Abstract
Periodontal disease is a pervasive and serious health issue, affecting millions globally and leading to severe oral and systemic health complications. This underscores the urgent need to thoroughly understand the complex host-microbe interactions involved. Developing models that allow crosstalk among various bacteria, periodontal component cells, and circulating immune cells is crucial for investigating periodontal disease and discovering new treatments. This study aimed to develop a biomimetic gum tissue model. Within four days, a bio-fabricated tissue with well-established barrier and immune functions was created. In this model, the key periodontal pathogen, Porphyromonas gingivalis, was observed to suppress the recruitment and migration of immune cells and dysregulate CD14 expression in THP-1 cells, leading to significant inflammation and tissue damage. Conversely, the probiotic Akkermansia muciniphila enhanced the host’s defensive immune response, highlighting its potential as a therapeutic agent in periodontal disease.
Background
Periodontal disease, characterized by inflammation of the tissues supporting teeth, is a widespread and serious oral health issue affecting millions globally.1-3 It includes conditions such as gingivitis and periodontitis, primarily triggered by microbial dysbiosis in the oral cavity.
A key pathogen in periodontal disease is Porphyromonas gingivalis (Pg), which uses sophisticated mechanisms to evade the host immune response and promote microbial imbalance.4-6 Pg produces virulence factors like SerB and gingipains that suppress the host’s immune response, degrade tight junction proteins and chemokines, and inhibit the recruitment and migration of immune cells—a process known as local chemokine paralysis.7-9 Additionally, Pg manipulates Toll-like receptors (TLRs) and the complement receptor C5aR to dysregulate neutrophil and macrophage functions, aiding its survival and the overgrowth of pathogenic bacteria.10-13 Probiotics, as live beneficial microorganisms, have shown potential therapeutic benefits for periodontal diseases by regulating immunity and modifying the microbiota.14-19 One of the next-generation probiotics, Akkermansia muciniphila (Am), has garnered significant attention. Since it was first isolated in 2004, numerous studies have indicated that supplementation with Am, its pasteurized form, or its extracted protein can improve various health conditions. For instance, extracellular vesicles from Am MucT (AmEVs) assist in regulating tight junctions, thus restoring gut barrier function. 20 Furthermore, Am can enhance the intestinal production of antimicrobial peptides like regenerating islet-derived protein 3 gamma (REG3G), which helps maintain microbiota homeostasis. 21 These findings support the potential clinical application of Am. Excitingly, several studies have also demonstrated its capacities in managing periodontitis. It was reported that Am could increase junctional markers (intergrin-β1, E-cadherin, and ZO-1) in gingival epithelial cells. 22 Meanwhile, Am and Amuc_1100 could increase chemokines expression (IL-8 and CXCL10) in gingival epithelial cells.22,23 Additionally, it could modify periodontal microbiome and suppress the virulence of periodontal pathogens like Pg and Fusobacterium nucleatum.22,24,25 Despite these promising results, translating these benefits from pre-clinical studies to clinical applications are challenging, due to the lack of human-relevant models for evaluating their effects. 26
The pathogenesis of periodontal disease involves intricate interactions between the host and microbial communities, engaging various periodontal cell types, including epithelial cells, fibroblasts, endothelial cells, and immune cells. This complexity poses significant challenges for disease modeling and therapeutic screening. Traditional two-dimensional in vitro models fall short in replicating the physiological relevance and intricate host-microbe interactions observed in vivo. Although animal models have been instrumental in understanding periodontal disease pathogenesis, they come with limitations, including ethical concerns, high costs, and time consumption. 27 Additionally, differences in bacterial communities and genetic variations between animals and humans further complicate the extrapolation of animal study results to human conditions. 28 Furthermore, specimens from animals are typically collected postmortem, which inhibits real-time analysis.
Recent advancements in organ-on-chip (OoC) technology, particularly microfluidic-based systems, offer a promising future for disease modeling and drug discovery. Compared with traditional two-dimensional models, these platforms can mimic the tissue structure and function of human organs, providing a more physiologically relevant environment. Transwell inserts are widely used in three-dimensional (3D) tissue engineering due to their simplicity. Nevertheless, constructing 3D cultures using Transwell inserts can be time-consuming as they build tissue layer by layer.29,30 Organoids represent an advanced technology for 3D culture, but the organization of the tissue structure highly relies on the nature of the cells. Additionally, insufficient nutrition and oxygen supply can easily lead to cell necrosis.31,32 Given the challenges of the traditional models, OoC technology can be a better alternative. Over the past two decades, OoC technology has made significant strides in periodontal research. Customization features of OoC design allow researchers to tailor devices and develop different interacting tissues based on specific needs, such as gingival crevice-on-chip, periodontal ligament-on-chip, tooth-on-a-chip, dental implant-on-a-chip, and gingiva-on-chip.33-39 Typically, in these studies, human-derived cells and periodontal microorganisms have been used, allowing for more precise extrapolation of the results. Additionally, by employing transparent materials like polydimethylsiloxane (PDMS), real-time visualization has been easily achieved. Furthermore, with a rich compartment design for medium exchange, a gingiva tissue with a lifespan of over 28 days was developed using this technology. 37 The fluid has also been utilized to replicate the interstitial fluid in periodontium, apply mechanical stress to periodontal ligament stem cells, induce interstitial pressure experienced under periodontal disease condition, and simulate the protective effects of gingival crevicular fluid flow.33,34,38-41 In these studies, OoC systems that integrate multi-cell type, multi-layered tissues with programmable flow control and functional matrices facilitate periodontal research. However, till date, all gingival/periodontal equivalents have only involved structural cells such as keratinocytes, epithelial cells, fibroblasts, and endothelial cells. To study the immune response in periodontal diseases, immune cells warrant integration into periodontal OoC models. The present model addresses these research gaps with its four-channel design, which allows for the accommodation of circulating immune cells. In addition, most studies have used single matrix components, like fibrin, which are less representative of physiological conditions. 33 Various extracellular matrix components can be combined and adopted.
