Abstract
Traumatic brain injury (TBI) is a major cause of mortality and morbidity, commonly leading to long-term impairments in cognition, sensorimotor function, and personality. While neuroprotective drugs have demonstrated some efficacy in vitro cultures and in vivo animal models, their clinical applications remain debated. Intranasal delivery to the brain parenchyma, bypassing the blood-brain barrier for more direct access to target sites, offers a favorable and safe approach. This review illuminates current advancements in intranasal delivery systems for TBI treatment. We begin with an overview of TBI and its current clinical treatment options. We then outline recent developments in intranasal delivery systems of molecules and cells, emphasizing their efficacy in animal models. Finally, we discuss future clinical perspectives on emerging trends, offering insights into leveraging intranasal delivery for effective TBI therapeutics.
Introduction
Traumatic brain injury (TBI) entails brain damage resulting from external mechanical forces, including rapid acceleration or deceleration, blast waves, crush injuries, impact, or penetration by a projectile. It can lead to temporary or permanent impairments in cognitive, physical, and psychosocial functions. 1 TBI is a leading cause of mortality and disability among individuals under 45 years old, with approximately 10 million deaths and/or hospitalizations attributed directly to TBI annually, affecting an estimated 57 million individuals globally. 2
TBI manifests as a complex disease process rather than a single pathophysiological event involving primary and secondary injury processes. 3 Primary injury, occurring immediately upon exposure to external forces, results in structural damage and dysfunction, such as axonal shearing, contusion, blood vessel destruction, and hemorrhage. 4 Following a primary injury, secondary injury ensues over minutes to months due to metabolic, cellular, and molecular cascades, culminating in brain cell death, tissue damage, and atrophy. 5
Secondary damage often arises from metabolic imbalances, including disruption in cellular calcium homeostasis, increased free radical production, lipid peroxidation, mitochondrial dysfunction, inflammation, apoptosis, and diffuse axonal damage. 6 These events lead to the loss of neurons, endothelial cells, glial cells, as well as degeneration of brain white matter, with cell death occurring minutes to months after injury, particularly in contused areas and subcortical regions. 7 Apoptosis also coexists with the gradual shrinkage of gray and white matter after TBI. 8
The extent to which cell death and sublethal neurobiological disturbances contribute to post-traumatic morbidities remains unclear. Sublethal cellular processes and systemic insults, such as hypoxia and hypotension, may eventually lead to cell death. Functional abnormalities following TBI result from both acute cell death and delayed apoptosis. Studies on humans and animals have indicated that even mild TBI, without obvious cell death, can lead to cognitive abnormalities likely associated with diffuse axonal damage.9–11 These findings suggest that post-traumatic cognitive deficits may arise from multifocal axonal and myelin abnormalities.
While primary injury prevention is limited to immediate trauma management, the prolonged nature of secondary injury development offers a therapeutic window for intervention to mitigate brain damage and improve long-term outcomes. However, despite promising preclinical results, translating prospective TBI treatments into successful clinical trials has been challenging. Pathophysiological heterogeneity among patients with TBI, inadequate pharmacokinetic analysis for determining optimal dosages, and drug delivery outside the therapeutic window may contribute to the failure of clinical trials. 12
Intranasal drug delivery presents a promising approach for administering medications in TBI and other neurological disorders, offering several potential advantages. This review explores recent advancements in intranasal TBI therapy delivery technologies, beginning with an overview of TBI and available clinical treatments. We then highlight the effectiveness of these systems in animal models and discuss recent advancements in intranasal delivery systems for molecules and cells. Finally, we offer insights into utilizing intranasal delivery for efficient TBI therapies, providing forward-looking clinical perspectives on developing advancements.
TBI and current medication: An overview
TBI pathology and key biological events
TBI is defined as a disruption in brain function or other evidence of brain pathology, caused by an external physical force. 13 According to estimates, there are 50 million cases of TBI worldwide each year, indicating that over half of the global population will experience a TBI at some point in their lives. 14 The annual cost of TBI to the global economy is estimated at 400 billion US dollars, equivalent to 0.5% of the gross world product. 14 TBI is a heterogeneous condition that reflects multiple underlying macroscopic modes of injury (e.g. diffuse axonal injury (DAI), contusion, and extrinsic compression from mass lesion), as well as various mechanisms that can cause neuronal injury in differing degrees and clinical patterns (e.g. apoptosis, mitochondrial dysfunction, cortical spreading depression (CSD), and microvascular thrombosis; Figure 1). 15 In up to 60% of cases, severe TBI results in major physical, neurological, psychological, and social impairments. The fatality rate of severe TBI ranges between 30% and 40%. 16

Overview of the pathophysiology, clinical outcomes, and therapeutic strategies in traumatic brain injury (TBI).
Given the complex and progressive nature of TBI, effective treatment strategies must address both acute neuroprotection and long-term neurorestoration. Understanding the underlying biological events informs potential therapeutic interventions. One crucial approach focuses on promoting cell survival by preventing apoptosis and necrosis, which can be achieved using calcium channel blockers, anti-excitotoxic agents, and metabolic support therapies.17–19 These interventions aim to stabilize neuronal function, mitigate further cellular damage, and enhance the brain’s capacity to recover. Another key strategy involves modulating neuroinflammation, which plays a pivotal role in secondary injury progression. Anti-inflammatory cytokines, immunosuppressants, and nanoparticle-based targeted drug delivery methods can help control the immune response and minimize the extent of inflammation-induced neuronal damage.20,21 By regulating inflammatory cascades, these therapies may help preserve brain tissue and promote functional recovery.
Beyond immediate neuroprotection, neurorestorative therapies are being explored to enhance neurogenesis and angiogenesis, which are essential for brain repair. Stem cell-based interventions, growth factor administration, and neurotrophic support are promising strategies for fostering neural regeneration and synaptic plasticity.22–24 These approaches aim to stimulate the brain’s inherent repair mechanisms, facilitating the replacement of damaged neurons and improving functional recovery in patients with TBI.
Importantly, the success of these pharmacological and biological interventions depends heavily on efficient drug delivery to the central nervous system (CNS). In this context, intranasal administration has emerged as a promising, non-invasive route that bypasses the blood–brain barrier (BBB) and enhances drug bioavailability in the brain. Innovative delivery platforms, such as nanoparticles, hydrogels, and ligand-targeted systems, enable precise, sustained, and localized release of therapeutic compounds, thereby maximizing therapeutic efficacy while minimizing systemic exposure. These advances represent a significant shift toward optimizing drug efficacy and improving outcomes across both acute and chronic phases of TBI.
Current pharmacological therapies for TBI
Despite decades of promising preclinical research, no neuroprotective treatment has yet been successfully translated into routine clinical use for TBI. The translational gap reflects several challenges, including fundamental biological differences between human and rodent TBI models, limited funding for mechanistic human studies, the need for precise patient stratification, and a lack of robust pharmacokinetic data in humans. Nevertheless, several pharmacological agents previously investigated for TBI are discussed below.
One of the earliest large-scale pharmacological trials in TBI was the Corticosteroid Randomization After Significant Head Injury (CRASH) study, conducted between 1999 and 2004. 25 This international, multi-center randomized controlled trial (RCT) enrolled 10,008 patients with moderate-to-severe TBI to receive either high-dose methylprednisolone or placebo for 48 h. The primary endpoint—2-week mortality—was higher in the treatment group (21.1%) compared to placebo (17.9%), with 173 additional deaths in the corticosteroid arm at 6 months (1248 vs 1075). The study did not systematically assess known corticosteroid-related complications such as immunosuppression and hyperglycemia. 25
Following the failure of corticosteroids, progesterone, a potent neurosteroid synthesized in the CNS, was evaluated due to its preclinical efficacy in reducing neuronal loss, cerebral edema, and behavioral deficits after experimental TBI. However, subsequent large-scale, phase III, double-blind, placebo-controlled RCTs (SYNAPSE and PROTECT III, both published in 2014) failed to demonstrate improvements in mortality or functional outcomes, dampening early enthusiasm.26–28 These trials did not show clinical benefit, due to in part to the heterogeneity of the patient population.
Another candidate, erythropoietin (EPO), a glycoprotein hormone primarily produced in the kidneys in response to hypoxia, has demonstrated neuroprotective effects in preclinical models via anti-inflammatory, anti-apoptotic, and neurotrophic mechanisms. However, clinical data remain mixed. A 2017 meta-analysis of six RCTs involving 1041 patients with moderate-to-severe TBI found that EPO significantly reduced mortality but did not improve functional outcomes. Rates of complications, including deep vein thrombosis, were not significantly different between groups.29–32 These findings underscore the need for further well-designed trials to determine optimal dosing strategies and identify suitable patient populations.
Amantadine hydrochloride, which acts as both an indirect dopamine agonist and NMDA receptor antagonist, has also been evaluated. In a multi-center, double-blind RCT involving 184 patients with post-traumatic disorders of consciousness (4–16 weeks post-injury), amantadine accelerated functional recovery during a 4-week treatment phase, as measured by the Disability Rating Scale, without increasing serious adverse events.33,34 However, smaller studies have yielded inconsistent results. One single-center RCT with 40 patients with severe TBI showed no mortality or functional benefit at 6 months, while another trial in 119 patients with chronic TBI (>6 months post-injury) suggested potential cognitive impairment with amantadine during the first 28 days of use.35,36 These discrepancies highlight the complexity of evaluating amantadine’s efficacy across different stages of TBI recovery.
