Abstract
The construction of bone organoids represents a transformative approach in tissue engineering, offering unprecedented opportunities for studying bone biology, disease modeling, and regenerative medicine. The intricate understanding of the skeletal microenvironment, or niche, which governs cellular behavior, tissue organization, and functional maturation, is critical important to construct bone organoid. This review explored insights into the skeletal microenvironment, including the roles of extracellular matrix components, mechanical cues, biochemical signaling, and cellular interactions. It also proposes a foundational strategy how advancements in biomaterials, extracellular matrix, and micro-structure have enabled the precise recapitulation of niche conditions, facilitating the development of physiologically relevant bone organoids. Furthermore, we highlight the applications of these organoids in drug screening, personalized medicine, and bone regeneration. By bridging the gap between niche biology and organoid engineering, this review underscores the potential of microenvironment-driven approaches to revolutionize bone tissue engineering and its translational impact.
Introduction
Bone-related diseases, including bone defects, osteoporosis, osteonecrosis, osteoarthritis, and bone cancer, pose significant challenges to quality of life and lead to considerable socio-economic burdens globally.1–4 Recent estimates suggest that 20%–33% of individuals across all age groups, particularly the elderly, are afflicted by bone disorders, leading to economic losses amounting to hundreds of millions of dollars annually. To effectively address these challenges, it is crucial to gain a deeper understanding of the mechanisms, etiology, development, and treatment of bone-related diseases, which necessitates the utilization of appropriate research model.
Traditionally, two-dimensional (2D) cell lines have served as the most established and widely used model systems for studying bone-related diseases, playing an important role in modern biomedical advancements. However, 2D cell lines lack the hierarchical structure, dimensionality, cellular diversity, and crucial cell-cell or cell-matrix interactions necessary to accurately simulate cellular functions found in tissues or organs. 5 While animal models can resolve some of these limitations, their utility in bone-related disease research is often restricted due to species differences, individual animal variation, the complexity of model preparations, substantial costs, and ethical concerns associated with animal experimentation.6,7 Three-dimensional (3D) cell models represent a feasible alternative to traditional 2D cell lines and animal models. However, mainstream 3D cell models typically rely on cell scaffolds, 8 which can disrupt cell-cell interactions and the natural arrangement of the extracellular matrix produced by cells. 9 Moreover, the use of scaffolds can provoke inflammatory reactions, induce fibrous encapsulation, and present material toxicity,10,11 thereby limiting the broader application of 3D cell models in bone research. The rapid advancement of organoid technology, defined as the replication of the intricate biological characteristics of original organs or tissues in vivo, offers promising solutions.
Organoid, composed of organ-specific cell types from stem cells, including both embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSC), self-organize through cell sorting and spatially restricted lineage commitment much like in vivo systems. 12 Although organoids are classified as a form of 3D cell model, they significantly differ from traditional models. Scaffold-free, self-assembled organoids can exhibit higher cell density, enhanced matrix deposition and strength, and improved biological function compared to scaffold-based 3D systems. These characteristics enable organoids to effectively mimic the micro-anatomy, physiological activity, and microenvironment crucial to encapsulating the heterogeneity, structure, and function of specific organs. Thus, organoids present substantial advantages as research models for studying organ development and organ-related diseases.12,13
To date, successful organoid development has been reported for various organs, including the lung,14,15 liver, 16 pancreas,16,17 and brain.18,19 Furthermore, techniques are continually evolving to create organoids that replicate the pathological features of tissues or organs under diverse disease conditions.20–22 At the same time, there have been reviews summarizing organoid engineering for medical applications, offering physiologically superior models compared to conventional techniques.23,24 However, the construction and application of bone organoids remain limited. 25 Many challenges persist, including a lack of unified standards and preparation protocols. These challenges hinder the application of organoids as an innovative biotechnology in the research of bone-related diseases. This review aims to provide a theoretical foundation for bone organoid preparation and emerging technologies in the field, thereby promoting their application in bone-related research.