This study aims to integrate various cell types from gingival tissue—epithelial cells, fibroblasts, endothelial cells, and circulating immune cells—along with the key pathogen Pg into a gum-on-a-chip platform. This approach seeks to create a stable platform for multi-species host cell-microbe interactions and simulate the immune response during disease, providing a more accurate and reliable foundation for drug discovery and evaluation. We propose using the gum-on-a-chip platform to investigate the therapeutic potential of Am in periodontal disease.
Materials and methods
Cell culture
Primary human gingival epithelial cells (HGECs) and their culture media, CnT-PRIME, were ordered from CELLnTEC (Switzerland). Primary human gingival fibroblasts (HGFs, American Type Culture Collection, ATCC, USA) were grown in fibroblast basal medium (ATCC, USA) supplemented with fibroblast growth kit-low serum (ATCC, USA). Primary human umbilical vein endothelial cells (HUVECs) were ordered from ATCC, which were grown in Endothelial Cell Medium (ScienCell, USA). THP-1 cells (ATCC, USA) were maintained in RPMI-1640 medium (ATCC, USA) containing 10% fetal bovine serum, 100 µg/mL normocin (InvivoGen, USA). All cells were cultured at 37°C in a 5% CO2 atmosphere.
Bacterial strains and culture conditions
Akkermansia muciniphila (Am, BAA-835) and Porphyromonas gingivalis (Pg, 33277 and W83) were obtained from ATCC (USA). Am was cultured on brain infusion agar plates (Difco, USA), while Pg was grown on blood agar plates (39 g/L Columbia agar base from Difco, 5% defibrinated horse blood from Hemostat, and 1% hemin and vitamin K1 solution) under anaerobic conditions at 37°C . Pg was also cultured in tryptic soy broth (TSB; 30 g/L tryptic soy broth from Difco, 5.0 g/L yeast extract from Difco, and 1% hemin and vitamin K1 solution), and Am in brain heart infusion (BHI, Difco) broth medium with 0.3% mucin. Bacterial cells in the log phase were harvested for further use.
Experimental workflow
As illustrated in Figure 1, a designed chip was first constructed using transparent and biocompatible PDMS, allowing for visualization during subsequent assays. A matrix gel was then developed, consisting of collagen Ⅰ, collagen Ⅳ, and fibronectin, to provide a biomimetic environment conducive to periodontal cell growth and proliferation, as well as a robust scaffold for periodontal cells to spread and migrate. HGECs and a mixture of HGFs and HUVECs were embedded in the matrix gel and injected into the respective channels. The characteristics, such as anatomical structure and barrier function, were verified as the tissue grew. After confirming the establishment of the gum-on-a-chip model, it was used to model Pg-induced disease and evaluate therapeutic agents.

Schematic illustration of the experimental design in gum-on-a-chip. The construction of the Gum-on-a-chip involves a two-step process: the fabrication of the microfluidic device, followed by the creation of the microtissues. The validation of the Gum-on-a-chip’s characteristics involved a comprehensive assessment of its anatomical structure, cell morphology, functional protein expression, and barrier function. Established gum-on-a-chip was used to evaluate Am’s influence on Pg and the host immune response.
Device fabrication and assembly
The gum-on-a-chip device was designed using AutoCAD (Autodesk 2024, USA), printed onto transparency photomasks, and fabricated using lithography techniques. The SU-8 silicon master was made according to a previous study. 42 A 4-inch silicon wafer was first dehydrated at 200°C for 10 min, then spin-coated with 10 g SU-8 2100 at 2000 rpm for 30 s. The photoresist underwent a baking process at 65°C for 60 s and then at 95°C for 360 s. It was subsequently exposed to 160 mJ/cm2 at 365 nm using a mask aligner with the transparent photomask. Following this, a post-exposure bake was conducted at 65℃ for 60 s and at 95℃ for 360 s. The wafer was then developed in mr-Dev 600 developer for 5 min, resulting in channel heights of 150 µm. Finally, the master mold was treated overnight with 1H, 1H, 2H, 2H-perfluorodecyltrichlorosilane to aid in the removal of the polymer cast. A degassed PDMS, Dow Corning, USA) mixture (w/w, 10:1) was poured onto the SU-8 wafer master, cured overnight at 65℃, and peeled off. The PDMS surface with channels was plasma-treated using a Harrick Plasma Cleaner (USA) at medium power (10.5 W) for 60 s and bonded with plasma-treated glass slides (CellPath, UK) to assemble the device. For sterilization, chips were autoclaved for 60 min at 110°C.
The device includes two central gel channels (width: 1.3 mm) and two lateral channels (width: 1 mm) separated by trapezoidal dams. The depth of the microfluidic channels is 150 µm. It includes inlets and outlets for fluid introduction and removal, as well as reservoirs for reagent storage (Figure 2).

