Abstract
Lymphedema has emerged as a significant health issue among cancer survivors. The primary goal of treatment is to restore lymphatic drainage function. Engineering vascularized lymphatic tissue offers a promising alternative to achieve this goal. Currently, lymphatic tissue engineering with the use of cell-seeded scaffolds incubated in high hemodynamic flow environments, such as AV loop chambers, has shown promising results for lymphatic vessel regeneration. In this study, lymphatic endothelial cells (LECs) and adipose-derived stem cells (ASCs) were incorporated into an AV loop chamber and cultured in the groin region of a rat model. Surprisingly, the level of lymphangiogenesis, indicated by increased expression of the lymphatic marker LYVE-1, was significantly higher in the group with LECs alone than in the group with both LECs and ASCs. The engineered lymphatic tissue was subsequently orthotopically transplanted into the area of lymph node dissection. This procedure restored lymphatic drainage and reduced local inflammation, with decreased levels of CD3, CD4, and CD8 expression. These findings provide the potential for creating a functional, organized lymphatic system through the engineering of vascularized lymphatic tissue via AV loop cultivation, offering an applicable treatment option for lymphatic defects.
Introduction
Recently, lymphedema has been identified as one of the top health issues in cancer survivors. The best treatments for lymphedema have yet to be developed. 1 The causes of lymphedema other than cancer surgery are congenital maldevelopment of part or all of the lymphatic system, filarial infection, and trauma. 2 The lymphatic system plays a crucial role in the body’s fluid homeostasis by counteracting the various mechanisms leading to fluid extrusion into and accumulation within the interstitial compartment. 3 When the lymphatic system cannot achieve this goal successfully, protein-rich fluid collections within the soft tissue, termed lymphedema, develop. Furthermore, lymphedema leads to compromised local immunity, tissue fibrosis and a decreased quality of life for the affected patient. 4 Although surgical treatments, including lymphaticovenous anastomosis (LVA), vascularized lymph node transfer (VLNT), and liposuction, can be effective, they are accompanied by overall procedural surgical risks such as infection, scars, and donor site morbidity or even a lack of suitable donor lymphatic tissue.5,6 None of the currently available treatments are considered the most ideal. Understanding the mechanism of lymphangiogenesis and identifying alternative treatment options have become major goals of lymphatic research. Engineering lymphatic tissue can be a good alternative to eliminate donor site morbidities and provide more options for treating congenital lymphedema.
Currently, lymphatic tissue engineering focuses on approaches that include cell-seeded scaffolds for lymphatic vessel reconstruction, stem-cell injection, the application of cytokines and chemokines to induce lymph vessel formation or a combination of different approaches.7,8 Very few studies have aimed to engineer a lymph node or lymphatic tissue. Moreover, the basic requirement for engineered tissue to be continuously viable is engineering a tissue containing both arteries and veins. The vascular pedicle provides a constant and sufficient blood supply, and the arterial inflow and venous outflow maintain the transportation of nutrients, oxygen, and other essential substances. For example, the arteriovenous (AV) loop model has been utilized in previous studies to increase angiogenesis and maintain vascularity in vascularized tissue engineering (VTE). 9 In our previous study, we demonstrated that, with a stronger hemodynamic force, the AV loop model provides superior lymphangiogenesis potential to form vascularized lymphatic tissue in comparison to another vascular model: the flow-through model. 10 The hemodynamic features of the AV loop model, together with its vascular properties, provide a good foundation for further investigations of lymphatic regeneration. 10 The combination of lymphatic endothelial cells (LECs) and fibrin-containing scaffolds in a flow chamber has been demonstrated to promote lymphangiogenesis with a tube-like organization in vivo. 11
In addition to LECs, adipose-derived stem cells (ASCs) have been shown to support the migration, proliferation and tube formation of LECs through direct contact as well as the secretion of lymphangiogenic factors.12,13 The combination of ASCs and LECs in a three-dimensional fibrin scaffold exhibited a positive effect on vasculature network formation, whereas without the addition of ASCs, the formation of primitive tube-like structures was not observed, 14 suggesting synergistic effects with the addition of ASCs. Upon stimulation with VEGF-C in vitro, ASCs presented a significant lymphangiogenic response after being implanted in a mouse model, which supports their role in lymphangiogenesis. 15 To use the knowledge of tissue engineering for tissue repair, one possible strategy is to engineer vascularized lymphatic tissue in an area where lymphatic tissue is lacking. Another strategy is to engineer vascularized lymphatic tissue that contains arteries and veins in vivo and transfer it as a microsurgical free flap to the defect. Considering both the concept of lymphatic tissue engineering and repair and the concept of a cellular component and consistent vascularity, we used a fibrin gel encapsulated with either a combination of ASCs and LECs or LECs only and placed them into an AV loop chamber in a rodent model to regenerate lymphatic tissue. Furthermore, the engineered vascularized lymphatic tissue generated via the AV loop model was harvested and transplanted orthotopically into the adjacent location where the lymph node dissection was performed and the lymphatic gap was present. Our goal was to explore its role in lymphangiogenesis and ability to bridge the lymphatic gap. Clinical and molecular investigations were conducted to investigate the efficacy of the engineered vascularized lymphatic tissue in terms of its lymphangiogenic properties.
Materials and methods
Study design
This study is the first to validate the regenerative potential of tissue-engineered constructs containing fibrin gel-encapsulated LECs alone or LECs combined with ASCs. These constructs were placed within an AV loop chamber and incubated in the groin region of rats. The regenerated lymphatic tissue was assessed using the lymphatic markers LYVE-1, podoplanin, VEGF-C, and VEGFR-3 2 weeks after. The second part of the study investigated the efficacy of the engineered vascularized lymphatic tissue in restoring lymphatic function following lymph node dissection. The tissue-engineered constructs cultivated within the AV loop chamber were removed from the chamber and transplanted into the site adjacent to where the lymph node had been dissected 2 weeks after the engineering and assessment performed another 2 weeks after transferring.
