foam cell formation under proatherogenic conditions (Figure 7(c)).
Wang et al. developed a novel 3D GelMA-based VOC that recapitulates the intima, media structure of human arteries and incorporates high-fat and inflammatory stimuli to investigate the dual role of zinc ions in atherosclerosis, a question motivated by the clinical development of absorbable zinc-based stents. 116 Their study revealed a biphasic dose-dependent response, where low concentrations of zinc ions were protective, while high concentrations exhibited toxicity. The researchers identified the SLC30A1-HMOX1-CD40 pathway as a key target influenced by zinc levels in this context. The chip was fabricated as a bidirectional hydrogel tube using 5% GelMA for the inner and medial layers, with a 0.5% GelMA transition layer. Atherosclerosis was induced by exposing the chip to ox-LDL for 12 h, followed by TNF-α/IL-1β stimulation for another 12 h, which resulted in a twofold increase in reactive oxygen species, cholesterol, and triglycerides. Subsequently, zinc ions were perfused through the lumen for 24 h. The chip’s functionality and cellular responses were assessed using assays such as live/dead staining, transepithelial electrical resistance, LDL permeability, and immunofluorescence for α-SMA and osteopontin. Furthermore, RNA-sequencing and quantitative polymerase chain reaction (qPCR) were employed to identify genes related to zinc transport and inflammation-induced ferroptosis. However, the model has several limitations: it lacks macrophages and lipid core structures, cannot replicate vulnerable or ruptured plaque morphology, and employs a 24-h zinc exposure that does not reflect the 3–6-month degradation timeline of zinc-based stents. Moreover, critical plaque stability parameters such as collagen content, fibrous cap thickness, and mechanical strength were not assessed, preventing direct conclusions regarding zinc-induced “cap stabilization.”
Current atherosclerotic chip models have made considerable progress in simulating early-stage lesions by incorporating multi-layered vascular structures, high-fat stimuli, and acute inflammatory cues (Table 2). A key advantage of these platforms is their integration of vascular stratification, cell-to-cell interactions, and real-time imaging within microfluidic systems. These advancements offer a controlled environment with tunable shear forces, enhanced visualization, and high throughput, thereby providing a rapid tool for mechanistic studies and drug screening. However, three key limitations persist. First, they lack the 3D mechanical characteristics of a lipid core, fibrous cap, and calcification, which hinders the simulation of mechanical instability in thin-cap fibrous atheroma. Second, the short culture periods are misaligned with the chronic nature of clinical plaque remodeling and scaffold degradation, which can span months to years. Third, available models often rely on limited cell sources and small donor pools, failing to capture patient-specific variations in immune polarization, genetic background, and disease susceptibility. To bridge these gaps, future research should focus on incorporating patient-specific geometries, extending dynamic culture periods to weeks or months, and enabling real-time tracking of collagen cross-linking and calcification evolution. Additionally, the use of multi-donor immune cells and whole blood perfusion is necessary to accurately recapitulate the entire progression from lipid streaks to plaque rupture, ultimately establishing a reliable in vitro evaluation system for personalized interventions.
Atherogenesis-on-a-chip models.

Plaque rupture-driven atherothrombosis
Atherothrombosis is a critical pathological event characterized by the rupture of atherosclerotic plaque and subsequent thrombus formation, constituting a major underlying cause of myocardial infarction. Upon plaque disruption, procoagulant factors are exposed to circulating blood, triggering platelet adhesion, activation, and aggregation. These processes initiate the coagulation cascade, ultimately promoting thrombus formation and potential vascular occlusion. 99 To model this process, Brouns et al. developed a novel microfluidic chip model exploring the mechanism of thrombus function in vivo. 117 Their study demonstrated that atherosclerotic plaques and associated vascular microenvironments exhibit elevated levels of pro-hemostatic components, such as collagen and tissue factor (TF 12). Their findings revealed that ECs exert only a limited modulatory effect on platelet activation and the initiation of the coagulation cascade, primarily through paracrine signaling and cell-surface interactions. These results suggest a localized mechanism of hemostatic regulation at the endothelial interface, providing insights into how thrombus formation may be spatially restricted to sites of vascular injury (Figure 7(d)).