In contrast, tranexamic acid (TXA), a synthetic antifibrinolytic agent derived from lysine, functions by inhibiting plasminogen activation and reducing active bleeding. Approximately 30% of patients with TBI exhibit coagulopathy, which contributes to cerebral edema and peri-hemorrhagic damage.37–40 In the CRASH-2 trial, involving 20,211 patients with trauma (excluding isolated intracranial injuries), early TXA administration reduced mortality, particularly when given promptly. These findings led to its inclusion in the WHO List of Essential Medicines.37,41 A nested analysis in CRASH-2 indicated a non-significant trend toward reduced hemorrhage progression and mortality among patients with TBI with abnormal CT findings. 42 These results underpin the ongoing CRASH-3 trial, which aims to evaluate the efficacy of TXA in patients with traumatic intracranial hemorrhage. 37
Citicoline, a cholinergic agent believed to enhance ATP production and stabilize cell membranes by supporting ATP-dependent ion pumps, was explored for its potential to attenuate secondary brain injury. However, the phase III COBRIT trial, a multi-center, double-blind RCT, failed to demonstrate any significant improvement in functional or cognitive outcomes at 90 days in patients with moderate-to-severe or complicated mild TBI.43,44 As such, citicoline is not currently recommended as a standard treatment.
Finally, recombinant interleukin-1 receptor antagonist (rIL-1ra), which blocks IL-1 receptor-mediated neuroinflammation, has shown neuroprotective potential across several neurological disorders. A phase II, single-center RCT demonstrated its safety and ability to modulate acute neuroinflammatory responses in TBI.45–47 Ongoing dose-ranging trials aim to optimize the timing and dosage for future clinical application. 48
Efficacy models to evaluate TBI therapeutics
The development of effective therapeutics for TBI relies heavily on model systems that accurately recapitulate the complex pathophysiology of injuries. While both in vivo and in vitro models are available across various species, in vitro systems offer superior experimental control, reproducibility, and ease of manipulation, making them the preferred platform for investigating cellular mechanisms and evaluating candidate treatments. 49 In addition to their mechanistic value, robust in vitro TBI models also play a crucial role in translational research by enabling early-stage screening of candidate therapeutics and toxicity profiles before proceeding to animal studies. This approach aligns with the 3Rs (Replacement, Reduction, and Refinement) in animal research, as it helps to minimize unnecessary animal use by filtering out ineffective or harmful agents early in the pipeline. Thus, in vitro models serve not only as mechanistic tools but also as ethically and scientifically advantageous platforms in the preclinical development of TBI therapies. Among in vitro models, mechanical injury paradigms, particularly those involving cellular stretch, are widely used to simulate the physical forces experienced during TBI. 50 One of the earliest and most influential models was developed by Ellis et al., who introduced the cell-injury controller (CIC) to deliver precise stretch-induced injury to cultured cells. 51 Since then, stretch-based injury has been extensively applied to various cell types, including neurons, astrocytes, and endothelial cells, to study cellular damage, inflammation, and repair pathways.52–54 Notably, the use of stretch injury cultured mouse brain endothelial cells has been instrumental in elucidating cellular and molecular mechanisms underlying TBI-induced BBB dysfunction. 55 This approach is based on the hypothesis that primary TBI damage arises from mechanical strain and strain rate experienced by the CNS during traumatic insult.
In addition to mechanical stretch models, oxygen-glucose deprivation (OGD) is commonly employed to mimic ischemic conditions, which frequently accompany TBI due to impaired cerebral perfusion. OGD replicates the metabolic crisis that occurs when blood flow to the brain is disrupted, resulting in reduced oxygen and glucose availability. Given that ischemia is a secondary yet prevalent component of TBI, combining stretch injury with OGD can more faithfully recapitulate the multifaceted pathophysiology observed in clinical settings. The choice of cell type is another critical factor in developing a physiologically relevant in vitro TBI model. Although astrocytes, neurons, and endothelial cells derived from murine, bovine, and human sources have all been utilized, brain microvascular endothelial cells are particularly valuable for studying alterations in the BBB following trauma.51,53,54
Disruption of the BBB is a hallmark of TBI, leading to increased cerebrovascular permeability, vasogenic brain edema, and neuroinflammation. These secondary events can significantly influence patient outcomes. Therefore, in vitro models incorporating brain endothelial cells provide a powerful platform to study BBB dysfunction and to screen therapeutic agents targeting both primary and secondary injury mechanisms.56,57
Given the wide clinical variability in TBI presentation, numerous animal models have been developed to replicate different aspects of human brain injury. Rodents remain the most commonly used species due to their small size, affordability, and the availability of standardized outcome measures. Although larger animals offer anatomical and physiological similarities to humans, rodent models provide logistical advantages and are suitable for high-throughput experimental studies. Importantly, while early TBI models primarily addressed the biomechanical mechanisms of injury, recent models have shifted toward understanding the complex biochemical and molecular cascades activated following traumatic brain insults.58–60 Among the most widely utilized modern TBI models are the weight drop injury (WDI), 61 fluid percussion injury (FPI), 62 controlled cortical impact (CCI) injury,63,64 and blast-like injury.65,66 Each provides unique advantages and limitations in replicating human TBI (as summarized in Figure 2).
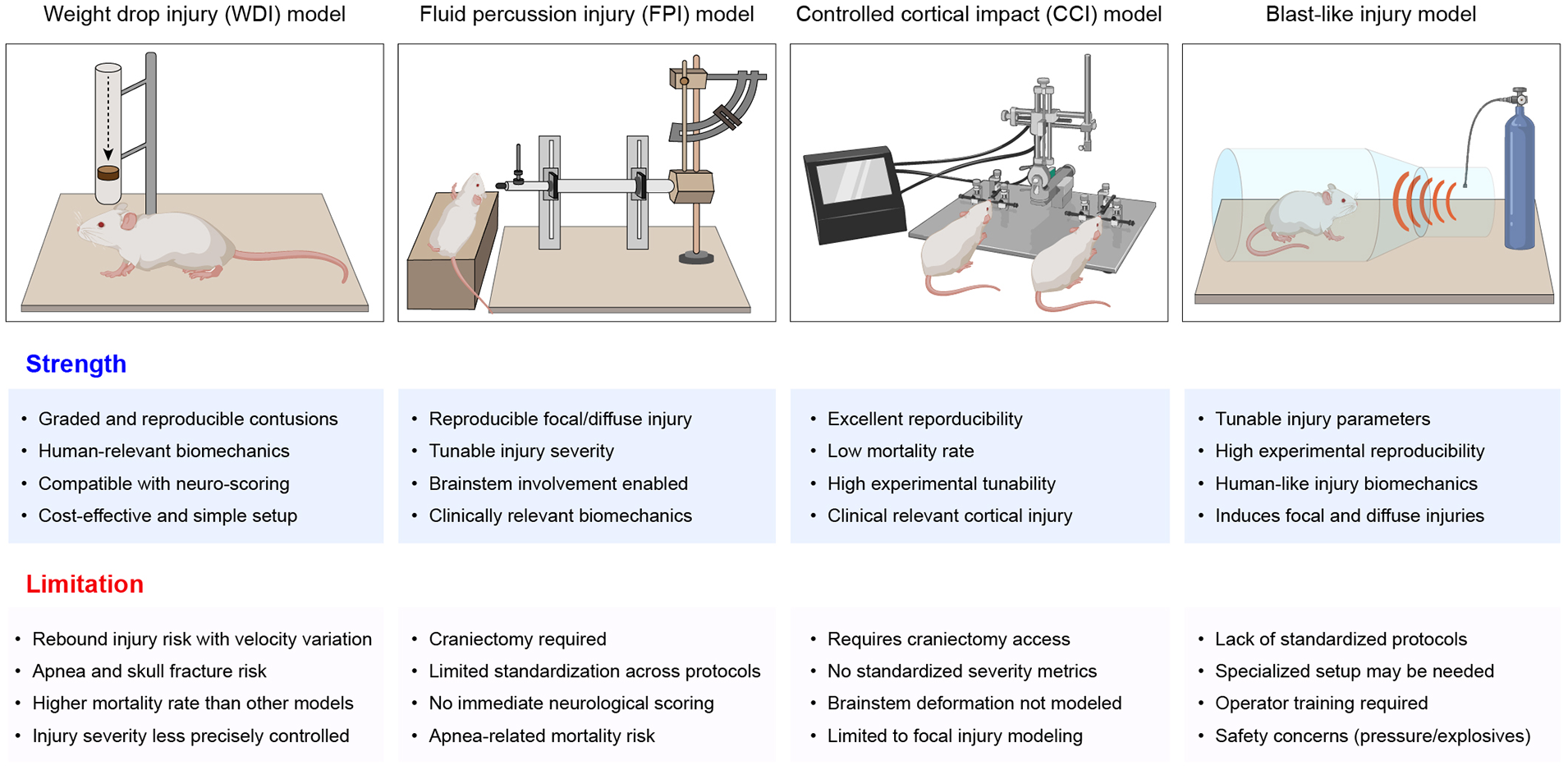
Rodent TBI models and their strengths and limitations.