Animal model
The study was conducted according to the animal research guidelines approved by the Institutional Animal Care and Use Committee (IACUC#: 2020122216 and 2023062706) of Chang Gung Memorial Hospital, Linkou, Taiwan and were performed in accordance with the institution’s animal research guidelines. Fifty male Lewis rats (aged 2 months) with average weights ranging from 350 to 400 g were obtained from BioLASCO Taiwan Co., Ltd. (Taiwan). The rats were housed in standard cages and kept under a 12-h dark-light cycle at a constant temperature of 21 ± 2°C with a constant humidity of 50 ± 10%. All the animals had access to water ad libitum and were able to move freely around their cages. The assignment of the animals to the groups was conducted at random.
Harvesting and preparation of LECs and ASCs
LECs were harvested from the thoracic duct of Lewis rats. The thoracic duct was surgically removed, washed with phosphate-buffered saline (PBS), cut into several small pieces, and subsequently digested with EBM-2/EGM-2 medium (Lonza, USA) containing 0.5% collagenase (Sigma, USA) at 37°C for 1 h. The samples were then centrifuged at 2000g, and the supernatants were removed. The remaining cells were resuspended and cultured in EBM-2/EGM-2 medium supplemented with 10% fetal bovine serum (FBS) at 37°C with a 5% CO2 concentration. The LECs were maintained by changing the culture medium every other day.
The ASCs were harvested from the groin fat pads of the rats as described previously. 16 The fat pad was minced and washed several times with PBS containing 100 units/mL penicillin and 100 μg/mL streptomycin (Gibco, USA). The fat tissue was digested with Dulbecco’s modified Eagle’s medium (DMEM; Gibco, USA) containing 0.5% collagenase at 37°C for 1 h. After preparation and centrifugation, the red blood cells were removed with lysis buffer containing ammonium chloride potassium (Sigma, USA). The cell debris was removed via a 100 μm strainer. After centrifugation, the cells were grown in DMEM with 10% FBS (Gibco, USA) in an incubator at 37°C with a 5% CO2 concentration. The culture medium was changed every 3 days.
Fibrin gel preparation
The fibrin gel was prepared immediately before the AV loop surgery. For this purpose, 500 μl of fibrinogen (10 mg/mL; Sigma, MO, USA) and thrombin (10 U/mL; Sigma, MO, USA) were mixed in a centrifuge tube and kept at room temperature for 1–3 min. In the LEC-only group, 2 × 104 LECs, and in the LEC+ASC group, 1 × 104 LECs and 1 × 104 ASCs were suspended in the fibrinogen‒thrombin mixture and allowed to gel for 3 min.
Tissue engineered construct in the AV loop chamber in an animal model
The AV loop model was created as described previously. 10 Briefly, a 5 cm incision was made in the groin area, through which the left femoral artery and vein were carefully exposed and dissected. The right femoral vein, approximately 1 cm long, was also identified and harvested as a graft. Following this, both the left femoral artery and vein were sectioned distally, and the harvested vein graft was utilized to establish a connection between them, forming the AV loop. The AV loop was subsequently placed in a customized AV loop chamber made with biocompatible dental implant material (MED610, 3D by Stratasys, Rehovot, Israel). Within the chamber, a mixture of LECs and ASCs or only LECs with fibrin gel was inserted (Figure 1(a)). Prior to sealing the chamber, the patency of the anastomosis was ensured. Finally, the chamber was affixed in place via 4-0 nylon sutures, and the incision was closed primarily. This engineered construct was then incubated for 2 weeks in the groin region of the rat and subsequently harvested for examination or transferred to extensive lymph node dissection area.

Investigation of the lymphatic regeneration potential of tissue-engineered constructs composed of fibrin gel with LECs or a combination of LECs and ASCs. (a) Tissue-engineered construct within the AV loop chamber in a rat model. (b) Hematoxylin and eosin staining of the tissue grown in the AV loop chamber with fibrin gel containing LECs only or the combination of LECs and ASCs. The letters A and V denote artery and vein, respectively. The scale bar equals 500 µm. (c) Immunofluorescence staining of the engineered tissue in the AV loop chamber containing LECs only or the combination of LECs and ASCs with LYVE-1 (green), αSMA (red), and DAPI (blue). Arrow: LYVE-1 labeled lymphatic vessel. Arrow head: blood vessel. Asterisk: functional lymphatic vessel. The scale bar equals 100 µm. Quantification of LYVE-1 and αSMA expression per area of the field of view as a percentage. The error bars denote the SEs.
Lymphadenectomy rat model
The rats were anesthetized via 2.5% isoflurane (Piramal Critical Care, USA). Once anesthetized, the surgical procedure for lymphadenectomy was performed on the basis of a modified protocol from a previous study. 17 The rats were placed in the supine position with their groin area exposed. A 3 cm transverse incision was made in the groin region, allowing access to both inguinal and popliteal lymph nodes. These nodes, along with the surrounding lymphatic tissue, were carefully excised, guided by the visualization of Evans blue-stained lymphatics.
Transfer of the engineered lymphatic tissue
The rats were anesthetized via 2.5% isoflurane (Piramal Critical Care, USA). After lymphadenectomy, the chamber for tissue engineering was carefully removed to keep the engineered lymphatic tissue and its artery and vein intact. The whole engineered vascularized lymphatic tissue was placed in the dead space left after the lymphadenectomy. The wounds were closed primarily.
Examination of lymphatic vessels with indocyanine green (ICG)
Under isoflurane anesthesia, 0.1 mL of 0.05% ICG (TAIYO Pharma, JP) was injected into the dorsal and plantar sides of the paw of the left hindlimb. Thirty minutes after injection, images were taken to observe the lymphatic flow, starting from the paw and extending to the inguinal area. The Pearl Impulse Small Animal Imaging System (LI-COR Biotechnology, Nebraska) was utilized to capture the lymphatic flow pattern in the operated legs of each thigh.