Device structure: (a) 3D perspective view. (b) The vertical view, measured in millimeters (mm), details the dimensions of the channel and dam. (c) The right view provides height of the reservoirs and channels.
Gum-on-a-chip Tissue Construction
In this study, a biomimetic extracellular environment was created in the chip using a combined extracellular matrix (ECM) gel. The matrix was composed of rat tail collagen Ⅰ (3 mg/mL, Thermo Fisher, USA), Matrigel (collagen Ⅳ constituting 30% of total protein content, Corning, USA, Cat# 354230), and fibronectin (1 mg/mL, Sigma Aldrich, USA, Cat# F0895). The protein level of Matrigel varies for each batch. The stock was adjusted with PBS on ice to a concentration of 3 mg/mL, based on the original concentration specified by the manufacturer for each batch. All the matrix gels were mixed in a 1:1:1 ratio in the following steps. One volume of fibronectin was mixed with one volume of Matrigel on ice. Then, NaOH was added to the collagen I solution, to induce its gelation. According to the manufacturer’s instruction, for each volume of collagen Ⅰ solution, add 0.025 volumes of 1N NaOH. Once gelation was induced, one volume of collagen Ⅰ was mixed with the fibronectin-Matrigel mixture on ice. This mixture was then immediately combined with one-fifth of its volume of cell suspension and injected into the channels within 3 min.
Example Calculation:
Combined ECM gel: 20.5 µL Collagen Ⅰ solution + 40 µL fibronectin-Matrigel mixture.Then mix every 25 µL of gel with 5 µL of cell suspension.
In this matrix gel, collagen Ⅰ is the predominant collagenous protein, collagen Ⅳ constitutes 9% of the total tissue composition, and the final concentration of fibronectin is 0.28 mg/ml.
A 10 µL suspension of HGECs (5 × 106 cells/mL) or a 10 µL mixture of HGFs (2.5 × 106 cells/mL) and HUVECs (2.5 × 106 cells/mL) was embedded in the matrix gel and injected into the two central channels. Then the device was placed in a 37℃ incubator to allow the curing of the matrix gel. After curing, the culture medium was injected into the lateral channels using a pipette. The medium in the sulcus channels was CnT-PRIME for HGECs, while the medium in the vessel channels was a 1:1 mixture of HGF and HUVEC media. The media was introduced into any two inlets on the same side of the two channels and allowed to diffuse naturally to the outlet channel. The culture media was perfused statically throughout the culture period, with daily medium changes.
Characterization of Cell Layers
For assessing the structure and viability of the cells cultured in the chip, different cells were labeled with CellTracker (Thermo, USA, Cat# C2110, C34552, and C7025) for tissue structure visualization. Their viability was monitored using the LIVE/DEAD viability/cytotoxicity kit (Invitrogen, USA). Images were captured with a Leica Microsystems DMi8 microscope with LED 8 and DFC9000 sCMOS fluorescence microscope and processed using LAS X office (1.4.4, Leica, Germany) and ImageJ (Fiji, 2.14.0/1.54f, National Institutes of Health, USA).
Immunofluorescence staining was performed to visualize the expression of F-actin, E-cadherin and occludin in the 3D-cultured gingiva equivalent. Samples were rinsed with PBS 5 times each for 3 min and fixed in 4% paraformaldehyde for 1 h followed by permeabilization in the solution of 0.1% Triton X-100 in PBS for 40 min and blocking in 10% normal goat serum solution (Life Technologies, Carlsbad, USA) for 1 h. Afterward, the samples were incubated with primary antibodies at 4℃ overnight prior to 2-h Alexa Fluor 488-conjugated anti-rabbit IgG secondary antibody staining. The cell nuclei were stained with DRAQ5 for 10 min. The antibodies used are as follows:
F-actin: Alexa Fluor™ 594 Phalloidin (Invitrogen, Cat# A12381, at a concentration of 0.2 µM).
E-cadherin: E-Cadherin (24E10) Rabbit mAb (Cell Signaling Technology, Cat# 3195 1:800 dilution).
Occludin: Occludin (E6B4R) Rabbit mAb (Cell Signaling Technology, Cat# 91131, 1:200 dilution).
Secondary antibody: Anti-rabbit IgG (H+L), F(ab’)2 Fragment (Cell Signaling Technology, Cat# 4412, 1:400 dilution).
DRAQ5: (Invitrogen, Cat# 4084, 1:500 dilution).
Permeability and Electrical Resistance
Regarding the barrier function of the constructed epithelial layer in the chip, the permeability of the gingiva-on-chip model was assessed using fluorescein isothiocyanate conjugated dextran (FITC-dextran, 40 kDa, Sigma-Aldrich) at a concentration of 0.2 µg/mL as an indicator. After culturing the tissue for 4 days, the medium was removed from the lateral channels and replaced with PBS containing FITC-dextran in sulcus channel and PBS alone in the vessel channel to maintain hydrostatic pressures. The green fluorescence of FITC-dextran flow was recorded every 10 s using Nikon Eclipse Ti-S inverted microscope equipped with D-LEDI fluorescence LED illumination system and processed by NIS Elements (4.20, NIKON, Japan).