The WDI model is valued for its simplicity, reproducibility, and cost-effectiveness, making it accessible to a wide range of laboratories. It involves dropping a calibrated weight from a defined height onto the animal’s skull, thereby simulating impact injuries such as those resulting from falls or blunt trauma. 67 However, inconsistencies in the angle and distribution of force can lead to variable injury patterns, and this model offers limited control over key parameters such as impact depth or velocity, restricting its use in studying region-specific injuries. 68
In contrast, the FPI model allows for more controlled injury induction, using a burst of fluid pressure applied to the intact dura to simulate TBI. It effectively reproduces several pathological features seen in human injury, including subarachnoid hemorrhage, intraparenchymal bleeding, and necrosis. 69 Importantly, injury severity can be finely tuned, allowing researchers to correlate trauma intensity with physiological and behavioral outcomes. However, this method requires specialized equipment and considerable technical expertise, and its ability to mimic chronic sequelae is still under investigation. Additionally, anatomical differences between rodent and human brains may limit translational validity.67,70
The CCI model is particularly valued for its precision and reproducibility. Utilizing a pneumatic or electromagnetic piston, researchers can control the depth, velocity, and dwell time of cortical impact, generating focal injuries that closely resemble human brain contusions, axonal injury, and neuronal loss.68,71 This versatility enables modeling of a range of TBI severities in various species. Nevertheless, the CCI setup is technically demanding, costly, and may not be accessible in all laboratories. Furthermore, while excellent for focal injuries, it may fall short of replicating the diffuse axonal damage common in human cases.68,72
The blast-like injury model has emerged in response to increasing military-related TBIs, simulating injuries from explosive blasts. It captures the complex overpressure dynamics and rapid acceleration-deceleration forces involved in such scenarios.65,66 This model is particularly useful for studying cognitive, behavioral, and emotional consequences of blast exposure. However, it poses significant technical and safety challenges, often requiring sophisticated instrumentation and rigorous protocols to ensure reproducibility. Injury severity can vary due to the intricate physics of blast waves, further complicating experimental outcomes. 73
TBI frequently impairs motor and cognitive functions due to disruptions in the complex neural circuitry connecting the cortex, spinal cord, and peripheral musculature. 74 Accordingly, sensorimotor and behavioral tests are commonly used to assess functional outcomes in animal models. These include the cylinder test, rotarod, grip strength assessment, staircase test, and skilled forelimb reaching, providing valuable insight into post-injury motor coordination and strength. 74 For closed-head injuries in rodents, the neurological severity score (NSS) and its modified version for unilateral injuries are widely used to evaluate motor and behavioral deficits.75–78 Since cognitive dysfunction is a hallmark of human TBI, numerous models have also demonstrated post-injury cognitive deficits, particularly after CCI, FPI, blast, and impact-acceleration injuries.10,76,79–83 Common cognitive assessment tools include the Morris water maze, object recognition tests, fear-conditioning paradigms, and memory tasks. 74 Beyond cognitive and motor domains, TBI is often accompanied by psychological and emotional disturbances such as anxiety and mood dysregulation. Consequently, advanced behavioral paradigms such as the elevated plus maze, open field tests, and exploratory behavior assays have been adopted to mirror the neuropsychiatric symptoms observed in clinical TBI populations.2,84–87
Despite their value, current animal models also have limitations. Key structural and functional differences between rodents and humans, such as brain geometry, craniospinal angle, cortical gyrification, and the white-to-gray matter ratio, introduce challenges in translating findings.88,89 Furthermore, even within rodent species, there are notable inter-strain differences in histological responses and behavioral outcomes following TBI.90–93 These biological variations underscore the need for caution in extrapolating preclinical findings and highlight the importance of complementary clinical and translational research.
Therapeutic approaches for TBI recovery
TBI initiates a complex cascade of pathological processes, beginning with the primary mechanical insult and progressing to secondary injury mechanisms such as oxidative stress, inflammation, excitotoxicity, vascular dysfunction, and apoptotic cell death. 94 Consequently, effective therapeutic strategies must be designed to intervene at various stages of injury progression, ideally mitigating immediate damage and delayed cellular and molecular deterioration. In this context, pharmacological and biological interventions have been developed to target specific components of TBI pathophysiology (Figure 3).95,96

Overview of therapeutic strategies for TBI. TBI induces neuronal loss, inflammation, vascular dysfunction, and impaired regeneration. Four major approaches—neuroprotective, anti-inflammatory, neurovascular, and neuroregenerative—target these pathological processes using distinct mechanisms and agents.
Neuroprotective approaches
Despite extensive research, many clinical trials investigating neuroprotective therapies in TBI have yielded disappointing results, prompting renewed scrutiny of their therapeutic potential. A key pathological mechanism following TBI involves the dysregulation of intracellular calcium homeostasis. Elevated levels of intracellular calcium contribute significantly to a cascade of deleterious events, including mitochondrial dysfunction, activation of proteases, and eventual cell death. Thus, modulation of calcium influx has emerged as a promising neuroprotective strategy.17–19
Among the pharmacological agents explored, L-type and N-type calcium channel blockers have shown potential in mitigating TBI-induced neuronal injury by limiting calcium accumulation within cells. For instance, nimodipine, an L-type calcium channel blocker, has demonstrated improved outcomes in patients with spontaneous subarachnoid hemorrhage. 17 However, a systematic review later challenged these findings, revealing no significant difference in mortality or morbidity between nimodipine-treated patients with TBI and those receiving a placebo. 18 In contrast, ziconotide (SNX-111), an N-type calcium channel inhibitor, has been evaluated for its efficacy in improving mitochondrial function when administered within a therapeutic window of 15 min to 6 h post-TBI. 19 While promising neuroprotective effects were observed, its use was also associated with notable side effects, including hypotension, which could limit its clinical applicability.
Amantadine, a dopamine agonist originally developed for Parkinson’s disease, has emerged as a therapeutic candidate for TBI. Functioning as an N-methyl-D-aspartate (NMDA) receptor antagonist, amantadine is capable of crossing into the frontal lobes, where it may counteract glutamate-mediated excitotoxicity in the acute phase of TBI. Several studies have demonstrated that administration of amantadine at doses ranging from 100 to 400 mg/day within 12 weeks post-injury can enhance arousal and cognitive function in patients with TBI.96,97
Another promising agent is EPO. Despite its high molecular weight, which exceeds the typical threshold for BBB permeability, exogenous EPO has been detected in the brain parenchyma, suggesting a potential for neuroprotective activity following brain injury. 98 EPO has been shown to possess anti-inflammatory, anti-excitotoxic, antioxidant, and antiedematous properties in TBI models.99–102 Notably, expression of the EPO receptor (EpoR) is significantly upregulated in neurons, glial cells, and endothelial cells after TBI. 103 Knockout studies in mice have revealed that the absence of EpoR leads to increased apoptosis and a reduced population of neural progenitor cells (NPCs), indicating the receptor’s importance in neuronal survival. 104
It has been demonstrated that the EPO/EpoR signal pathway contributes to neuroprotection in pathological settings.102,105 Following TBI, there is a marked upregulation of EPO receptor expression in neurons, glia, and endothelial cells, indicating a potential role in the endogenous repair response. 19 Upon binding to EpoR, EPO activates key intracellular cascades, notably the Janus kinase 2 (JAK-2)/nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) and phosphoinositide 3-kinase (PI3K) signaling pathways, which are implicated in cell survival and anti-inflammatory responses.19,106 In particular, phosphorylation of JAK-2 leads to the homodimerization of signal transducer and activator of transcription-5 (STAT-5), a process associated with anti-apoptotic and neurotrophic effects. This, in turn, further activates downstream signaling through the PI3K/AKT and Ras/mitogen-activated protein kinase (MAPK) pathways, enhancing cell survival and neurogenesis.30,107,108 Despite these promising molecular mechanisms, clinical translation has been challenging. A recent double-blind randomized controlled trial found that EPO administration did not significantly affect the proportion of patients experiencing severe neurological dysfunction, and its influence on mortality outcomes in moderate to severe TBI remains inconclusive. 106
Glial cells are the primary source of the calcium-binding protein S100B, which becomes detectable in the serum following TBI, particularly when BBB integrity is compromised. S100B exerts a dose-dependent dual effect on neurons: at low concentrations, it acts as a neurotrophic factor, supporting neuronal survival and repair. In contrast, at high levels, it promotes neuroinflammation and can impair neural viability, thereby exacerbating brain injury. 19
Mesenchymal stem cells (MSCs) have also emerged as promising candidates for neuroprotective therapy. Although the precise mechanisms by which MSC transplantation facilitates recovery after TBI are not yet fully elucidated, current evidence suggests that neurorestoration, rather than direct neuroreplacement, is the principal mechanism. This is supported by findings that MSCs secrete a variety of neurotrophic factors, such as brain-derived neurotrophic factor (BDNF), vascular endothelial growth factor (VEGF), and fibroblast growth factor 2 (FGF-2). 109 These factors play critical roles in synaptogenesis, angiogenesis, and neurogenesis, collectively enhancing functional recovery following TBI.19,110
Anti-inflammatory approaches
Following TBI, secondary injury is characterized by a robust neuroinflammatory response, which is driven by the activation of microglia and astrocytes, infiltration of peripheral immune cells, and the release of pro-inflammatory cytokines, including IL-1β, tumor necrosis factor-alpha (TNF-α), and IL-6. 111 Collectively, these events contribute to BBB disruption, exacerbation of neuronal injury, and the development of chronic neurological impairments. Accordingly, anti-inflammatory strategies are designed to dampen this cascade, ideally during the acute to subacute phases of TBI.
Among such strategies, melatonin, a neurohormone with potent antioxidant and anti-inflammatory properties, has shown promise in preclinical models. It exerts its effects by reducing reactive oxygen species (ROS) and inhibiting NF-κB activation, thereby attenuating neuroinflammation.112,113 Animal studies have demonstrated that melatonin administration results in decreased activation of microglia and astrocytes, reduced cerebral edema, and enhanced neurological recovery. Additionally, melatonin has been shown to alleviate cognitive deficits induced by repeated mild TBI by inhibiting astrocyte reactivation. However, translation into clinical practice has been challenging, as clinical trials in adults have yielded mixed results regarding its efficacy.
Another notable approach involves using IL-1Ra, such as anakinra, to block IL-1β-mediated inflammatory signaling. In preclinical models, IL-1Ra treatment has been associated with reduced microglial activation, preservation of white matter integrity, and improved functional recovery. 114 Early-phase clinical trials suggest that IL-1Ra is safe and potentially effective, though larger, well-powered studies are required to confirm its therapeutic benefit in patients with TBI.