Histological examination and immunofluorescence staining
The engineered tissue around the AV loop and the skin above the AV loop were harvested and used as samples for this investigation. Fifteen minutes before the skin was harvested, an injection of PBS with 100 µg of tomato lectin (Invitrogen, CA, USA) into the left foot paw was performed. The harvested tissues were fixed in 10% paraformaldehyde (Sigma, USA), dehydrated, embedded in paraffin and sectioned into 4 μm sections. The slides were baked at 60°C and rehydrated with Xylene, followed by serial steps of alcohol (100%, 95%, and 70%; Sigma, USA) to remove the paraffin. Next, citrate acid was used to reactivate protein activity, and then the primary antibody was applied. The levels of the lymphatic duct markers LYVE-1 (ab10278, Abcam, MA, USA), podoplanin (14-5381-85, Invitrogen, MA, USA), and α-SMA (ab7817, Abcam, MA, USA) were measured. Furthermore, the levels of CD3 (ab699, Abcam, MA, USA), CD4 (ab133616, Abcam, MA, USA), CD8 (ab4055, Abcam, MA, USA), eNOS (ab5589, Abcam, MA, USA), iNOS (ab283655, Abcam, MA, USA), VEGFR-3 (ab27278, Abcam, MA, USA), and VEGF-C (E-AB-64230, Elabscience, TX, USA) were also investigated. Fluorescent dye or horseradish peroxidase (HRP; Dako, CA, USA) was used after removal of the primary antibody. Images were captured via AxioVision (Carl Zeiss Microimaging, NY, USA). The quantification of tissue immunoreactivity was conducted via the ImageJ plugin.
Protein analysis (Western blotting)
Protein analysis was performed through Western blot analysis. The tissue was suspended in RIPA lysis buffer (Thermo Fisher, MA, USA), kept on ice for ten minutes and then centrifuged at 15,000g at 4°C for 15 min. The protein concentration was determined with Pierce BCA protein assay reagent (Thermo, USA). Proteins were resolved by 10% sodium dodecyl sulfate‒polyacrylamide gel electrophoresis (SDS-PAGE) and electrophoretically transferred to 0.45 μm PVDF membranes. The blotted membrane was blocked with PBS containing 0.1% Tween 20 (Sigma, USA) and 3% skim milk. Primary antibodies (1:1000) against VEGF-C (E-AB-64230, Elabscience, TX, USA), VEGFR-3 (ab27278, Abcam, MA, USA), LYVE-1 (ab10278, Abcam, MA, USA), podoplanin (14-5381-85, Invitrogen, MA, USA), Prox1 (ab199359, Abcam, MA, USA), eNOS (ab5589, Abcam, MA, USA), iNOS (ab283655, Abcam, MA, USA), and actin (ab8227, Abcam, MA, USA) were added and incubated at 4°C overnight with gentle shaking. To remove the primary antibodies, the membranes were washed with PBST three times and then incubated with an HRP-conjugated secondary antibody (Abcam, MA, USA) at room temperature for 2 h. The membrane was then further washed with PBST three times, and the reactive protein bands were visualized by enhanced chemiluminescence detection reagents with exposure to chemiluminescence light film. The protein expression level was quantified via ImageJ software (National Institutes of Health, USA).
RNA analysis
The expression of lymphangiogenesis-related genes was analyzed by real-time qPCR. The expression of eNOS and iNOS was evaluated. Primers were designed and customized by GeneDireX (USA). The sequences are presented in Table S1. The tissue was minced and suspended in TRIzol (Invitrogen, CA, USA). Total RNA was isolated via a RNeasy Mini Kit (QIAGEN, Germany) according to the manufacturer’s protocol, and cDNA was generated via reverse transcription of RNA via a high-capacity cDNA reverse transcription kit (Applied Biosystems, MA, USA). The cDNA was used for real-time PCR with Fast SYBR green master mix (Applied Biosystems, MA, USA) in a QuantStudio 5 Real-Time PCR System (Applied Biosystems, MA, USA). The RNA expression data were normalized to those of GAPDH.
Statistics
All of the experimental data were analyzed with Prism software (Graph Pad, MA, USA). The Student’s t test was applied depending on the groups to be compared. A p value less than 0.05 was considered statistically significant, and a p value less than 0.01 was considered highly statistically significant.
Results
ASC presents no lymphatic regeneration when included with healthy LECs
To explore the role of ASCs in promoting the lymphangiogenesis of LECs, tissue-engineered constructs comprising fibrin gel with LECs or a combination of LECs and ASCs were implanted into the AV loop chamber for culture for 2 weeks (Figure 1(a)). More adipose tissue was observed via H&E staining in the combination LEC and ASC group than in the LEC only group (Figure 1(b)). Unlike adipogenesis, the level of lymphangiogenesis, as determined by the expression of the lymphatic vessel marker LYVE-1, was significantly greater in the LEC only group (LEC only vs LEC+ASC: 1.6 ± 0.09 vs 0.82 ± 0.06 vessels/μm2, p < 0.0001; Figure 1(c), arrow). The number of functional lymphatic vessels with double presentation of αSMA and LYVE-1 was significantly reduced when ASCs were included with LECs in the AV loop chamber (LEC only vs LEC+ASC: 2.77 ± 0.22 vs 0.5 ± 0.22 vessels/μm2, p < 0.0001; Figure 1(c), asterisk). Interestingly, the vessel density was drastically increased with the addition of ASCs (LEC only vs LEC+ASC: 10.83 ± 0.52 vs 28.17 ± 2.3 vessels/μm2, p < 0.0001; Figure 1(c), arrowhead). In summary, when ASCs were incorporated into LECs within AV loop engineered constructs rather than primarily enhancing lymphatic vessel regeneration, their predominant role was to promote angiogenesis and adipogenesis within the regenerated tissue.