To measure the permeability of the tissue layers, TEER (Transendothelial/Epithelial Electrical Resistance) measurements were performed using a volt ohm meter (EVOM, World Precision Instruments, USA). Modified electrode sets were used, where two thin enameled copper wires (electrode set 1/2) were separately attached to two electrodes on each arm of the EVOM STX2 electrodes. The insulation coating was removed at the wire ends that are attached to the electrodes, and they were fixed with electrical tape. The wires in each set were tightly tied together to ensure proximity between the current-carrying and voltage-sensing electrodes. One electrode set was connected to the gingival sulcus channel, and the other to the blood vessel channel. Chips with medium only were used to obtain baseline levels.
Utilizing gum-on-a-chip to explore Am-modulated immune-inflammatory responses disrupted by Pg
After four-day culturing, THP-1 cells, labeled CellTracker Green, were introduced into the vessel channels, while Am and Pg were added into the sulcus channels at the concentration of 1.5 × 107 CFU/mL. THP-1 migration in the gingiva-on-chip was investigated. After 16 h, the microtissues were fixed and stained with DRAQ5. The migration of THP-1 in tissues was visualized by scanning the tissues under ZEISS LSM 900 with Airyscan 2 confocal microscope (Carl Zeiss AG, Germany). To investigate CD14 expression of THP-1 in different groups, mRNA was extracted from the cells in the vessel channels at different time points. Two micrograms of RNA were reverse transcribed into cDNA using the TB Green® Premix DimerEraser™ Kit (Takara, Japan). Quantitative PCR (qPCR) was performed on the ABI Prism 7700 (Applied Biosystems, USA). The primer sequences for used were: CD14 forward: 5’- CTGGAACAGGTGCCTAAAGGAC-3’, and CD14 reverse: 5’- GTCCAGTGTCAGGTTATCCACC-3’, and ACTB forward: 5’- CACCATTGGCAATGAGCGGTTC-3’, and ACTB reverse: 5’- AGGTCTTTGCGGATGTCCACGT-3’.
The supernatants of tissue challenged by different bacteria were collected from the side channels, and debris was removed by centrifugation at 100 × g for 5 min. The selected cytokine or chemokine levels were quantified using ELISA kits (R&D Systems, USA). The supernatants after 24-hour treatment were harvested and tested using Proteome Profiler Human XL cytokine array kit ARY022B (R&D Systems, USA). Array panels were visualized using iBright 1500 (Invitrogen, USA), and the results were analyzed using HLImage++ (Western Vision Software, USA).
Statistical analysis
All experiments were repeated at least three times independently. The one-way analysis of variance (ANOVA) with Tukey’s multiple comparisons test was used for statistical evaluation of all results. A p-value <0.05 was considered statistically significant.
Results
Design and optimization of a four-channel microfluidic chip for periodontal disease modeling
The periodontal environment involves a complex ecosystem, including oral microbiota inhabiting the gingival sulcus, adjacent epithelial layers, underlying connective tissues, and immune cells circulating in the blood vessels (Figure 3(a)). According to the anatomy of periodontal tissues, a four-channel microfluidic chip was designed. The four channels function as the gingival sulcus, gingival epithelial tissue, connective tissue, and blood vessels respectively (Figure 3(b)). Gingival component cells habitat in the epithelium channel (HGECs) and connective tissue channel (HGFs and HUVECs). Fluidic flow in the gingival sulcus and blood vessel channels supplies the necessary nutrients to cells while accommodating bacterial and immune cell suspensions.

Device fabrication: (a) Illustration of Periodontal environment. (b) Schematic illustration of the four-channel microfabricated gum-on-a-chip model as gingiva equivalents with microorganisms cultured in the gingival sulcus channel, HGECs in the epithelium channel, HGFs and HUVECs in the connective tissue channel, and THP-1 monocytes in the blood vessel channel. (c) The macroscopic view of custom-fabricated gum-on-a-chip device. (d) Flow chart showing the whole process of the bio-fabrication and phase-contrast images of the formed cell clusters and connections over time.
Type Ⅰ collagen, a principal component of the periodontium organic matrix, is prone to shrinkage and collapse due to cell-collagen interaction-induced contraction, compromising tissue integrity. 43 Although Matrigel commonly employed in 3D cultures, its batch-to-batch variability poses significant challenges. 44 Fibronectin, while crucial for cell adhesion, is predominantly associated with clots and granulation tissue in wound healing. To provide an appropriate biomimetic physiological scaffold with good mechanical strength for in vitro periodontium culture, a mixed matrix gel consisting of collagen Ⅰ, Matrigel, and fibronectin was developed, facilitating the three-dimensional distribution of cells. Collagenous proteins constitute the predominant component of the matrix proteins within periodontal tissues. 45 Type Ⅰ collagen is the main collagen species in gingival connective tissue, comprising approximately 80% of the total collagen, while Type Ⅳ collagen constitutes around 1% to 8%.45,46 Fibronectin is distributed throughout the gingival connective tissues. 47 The fibronectin level in circulating blood is about 200–400 µg/mL. 48 However, the specific amount of fibronectin relative to the total protein content in gingival tissue can vary based on factors such as inflammation and smoking.47,49 In our physiologically relevant ECM gel, collagen I constituted the predominant collagenous protein, collagen IV made up a small percentage of the total tissue composition, and fibronectin was present at a biomimetic level.
In this gum-on-a-chip device, human-derived cells were adopted (HGECs, HGFs, and HUVECs) and suspended in the mixed matrix gel before being introduced into the device. The growth of the cells was visualized, and they spread fast in the matrix gel, formed clusters and connected after a three-day culture without structural collapse (Figure 3d).