Von Willebrand factor (vWF) is a large glycoprotein produced and secreted by ECs and megakaryocytes—precursor cells of platelets—and is abundant in both the bloodstream and the vascular endothelium. It is a key player in blood coagulation and platelet aggregation, serving as a critical component in maintaining normal hemostasis. Under conditions of high shear stress, vWF undergoes depolymerization, significantly enhancing platelet aggregation and thrombus formation. Cho and Park and associates engineered a micro-wrinkled arterial model to investigate the impact of wall shear stress on the expression of vWF in cells. 43 The microfluidic chip features a modular architecture comprising multiple functional units, each integrating four microchannels designed for co-culture applications. This modular design enhances the experimental reproducibility and stability while permitting flexible module interchangeability to accommodate diverse research requirements. To engineer the microtopographical features, researchers employed customized 3D-printed fixtures to mechanically strain PDMS substrates followed by air plasma treatment, generating precisely organized micro-wrinkle patterns along channel surfaces. These engineered topographical cues effectively guide the circumferential alignment of SMCs through contact guidance mechanisms. Subsequent precision alignment and bonding of two semicircular PDMS components yielded a functional device incorporating multiple 1 mm diameter circular microchannels. Cellular organization demonstrated distinct patterns under different culture conditions: in static cultures, HUVECs adopted circumferential arrangements mirroring the underlying circular organization of HASMCs, whereas perfusion conditions induced unidirectional alignment of HUVECs into confluent linear monolayers. Under static culture conditions, the expression levels of vWF remained relatively consistent across channels with varying degrees of stenosis, showing no significant differences. However, in channels with 50% stenosis, the expression of vWF was markedly elevated compared to other stenotic channels, particularly within the stenotic regions. This observation further underscores the regulatory influence of local shear stress on vWF expression (Figure 7(e)).
Akther et al. developed a dual-channel microfluidic model to emulate multiple in vivo atherosclerotic conditions, including hyperglycemia, hyperlipidemia, inflammatory states, and vessel stenosis. 118 The device comprises an upper channel mimicking a stenosed lumen and a lower channel simulating a ruptured plaque environment. These channels are interconnected via a 400 μm region containing 2 μm micropores, enabling cross-communication and clot induction. A composite hydrogel composed of 10 wt% gelatin and 0.02 wt% type I rat tail collagen (GEL-COL) was introduced into the lower channel to replicate the thrombogenic extracellular matrix exposed after plaque rupture. which facilitated platelet adhesion and aggregation. Subsequently, venous blood from healthy donors-supplemented with varying concentrations of D-glucose, cholesterol, and TNF-α-was introduced to simulate pathological states associated with diabetes, dyslipidemia, and inflammation. Platelet aggregation was visualized using DiOC6 staining and fluorescence microscopy, allowing real-time assessment of thrombogenic potential under diverse pathophysiological conditions. Afterward, they tested the effects of different aspirin concentrations on platelet aggregation. Under hyperglycemia conditions, 50 and 100 μm aspirin significantly reduced platelet aggregation, while only 100 μm aspirin was effective under hyperglycemia + TNF-α conditions. Overall, this microfluidic model can simulate the impact of various disease factors on platelet aggregation and thrombosis and evaluate individualized antiplatelet therapy strategies (Figure 8(a)).