Anti-TNF agents, such as etanercept, have also demonstrated promising effects in reducing glial activation and promoting neurogenesis in animal models. In rodent models of TBI, etanercept treatment has been associated with significant improvements in cognitive and motor functions, as well as a reduction in pro-inflammatory markers in the brain. 115 While preliminary human data suggest potential benefits for post-TBI recovery, the evidence remains limited, with controlled clinical studies still being relatively scarce. 116
Neurovascular approaches
TBI disrupts the neurovascular unit, impairing blood flow, damaging the BBB, and limiting the delivery of oxygen and nutrients to injured tissue. These changes contribute to secondary injury cascades and hinder recovery. Therefore, therapeutic strategies that restore vascular function and support angiogenesis are essential for neuroprotection and regeneration.
Intriguingly, statins, primarily known for their ability to reduce cholesterol by inhibiting its production, have shown promising effects in promoting angiogenesis, neurogenesis, and synaptogenesis, as well as improving functional recovery after TBI in various animal studies.117–119 Importantly, these positive effects are not directly related to their cholesterol-lowering properties. Instead, the beneficial actions of simvastatin may be mediated through the activation of key signaling pathways, including nuclear factor-κB, Forkhead transcription factor 1, and Akt. These pathways contribute to the prevention of caspase-3 activation and apoptotic cell death, ultimately supporting the restoration of neuronal function following TBI. 120 In rat models of TBI, simvastatin treatment resulted in sustained functional improvements for up to 3 months post-injury. 121
Moreover, statins may enhance the effectiveness of stem cell transplantation in TBI recovery. For example, atorvastatin has been shown to increase MSC access and survival within the injured brain, leading to greater functional recovery when combined with MSC therapy, compared to either treatment alone. 122 Given their widespread use, favorable safety profile, and positive preclinical and clinical data, further clinical trials are necessary to fully evaluate the neuroprotective and neurorestorative properties of statins in the context of TBI. 123
Neuroregenerative approaches
Neuroregeneration refers to the process of replacing or repairing neurons, oligodendrocytes, and other neural cells lost or damaged following TBI. While endogenous neurogenesis occurs to a limited extent in adult mammals, primarily in the subventricular zone (SVZ) and the hippocampal dentate gyrus, this process is often insufficient to compensate for widespread injury. To address this gap, therapeutic strategies have been developed to enhance the regenerative capacity of the brain through exogenous stimulation and transplantation approaches.
One such strategy involves the 43-amino acid polypeptide thymosin beta 4 (Tβ4), which was initially discovered in thymus tissue and later identified in all mammals. The primary intracellular role of Tβ4 is the sequestration of G-actin, an essential component for organogenesis and cell motility. 124 Beyond this, Tβ4 has shown promise in enhancing tissue healing in various organs, including the skin, cornea, and heart, while simultaneously preventing inflammation and apoptosis. 125 Additionally, Tβ4 acts as a paracrine factor, critical for epicardial progenitor cells in promoting angiogenesis after ischemic injury. 126 Tβ4 supports several key cellular functions, including motility, axonal pathfinding, neurite outgrowth, proliferation, and neuronal survival.125,127 For example, Tβ4 has demonstrated positive outcomes in rats with embolic stroke, 128 mice with experimental autoimmune encephalomyelitis, 129 and TBI models. In one study, delayed Tβ4 treatment significantly improved sensorimotor recovery, spatial learning, angiogenesis, and neurogenesis while reducing hippocampal cell loss in rats with TBI. 130 Compared to saline treatment, delayed Tβ4 administration showed enhanced sensorimotor function, neurogenesis in the injured cortex and hippocampus, and increased oligodendrogenesis in the CA3 region, ultimately improving histological and functional outcomes. These findings suggest that Tβ4 holds significant therapeutic promise for improving recovery in patients with TBI.
Endogenous neurogenesis can also be pharmacologically enhanced through the administration of specific growth factors. Molecules such as BDNF, FGF-2, and insulin-like growth factor 1 (IGF-1) play central roles in promoting neuronal survival, differentiation, and plasticity. 131 In experimental TBI models, these agents have been shown to increase the proliferation of neural progenitor cells, enhance synaptogenesis, and contribute to cognitive recovery.
Neural stem/progenitor cells (NS/PCs) reside in neurogenic niches such as the hippocampus, subventricular zone (SVZ), and the ependymal lining of the mammalian brain. 132 Recent studies have demonstrated that stimulating the growth and differentiation of these endogenous NS/PCs can stabilize the cortical microenvironment and improve functional recovery after TBI.133,134 Moreover, exogenous NSC transplantation has been found to improve functional outcomes, increase hippocampal neurogenesis, and provide neuroprotective effects. 135 Emerging research has also shed light on the interplay between NSCs/NPCs and the immune system. Transplanted NS/PCs interact with resident and peripheral immune cells, promoting the functional integration of the grafted cells and enhancing endogenous regenerative responses. 136 This cross-talk underscores the therapeutic potential of NSC-based strategies for restoring neural integrity and function following TBI.
Oligodendrocyte regeneration is essential for restoring myelin integrity and white matter connectivity following TBI. 137 Multiple studies have demonstrated that NS/PCs and oligodendrocyte progenitor cells (OPCs) can differentiate into mature oligodendrocytes in vivo, contributing to remyelination and the restoration of axonal conduction. 138 Therapies such as Tβ4 and MSC-derived exosomes appear to potentiate this regenerative process by modulating the injury microenvironment, promoting oligodendrogenesis, and reducing inflammation.
Embryonic stem cells (ESCs) have also been explored as a potential therapeutic platform in TBI models.139,140 While preclinical studies have indicated neuroprotective and regenerative effects, the long-term safety, tumorigenicity risk, and ethical concerns surrounding ESC use necessitate further investigation before clinical translation.
In summary, neuroregenerative strategies offer significant promise for long-term functional recovery by reestablishing neuronal circuitry, promoting axonal integrity, and facilitating white matter repair. Future research should prioritize enhancing the survival, targeted differentiation, and functional integration of transplanted or reprogramed cells. Additionally, optimizing the timing, delivery route, and microenvironmental cues will be critical to maximizing therapeutic efficacy in patients with TBI.
Intranasal delivery: Alternative route to bypass the BBB
Intranasal administration offers a promising route for delivering therapeutic agents directly to the CNS by bypassing the BBB. This method enables substances to enter the brain through olfactory and trigeminal neural pathways (as illustrated in Figure 4), making it an attractive alternative to systemic or intracerebroventricular (ICV) routes.

Mechanism of intranasal delivery. Schematic illustration of nose-to-brain transport following intranasal administration of biomolecules or nanoparticles. Upon delivery into the nasal cavity as a spray or drop, therapeutic agents interact with the olfactory mucosa and reach the brain through paracellular, transcellular, and intracellular (axonal) pathways. The paracellular pathway enables passive diffusion of small molecules or nanoparticles through tight junctions between epithelial cells. The transcellular pathway involves endocytic uptake and vesicular transport across epithelial cells, allowing receptor-mediated or adsorptive delivery of macromolecules. In the intracellular pathway, substances are internalized by olfactory sensory neurons and transported via axonal projections into the olfactory bulb and brainstem. These pathways enable direct access to the central nervous system while bypassing the blood-brain barrier, supporting efficient delivery of therapeutics to target brain regions.
Intranasal delivery has several advantages. It enables targeted CNS access while minimizing systemic exposure and potential side effects, as only a small fraction of the administered dose typically enters the bloodstream. Additionally, intranasal administration can avoid enzymatic degradation by serum proteases and is relatively non-invasive, making it particularly suited for repeated or chronic therapeutic regimens. Compared to intravenous (IV) or peripheral routes, intranasal delivery often achieves higher CNS bioavailability without the need for surgical intervention. While many treatments have shown promising preclinical efficacy via ICV or systemic routes, intranasal administration combines the key advantages of non-invasiveness, selectivity for the CNS, and reduced systemic toxicity. 141 The cribriform plate serves as the primary anatomical target for CNS-directed intranasal delivery.
However, variations in nasal cavity anatomy between species influence administration protocols. In rodents, two common methods are employed: The first and most popular administration method involves using a pipette tip to place the substrate (⩽20 μL for mice and up to 50 μL for rats) on the edge of the nares, which is subsequently inhaled.142,143 To optimize CNS exposure without exceeding safe volume limits, dosing is typically repeated alternately between nostrils. Modifications to this technique, such as gently sealing the mouth and contralateral nostril, can further improve deep nasal penetration and delivery efficiency. 144 In non-human primates, larger volumes (0.1–0.5 mL per nostril) can be delivered using flexible tubing positioned approximately 25–30 mm from the cribriform plate.145,146 This method provides more consistent and localized deposition of therapeutics near olfactory structures. An alternative delivery strategy involves misting the target substrate in front of the animal’s nostril with a controlled, pulsed, pressurized atomizer—a technique akin to that employed in human clinical trials.147,148 Nebulizers, also known as atomizers, are frequently used for intranasal drug delivery to humans. These devices generate pressurized air that transforms liquid formulations into a fine aerosol spray, which can enhance drug deposition in the narrow, upper regions of the nasal cavity. Compared to conventional spray pumps, atomizers improve the penetration and dispersion of drugs in regions more closely connected to the olfactory and trigeminal pathways. To optimize absorption, dosing is typically alternated between nostrils, allowing for consistent drug uptake while avoiding mucosal saturation.32,149 However, aerosolization introduces shear stress that may affect the structural integrity of sensitive biologics. 150 While small-molecule drugs generally remain stable during nebulization, macromolecules such as peptides and proteins may undergo partial denaturation or aggregation under high-pressure atomization. To address this, formulations often incorporate stabilizing excipients (e.g. sugars, surfactants), and device designs have been optimized to minimize mechanical stress. 151 For cell-based products, direct aerosolization is not feasible due to the high risk of mechanical damage; therefore, therapeutic strategies typically rely on delivering stem cell-derived secretomes or extracellular vesicles encapsulated in protective carriers such as nanoparticles or hydrogels to maintain stability during administration
After intranasal administration, drugs are believed to primarily enter the brain via retrograde axonal transport along olfactory and trigeminal neurons, and direct passage into the CSF through the cribriform plate. 152 Early studies highlighted the olfactory bulb as the primary site of deposition. However, more recent investigations have demonstrated widespread CNS distribution, with certain compounds preferentially accumulating in regions such as the hippocampus or hypothalamus, depending on their physicochemical and pharmacokinetic properties.153,154 For example, insulin, a water-soluble peptide that is rapidly degraded in circulation, shows preferential CNS uptake with minimal systemic exposure when administered intranasally. In contrast, progesterone, a small lipophilic molecule, exhibits more diffuse systemic distribution, with tissue-specific concentrations influenced by local metabolism, sequestration, and tissue affinity.