VEGF-C is a crucial growth factor and is essential throughout lymphatic vessel development. 18 It has a specific and unique role in lymphangiogenesis but is not essential for angiogenesis. 19 Immunofluorescence (IF) confirmed that the expression of VEGF-C was reduced when ASCs were loaded with LECs inside the hydrogel construct (LEC only vs LEC+ASC: 1.49 ± 0.08 vs 0.89 ± 0.04%, p < 0.0001; Figure 2(a)). In the presence of VEGF-C, a significant reduction in the expression of the VEGF-C receptor, namely, VEGFR-3, was also noted in the ASC+LEC group (Figure 2(b)). Furthermore, the protein expression of lymphangiogenesis-related genes presented a similar trend (Figures 2(c) and S1). A reduction in the expression of Prox1, LYVE-1, and podoplanin was found in the engineered construct with a combination of LECs and ASCs. Similar findings were observed for the essential growth factor VEGF-C and its receptor, VEGFR-3, which presented lower protein levels. These findings align with a decrease in the regenerative capacity of the lymphatic system with the reduction in LYVE-1 expression and the number of functional lymphatic vessels in the LEC+ASC group (Figure 1(c)).

Immunohistochemical staining of VEGF-C and VEGFR-3 in the tissue-engineered constructs composed of fibrin gel with LECs or a combination of LECs and ASCs. (a) Immunohistochemical staining of VEGF-C (brown color) in the LEC only and LEC+ASC groups. The quantification of VEGF-C expression (brown staining area) is presented. (b) Immunofluorescence staining of VEGFR-3 (red) in the LEC only and LEC+ASC groups. The quantification of VEGFR-3 expression is presented. DAPI staining nucleus marked as blue color. Scale bar equals 100 µm. (c) The protein expression of VEGFR-3, Prox1, LYVE1, VEGF-C, podoplanin, and β-actin was analyzed by Western blot analysis. The error bars denote the SEs.
ASCs are associated with immune response reduction within engineered lymphatic tissue
Mesenchymal stem cells exhibit immunomodulatory properties, as evidenced by their secretion of cytokines, growth factors, and chemokines, which effectively suppress immune reactions.20,21 The anti-inflammatory function of ASCs has been utilized in the treatment of inflammatory diseases. 22 Here, we examined the presentation of CD3 T cells, CD4 T helper cells, and CD8 cytotoxic T cells in the engineered tissue constructs that were implanted within AV loop chambers. As expected, drastic reductions in the numbers of CD3, CD4, and CD8 T cells were observed in the presence of ASCs in the engineered constructs (CD3: 2.36 ± 0.50 and 0.66 ± 0.51 cells/µm2 in LEC only and LEC+ASC, respectively, p < 0.0001; CD4: 1.62 ± 0.51 and 0.83 ± 0.75 cells/µm2 in LEC only and LEC+ASC, respectively, p = 0.0374; CD8: 1.42 ± 0.53 and 0.66 ± 0.81 cells/µm2 in LEC only and LEC+ASC, respectively, p = 0.0682; Figure 3).

Immunofluorescence staining of the inflammatory markers CD3, CD4, and CD8 in the different groups. The expression of CD3, CD4, and CD8 is marked in red; DAPI (blue) was used to mark the nuclei. The quantification of the number of CD3-, CD4-, and CD8-expressing cells is presented. The scale bar equals 100 µm. The error bars denote the SEs.
Nitric oxide is involved in lymphatic vessel regeneration
Nitric oxide (NO) is involved in regulating the growth of lymphatic vessels. 23 Endothelial NO synthase (eNOS) is known to be involved in angiogenesis and angiogenic sprouting.24,25 Furthermore, the isoform of eNOS is required for VEGF-C-mediated lymphangiogenesis and lymphatic valve specification via B-catenin signaling.23,26 The synthase of inducible NOS (iNOS) is correlated with VEGF-C-mediated lymphangiogenesis. 27 Compared with that in the LEC only group, the expression of eNOS increased significantly when ASCs were included (1.1 ± 0.33% and 4.4 ± 0.61% in LEC only and LEC with ASC groups, p < 0.001; Figure 4(a)). Conversely, iNOS expression decreased in the ASC+LEC group (2.3 ± 0.25% and 1.0 ± 0.15% in LEC only and LEC with ASC groups, p < 0.001; Figure 4(b)). The eNOS and iNOS RNA expression results were consistent with those of the immunostaining analysis (eNOS: p = 0.0002; iNOS: p = 0.1003; Figure 4(c)). These results suggest that eNOS and iNOS each contribute differently to the pathways of lymphangiogenesis and angiogenesis and that ASCs can alter their presentation.

Immunofluorescence staining of the NO pathway markers eNOS and iNOS in the LEC only group and group with the combination of LECs and ASCs. (a) Immunohistochemical staining of eNOS in the LEC only and LEC+ASC groups. The quantification of the area affected by eNOS expression (%) is presented. (b) Immunohistochemical staining of iNOS in the LEC only and LEC+ASC groups. The quantification of the iNOS expression area (%) is presented. (c) The relative gene expression levels of eNOS and iNOS were quantified. The scale bar equals 100 µm for all images. The error bars denote the SEs.
Engineered lymphatic tissue successfully bridges lymph node dissection defects
After confirmation of the unique role of LECs in enhancing lymphangiogenesis through the presentation of VEGF-C and VEGFR-3 and promoting lymphangiogenesis without the inclusion of ASCs, our next interest was to investigate the capacity of the engineered vascularized lymphatic tissue to regenerate in the lymphatic defect location via the AV loop model. The AV loop chamber with only LECs was incubated in vivo in the groin region for 2 weeks. Then, extensive groin lymph node dissection was performed on the ipsilateral side. The chamber was subsequently removed, and the engineered tissue inside the chamber was subsequently transplanted as a pedicle flap to the space where the lymph node dissection was performed and removed another 2 weeks later (Figure 5(a)–(c)). The transferred tissue was left for another 2 weeks. Before harvest, ICG was injected into the feet and traced to confirm its presence inside the engineered lymphatic tissue (Figure 5(d)). The presence of ICG inside the engineered lymphatic tissue revealed a prominent spot in the transplanted area and some linear lymphatics connected to the spot, suggesting that regenerated lymphatics connected from the feet to the groin area. The whole picture of the ICG drainage of the transferred group is also closer to the ICG picture of a normal, non-operated rat.