Application of OOC in atherothrombosis and aneurysm. (a) (i) 2D schematic diagram and a cross-sectional view of the stenosis region. (ii) Calculating fluorescence intensity to determine the corresponding reduction of platelet aggregation in different aspirin concentrations compared to the positive control. (b) (i) Representative confocal microscopy images show that endothelial-specific proteins VE-cadherin and PECAM-1 after 2 months of culture in the microvasculature-on-a-chip model. (ii) Representative confocal microscopy images show that ECs continue to express VE-cadherin and PECAM-1 after 16 h of exposure to 10 ng ml−1 TNF. (c) (i) Assessment of thrombus formation extent under varied laser durations. (ii) Sequential irradiation first on the right, then on the left side, achieving thrombus formation specifically at these two locations. (d) (i) Analysis of CD31 expression in the vessel and bulge regions following 10 days medium perfusion. (e) Fluorescence confocal micrographs of cylindrical VOC and aneurysm chips. (green: VE-cadherin; blue: nuclei). (f) (i) Photograph of the microfluidic chip. (ii) Aneurysm cross-sectional. (iii) 3D views in a cross-section (top) and along the channel axis (bottom) for HCAECs monolayer immunostained. (VE-cadherin: red, Nucleus: blue).
While the preceding model primarily addresses coronary macroangiopathy, the thrombo-inflammatory cascade within distal microvessels also significantly influences perfusion outcomes. A seminal study documented the temporal and spatial dynamics of microvascular thrombolysis, although which research has utilized microvessel models, the insights regarding the timing of thromboinflammation and fibrinolysis, as well as mechanical regulation, remain pertinent to distal coronary embolism and microcirculation reperfusion injury. Initially, Qiu et al. developed a three-layer PDMS chip fabricated with an extractable rod mold to create bifurcated microvascular lumens with diameters ranging from 200 to 400 µm. 119 These lumens were perfused with HUVECs for 7 days, establishing a complete monolayer, and the chip supported continuous culture for over 30 days. Endothelial activation was induced by 16-h TNF-α exposure under a physiological shear stress of 1 dyn·cm−2 (0.1 Pa), leading to von Willebrand factor string formation. Perfusion of healthy whole blood then triggered platelet–leukocyte–fibrin clot formation. Using continuous confocal z-stack imaging and multiplex RNA-FISH over 30 days, the team quantified fibrin volume, platelet–leukocyte dynamics, and endothelial barrier resistance in real time. In microfluidic thromboinflammation models, intraluminal delivery of tissue plasminogen activator (t-PA; 5 µg mL⁻¹) within three hours dissolved fibrin and restored endothelial barrier integrity. Delayed t-PA administration at six hours still achieved fibrinolysis but provoked hemorrhage-like leakage. Enoxaparin, administered either prophylactically (0.01 or 1 mg mL⁻¹ for 16 h before clotting) or therapeutically (1 mg mL⁻¹ post-thrombosis), consistently reduced fibrin deposition, suppressed platelet aggregation, and accelerated clot resolution independently of its anticoagulant activity. These results delineate a three-stage thrombolysis timeline (platelet loss → fibrinolysis → endothelial recovery) that mirrors in vivo dynamics, providing the first microfluidic evidence that early t-PA treatment enhances endothelial junctional integrity and limits platelet recruitment, while enoxaparin preserves barrier function and expedites thromboinflammatory resolution (Figure 8(b)). However, the chip employed a uniform circular bifurcation geometry lacking patient-specific stenotic features or residual stress. The model also relied exclusively on HUVECs, omitting SMCs, lipid cores, and donor variability. Potential mechanical drifts in the mechanical microenvironment due to PDMS creep and gel shrinkage during the 30-day culture period were not corrected in real time. Future integration of iPSCs-derived disease-specific ECs, coronary-scale stenosis geometries, and online calibration of elastic modulus could enable the extrapolation of this “microvascular time axis” to individualized treatment decisions for distal coronary embolization.
Liu et al. introduced a photothrombosis-on-a-chip platform that integrates ECs, whole blood, and hemodynamics. 120 This system utilizes targeted photothrombosis to induce site-specific thrombus formation using Rose Bengal as a photosensitizer, which generates reactive oxygen species (ROS) upon illumination with 561 nm green laser light. Whole blood supplemented with the photosensitizer was perfused through a microfluidic chip at a speed of 30 μL·min−1. Localized laser irradiation allowed precise induction of thrombi at defined regions within the microchannel. By modulating laser intensity and exposure duration, the severity of thrombus formation could be finely tuned, enabling simulation of various levels of vascular occlusion (Figure 8(c)).