Validation of cell viability and structural integrity in a co-cultured gum-on-a-chip model
To confirm the survival of cocultured cells, we stained them with LIVE-DEAD dye twelve days post-seeding. Fluorescence microscopy was then employed to assess overall cell viability. Only a few dead cells (in red) were detected in both channels (Figure 4a), indicating a well-established co-culturing system of epithelium (approximate size: H 0.15 mm × L 19 mm × W 1.3 mm) and connective tissue (approximate size: H 0.15 mm × L 19 mm × W 1.3 mm) within our chip model, using a mixed matrix gel and mixed culture media.

Characterization of bio-fabricated tissue: (a) HGECs in the epithelium channel and HGFs and HUVECs in the connective tissue channel stained with both calcein-AM (live, green) and propidium iodide (dead, red) were examined under a fluorescent microscope after a twelve-day culture in the chip. (b) The interface between epithelial layer and connective tissue layer. (c) HGECs labeled with CellTracker Blue, HGFs labeled with CellTracker Red, and HUVECs labeled with CellTracker Green were imaged using a confocal microscope to show the cell distribution and structure of the biomimetic gum tissue.
Figure 4(b) illustrates the orderly distribution of different cell types, with an interface present between the epithelial layer (depicted in blue) and the connective tissue layer (with HGFs in red and HUVECs in green). Epithelium and connective tissue interwove in the gaps between the dams. The matrix gel provided extensively porous and stiff scaffolds allowing cells to grow in a three-dimensional way. No structural deficiency was observed (Figure 4(c)).
Assessment of barrier function and permeability in the gum-on-a-chip model
Bacteria in the oral cavity are initially blocked by the gum tissue, which acts as a barrier to prevent them from invading deeper tissues and entering the bloodstream. Cells within the gum tissue interact with each other to form intercellular junctions, supporting this barrier function by selectively allowing certain substances to pass through. To confirm the barrier function of this fabricated tissue, junction-associated proteins were stained with specific antibodies. The expression of junctional proteins, including E-cadherin (in green) and Occludin (in yellow), was positive in both the epithelial and connective tissue layers (Figure 5(a)). The cytoskeleton (F-actin) in red indicated that the cells were well-spread. The cell nuclei were depicted in blue.
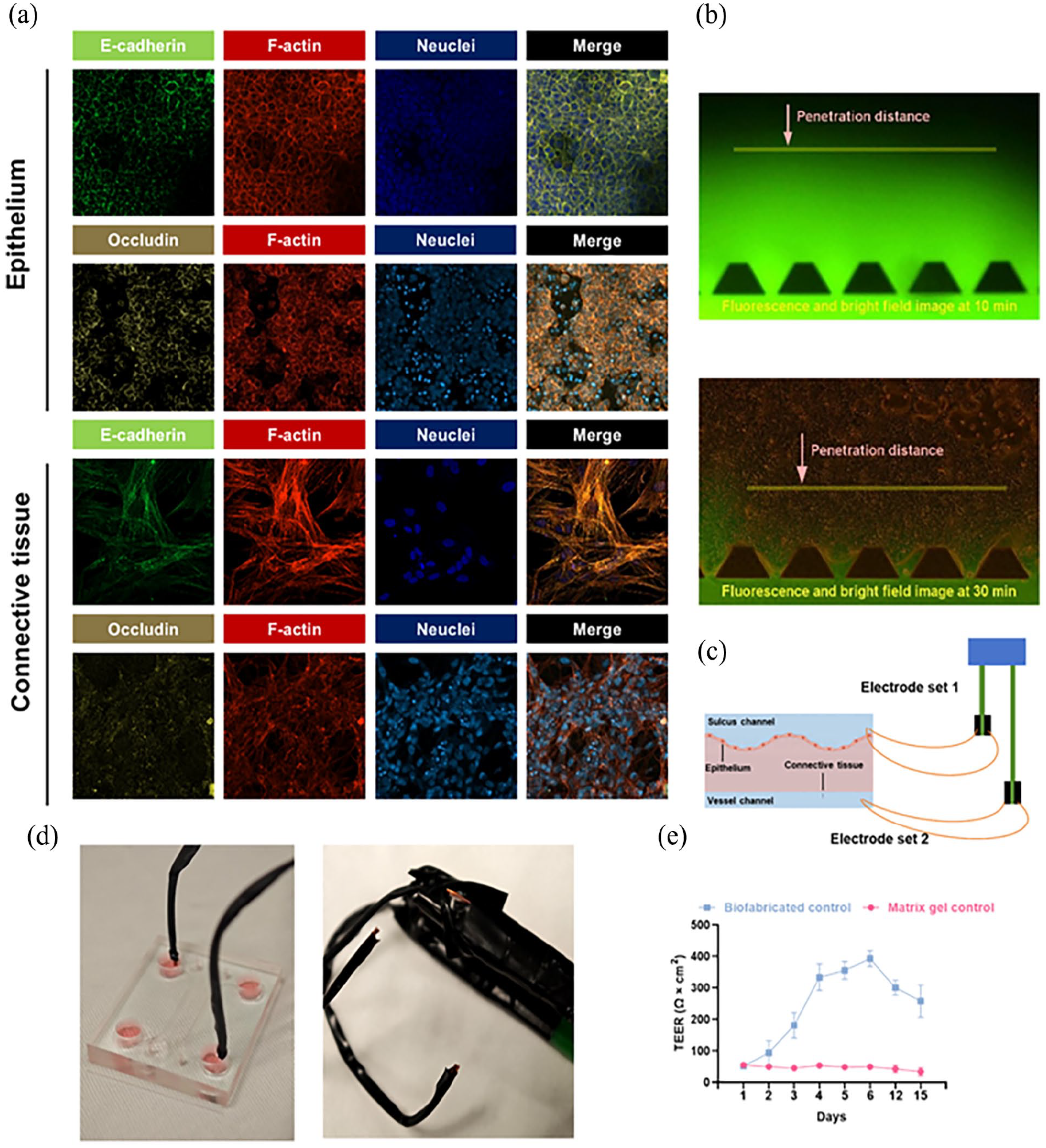
Barrier function of the gum tissue. (a) Immunofluorescence micrographs showed the tight junction of the fabricated tissue on day 4 (green, E-cadherin; Yellow, Occludin; red, F-actin; blue, nucleus). (b) Penetration distances of FITC-dextran in the HGEC-cultured epithelium channel and blank ECM gel channel were recorded at 30 and 10 min respectively after injecting FITC-dextran into the sulcus channel. (c) Illustration of the TEER assessment. (d) Modified electrodes. (e) The electrical resistance (TEER) of the full-thickness gum tissue was assessed over time.