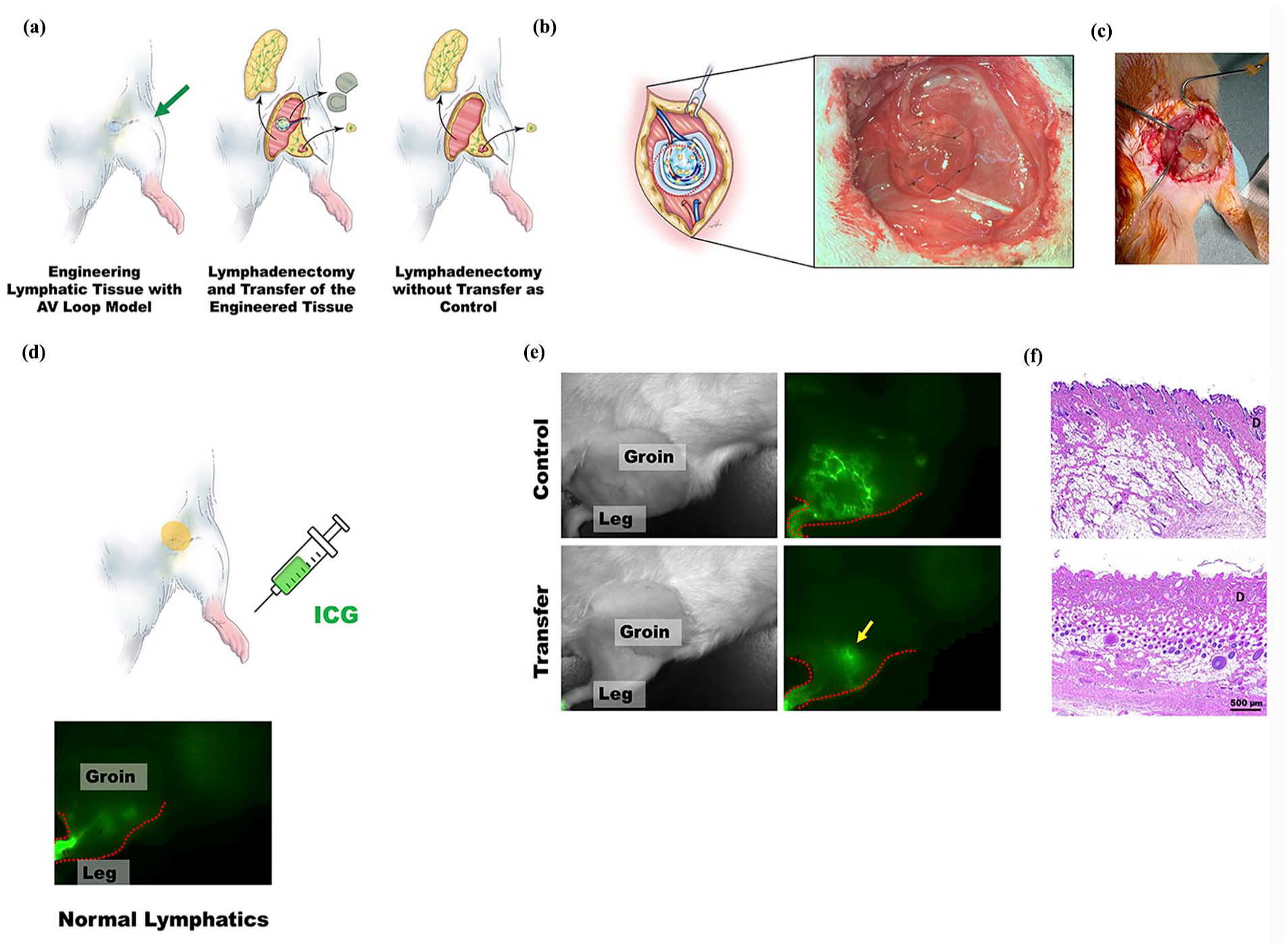
The engineered vascularized lymphatic tissue with the AV loop model in a lymphatic defect location. (a) Sketches showing transfer of engineered tissue inside the AV loop chamber into the space where lymph node dissection (groin and popliteal area) was performed and control group in which lymph node dissection was done without transferring. (b and c) The engineered lymphatic tissue transferred to the defect of lymph node dissection. (d and e) The ICG injection site at the feet showing the tracing and confirmation of the engineered lymphatic vessel tissue in the lymphatic defect location 2 weeks after orthotopic transfer of the engineered tissue around the AV loop. The ICG below the sketch is an example of a normal ICG picture of a normal rat. (f) Hematoxylin–eosin-stained sections of the skin tissue surrounding the lymphadenectomized area of the control rats and the rats with the engineered vascularized tissue with the AV loop after orthotopic transfer. D: dermis layer. The scale bar equals 500 µm.
This means that the engineered lymphatic tissue was able to successfully bridge the lymphatic defect after lymph node dissection (Figure 5(e)). In the control group, the remaining green ICG signal observed in the groin region reflected lymphatic accumulation. The visible lymphatic network indicated that the ICG-labeled lymph was retained within the lymphatic vessels, suggesting impaired or slowed drainage in that region (Figure 5(e)). Similarly, more tube-like structures were found in the dermis of the skin via H&E staining (Figure 5(f)).