The dosage above align with (or remain within twofold of) the unbound Cmax reported for healthy volunteers after standard clinical doses: ≈5–10 µM free aspirin, 121 0.38–0.45 IU·mL⁻¹ anti-Xa activity for enoxaparin, 122 and 1–2 µg·mL⁻¹ free alteplase, 123 supporting the physiological relevance of the observed on-chip efficacy (Table 3).
Alignment of on-chip effective concentrations with clinical exposure ranges.

The on-chip conc. of 1 mg mL⁻¹ (≈ 1 000 IU mL⁻¹) is used only for mechanistic verification and far exceeds clinical exposure; therefore, the fold value (2–3×) is calculated relative to 0.38–0.45 IU mL⁻¹.
The six chip studies discussed above collectively incorporate mechanical, biochemical, and pharmacological factors into in vitro thrombosis modeling (Table 4). They demonstrated stenosis-induced upregulation of vWF under high shear stress, the impact of multiple risk factors on platelet aggregation, laser/photochemical induced occlusion, and extended thrombolysis dynamics in microvessels. Together, they provide a high-throughput platform for investigating antithrombotic mechanisms and personalized medication. Nevertheless, these models share limitations including geometric simplification, short culture periods, and the use of single donor cells. They lack patient-specific stenosis, long-term collagen remodeling, and interactions involving multiple blood cell lineages. To advance beyond current fragmented thrombus models and achieve a comprehensive in vitro space-time continuum from coronary stenosis to distal embolism, it is imperative to integrate imaging and 3D printing, implement dynamic cultures spanning weeks to months, and enable online calibration of the mechanical microenvironment. Such integration is crucial for the realization of truly individualized anti-thrombotic and anti-inflammatory program verification.
Atherothrombosis-on-a-chip models.

Coronary artery aneurysm
Coronary artery aneurysm (CAA) is a relatively rare cardiovascular abnormality characterized by focal dilation of a coronary artery segment to more than 1.5 times the diameter of an adjacent normal vessel. In adults, the predominant etiology is coronary atherosclerosis, which leads to endothelial injury, lipid accumulation, fibrous tissue proliferation, and calcification. These pathophysiological changes compromise vascular elasticity and structural integrity, eventually resulting in aneurysm formation. In contrast, Kawasaki disease is the most common cause of coronary artery aneurysms in pediatric and adolescent populations. During the acute phase of Kawasaki disease, intense vascular inflammation causes endothelial damage and SMC apoptosis, culminating in aneurysmal dilation of the coronary arteries.
The development of VOC platforms has increasingly relied on advanced 3D printing techniques to replicate the geometric and biomechanical features of aneurysms. Yong et al.’s team developed an intracranial aneurysm model based on 3D printing and hydrogel, which offers significant translational value for coronary applications, due to the conserved architecture of arterial vessels. 124 Using a technique termed liquid-assisted injection molding, the team fabricated anatomically and biomechanically relevant aneurysm models with biocompatible materials such as GelMA. A tailored hydrogel, engineered through precise modulation of its composition and cross-linking density, was developed to closely approximate the mechanical properties of native aneurysmal tissue under different pathological states, including healthy, inflamed, and ruptured conditions. ECs and SMCs were subsequently seeded within the hydrogel scaffold to recapitulate the cellular microenvironment of the aneurysmal wall. These models successfully recapitulated critical physical parameters such as wall compliance and stress distribution (Figure 8(d)).
Yu et al. developed a photopolymerization-based method to fabricate hydrogel-based vascular lumens within PDMS microfluidic channels. 125 By precisely modulating the diffusion time of a photoinitiator (PI) and the exposure duration to ultraviolet (UV) light, vascular geometries including stenoses, aneurysms, bifurcations, and curvatures were formed with high spatial fidelity. This diffusion-controlled polymerization technique enabled fine-tuning of hydrogel stiffness and elasticity, thus replicating both physiological and pathological mechanical environments. HCAECs were cultured on the lumen surface, and pulsatile flow was applied to simulate physiological blood flow dynamics. This platform is capable of replicating four critical mechanical forces-shear stress, intraluminal pressure, cyclic strain, and Young’s modulus of which can be independently modulated to mimic disease-specific vascular mechanics (Figure 8(e)).