To validate the tissue perfusion, the permeability of the epithelial layer to FITC-dextran was examined using a fluorescent microscope on day 4. Figure 5(b) shows the distance that FITC-dextran has traveled through the HGEC-cultured epithelium channel compared to the blank matrix gel-filled channel. The penetration distance in the HGEC-cultured epithelium channel is notably shorter than in the blank matrix gel-filled channel, suggesting a decreased permeability and sound integrity of the epithelial tissue. To quantify the permeability of full-layer gum tissue, electrical resistance was measured over time, from the first day after cell seeding to 15 days post-seeding (Figure 5(c)). Compared to the TEER of cured matrix gel alone, the TEER of the fabricated gum tissue steadily increased from the first day after seeding, plateaued from day 4, and slightly decreased from day 12. This suggests that cells suspended in the matrix gel proliferated and gradually reached confluency from the first day post-seeding, maintaining stable permeability from day 4 to day 12. The decrease in TEER from day 12 to day 15 might be due to cell apoptosis in the densely compacted tissue.
Pg dampened migration and CD14 expression in THP-1 cells, reversed by Am
Immune cells, such as monocyte/macrophage, are the key player during the host defensive or pathological process, where they act by engulfing pathogens and secreting cytokines to orchestrate the immune response. To recapitulate the immune response in the periodontal environment under various bacterial challenges, including the key periodontal pathogen Pg and potentially beneficial probiotics Am, bacteria were introduced into the sulcus channel with THP-1 cells introduced into vessel channel on day four (Figure 6(a)). To evaluate the influence of bacteria on THP-1 migration within the tissues, CellTracker-labeled THP-1 cells (in green) were captured under a confocal microscope after 24 h of infection treatment. A trend was observed wherein a greater accumulation of THP-1 was found in tissues exposed to Am with/without Pg though no statistically significant differences were detected between any of the four groups (Figure 6(b) and (c)). CD14, a protein primarily found in macrophages, plays a crucial role in the body’s defense against bacteria by acting as a co-receptor for detecting bacterial lipopolysaccharide. Compared to each control group at various time points, the administration of Am or the bacterial mixture increased the relative expression of CD14 over time (Figure 6(d)). Specifically, Am upregulated the expression of CD14 from the 12th hour onwards compared to the control group (p = 0.0003). However, Pg showed a trend of downregulating CD14 expression at 12 h, followed by an accelerating increase in CD14 expression over the next 24 h. After 36 h, a statistically significant increase in CD14 expression was observed in the Pg group compared to the control group (p < 0.0001). Pg has been reported to weaken CD14 expression, thereby reducing the immune response, which aligns with our findings.50,51 In contrast, the probiotic Am exhibited an opposing effect on gum tissue, enhancing the innate immune response against infection. The integrity of the gum tissue under bacterial challenges was investigated using TEER (Figure 6(e)). Before introducing the bacteria, the electrical resistance of the tissue was high across all groups. Twenty-four hours after bacterial introduction, a statistically non-significant reduction in electrical resistance was observed in the Pg group. Forty-eight hours post-introduction, Pg significantly increased tissue permeability (p = 0.009), whereas Am neither increased permeability on its own nor restored the tissue damage induced by Pg.

Pg suppressed the migration and recruitment of THP-1 cells, reversed by Am: (a) Experimental design for assessing the effects of Am in the gum-on-a-chip. Cells were seeded on day 1, and THP-1 and Am was introduced into the gingival sulcus channel on day 4, with or without Pg. Supernatants were collected at different time points, and THP-1 migration was visualized 16 hours after Pg introduction. (b) CellTracker Green-labeled THP-1 cells were administered in the blood vessel channel with microbes introduced into the gingival sulcus simultaneously. After 16 hours, the fabricated tissue was stained with DRAQ5, and THP-1 migration in the epithelial layer was captured under a confocal microscope (blue, nucleus; green, THP-1). Images were taken from three independent fabricated tissues for each group. THP-1 cells in regions of interest were counted. (c) Randomly selected areas from three independent chips of each group were used for statistical analysis. (d) The mRNA of THP-1 cells was extracted at various time points to assess the relative expression of CD14 mRNA in bacterial-challenged groups compared to control groups. (e) The tissue damage under bacterial challenge over time was assessed using TEER. The one-way analysis of variance (ANOVA) with Tukey’s multiple comparisons test was used for statistical evaluation of all results. Data are presented as mean ± SD. *P < 0.05, **P < 0.01, ***P < 0.001, and ****P < 0.0001 versus the control and matched groups. Ns, not significant (P > 0.05).