Cellular composition and microenvironment of bone
Bones are intricate tissues that protect and support soft tissues, facilitate mineral homeostasis, and regulate the microenvironment within the medullary cavity. These functions dependent on bone’s unique architecture and physiological processes, which require precise interactions between cells with their surroundings (Figure 1). A comprehensive understanding of the cellular composition and microenvironment of bone is essential for developing bone organoids.

The composition of the bone microenvironment.
The bone microenvironment has a dynamic composition characterized by orchestrated interactions between cellular components (skeletal cells, stem cells, vascular cells, immune cells, etc.) and noncellular components (e.g. extracellular matrix, soluble signals, and vascular networks).
The skeletal lineage
Bone cells can be primarily categorized into two primary groups based on lineage origin and function: the skeletal lineage and the hematopoietic lineage. The skeletal lineage including osteoblasts, osteocytes, chondrocytes, and marrow stromal cells participates in the formation of bone and cartilage and plays a crucial role in maintaining and repairing skeletal homeostasis.26,27
Osteoblasts
Osteoblasts, the primary bone-forming cells, are polarized, cuboidal, mononuclear cells abundant in organelles. They synthesize and secrete a distinctive extracellular matrix composed of osteocalcin, osteopontin, alkaline phosphatase, significant amounts of type I collagen, and growth factors such as bone morphogenetic proteins (BMPs) and transforming growth factor-β (TGF-β). The initially deposited extracellular matrix, known as osteoid, undergoes mineralization through the accumulation of calcium phosphate, primarily in the form of hydroxyapatite, culminating in the formation of bone’s main component: a hard yet lightweight composite of organic and inorganic materials. 26 Within the bone microenvironment, intricate cellular interactions orchestrate various functions, with osteoblasts emerging as key regulators. Notably, osteoblasts influence osteoclast differentiation and bone resorption by expressing genes such as MCsf and Rankl. 28 Moreover, as vital constituents of the hematopoietic stem cells (HSCs) niche, osteoblasts mediate the expansion of HSCs, underscoring their significance in maintaining bone marrow homeostasis.29,30
The development of osteoblast lineage cells, including mesenchymal progenitors, preosteoblasts, mature osteoblasts, bone-lining cells, and osteocytes, follows a complex and dynamic pathway. 29 Osteoprogenitor cells exhibit heterogeneity, with evidence suggesting contributions from both mesenchymal stem cells (MSCs) and skeletal stem cells (SSCs), notably marked by the transcription factor SOX9. The expression of Runt-related transcription factor 2 (RUNX2) in osteoprogenitor cells signifies commitment to the preosteoblast stage. 31 WNT-βcatenin signaling affects preosteoblasts throughout the maturation stage by promoting the production of osterix (OSX; also known as SP7), which specifies the development into an osteoblast. 32 Ultimately, the expression of RUNX2 and OSX marks the commitment to mature osteoblasts. 33 Osteoblasts do not represent the terminal stage of the osteoblast lineage. Upon cessation of active bone matrix formation, osteoblasts may follow one of three potential fates: transition into a quiescent bone, lining cells on the bone surface 34 ; undergo programed cell death via apoptosis 35 ; or become embedded in their own osteoid and differentiate into an osteocyte.36,37
With the rapid development of 3D cell culture technology, a wide variety of 3D osteoblast models have been developed over the last decade, such as spheroids, cell sheets, scaffolds, hydrogels, bioreactors, and microfluidics. 38 Among scaffold-free 3D models, osteoblast spheroids stand out as early endeavors to create bone organoids composed of osteoblasts. For instance, co-spheres comprising human osteoblast-like cells (hOB) and human umbilical vein endothelial cells (HUVECs) were generated using the liquid overlay technique and subsequently embedded in collagen gels to investigate in vitro angiogenesis process. 39 Additionally, Stahl et al. 40 as well as Restle et al. 41 both demonstrated the successful construction of cell spheroids, referred to as bone organoids, using osteoblasts through a similar approaches.