In addition to hydrogels, collagen is frequently employed to create deformable elastic channels. Lee et al. introduced a unified method based on gravity channel shaping for constructing complex, living 3D vascular structures within a collagen matrix. 126 This versatile method allowed for the replication of various vascular geometries, including linear vessels, aneurysms, stenotic segments, bifurcations, and tortuous configurations. The process began with computer-aided design to define the target vascular morphology, followed by mold fabrication using 3D printing and subsequent PDMS casting to generate the microfluidic device. Collagen was then injected into the channel system under GLP regulation to create lumens with variable diameters and morphologies. Channels with expansions of 1 and 2 mm simulated aneurysmal dilations, while constrictions of 25%, 50%, and 75% were used to model different grades of stenosis. Branching angles of 30°, 60°, and 90°, as well as sinuous vessels with peak-to-peak amplitudes of 1.5–2.5 mm, allowed simulation of complex vascular topographies. Upon seeding with HUVECs, the model exhibited EC morphology and hemodynamic behavior closely resembling in vivo conditions (Figure 8(f)). The above microfluidic platforms, leveraging advanced techniques such as liquid-assisted injection molding, gravity channel shaping, and photopolymerization, enable highly realistic simulation of coronary artery aneurysms and other complex cardiovascular conditions, thereby providing invaluable tools for elucidating disease mechanisms and developing and testing targeted therapeutic strategies.
Given the significant overlap in geometric complexity and hemodynamic behavior between coronary and intracranial aneurysms, a study that developed a 3D printed chip for patient-specific internal carotid aneurysms offers a readily applicable experimental framework for the patient-specific mechanical analysis of coronary aneurysms. 127 This approach integrates in vitro particle image velocimetry with in vivo computational fluid dynamics to provide closed-loop verification within a heterotypic vessel model. Using patient-specific digital subtraction angiography data, the researchers fabricated a transparent rigid resin chip that accurately replicated the aneurysm geometry. High-speed particle image velocimetry (PIV) was employed to characterize the velocity field and vortex trajectories under pulsatile flow conditions (1 Hz). The study made its STereoLithography file format files, boundary flow conditions, and PIV parameter scripts openly available, facilitating their application to other vascular systems, such as coronary aneurysms or renal artery stenosis. However, the use of rigid WaterShed resin prevents the simulation of arterial viscoelasticity and the coupling of stress and blood flow. The k-omega shear stress transport turbulence model file format turbulence model, while suitable for low Reynolds numbers, requires recalibration for the high-shear and dynamic oscillation characteristic of the coronary environment, particularly concerning near-wall treatments and turbulence modeling. Moreover, the current experimental setup lacks an endothelium-matrix layer, a critical component for physiological relevance. Future advancements should emphasize elastic chips based on PDMS or hydrogels, incorporating multicellular layers comprising ECs and SMCs from multiple donors within a collagenous matrix. Coupled with real-time correction for material creep and shrinkage, such advances would shift the paradigm from mere mechanical analogy toward biological fidelity, a crucial step toward developing physiologically representative in vitro models for assessing coronary aneurysm rupture risk or stent performance.