Immuno-modulatory effect of Am on Pg-perturbed host response in the gum-on-a-chip model
Following the treatments demonstrated in Figure 5(a), the supernatants from the two channels were collected at various time points. Upon exposure to Am, the tissue exhibited an increasing secretion of IL-8 (Figure 7(a)) and MCP-1 (Figure 7(b)) over time in both sulcus and vessel channels. Specifically, during the initial 6 h, the levels of IL-8 and MCP-1 in the vessel channels remained relatively low. Subsequently, an increase in cytokine and chemokine levels was observed in the vessel channel, potentially attributable to the time lag in signal transduction and protein expression within the chip. The infection of Pg alone induced a response pattern distinct from other treatments: the IL-8 and MCP-1 levels were not increased within the first 12 h, while a higher inflammatory response was triggered in the sulcus compared to the vessel channel after 24 h. It aligns with the immune subversion and evasion strategies employed by Pg. 7 When exposed to the mixture of Am and Pg, the tissue released more IL-8 compared to that treated with Pg alone. Notably, these results showed spatial and temporal gradients of the cytokine and chemokine levels in different compartments of the gum-on-a-chip model.

Am modulated immune-inflammatory response perturbed by Pg in the gum-on-a-chip model. (a, b) Supernatants from the gingival sulcus and blood vessel channels of four groups exposed to different bacterial challenges were collected at various time points. ELISA assays determined IL-8 and MCP-1 levels at different post-infection times. (c) After 24-hour bacterial challenges, supernatants from the blood vessels were evaluated using the Proteome Profiler Human XL Cytokine Array Kit. (d, e) A heat map and bar chart present 27 out of 105 proteins with the most significant differences.
To get more details of the immuno-modulatory effect of Am on Pg-perturbed host response in the chip model, the cytokine profiles of supernatants collected from the vessels at 24 h showed that cytokines involved in the recruitment or migration like MCP-1 and CD31 were decreased by Pg, while pro-inflammatory cytokines such as IL-6 and IL-1β were upregulated in all infected groups (Figure 7(c)-(e)). C5a, in conjunction with TLR2 crosstalk, is a key mechanism by which Pg induces dysregulation of macrophage and neutrophil phagocytosis function. Noteworthily, Am decreased the C5a level which is in stark contrast with the Pg and mixture groups.
The weak secretion of IL-8 and MCP-1, along with increased C5a levels in response to Pg exposure, suggests that Pg suppresses the recruitment of immune cells to the infection site and disarms immune cell function, ultimately leading to potent inflammation. In contrast, Am enhances the defensive response, indicating its immune-modulatory potential in periodontal disease.
Discussion
Periodontal health is significantly influenced by the intricate balance between microbial symbiosis and immune homeostasis. The advent of OoC technology, which integrates fluid flow, biomaterials, various cell types, and microorganisms, offers a versatile and innovative tool for exploring these complex interactions. In this study, we designed and fabricated a four-channel microfluidic chip that integrates microorganisms, structural tissues (epithelial and connective tissues), and immune cells to investigate the local immune responses to the key periodontal pathogens Pg and the potential probiotics Am. It was observed that Am could enhance the immune response paralyzed by Pg.
Recent applications of OoC technology in periodontal research have demonstrated its potential to significantly enhance our understanding of periodontal disease mechanisms and treatment strategies. Compared with traditional models, OoC technology has expanded the scope of periodontal research. For instance, a previous study has integrated the spheroid culture of periodontal ligament stem cells (PDLSCs) with microfluidic perfusion platform to explore the influence of interstitial fluid stress on the osteogenic differentiation. 40 Another model, termed “PDL-on-chip,” cocultured patient-derived periodontal ligament cells and endothelial cells in a microfluidic chip to investigate ligament cells’ effect on the vascularization. 33 Additionally, a “gingival crevice-on-chip” model, utilized hydrostatic pressure differences in the microfluidic chip to mimic the flow of gingival crevicular fluid from connective tissue to the gingival crevice. 33 This approach facilitated the exchange of culture media while avoiding disruption of the microbial biofilm, enabling the long-term co-culture of oral commensal Streptococcus oralis with the gingival fibroblast-based gingival tissue equivalent.
Monocytes/macrophages, key players in periodontal immunity, should be involved in periodontal research. A recent study integrated reconstructed human skin with the monocyte-like MUTZ-3 cells for dermal toxicology research. 52 However, models that enable crosstalk among microorganisms, periodontal structural cells, and immune cells have yet to be established. Here, the four-channel device addresses this gap by accommodating periodontal microbes, epithelial layers, connective tissues, and immune cells independently yet communicatively. Signals from the epithelial and connective tissues were transferred to remote immune cells, inducing their migration and differentiation. This setup allows for the investigation of both structural cells’ and immune cells’ responses to microbes.