Given the promising potential of intranasal delivery to target the brain efficiently and non-invasively, our analysis focuses on therapeutic compounds delivered intranasally for the treatment of TBI. These agents fall into six major categories: immunosuppressants, cytokines, growth factors, vitamins and metabolites, exosomes, and stem cell therapy. In the following sections, we discuss both preclinical studies and clinical case reports, with a focus on the behavioral, biochemical, and histological outcomes associated with these intranasal therapies for TBI.
Candidate therapeutic molecules for intranasal delivery
Growth factors and cytokines
In most preclinical studies cited in this section, intranasal administration was performed by delivering a small volume of the therapeutic agent (typically 10–20 µL per nostril for mice or 20–50 µL for rats) using a micropipette or syringe while the animal was placed in a supine position. This method ensures direct deposition onto the nasal mucosa and facilitates uptake through the olfactory and trigeminal pathways. In contrast, in studies involving human subjects, intranasal administration was performed using spray-based devices (e.g. nasal sprays or atomizers), which enable non-invasive and controlled delivery to the upper nasal regions. IGF-1 plays a central role in supporting neuronal survival and reducing injury in the hippocampus following cerebral ischemia (Figure 5(a)). Upon intranasal administration, IGF-1 becomes detectable in the CNS within approximately 20 min, with its highest concentration observed in the olfactory bulb. 144 Several animal studies have demonstrated that intranasal IGF-1 delivery attenuates neuroinflammation and enhances motor function after stroke.144,155 Given its well-established neuroprotective and regenerative effects in TBI animal models, the intranasal administration of IGF-1 represents a promising, non-invasive strategy for treating TBI-related pathology.156,157

Mechanisms of therapeutic molecules and candidates in TBI. Schematic overview of representative molecular and cellular mechanisms underlying therapeutic strategies for TBI. (a) IGF-1 exerts neuroprotective effects by engaging IGF1R and activating multiple signaling pathways. The RAS–ERK–Akt axis suppresses neuronal apoptosis, while the PI3K–Akt–HO-1 pathway attenuates oxidative stress and inflammation. Concurrently, PI3K–Akt–GLUT signaling enhances glucose uptake and metabolic homeostasis. IGF1R also modulates ion channel activity and neurotransmission through ionic current regulation. (b) BDNF binds to TrkB and initiates downstream cascades that support neuronal growth and plasticity. The PI3K–Akt–mTOR pathway promotes dendritic development and cell survival, whereas the Ras–MEK–ERK and PLC–CaMKII pathways converge on CREB to induce transcriptional programs involved in synaptic remodeling, differentiation, neurotransmitter regulation, and myelination. (c) Exosomes and extracellular vesicles (EVs) deliver bioactive cargos such as miRNAs, lncRNAs, growth factors, and cytokines to injured neural tissue. These components promote neuroregeneration, suppress neuroinflammation (e.g. via IL-4, IL-10), reduce apoptosis through autophagy regulators (LC3B, Beclin-1), and enhance angiogenesis via VEGF-mediated signaling. (d) Stem cells modulate the post-injury brain microenvironment primarily through paracrine mechanisms. They release EVs, trophic factors, and cytokines that collectively support vascular, neuronal, and immune recovery. EV-associated IGF-1, VEGF, ANG-1, and HGF promote angiogenesis; neurotrophic factors (BDNF, NGF, GDNF) facilitate axon growth and myelination; and immunomodulatory cytokines (IL-4, IL-10, IL-13) contribute to neuroprotection, neurite outgrowth, and neurogenesis.
Recent work has investigated the CNS impact of insulin administered through various delivery routes. 158 Initially, insulin was shown to dampen peripheral inflammation by reducing TNF-α and IL-1β. 159 More recent findings indicate that insulin also modifies CNS inflammation.160,161 Specifically, intranasal insulin can modulate inflammatory signaling in the hippocampus of Alzheimer’s disease mouse models, influencing processes such as T cell receptor signaling, cytokine–cytokine receptor interactions, and cell adhesion pathways. 162 In one in vivo study, intranasal insulin therapy reduced lesion volume and enhanced glucose uptake and memory function in the ipsilateral hippocampus. 163 Using a pipette-based approach, intranasal insulin effectively reached the cortex, hippocampus, brainstem, and cerebellum following a CCI. Notably, it significantly reduced microglial activation, as indicated by diminished Iba1 staining in the CA1 region of the hippocampus. This evidence highlights the promise of intranasal insulin for treating TBI-related metabolic dysfunctions, mitigating microglial-driven inflammation, and improving memory outcomes.
Decreases in blood and brain levels of BDNF have been associated with inflammatory injuries, including stroke and TBI, suggesting a correlation with injury severity in animal and human studies.164,165 Jiang et al. reported that intranasal delivery of BDNF, although not altering lesion volume post-stroke, shifted the neuroinflammatory landscape and protected neurons. 166 Such findings indicate that BDNF may exert a protective effect partly through immunomodulatory pathways, supporting the idea that intranasal administration of BDNF could be beneficial in brain injury management (Figure 5(b)).
Nerve growth factor (NGF) is another neurotrophin that fosters neuronal growth, differentiation, survival, and repair after neuronal insult. 167 In rat TBI models, intranasal NGF notably curbed edema and reduced cell death. 168 Similarly, a case study illustrated that intranasal NGF administration improved neurological function in a 4-year-old boy with TBI. 169 This improvement was accompanied by an increase in both doublecortin protein (a neurogenesis marker) and NGF levels in the CSF.
Toll-like receptors (TLRs) can be activated by CNS traumas such as stroke and TBI, subsequently inducing pro-inflammatory signaling cascades, such as NF-κB-mediated cytokine production. In a rat stroke model, intranasal delivery of a TLR/NF-κB pathway inhibitor effectively reduced infarct volume and improved neurological function. 170 Transforming growth factor beta (TGF-β) is an anti-inflammatory cytokine capable of mitigating NF-κB signaling.20,21 Administration of TGF-β has been shown to promote neurogenesis and aid in functional recovery in a mouse model prone to stroke. 171 TLR activation also elevates granulocyte-colony stimulating factor (G-CSF), a cytokine that stimulates the innate immune response and cell division. 172 G-CSF is regarded as neuroprotective and is significantly increased in various inflammatory conditions and diseases. 173 Interestingly, its plasma levels peak around 12 h following TBI, suggesting potential diagnostic utility. 174 These observations support the idea that intranasal administration of cytokines modulating immunity and cell proliferation may offer an effective approach to TBI therapy.
Endogenous cytokines can impact both innate and adaptive immune processes. The anti-inflammatory cytokine interleukin 13 (IL-13) is secreted by T helper 2 (Th2) cells. 175 IL-13 can stimulate neurons and endothelial cells through IL-13 receptor α1-dependent signaling.176,177 Studies have found that Th2-derived IL-13 levels are increased in the CSF of patients with relapsing multiple sclerosis, implying a major role for IL-13 in immune-cell recruitment under inflammatory stress.178,179 Growth factors such as BDNF are linked to IL-13 and may have their expression upregulated by it. 180 As noted earlier, neurotrophic factors like BDNF can be delivered intranasally in models of CNS disease; however, delivering cytokines that boost endogenous neurotrophic factor production in the CNS is another viable strategy. Indeed, recent work demonstrated that intranasal IL-13 reduces neuroinflammation and improves recovery in a rat TBI model. 181 Therefore, intranasal administration of anti-inflammatory cytokines might enhance neurotrophic factor activity and alleviate various CNS disorders.
Another key anti-inflammatory cytokine, interleukin 10 (IL-10), is secreted in response to brain injury to moderate inflammatory cascades that might otherwise exacerbate tissue damage. Although preclinical TBI models suggest a neuroprotective role for IL-10, clinical trials have shown divergent outcomes. One study in Sprague-Dawley rats found that IL-10 mRNA levels in the brain rise immediately after TBI, whereas IL-10 protein remains stable until a sharp increase occurs 2 h post-injury. 182 These data indicate that local IL-10 synthesis, rather than leakage of IL-10 from systemic circulation, is primarily responsible for the early rise in IL-10. Research on IL-10−/− mice confirms the benefits of IL-10 following TBI, though the therapeutic outcomes of exogenous IL-10 administration can vary based on experimental protocols and delivery routes. After CCI-induced TBI, IL-10−/− mice displayed elevated BBB disruption, enhanced apoptosis, greater lesion size, more pronounced edema and inflammation, and worsened motor and cognitive outcomes at 4 weeks. 183 Subcutaneous and intracerebroventricular IL-10 injections have yielded positive effects in TBI animal models,183,184 and intranasal delivery may similarly facilitate direct CNS access without crossing the BBB.