Osteocytes
Osteocytes, which account for up to 95% of total bone cells in mineralized bone, are the terminally differentiated cell type of the osteoblastic lineage and play a pivotal role in maintaining skeletal homeostasis.36,42 As robust responders and translators of mechanical force applied to bone, osteocytes express receptor activator of nuclear factor kappa B ligand (RANKL) during mechanical unloading, which activates osteoclasts, thereby promoting bone resorption. 43 Conversely, in response to mechanical loading, osteocytes reduce the production of Dickkopfrelated protein 1 (DKK1) and sclerostin, which stimulate osteoblasts to produce more bone by activating WNT-β-catenin signaling. 37 In addition to their pivotal roles in regulating bone metabolism and remodeling, osteocytes function as endocrine cells that exert influence not only over bone cells but also over distant organs.36,37 For instance, osteocytes generate osteocalcin, a bone-derived hormone that has recently been discovered to target muscle and affect male fertility, cognitive function, and energy metabolism. 44
There has been a paucity of reports on the direct incorporation of osteocytes into bone organoids. Notably, Wang et al. 45 devised a novel approach in which decellularized matrix (DM) was prepared, employing osteocytes with dominant active WNT-βcatenin signaling (daCO), utilizing a process involving freeze-thaw cycling and DNase I treatment. They found that this decellularized matrix (daCO-DM) exhibited a notable capacity to increase the expression of RANKL, MCSF, Vegfa, Angpt1, and Ngf in bone marrow-derived stem cells (BMSCs), which contribute to osteogenesis, angiogenesis, and neurogenesis, respectively. Then they fabricate metabolic and neurovascularized bone organoids with daCO-DM to accelerate the repair of bone defects. These findings illuminate the potential of harnessing osteocytes, not merely as a direct addition to organoids but also as a resource for the acquisition of decellularized matrixes.
Chondrocytes
Although absent from mineralized bone tissue, chondrocytes are crucial for bone regeneration and repair. Beyond intramembranous ossification, endochondral ossification represents an alternative modality for bone development and regeneration. This process predominantly occurs within the articular/epiphyseal and metaphyseal growth plates and at secondary centers of ossification. Characterized by bone formation via a cartilage intermediary, endochondral ossification differs significantly from intramembranous ossification, where mesenchymal progenitor cells condense and differentiate directly into osteoblasts. In endochondral ossification, mesenchymal progenitors differentiate into chondrocytes and perichondral cells that envelop the cartilage primordium. 46 Chondrocytes within this primordium initially proliferate but eventually exit the cell cycle and undergo hypertrophy after secreting the cartilage matrix. Osteoblasts then differentiate from the perichondral cells. Chondrocyte hypertrophy triggers the osteoblast differentiation, followed by vascular invasion of the cartilaginous tissue and the progression of endochondral ossification. 26
Considering the significant role of chondrocytes in bone development and remodeling, the establishment of cartilage organoids containing chondrocytes or their progenitor cells, also known as bone callus organoids, provides a new strategy for the construction of bone organoids. In prior studies, cartilage organoids were in vivo transplanted, permitting the vascular infiltration that initiated the endochondral ossification process, and ultimately resulting in the successful ossification of the cartilage organoids. These findings imply that the bone organoids development may be achieved through a biphasic approach: cultivating cartilage organoids in vitro, followed by transplantation into an in vivo environment with a well-developed vascular network conducive to ossification. A similar biphasic approach has been demonstrated in the fabrication of brain organoids. Mansour et al. 47 explored a specific xenotransplantation strategy where preliminary human brain organoids were transplanted into the brain of an animal host. The grafts developed functional neuronal networks and blood vessels, indicating the transplanted brain organoid were both matured and vascularized.