Engineered lymphatic tissue restores lymphatic defects and enhances lymphangiogenesis
Tomato lectin was injected subcutaneously into the feet of each rat before the animal was sacrificed. Later, tomato lectin was identified in the skin as well as in the engineered lymphatic tissue (Figure 6(a)). Functional lymphatic vessels, which were identified with double staining of LYVE-1 and αSMA, were counted. Compared with the non-transplanted area, the lymphadenectomy area featuring the transplanted engineered lymphatic tissue presented a greater number of capillary lymphatics and functional lymphatic vessels (LYVE-1: 0.7 ± 0.05% and 1.5 ± 0.09% in control and transfer group, respectively, p < 0.0001, and LYVE-1 and αSMA: 0.7 ± 0.33% and 2.2 ± 0.30% in control and transfer group, respectively, p = 0.0079; Figure 6(b) upper). Similar to the enhancement of lymphangiogenesis, the expression of αSMA was greater in the transferred group than in the control group, suggesting an increase in angiogenesis in the transferred group (αSMA: 10.8 ± 0.83% and 23.7 ± 2.03% in control and transfer group, respectively, p = 0.0002). The transfer of the engineered lymphatic tissue seemed to promote not only lymphangiogenesis but also angiogenesis (Figure 6(b)).

Examination of the lymphatic regeneration capacity of the engineered vascularized lymphatic tissue with the AV loop model in a lymphatic defect location. (a) The tomato lectin injection site at the feet, which was used to confirm the flow of the engineered lymphatic vessel. (b) Immunofluorescence staining of αSMA (red), LYVE-1 (green), tomato lectin (green), and DAPI (blue) in the tissue and skin around the transferred engineered vascularized tissue with the AV loop. The scale bar equals 100 µm. The quantification of the LYVE-1 and/or αSMA (%) is presented. The quantification of lymphatic vessels with tomato lectin expression (number/µm2) is presented. (c) Upper: Immunofluorescence staining of LYVE-1 (red), tomato lectin (green) of the engineered lymphatic tissue after transfer. Lower: Immunofluorescence staining of podoplanin (red), tomato lectin (green) of the engineered lymphatic tissue after transfer. The scale bar equals 100 µm.
Along with the presence of more functioning lymphatic vessels, the presence of tomato lectin of the skin of the proximal hindlimb was greater in the transferred group than in the lymph node dissection only group, suggesting a more rapid lymphatic regeneration and drainage in the transferred group (Lectin: 4.5 ± 0.43 and 10.7 ± 0.84 vessels/µm2 in control and transfer groups, respectively, p < 0.0001; Figure 6(b) lower) The presence of the tomato lectin within lymphatic vessels in the engineered lymphatic tissue was confirmed (double positive of tomato lectin and LYVE-1 or tomato lection and Podoplanin), suggesting that the engineered lymphatic tissue successfully bridged the lymphatic gap in the lymph node dissection area and restored lymphatic drainage from the feet to the groin (Figure 6(c)). The expression of subcutaneous VEGF-C and VEGFR-3 was also elevated in the transferred group, suggesting that the VEGF-C pathway was promoted to enhance lymphangiogenesis after the transfer of the engineered lymphatic tissue (VEGF-C, 0.8 ± 0.05% and 2.1 ± 0.08% in control and transfer group, respectively, p < 0.0001; VEGFR-3, 0.7 ± 0.07% and 1.1 ± 0.04% in control and transfer group, respectively, p = 0.0003; Figure 7(a)).

The immunohistochemical and immunofluorescence staining of VEGF-C, VEGF-R3 and the inflammatory markers CD3, CD4, CD8, and CD68 after implantation of the engineered tissue. (a) Upper: In the Immunohistochemical staining of VEGF-C in the control and transferred groups. The quantification of VEGF-C expression is presented. Lower: Immunofluorescence staining of VEGFR-3 in the control and transferred groups. The quantification of VEGFR-3 expression is presented. The scale bar equals 100 µm. The percentages of VEGF-C and VEGFR-3 expression per field of view are presented. (b) Western blot showing the relative protein expression of VEGFR-3, Prox1, LYVE-1, VEGF-C, podoplanin, and ß-actin in the control and transferred groups. (c) Immunofluorescence staining of the inflammatory markers CD3, CD4, CD8, and CD68 in the tissue surrounding the AV loop-engineered tissue in the control and transferred groups. The scale bar equals 100 µm for all images. The error bars denote the SEs.
The protein expression of lymphangiogenesis-related genes was further confirmed via Western blot analysis. The expression levels of VEGFR-3, LYVE-1, podoplanin, Prox1, and VEGF-C were increased in the group with transferred tissue compared with those in the control group (Figures 7(b) and S2). Overall, immediately transferring vascularized engineered lymphatic tissue to the defect after lymphadenectomy significantly enhanced the regeneration of both lymphatic and blood vessels.
The transfer of engineered lymphatic tissue reduces local inflammation
The immune cells marked with CD3, CD4, CD8, and CD68 were investigated after the transfer of the engineered LEC tissue. There was a greater reduction in the number of immune cells present in the transferred area than in the non-transferred area (CD3: 3.8 ± 0.31 and 1.0 ± 0.26 cells/µm2 in control and transfer groups, p < 0.0001, respectively; CD4: 4.3 ± 0.42 and 0.8 ± 0.31 cells/µm2 in control and transfer groups, respectively, p < 0.0001; CD8: 5.0 ± 0.37 and 1.5 ± 0.56 cells/µm2 in control and transfer groups, respectively, p = 0.0004; CD68: 35.83 ± 8.01 and 17.33 ± 4.41 cells/µm2 in control and transfer groups, respectively, p = 0.0006; Figure 7(c)). Furthermore, the transferred group showed a significant reduction in macrophage numbers, suggesting that implantation of the engineered lymphatic tissue did not trigger an inflammatory response (Figure 7(c)). These findings suggested that the transfer of LEC-engineered lymphatic tissue effectively reduced the inflammatory response, which was triggered by the removal of lymphatic flaps in the groin and popliteal areas.