TNF-α is a pro-inflammatory cytokine that signals through TNFR1 or TNFR2. It exists in two bioactive forms: a soluble form generated by proteolytic cleavage of its membrane-bound counterpart and a membrane-bound form involved in cell–cell interactions. 185 Inhibiting TNF-α has shown therapeutic benefits for TBI in animal studies,186,187 and there are several FDA-approved biologics targeting TNF-α include recombinant fusion proteins (entanercept) and monoclonal antibodies (adalimumab, certolizumab, golimumab, and infliximab). 188 One example investigated intranasal administration of a TNF-α-inhibitory single-chain variable fragment (ESBA105). Even at a tenfold higher intranasal dose, systemic exposure remained about 33-fold lower than that of systemic injection. Incorporating a penetration-enhancing peptide further improved ESBA105 transport to the cerebrum and olfactory bulb without raising systemic levels. 189
Extracellular vesicles (EVs)
Extracellular vesicles (EVs), including exosomes measuring approximately 50–200 nm, are formed from endosomes and play pivotal roles in regulating immune responses, among other functions.190,191 Cell-derived exosomes have recently emerged as an innovative treatment option for post-TBI neural injury. 192 Intranasal delivery of exosomes derived from birth-associated MSCs attenuated neuronal death and promoted neurological healing in preclinical studies.193,194 Moss et al. found that MALAT1, a long noncoding RNA abundant in MSC-derived exosomes, suppressed microglial activation through TrkC signaling. Furthermore, in a spinal cord injury animal model, intranasal administration of MSC-derived exosomes loaded with phosphatase and tensin homolog siRNA (ExoPTEN) effectively eased neurological deficits. 195 Exosomes can cross the BBB and are extensively used as drug carriers,196–198 suggesting that intranasal exosome therapy might mitigate harmful neuroinflammatory processes. However, their systemic distribution after intranasal application is not yet fully elucidated. Some CNS-derived exosomes reportedly contain efflux-related proteins, such as tau and α-synuclein, which implies that brain-to-blood clearance could shape their biodistribution. 199 More research into how donor cell characteristics and exosome composition affect their biodistribution and function will help harness exosomes as an intranasal delivery method for CNS repair (Figure 5(c)).
Stem cells
Stem cell-based approaches have been proposed as therapeutic interventions for neurological disorders (Figure 5(d)).152,200,201 Early efforts delivered stem cells directly to the CNS to trigger their neuroprotective effects. Several animal studies indicate that MSCs administered intranasally can benefit conditions including TBI, stroke, Parkinson’s disease, and brain cancer.202–204 While preclinical models are encouraging, safety considerations warrant caution for eventual clinical applications.205–207 To mitigate proliferative risks, researchers have explored capturing protective paracrine activities of stem cells without the uncontrolled cell growth traditionally associated with them.208,209
By administering the anti-inflammatory and neurotrophic molecules secreted by MSCs intranasally, these beneficial effects can be directed more safely and precisely to the CNS.210,211 Investigators are currently delineating which soluble mediators or vesicles underlie the positive outcomes of stem cell therapy. 212 MSC-derived extracellular vesicles (MSC-EVs), in particular, have garnered attention for immunomodulation in CNS diseases.208,210,213 It has been shown that MSC-EVs integrate into neurons and microglia once they enter the brain intranasally in a in vitro study. 214 Strategies such as preconditioning MSCs with inflammatory cytokines or hypoxic conditions can increase their EV production and therapeutic potency.215,216 Other methods involve exposing MSCs to substances like the Rho-kinase inhibitor fasudil, which was found to reduce dopaminergic neuron loss in a Parkinson’s animal model. 217 Genetically modifying MSCs to secrete various neuroprotective growth factors further broadens their clinical promise. 218 Additionally, guiding MSCs to differentiate into cell types specifically compromised in a neurological disease may enhance treatment outcomes. For instance, in an experimental multiple sclerosis model, conditioned medium from MSC-derived oligodendrocytes promoted myelination and curbed inflammation. 219 Moving forward, it will be important to optimize incubation and differentiation protocols that maximize MSC-based therapies’ delivery and benefits to the injured CNS. Carefully matching the diseased cell population with the appropriate preconditioning and differentiation strategy may yield safer, more targeted clinical outcomes.
Taken together, these findings underscore how intranasal administration of diverse therapeutic agents, from cytokines and growth factors to exosomes and stem cells, can circumvent traditional delivery barriers and directly target injured neural tissue. It is important to acknowledge that the efficiency of intranasal delivery is highly dependent on the physicochemical properties of the therapeutic agent. Small molecules and peptides (<1 kDa) generally traverse the nasal epithelium through paracellular or transcellular pathways. In contrast, macromolecules such as cytokines and growth factors require receptor-mediated or adsorptive endocytosis, which limits bioavailability. For whole cells, such as mesenchymal stem cells (MSCs), direct penetration across the nasal epithelium into the central nervous system is virtually impossible due to their large size (10–20 μm) and lack of motility across the olfactory barrier. Therefore, the therapeutic effects observed in preclinical studies following intranasal MSC administration are believed to primarily result from their paracrine actions, including secretion of neurotrophic factors and extracellular vesicles, rather than direct engraftment within the brain parenchyma.152,204 These considerations underscore the importance of alternative strategies, such as leveraging MSC-derived secretomes or exosomes and incorporating them into advanced carriers like mucoadhesive nanoparticles or thermoresponsive hydrogels, to achieve effective and sustained delivery.
Biomaterials-based intranasal drug delivery systems for TBI
Nanoparticle-based delivery: Rationale and strategies
Intranasal drug delivery has emerged as a promising non-invasive strategy to bypass the BBB and directly target the CNS, offering therapeutic potential for neurological disorders, including TBI. Among various delivery vehicles, biomaterials-based nanoparticles have gained attention due to their ability to enhance retention in nasal tissues, penetrate biological barriers, and increase drug bioavailability even at low doses.220,221 These systems enable site-specific delivery to injured brain regions, potentially improving therapeutic efficacy while minimizing systemic exposure.
The effectiveness of nanoparticle-mediated nose-to-brain delivery depends largely on the design and interaction of the nanocarriers with anatomical and biological barriers. Mucosal surfaces within the nasal cavity present the first major obstacle. Nanoparticles with mucoadhesive properties, particularly those carrying a positive surface charge, can adhere to the negatively charged mucus layer and resist mucociliary clearance. 222 However, overly adhesive particles may become entrapped. Inspired by viral surface characteristics, nanocarriers engineered with positive and negative charges or hydrophilic coatings can achieve balanced interactions, enhancing mucus penetration without excessive retention.223–225
Particle size also significantly influences transport efficiency. Nanoparticles smaller than 200 nm generally show improved diffusion through the nasal mucosa and better access to neural pathways. For instance, size-dependent uptake has been observed in the trigeminal nerve, where smaller particles (~100 nm) showed higher transport efficiency compared to larger counterparts.226,227 Additionally, surface charge affects the preferred neural transport route: negatively charged particles tend to favor the olfactory pathway, while neutral and positively charged particles more often utilize the trigeminal route.228,229
The performance and safety of nanoparticle-based delivery systems are strongly influenced by their physicochemical characteristics. For polymeric systems, factors such as molecular weight and degradation rate determine drug release kinetics and mucosal residence time. 230 Inorganic nanoparticles exhibit property-dependent behavior: smaller metal particles and specific shapes (e.g. rods, plates) can enhance olfactory transport but may also increase surface reactivity and cytotoxicity.231,232 Furthermore, the release of metal ions in contact with biological fluids raises concerns about oxidative stress and inflammatory responses.233,234 Prolonged accumulation of nanoparticles in neural tissues may further elevate toxicity risks, necessitating rigorous optimization of size, shape, and surface chemistry. 235 To mitigate these challenges, strategies such as PEGylation, biodegradable polymers, and surface functionalization have been explored to improve biocompatibility while maintaining effective nose-to-brain transport.
To further improve brain targeting, various materials and surface modification strategies have been developed. Polymeric or lipid-based nanoparticles may be functionalized with surfactants, PEGylation, or targeting ligands to enhance mucus permeability and epithelial penetration.236,237 Mucoadhesive or thermosensitive gels have also been combined with nanoparticles to prolong nasal residence and facilitate sustained release, allowing more efficient drug transport to injured brain tissues.238–241
Another strategy involves engineering nanoparticles to exploit the olfactory and trigeminal nerve pathways for intracellular transport. The restricted size of olfactory neurons (~100–700 nm) necessitates appropriately scaled nanoparticles to achieve efficient neuronal entry. 242 Furthermore, some advanced biomaterial systems incorporate deformable or magnetically responsive components to enhance directional delivery across these pathways.243,244
At the epithelial level, transient modulation of tight junctions (TJs) offers a route for paracellular transport. Natural and synthetic agents, such as surfactants, bile salts, or cationic polymers, can reversibly open TJs and improve nanoparticle permeation without causing permanent damage.245–248 However, safety concerns regarding cytotoxicity and inflammatory responses remain. 249
In addition to neuronal and paracellular transport, absorption via the lamina propria provides a supplementary mechanism for systemic and CNS access. This route, although less efficient due to BBB limitations, may still contribute to therapeutic effects, particularly for small lipophilic drugs. 250 However, the presence of immune cells in this region necessitates careful assessment of nanoparticle biocompatibility to avoid adverse inflammatory reactions. 251
Collectively, studies highlight the importance of rational nanoparticle design, considering size, surface chemistry, deformability, and muco-interactive properties, to effectively engage with the nose-to-brain delivery route (Figure 6). For TBI treatment, where timely and localized intervention is crucial, biomaterial-based intranasal systems offer a multifaceted platform to deliver therapeutics, mitigate inflammation, and promote neural repair.252–254 Continued exploration of nanoparticle-tissue interactions and biomimetic strategies inspired by natural intranasal transport processes may yield next-generation delivery systems with enhanced precision and safety. Despite encouraging results in preclinical models, no nanoparticle-based intranasal formulations have yet been clinically tested or approved for TBI in humans. A few early-phase trials have explored intranasal nanoparticle or exosome-based therapies for other CNS disorders, such as Alzheimer’s and Parkinson’s disease, but these remain limited and largely focus on safety assessment. Therefore, the clinical translation of intranasal nanoparticle delivery for TBI remains a significant unmet challenge requiring further investigation.