Collectively, the studies discussed above trace the evolution of coronary aneurysms and atypical blood vessels from mere geometric replication to the integration of mechanical and biological factors. Advanced technique-liquid injection molding, light-diffusion polymerization, and gravity channel formation, enable rapid customization of vascular features such as bifurcations, stenoses, tortuosity, and cystic dilations within a single microfluidic chip. The capacity to apply four distinct mechanical parameters, namely pulsatile flow, shear stress, pressure and strain, enables real-time and independent complex dynamic modeling. Furthermore, the incorporation of elastic hydrogels or collagen matrices also enables the co-culture of ECs and SMCs, thereby establishing a high-throughput platform for subsequent assessments of rupture risk or scaffold mechanical properties. Nevertheless, current models remain constrained by three principal limitations. First, the inherent “rigidity” of materials and geometries, with common use of hard resins or temporary gels, fails to replicate the viscoelastic deformation of arteries and the bidirectional coupling between stress and blood flow. Second, the “simplification” of cellular environments is notable, as most models employ only HUVECs monolayers, neglecting the co-culture of ECs, SMCs, and macrophages from diverse donors and disease backgrounds. This deficiency hinders the simulation of chronic inflammation, lipid core development, or fibrous cap remodeling. Third, the “fragmentation” of the temporal scale is a significant concern; endpoint assessments typically last only 7–30 days, which is incongruent with the clinical timelines of several months to years for stent degradation and calcification evolution. Moreover, the creep of PDMS/gel materials remains uncorrected in real-time, leading to a continuous drift in the mechanical microenvironment. Future integration of patient image-driven elastic hydrogel chips, multi-donor iPSCs-derived disease modeling, and real-time calibration of wall strain and calcification progression could shift the paradigm from “mechanical similarity” toward “biological similarity,” thereby enabling a more accurate in vitro assessment of coronary aneurysm rupture risk or personalized stent performance (Table 5).
Coronary artery aneurysm-on-a-chip models.
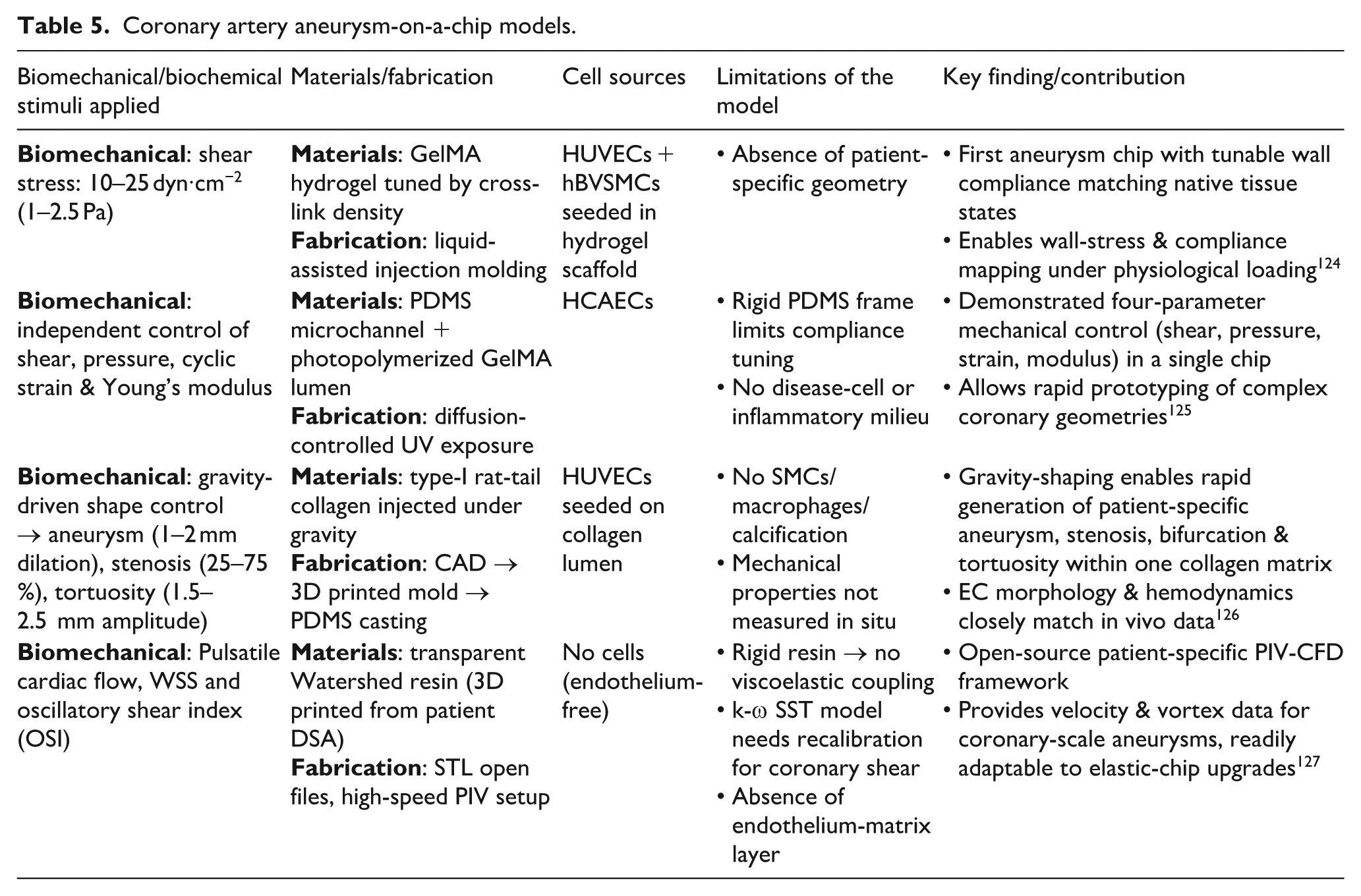
Challenges and opportunities
CAD-on-a-chip technology represents a significant advance in modeling coronary artery pathophysiology, yet its full potential is currently limited by persistent challenges. To systematically address these issues, the following sections outline key limitations and promising avenues for future research and development.
Vascular complexity and microarchitecture