In addition, the microscale and controllable features of OoC technology reduce sample requirements and improve reproducibility in periodontal research. For instance, a gingiva model with 96 mini compartments, each equipped with individual microfluidic pumps, provided a high throughput platform within a small chip. 37 Another study cultured gingival epithelial and endothelial cells on opposite sides of a porous membrane to construct the gingival barrier function in a coin-sized chip. 39 These two studies mimic the physical anatomy of gingival soft tissue. However, they did not incorporate ECM components, which are crucial for tissue integrity and resilience.45,53 ECM regulates cellular functions such as adhesion, migration, proliferation, and differentiation, essential for tissue maintenance and repair.54,55 Additionally, ECM forms a protective barrier against microbial invasion and mechanical stress, crucial in the oral cavity, and regulates the diffusion of water, nutrients, and toxic materials. 56
In this study, to provide a robust scaffold that supports the three-dimensional distribution of cells, mimics the natural environment of periodontal tissues, and ensures good mechanical strength for in vitro periodontium culture, we combined collagen I, Matrigel, and fibronectin as the extracellular matrix for the fabricated gingival tissue. Similarly, another study utilized a fibrin-based matrix to develop the connective tissue of the gingiva. 38 However, the epithelium was created using an air-liquid culture to achieve stratified, differentiated layers over a 20-day period. While the epithelial layer in the gingiva equivalent is highly relevant in terms of its anatomical properties, developing it can be time-consuming. To accelerate the development of the gum-on-a-chip model, we used ECM to provide anchor sites for cells in both the epithelial and connective tissue channels in a 3D manner. A confluent integrated and reproducible tissue with low permeability can be developed within 4 days.
Previous studies indicated immune evasion is a vital strategy of Pg. Its LPS is a weaker cytokine stimulator and can antagonize the cytokine-stimulating ability of other pathogens.57-59 It can also decrease NF-κB expression, cause ‘chemokine paralysis’ by preventing IL-8 production, and reduce neutrophil migration by lowering E-selectin expression.7,60-62 However, high levels of tissue inflammation and severe immune cell infiltration are typically observed during periodontitis. 63 The conflict arises because immune paralysis occurs only in the very early stages of host cell exposure to Pg, and the time window to capture the protein level shift is transient. Using this gum-on-a-chip model, we observed that Pg tended to inhibit the accumulation of THP-1 cells. This observation aligns with the cytokine profile, which showed that Pg induced a less potent secretion of IL-8 and MCP-1 upon initial exposure, followed by an increased expression of these cytokines later. This study, for the first time, captured this shift, providing evidence for the pathogenesis of periodontitis. Host response to Am was also investigated in this study. It showed that Am could increase the expression of chemokines thereby facilitating the migration of THP-1, which is consistent with previous studies.22,23 Moreover, Am increase CD14 expression of THP-1. CD14 functions as a co-receptor for detecting bacterial LPS, a component of the outer membrane of Gram-negative bacteria. Upon binding to LPS, CD14 aids in the activation of Toll-like receptor 4 (TLR4), which triggers an inflammatory response. This response is crucial for defending against pathogens and activating adaptive immunity. This might be due to Gram-negative Am’s ability to activate TLR receptors, thereby inducing a downstream host protective response.64,65 The results indicated the potential of Am in mobilizing host defensive mechanisms against pathogens.
Despite the promising findings, there are limitations to this study. One limitation is the distortion of the recapitulation of gingival epithelial tissue. To develop a timesaving tissue, an ECM scaffold was added, which is inconsistent with the natural epithelium where cells are compacted with little intercellular ECM. Additionally, without the air-liquid method, the differentiation and stratification of the epithelial tissue fail to be achieved. Also, in the assay evaluating epithelial permeability, technical limitations affected the accuracy of our measurements. Prolonged laser exposure during long-term recording led to FITC quenching, preventing the calculation of a specific permeability coefficient. Future studies should use a fluorescent microscope setup with longer intervals between captures and reduced laser power to accurately evaluate the permeability coefficient of epithelial tissue. Furthermore, this model is suitable for many additional investigations involving other immune cells, such as neutrophils, which can be incorporated in the future. Moreover, to gain a deeper understanding of the potential beneficial effects of Am, the mechanisms behind the immune response induced by Am should be further investigated.
In summary, we developed a straightforward gum-on-a-chip model that establishes gum barrier function within a short period (4 days) and maintains tissue viability for an extended duration (over 12 days). This model facilitates interactions among various bacteria, multiple periodontal cells, and peripheral immune cells. Using this model, we recapitulated the pathogenesis of Pg-induced periodontal disease and evaluated the immunomodulatory effects of the probiotic Am. Real-time observation on this chip revealed the transient immune paralysis induced by Pg and its restoration by Am. As a bioreactor, the gum-on-a-chip efficiently mimics the periodontal environment under human conditions, providing a strategic tool to understand the pathogenesis of periodontal and related infectious diseases, and enabling high-throughput, stable, and accurate drug discovery.
Footnotes
Acknowledgements
None.
Author contributions
In this study, Qin Hu took charge of methodology, data curation, and drafting the original manuscript. All authors were involved in formal analysis, as well as reviewing and editing the manuscript. George Pelekos and Wai Keung Leung were responsible for conceptualization and supervision.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Oral Health Research and Innovation Fund.
Ethical approval
Not applicable.
Informed consent
There are no human participants in this article and informed consent is not required.
Human rights
There are no human participants in this article.