Overview of biomaterial-based intranasal delivery systems for brain-targeted therapy. Schematic summary of nose-to-brain delivery using biomaterial-based systems, outlining overall advantages and challenges of the approach, along with comparative features of representative nanocarrier platforms.
In vivo findings: Nanoparticle-based intranasal delivery systems in TBI
Lipid-based nanoparticles
Considering the safety and neurotoxicity concerns associated with nanoparticle-based drug delivery, lipid-based nanocarriers offer significant advantages for treating CNS diseases due to their low toxicity, high stability, and ability to cross the BBB. 255 Various formulations using physiological lipids, such as liposomes, nanoemulsions (NEs), nanostructured lipid carriers (NLCs), and solid lipid nanoparticles (SLNs), have been developed to encapsulate and protect lipophilic drugs, exhibiting high biocompatibility and biodegradability.226,256,257
In the context of TBI, where the BBB presents a major obstacle to therapeutic delivery, lipid-based nanoparticles provide a minimally invasive strategy for enhancing brain targeting. Additionally, surface modifications such as ligand conjugation or hydrophilic coatings can further improve brain accumulation and reduce off-target effects.258,259
Liposomes, the first developed lipid-based nanocarriers, consist of one or more phospholipid bilayers enclosing an aqueous core. 260 Their ability to carry both hydrophilic and hydrophobic agents makes them versatile tools for drug delivery. In TBI models, liposomes have shown promise for localized delivery of neuroprotective agents, reducing systemic side effects and improving functional outcomes. For example, intranasal delivery of interleukin-4-loaded liposomes in a murine TBI model promoted white matter repair and improved sensorimotor function (Figure 7(a)). 261 Similarly, phosphatidylserine-enriched liposomes activated the CD36/TGF-β1 signaling pathway in a surgical brain injury model, attenuating neuroinflammation and cerebral edema. 262

Intranasal delivery of biomaterial-based therapeutics for targeted brain treatment after TBI. (a) Intranasally administered IL-4–loaded liposomes reach the injured brain via the olfactory pathway. The released IL-4 activates PPARγ signaling in damaged white matter, promoting oligodendrocyte differentiation and myelin regeneration, thereby improving sensorimotor function. (b) Intranasal delivery of cerebrolysin-loaded PLGA nanoparticles facilitates brain targeting through mucoadhesion and epithelial penetration, followed by transport via the olfactory and trigeminal pathways. Sustained cerebrolysin release within injured regions stabilizes the BBB, reduces neuroinflammation, enhances cognitive function, and protects neural tissue. (c) PEGylated gold nanoparticles (AuNPs) administered intranasally traverse the nasal epithelium via paracellular transport and distribute to multiple brain regions, including the olfactory bulb, hippocampus, brainstem, entorhinal cortex, and periaqueductal gray. In injured tissues, AuNPs downregulate NF-κB–mediated cytokine production and upregulate antioxidant enzymes, attenuating neuronal apoptosis and supporting neuroimmune stabilization, neuroprotection, and behavioral recovery. (d) A thermoresponsive hydrogel encapsulating neuroprotective agents is administered intranasally in liquid form. Upon contact with the nasal mucosa, it undergoes in situ gelation on the olfactory epithelium, forming a mucoadhesive layer that enables sustained drug release. The agents reach the brain via the olfactory nerve pathway, modulating neuroinflammatory responses, promoting neuronal resilience, enhancing cognitive performance, and stabilizing neurovascular function.
Among newer delivery systems, oil-in-water (O/W) NEs are receiving attention due to their small droplet size, enhanced surface area, and ability to facilitate drug transport across the nasal mucosa. These characteristics make them suitable for nose-to-brain delivery, particularly for poorly water-soluble drugs.263–265 Various studies have demonstrated the potential of intranasal NEs for targeting CNS conditions such as migraine, neuroinflammation, and depression. For instance, a mucoadhesive NE containing zolmitriptan and chitosan enhanced nasal retention and mucosal permeation, resulting in a faster onset and greater bioavailability than conventional formulations—features desirable for acute migraine treatment. 266 Likewise, Pathak et al. developed an in situ gelling NE system for nimodipine, achieving significantly increased drug permeation and higher brain concentrations, which may be relevant in treating neurovascular complications like senile dementia and cerebrovascular spasms, often associated with TBI. 267 Intranasal NEs have also been formulated for selective serotonin reuptake inhibitors (SSRIs) such as paroxetine and sertraline to overcome first-pass metabolism and low oral bioavailability. Paroxetine-loaded NEs enhanced drug permeation and brain uptake in animal models, while sertraline NEs demonstrated significantly increased solubility and mucosal transport in ex vivo studies. 268 Yadav et al. reported that intranasal administration of cyclosporine-A-loaded NEs significantly improved brain targeting and reduced systemic exposure compared to oral and IV routes. 269 High drug accumulation in the olfactory bulb and midbrain supported effective nose-to-brain delivery, with a rostral-to-caudal transport gradient observed. 269 In a related study, cationic NE-encapsulated TNF-α-targeting siRNA achieved greater delivery to the midbrain, indicating the potential of NE systems for anti-inflammatory RNA therapies in neurodegenerative conditions. 270
To further address the limitations of conventional CNS therapeutics, SLNs and NLCs have emerged as stable and efficient nanocarrier systems. SLNs contain a solid lipid matrix, while NLCs combine solid and liquid lipids, improving drug loading and storage stability. Both systems offer enhanced brain penetration, sustained drug release, and better biocompatibility compared to polymeric or inorganic nanoparticles.271–273 Recent studies demonstrate the neurotherapeutic promise of these systems. Sun et al. formulated an in situ gel incorporating paeonol-loaded SLNs, which showed low cytotoxicity and effective brain accumulation after intranasal administration. 274 Abourehab et al. reported that nicergoline-loaded NLCs, developed with sesame oil, achieved superior brain bioavailability and targeting efficiency. 275 Abo El-Enin et al. developed a chitosan-coated berberine-loaded NLC that significantly increased brain accumulation and showed potential to mitigate TBI-associated neuroinflammation. 276 Collectively, these findings highlight the potential of lipid-based nanocarriers, especially liposomes, NEs, SLNs, and NLCs, for non-invasive, nose-to-brain delivery of neuroprotective agents in the treatment of TBI and related CNS disorders.
Polymeric-based nanoparticles
Polymeric nanoparticles have been widely explored for intranasal drug delivery due to their ability to enhance drug absorption and facilitate brain targeting, making them particularly relevant for TBI therapy. These carriers adhere to the nasal mucosa, extend residence time, and protect therapeutic agents from enzymatic degradation and mucociliary clearance, ultimately improving uptake across the nasal epithelium.277–279 Their biocompatibility, modifiable surface properties, and ability to cross the BBB make them excellent candidates for therapeutic and diagnostic applications in neurotrauma.
One of the most widely studied polymeric systems is poly(butyl cyanoacrylate; PBCA). Particularly when coated with surfactants such as polysorbate 80, PBCA nanoparticles exhibit enhanced uptake by brain endothelial cells, facilitating effective drug transport across the BBB. This surface modification facilitates rapid uptake by endothelial cells, allowing timely delivery of neuroprotective and anti-inflammatory agents, which are critical in the acute phase of TBI. Surface properties were found to be more influential than particle size in determining brain uptake, with polysorbate 80-coated PBCA nanoparticles showing the highest efficacy.280–282 In parallel, poly(lactic-co-glycolic acid; PLGA) has also been an attractive drug delivery vehicle due to its controlled biodegradation rate and safety profile. In preclinical models, PLGA nanoparticles encapsulating cerebrolysin reduced BBB disruption and brain edema as early as 8 h post-injury, even when administration was delayed (Figure 7(b)). 283 These systems are also being investigated for theranostic applications. For instance, PLGA nanoparticles labeled with near-infrared dyes demonstrated size-dependent diffusion into deeper brain structures, while PX-coated formulations improved cognitive outcomes and decreased systemic clearance.284,285 Modifications such as iRGD peptides and olfactory-targeted coatings have further enabled efficient nasal uptake and direct CNS delivery through olfactory ensheathing cells and lymphoid tissue.286,287
To achieve even greater precision in targeting injured brain regions, dendrimer-based nanoparticles have been explored for their architectural uniformity and customizable surface functionality. Among these, polyamidoamine (PAMAM) dendrimers are particularly well suited to TBI applications. Their small size (~4 nm), neutral charge, and responsiveness to pathological stimuli allow them to accumulate in activated glial cells, a hallmark of TBI-induced neuroinflammation. Drug-loaded dendrimers have demonstrated significantly higher CNS accumulation—up to 100-fold compared to free drugs—and the ability to respond to injury-specific triggers such as pH changes and enzymatic activity supports their role in sustained, site-specific drug release.288–290
Recent work has also focused on optimizing nose-to-brain transport through the trigeminal nerve pathway. Polycaprolactone (PCL) nanoparticles, including PEGylated forms, have been employed for this purpose. For example, curcumin-loaded PCL systems (with PEG) reached the nasal mucosa and entered the trigeminal route, though initial olfactory delivery remained limited. 291 The addition of cell-penetrating peptides enabled broader CNS access via olfactory and trigeminal pathways within 15 min. 292 Notably, PCL-based carriers loaded with aripiprazole delivered drug levels to the brain nearly twice as high as those achieved with IV administration, 293 while surface modifications such as polysorbate 80 or sodium caproyl hyaluronate further enhanced permeability and bioavailability. 294
Enhancing mucosal adhesion and epithelial permeability has also been a focus of chitosan-based nanoparticle development. These mucoadhesive systems facilitate drug transport across the nasal epithelium while resisting mucociliary clearance. In one study, a chitosan platform delivering Galectin-1 siRNA modulated epithelial tight junctions and successfully transported its payload to the brain via the olfactory route. 295 Hybrid systems combining chitosan with lecithin have shown similarly improved mucosal retention and rapid CNS absorption. 294
Inorganic-based nanoparticles
Inorganic nanoparticles offer unique advantages for intranasal drug delivery in TBI, owing to their tunable size, surface properties, imaging functionality, and structural stability. Unlike many polymeric systems, these nanoparticles are inherently suitable for therapeutic and diagnostic applications, allowing for simultaneous drug delivery and real-time tracking. Their capacity to bypass the BBB via the olfactory and trigeminal pathways further supports their growing use in TBI research, particularly for targeting neuroinflammation and focal injury sites.