Despite ongoing advancements, current in vitro models for atherosclerosis, including microfluidic systems, still fall short in replicating the multifactorial and progressive characteristics of cardiovascular disease, largely because they cannot fully capture intricate biological interactions and environmental cues that drive pathogenesis in vivo. Specifically, the intricate interplay governing hemodynamic forces, inflammatory responses and lipid metabolism, critical elements in plaque development, remains a substantial hurdle for complete recapitulation within simplified microfluidic platforms. Existing OOC platforms encounter difficulties in replicating the native coronary network’s hierarchical branching, varying caliber, and region-specific hemodynamics. Most designs utilize straight conduits or basic bifurcations, overlooking the spatial gradient of oscillatory shear that contributes to the predisposition of thin cap fibroatheroma. While recent innovations in viscous-finger patterning and laser-etched glass capillaries permit patient-specific tortuosity, these methods remain low-throughput and often lack the elastic wall properties necessary to simulate cyclic strain.
Cell immaturity and standardization
The vascular system’s intricate branching, diverse cellular composition, and dynamic biomechanical forces complicate accurate disease modeling in simplified in vitro settings. Maintaining the physiological relevance of cellular phenotypes and ensuring standardization across various microfluidic platforms remain critical challenges for widespread adoption and clinical translation. These challenges underscore the persistent need for advanced microfabrication techniques and robust cell culture protocols to faithfully mimic the complex microenvironment of coronary arteries and associated pathologies. This involves replicating the mechanical forces experienced by blood vessels and maintaining cell differentiation for accurate disease modeling. While iPSCs-ECs and SMCs offer patient specificity, their differentiation protocols yield cells with fetal-like phenotypes, characterized by low endothelial nitric oxide synthase phosphorylation, immature calcium-handling proteins, and minimal contractile apparatus, necessitating protocol harmonization. To address these issues, we recommend establishing standardized analytical benchmarks, including consensus thresholds for transendothelial electrical resistance, agonist-induced vasoreactivity dose-response curves, and sprouting assay protocols, ideally validated through multi-laboratory collaborations akin to hematology analyzer standardization efforts.
Long-term multiculture dynamics
Sustaining functional co-cultures under physiologically relevant flow conditions remains a major obstacle for studying chronic cardiovascular processes. Endothelial-smooth muscle-fibroblast communication often deteriorates within 4 weeks, due in part to nutrient gradients and PDMS creep. Overcoming these limitations will require advanced biomaterials and dynamic culture systems that better replicate in vivo conditions. Incorporating tunable hydrogels and bioreactor designs may improve construct stability and enable prolonged observation of disease progression and drug responses. In parallel, real-time, non-invasive monitoring of key biomarkers would provide deeper insight into cellular behavior and therapeutic effects.