Discussion
Lymphedema poses a significant therapeutic challenge, as its surgical treatment frequently does not yield the desired improvement and has inherent procedural risks and complications. In advanced stages, the affected tissue undergoes irreversible changes, losing its natural lymphatic properties and experiencing a depletion of healthy LECs. Research is needed to regenerate or engineer new, functional lymphatic tissue to reestablish overall lymphatic function. Various approaches, employing both in vitro and in vivo models, have been explored, incorporating different cell combinations and the addition of growth factors.28,29 In tissue engineering, many existing approaches lack sustainable vascularization, whereas graft vascularization in vivo could enable the creation of a larger tissue volume with consistent tissue survival.30,31 Moreover, the establishment of a vessel network eliminates the dependence of engineered tissue on diffusional properties for nutrients and oxygen, potentially enabling transplantation to specific sites with long-term graft survival. These attributes highlight the importance of the AV loop model 32 as a highly pertinent model for translational research with potential applications in clinical practice. Our previous work demonstrated that the use of an AV loop model in engineering lymphatic tissue resulted in superior lymphangiogenesis compared with subcutaneous or flow-through pedicle models with less hemodynamic flow or no hemodynamic stimuli. 10 The AV loop model presents a promising approach for engineering lymphatic tissue in the clinical treatment of lymphedema.
Previous studies have demonstrated that lymphangiogenesis begins during embryonic development and requires LECs to bud from the cardinal vein. This highlights the critical role of LECs and the vasculature in sending out signals with the extracellular matrix to facilitate differentiation, proliferation, and migration.33,34 Considering the lack of healthy LECs in lymphedema, research has also explored the involvement of ASCs, which have significant potential in vascular generation, in lymphangiogenesis. 35 Direct contact between ASCs and LECs is crucial for ASCs to differentiate into LECs and contribute to lymphangiogenesis in vitro. 14 In certain lymphedema conditions where regional healthy LECs are insufficient, the combination of LECs and ASCs provides a significantly ample cell source for treating lymphatic disorders. In our findings, the group that included LECs alone presented significantly greater expression levels of LYVE-1 and podoplanin and greater coexpression of αSMA and LYVE-1 than did the group that received the combination of ASCs and LECs. These findings indicate that the presence of LECs alone had a more pronounced positive effect on lymphangiogenesis than the simultaneous addition of ASCs and LECs. Interestingly, our results did not show the same trend of enhanced lymphangiogenesis as mentioned in most of the literature when we added ASCs to the scaffold-LEC construct. However, there are some differences between our approach and most of the published methods. We used an in vivo vascular model, while most of the published studies were either in vitro, in the lymphedema area where the LECs were possibly not healthy, or in a nonvascularized model. In addition to these differences, other factors are required to trigger the effect, such as the inclusion of growth factors and the different roles of vascularity. The microenvironment may also induce the different cells to present differently. These issues deserve further study. One more limitation on our study was that we did not include different numbers of ASCs in the study. Further studies can be conducted in the future to investigate its role in an in vivo model.
In addition to their effects on lymphatic regeneration, the addition of ASCs resulted in greater immunomodulation properties. The number of immune cells (CD3-, CD4-, and CD8-expressing cells) was reduced in the group with the addition of ASCs, suggesting that immune reactions were effectively suppressed. Moreover, the inclusion of ASCs in engineered constructs resulted in enhancement primarily at the level of angiogenesis rather than lymphangiogenesis. These results suggested that the combination of LECs and ASCs was not sufficient to promote the process of lymphatic vessel differentiation. In other rodent models, studies have demonstrated that ASCs, in combination with the growth factor VEGF-C, support and augment lymphangiogenesis. 36 Moreover, temporary exposure of ASCs to VEGF-C leads to elevated expression levels of VEGF-A, VEGF-C, and Prox-1, resulting in a notable enhancement of the lymphangiogenic response upon in vivo implantation. 15 Our data further revealed a significant increase in VEGF-C and VEGFR-3 in the LEC-only group compared with the group with both LECs and ASCs. Together, the supply of VEGF-C in the LEC+ASC group might be required for the induction of lymphatic vessel regeneration.
A significant increase in eNOS expression was observed in the group treated with ASCs and LECs, whereas the level of iNOS was lower. In contrast, eNOS and iNOS presented opposite patterns in the LEC only group. The correlation between iNOS and lymphangiogenesis parallels our previous work. 10 The substantial role of iNOS in enhancing lymphangiogenesis is significant and can further be promoted in higher hemodynamic flow and shear stress models and in the presence of LECs. In the present study, the results further suggested that the presence of ASCs can possibly modulate eNOS and iNOS expression in different ways and further determine the differentiation process of lymphangiogenesis and angiogenesis. However, more evidence is required to clarify it. Further study to focus on this issue can be conducted in the future.
For the purpose of treating lymphedema, our next goal was to reestablish the lymphatic system. To do so, extensive groin lymph node dissection followed by orthotopic transfer of the engineered vascularized lymphatic tissue was performed after removing the chamber. As expected, in comparison with lymph node dissection without transfer, the engineered lymphatic tissue successfully enhanced lymphatic regeneration and lymphatic reconnection. Evidence of lymphangiogenesis was based on the increased expression of LYVE-1, podoplanin, and tomato lectin, the double-positive presentation of LYVE-1 and αSMA, and the double-positive expression of tomato lectin and αSMA in the skin histological examination, which was further supported by their protein expression. Moreover, the notable presentation of tomato lectin in the transferred tissue suggests that the lymphatic channel was successfully reestablished from the foot paw all the way proximally to the groin bridging the gap, since the tomato lectin was injected into the foot paw to trace its pathway within the lymphatic channel. Importantly, distal lymphangiogenesis, as the primary response to lymphatic obstruction, was promoted, and the engineered lymphatic tissue successfully bridged the lymphatic gap. The transfer of the engineered lymphatic tissue effectively reconnected ruptured lymphatics to maintain their integrity. Additionally, a local anti-inflammatory effect, as evidenced by a significant reduction in CD3, CD4, and CD8 expression in the transplanted engineered tissue group compared with the group with lymphadenectomy only, suggested the presence of lymphatic functionality in the transplanted area. These findings highlight the creation of an organized functional lymphatic system via our approach. The lymphangiogenesis within the engineered vascularized lymphatic tissue was promoted by the strong arterial flow into the vein graft and the subsequent increased presentation of VEGF-C, VEGFR-3, and iNOS. The whole engineered structure further enhanced lymphangiogenesis and lymphatic reconnection when it was placed in the defect after lymph node dissection (Figure 8). Notably, long-term effects of engineered lymphatic tissue can be expected since engineered lymphatic tissue is vascularized. This approach not only provides ideas for lymphangiogenesis but also opens the possibility of future treatment strategies involving tissue engineering in situ to overcome tissue insufficiency.