Among various inorganic systems, gold-based nanoparticles have been extensively studied due to their inert nature and high surface-area-to-volume ratio (Figure 7(c)). In comparative experiments involving PEGylated nanospheres and nanoprism structures functionalized with D1 peptides, nanospheres demonstrated superior brain translocation following intranasal administration, despite similar physicochemical characteristics. These particles reached diverse brain regions, including the olfactory bulb, hippocampus, and entorhinal cortex, suggesting effective neural tissue penetration. Studies combining focused ultrasound and microbubble-mediated delivery further enhanced the targeting of gold nanoparticles to the brainstem via the trigeminal nerve, highlighting their potential in localized delivery strategies. 296
Iron oxide nanoparticles, particularly PEGylated forms, have also been utilized to map nose-to-brain transport routes. After administration, these particles cross the nasal epithelium and travel through the perineural spaces of the olfactory and trigeminal nerves. Once they reach the subarachnoid space, CSF flow facilitates broad dispersion across cerebral cisterns and into parenchymal tissues. 297 This widespread distribution, combined with the magnetic properties of iron oxide, positions them as useful agents for both therapeutic and image-guided delivery.
Graphene oxide nanosheets have added further diversity to the inorganic delivery toolkit. Owing to their high surface area and mechanical flexibility, ultrathin nanosheets show efficient CNS translocation when delivered intranasally. Notably, the smallest sheets achieved the highest regional distribution, particularly in the olfactory bulb, and were observed to accumulate within microglial cells. 283 This suggests potential for modulating inflammation, a key component of secondary injury in TBI, using graphene oxide as a therapeutic platform.298,299
Quantum dots (QDs), such as CdSe/ZnS (with sizes of ~15 nm), have shown rapid accumulation in the olfactory bulb and tract when administered as an aerosol. The intrinsic fluorescence of QDs allows for straightforward visualization via fluorescence microscopy and electron imaging, offering dual utility for cargo delivery and biodistribution analysis. 300 However, concerns over heavy metal toxicity remain a significant barrier to broader applications of QDs, especially for chronic or repeated dosing in clinical settings.
To address safety concerns while retaining optical functionality, attention has turned to carbon dots (CDs). These sub-10 nm particles are known for their strong fluorescence, excellent water solubility, and biocompatibility. Functionalization with targeting moieties, such as transferrin, has enabled CDs to exploit receptor-mediated transcytosis at the BBB and deliver agents like doxorubicin to brain tumor cells. 301 Other studies report that CDs with a quantum yield of 51% and low cytotoxicity can effectively penetrate in vitro BBB models composed of endothelial and astrocyte co-cultures. Moreover, modifying CDs with cationic polymers such as polyethyleneimine further enhances brain penetration by promoting interactions with negatively charged cellular membranes. Their tunable fluorescence and ability to respond to environmental cues make CDs especially well-suited for theranostic approaches, combining imaging with site-specific therapy.302,303
Intranasal hydrogel delivery and therapeutic efficacy
While nanoparticle-based intranasal delivery systems offer significant advantages in enhancing drug transport to the brain, hydrogel-based approaches provide a complementary strategy with distinct benefits. Unlike nanoparticles, which primarily rely on cellular uptake and nerve pathway penetration, hydrogels can form localized drug reservoirs that offer sustained, controllable release profiles and minimize systemic exposure.299,304 Their ability to conform to the architecture of the nasal cavity and maintain close contact with the mucosa enhances residence time, drug stability, and delivery precision—important factors for managing the complex pathology of TBI, including neuroinflammation, oxidative stress, and neuronal apoptosis.305,306
Although acute interventions may limit secondary damage, long-term neuroprotection often requires continuous therapeutic support. Injectable hydrogels, especially those that are thermoresponsive or stimuli-sensitive, have emerged as ideal carriers in this context due to their ability to encapsulate diverse therapeutic agents, including small molecules, proteins, and biologics, while enabling extended-release kinetics (Figure 7(d)).307–309 These hydrogels remain in a liquid state at lower temperatures and undergo a sol-to-gel transition at body temperature, ensuring close adherence to the administration site. Intranasal delivery, in particular, offers a direct route to the brain via the olfactory and trigeminal nerve pathways, thus avoiding systemic circulation and first-pass metabolism.310–312 Once in place, hydrogel scaffolds release drugs more steadily compared to simple solutions, while also reducing adverse systemic effects.
One illustrative study used a thermoresponsive poloxamer (P407/P188) formulation that complexes tetrandrine with hydroxypropyl-β-cyclodextrin (HP-β-CD) to treat a microwave-induced brain injury model in Wistar rats. 313 Upon intranasal administration, the hydrogel formed a semi-solid gel in the nasal cavity and gradually released tetrandrine, thereby lowering inflammatory mediator levels in the injured brain region. This strategy also yielded functional benefits, with improvements in neuronal viability and spatial memory relative to control groups. The sustained-release profile is partly attributed to the mucoadhesive properties of the poloxamer hydrogel, which enables the formulation to remain at the nasal epithelium for a longer period.
Despite these advances, several challenges exist. The limited volume of the nasal cavity constrains the quantity of hydrogel and drug that can be administered at one time. 314 Meanwhile, normal mucociliary clearance in the nasal epithelium can reduce the contact time and the effective dose of encapsulated therapeutics.315,316 To address this, strategies such as incorporating strong mucoadhesive polymers and optimizing poloxamer ratios have been employed to improve retention and ensure ideal sol-gel transitions.317,318 Moreover, species-specific differences in nasal anatomy necessitate careful consideration when translating formulations from animal models to humans. The intranasal route may be particularly suitable for patients with mild to moderate TBI during the subacute or chronic stages, where self-administration is feasible and non-invasive delivery is preferred. In contrast, patients with severe TBI in the acute phase may have impaired consciousness or require assisted ventilation, making nasal delivery impractical. From a compliance standpoint, intranasal sprays and nebulizers offer potential advantages over injections or oral tablets in outpatient or long-term use. However, factors such as nasal irritation, mucosal variability, and device handling difficulty may influence patient adherence and treatment acceptability.
Future directions in hydrogel development aim to incorporate responsiveness to temperature and pathophysiological cues such as pH shifts, enzyme activity (e.g. MMP upregulation), and ROS.319–321 These multifunctional systems can co-deliver neuroprotective agents with growth factors, anti-inflammatory drugs, or genetic material to enable synergistic effects. Concurrent advances in polymer chemistry and in vivo imaging are expected to support more precise control over drug release and dosing, enhancing therapeutic outcomes across both acute and chronic phases of TBI.
Additionally, recent studies have explored the integration of antioxidant enzymes, neurotrophic factors, and EVs (such as exosomes) into intranasal hydrogels to improve tissue regeneration and alleviate neuroinflammation.322–325 These advanced hydrogel systems leverage mucoadhesive and stimuli-responsive properties to (i) enhance payload stability, (ii) target therapeutic agents to the lesion site for extended durations, and (iii) enable controlled, on-demand release in response to the dynamic changes in the TBI microenvironment. However, additional in vivo studies are necessary to fully understand the distribution of these nanocomposite- or biologics-loaded hydrogels within the nasal passages and their ability to effectively deliver therapies to the injured brain regions.
Concluding remarks
TBI often leads to long-term cognitive, physical, and psychosocial impairments, contributing to significant global morbidity and mortality. Effective therapies that reduce secondary brain damage and promote neuronal regeneration are thus crucial. In this context, intranasal administration, offering non-invasive, rapid delivery of therapeutics directly to the brain while bypassing the BBB, shows promise as a therapeutic approach for TBI. This review highlights recent advances in intranasal delivery as potential strategies for TBI. Preclinical studies have explored a variety of molecules, such as growth factors (BDNF, NGF, IGF-1), anti-inflammatory cytokines (IL-10, IL-13), stem cell-derived exosomes, and metabolites, demonstrating protective effects by reducing neuronal apoptosis, inflammation, and edema, while enhancing sensory and cognitive functions. Nanoparticle-based delivery systems are also being developed to improve drug absorption and controlled release.
Despite promising results in animal models, the clinical application of intranasal therapies for TBI faces several challenges. Key issues include determining the optimal therapeutic time window, assessing the long-term toxicity of high-dose treatments, and improving the diffusion of larger molecules or nanoparticles in the human nasal mucosa. Biodegradable and mucoadhesive nanoparticles may offer a better route for penetration. Furthermore, it remains unclear whether combination therapies or single treatments will yield the most effective outcomes against complex secondary cascades of TBI.
In summary, intranasal delivery offers great potential for improving TBI treatment by targeting molecular pathways involved in neuroinflammation and neurodegeneration. As personalized medicine advances, intranasal therapies could be tailored to the specific injury characteristics and genetic profiles of patients. However, more translational research is needed to support the clinical application of these therapies for better TBI management.
Footnotes
Author contributions
SHY, SCH, JSB, and JCK contributed to the concept of the work, data acquisition, and drafting of the manuscript and revised the article critically for important intellectual content. JHL and HWK designed the study, drafted the manuscript, procured funding, and supervised the study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation funded by the Korean Government (Grant Nos. RS-2021-NR060095, RS-2024-00348908, RS-2022-NR072135). This work was also supported by the Bio and Medical Technology Development Program of the National Research Foundation funded by the Korean government (Grant Nos. RS-2023-00220408, 2020-NR049585, 2022-NR075747).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