Cost barriers and fabrication bottlenecks
High material and equipment costs, combined with complex fabrication processes, pose significant accessibility challenges. The use of cleanroom SU-8 masters, precision milling, and automated alignment increases per-device costs beyond $500, excluding numerous global laboratories and thereby limiting accessibility. Furthermore, primary cell isolation, iPSCs differentiation, and quality control add over $200 per donor batch. Limited commercial availability of patient-specific coronary cells further increases expenses.
AI-driven opportunities
Artificial intelligence (AI), particularly deep learning, can address the limitations of microfluidic platforms by automating the analysis of complex biological data. AI can enhance predictive accuracy and accelerate drug discovery, especially for cardiovascular conditions where traditional models lack clinical relevance. By processing large datasets generated by OOC systems, AI can improve screening efficiency and mitigate time and cost constraints in preclinical development. Current applications include convolutional neural networks predicting thrombotic risk with 92% accuracy from shear-stress maps, reinforcement learning algorithms adjusting antiplatelet doses in silico in real time to reduce variability, and federated learning platforms enabling multi-institutional model improvement without sharing raw patient data.
Multi-organ and clinical translation
The integration of multiple organ systems onto a single microfluidic platform represents a major step forward in modeling systemic disease interactions and enhancing the translational validity of preclinical drug testing. Connecting the coronary chip in series with modules representing the heart or liver and adipose tissue, researchers can effectively model the entire metabolic pathway of conditions such as “high-fat diet → hepatic LDL oxidation → systemic inflammation → coronary endothelial activation” within a unified perfusion circuit. This systemic approach enables study of metabolic syndrome-driven atherosclerosis. As microfluidic device costs decline and automation becomes more widespread, future integration of multi-organ, multi-marker chips into catheterization labs could support bedside antiplatelet titration and personalized stent selection.
Conclusion
This comprehensive review surveys the evolving landscape of OOC technology in modeling CADs, offering a forward-looking perspective on its scientific, clinical and translational potential. To ensure a timely and focused scope, we performed a narrative search of PubMed and Web of Science (recent 5 years) using keyword combinations: (“coronary” OR “coronary artery”) AND (“organ-on-a-chip” OR “microfluidic”) AND (“shear stress” OR “atherosclerosis”). We began by outlining foundational materials and fabrication strategies, emphasizing innovations in PDMS microfluidics and 3D bioprinting for constructing physiologically relevant vascular microenvironments. The discussion then focused on cellular components essential for recapitulating coronary lesions, including ECs, SMCs, fibroblasts, and other stromal cells with particular attention to advances in patient-derived iPSCs differentiation protocols. Consequently, contemporary applications of OOC platforms were critically evaluated, highlighting their capacity to model physiological hemodynamics, endothelial mechanotransduction, and complex disease progression, from endothelial dysfunction and early atherogenesis to plaque rupture and thrombosis. Our analysis of current limitations underscored pressing challenges in vascular network maturation, long-term co-culture viability, and the need for standardized validation methodologies. Addressing these issues will require interdisciplinary collaboration to enhance the fidelity, reproducibility, and translational relevance of OOC models. Looking forward, OOC technology holds transformative potential through three key trajectories: (1) integration of multi-organ systems to study systemic cardiovascular interactions, (2) artificial intelligence-driven predictive modeling of disease progression and drug responses, and (3) clinical translation for personalized risk stratification and therapeutic optimization. By bridging in vitro models with in vivo complexity, OOC platforms are redefining preclinical research paradigms. Their ability to emulate human-specific disease mechanisms and permit real-time interrogation of molecular and biomechanical phenomena positions OOC systems as indispensable tools in the pursuit of precision medicine for CAD.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the “Pioneer” and “Leading Goose” R&D Program of Zhejiang (2023C03030), the National Key Research and Development Program of China (2022YFA1405002 and 2023YFF0613600), the National Natural Science Foundation of China (82370509 and 12325405), and the Wenzhou Basic Scientific Research Project (Y20240116).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