Proposed mechanism of lymphangiogenesis of the engineered lymphatic tissue and its role in enhancing lymphatic reconnection after lymph node dissection.
Although promising results can be seen in our study, there remain limitations and some unknown issues to be resolved. Even though the coculture of LECs and ASCs does not result in improved lymphangiogenesis, this effect might be limited to a series of factors, such as the inclusion of growth factors, the cell ratio, the contact time, the model, and the vascularity. Further studies should be conducted to investigate these differences in the future.
Conclusions
In conclusion, the use of LEC-cultivated tissue in AV loop chambers effectively regenerates vascularized lymphatic tissue, marked by increased expression of LYVE-1, podoplanin, SMA, VEGF-C, VEGFR-3, and iNOS. When this engineered lymphatic tissue containing LECs is orthotopically transferred, it has significant potential to connect newly regenerated lymphatic vessels to existing vessels, thus maintaining lymphatic transport following lymph node dissection (Figure 8). This promising concept provides a valuable engineered approach for regenerating lymphatic tissue. The utilization of vascularized engineered tissue not only mitigates the risks and morbidities associated with donor site issues but also allows for the creation of larger constructs customized to meet the varying degrees of severity of lymphedema in patients.
Supplemental Material
sj-docx-1-tej-10.1177_20417314251360755 – Supplemental material for Transplantation of engineered vascularized lymphatic tissue using LEC and in vivo AV loop model to enhance lymphangiogenesis and restore lymphatic drainage in a lymphadenectomy rat model
Supplemental material, sj-docx-1-tej-10.1177_20417314251360755 for Transplantation of engineered vascularized lymphatic tissue using LEC and in vivo AV loop model to enhance lymphangiogenesis and restore lymphatic drainage in a lymphadenectomy rat model by Gina A. Mackert, Hui-Yi Hsiao, Yung-Chun Chang, Robin T. Wu, Rushil R. Dang, Richard Tee and Jung-Ju Huang in Journal of Tissue Engineering
Supplemental Material
sj-tiff-2-tej-10.1177_20417314251360755 – Supplemental material for Transplantation of engineered vascularized lymphatic tissue using LEC and in vivo AV loop model to enhance lymphangiogenesis and restore lymphatic drainage in a lymphadenectomy rat model
Supplemental material, sj-tiff-2-tej-10.1177_20417314251360755 for Transplantation of engineered vascularized lymphatic tissue using LEC and in vivo AV loop model to enhance lymphangiogenesis and restore lymphatic drainage in a lymphadenectomy rat model by Gina A. Mackert, Hui-Yi Hsiao, Yung-Chun Chang, Robin T. Wu, Rushil R. Dang, Richard Tee and Jung-Ju Huang in Journal of Tissue Engineering
Supplemental Material
sj-tiff-3-tej-10.1177_20417314251360755 – Supplemental material for Transplantation of engineered vascularized lymphatic tissue using LEC and in vivo AV loop model to enhance lymphangiogenesis and restore lymphatic drainage in a lymphadenectomy rat model
Supplemental material, sj-tiff-3-tej-10.1177_20417314251360755 for Transplantation of engineered vascularized lymphatic tissue using LEC and in vivo AV loop model to enhance lymphangiogenesis and restore lymphatic drainage in a lymphadenectomy rat model by Gina A. Mackert, Hui-Yi Hsiao, Yung-Chun Chang, Robin T. Wu, Rushil R. Dang, Richard Tee and Jung-Ju Huang in Journal of Tissue Engineering
Footnotes
Acknowledgements
We would like to thank Professor Wayne Morrison for sharing the design of the chamber used in this study. We also would like to acknowledge the Laboratory Animal Center, Chang Gung Memorial Hospital, Linkou, Taiwan, for its assistance in animal housing and care. We sincerely thank Miss Ingrid Kuo and the Center for Big Data Analytics and Statistics at Chang Gung Memorial Hospital for creating the illustrations used herein.
Ethical considerations
All animal procedures were approved by the Institutional Animal Care and Use Committee (IACUC#: 2020122216 and 2023062706) of Chang Gung Memorial Hospital and were performed in accordance with the institution’s animal research guidelines.
Author contributions
Conceptualization: HYH, RT, and JJH; data curation: HYH, YCC, and JJH; formal analysis: GAM, HYH, YCC, and JJH; funding acquisition: HYH and JJH; investigation: GAM, HYH, YCC, RTW, RRD, and JJH; methodology: HYH, YCC, RTW, RRD, RT, and JJH; supervision: HYH and JJH; writing—original draft, GAM, HYH, YCC, RTW, RRD, and RT; writing—review and editing: GAM, HYH, YCC, and JJH. All authors have read and agreed to the published version of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Science and Technology Council in Taiwan [grant: NSTC 110-2314-B-182-047-MY3] and Chang Gung Memorial Hospital [grant: CMRPG3N0951] in Taiwan.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The raw datasets generated within this study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