In addition to their role as a precursor for bone organoids, cartilage organoids have emerged as a novel strategy for bone repair. They exhibit multifaceted functionalities in wound healing, fracture stabilization, paracrine factor secretion, bone precursor recruitment, bone defect reparation, and facilitation of osteointegration. For instance, Tam et al. 47 generated glycosaminoglycan-rich cartilage organoids utilizing hiPSCs in chondrogenic differentiation medium. These organoids functioned as wound-healing tissues, facilitating scaffold-free repair of long bone defects of critical size. Xie et al. 48 fabricated cartilage organoids with stage-specific gene expression patterns of chondrocytes during endochondral ossification. Combined with BMSCs-loaded hydrogel microspheres (MSs) produced by digital light-processing (DLP) printing technology and stepwise induction, these organoids facilitated rapid bone regeneration.
Marrow stromal cells
Marrow stromal cells(MSCs), which were also referred to as mesenchymal stem cells in the past, are present in the bone marrow cavity. Under specific conditions, they can differentiate into multiple cell types, including osteoblasts, adipocytes, and chondrocytes, 49 which play an important role in tissue repair through various pathways. MSCs can regulate the functions of immune cells, provide a repair microenvironment, and reduce the inflammatory response in damaged areas. When injury occurs, MSCs migrate to the site of injured tissue with the help of chemokines and adhesion molecules, differentiate into specific cells to participate in tissue remodeling, and simultaneously release cytokines and growth factors to engage in tissue repair. 50 The regulation of adaptive immunity intensity is achieved through the secretion of various immunomodulatory factors such as IDO and PGE-2 to inhibit the activation and proliferation of T cells and B cells.51,52 Evidence has shown that MSCs inhibit the maturation and function of myeloid dendritic cells (DCs), promote plasmacytoid DCs (pDCs) to secrete the anti-inflammatory cytokine IL-10, enhance the immunosuppressive microenvironment, and reduce their antigen-presenting ability, thereby exerting an anti-inflammatory effect. 53
In addition, the immunomodulatory function of MSCs exhibits plasticity under different inflammatory conditions. It can either exert immunosuppressive effects or display immunopotentiation effects. When high levels of inflammatory factors (e.g. IFN-γ and TNF-α) are present, MSCs are activated to upregulate immunomodulatory factors and exert immunosuppressive effects. Conversely, when the pro-inflammatory stimulation is insufficient, the immunosuppressive ability of MSCs cannot be manifested, and an immunopotentiation phenomenon may occur, leading to hindrance of tissue repair.50,54 As we know, studies about bone organoids are limited by the inability to replicate skeletal microenvironment, and this challenge may be gradually addressed in the future with increased understanding of MSCs. Zhang et al. combined bone MSCs with hydrogels to form bioinks, employing advanced 3D printing technology to fabricate biologically active bone organoids. After transplantation into an in vivo model, completely differentiated and vascularized bone tissue was eventually formed. 55
The hematopoietic lineage
In addition to the skeletal lineage cells, bone harbor a significant population of hematopoietic lineage cells derived from HSCs, primarily composing myeloid and lymphoid lineages (Figure 2). 56 While skeletal lineage cells play the pivotal role in bone metabolism homeostasis, the hematopoietic lineage, except osteoclasts, predominantly contributes to the mature blood cell population and participates in the inflammatory and immune responses, regulating the equilibrium of the hematopoietic system. Notably, osteoclasts, responsible for bone resorption, are derived from the hematopoietic lineage, whereas skeletal lineage cells are key in forming bone and cartilage. 33

Differentiation scheme of hematopoietic lineage cells.
Hematopoietic stem cells generate all blood components by differentiating into myeloid or lymphoid progenitors. Myeloid progenitors produce platelets, granulocytes, or monocytes, which further transform into dendritic cells, macrophages and osteoclasts. Lymphoid progenitors give rise to T cells, B cells, and NK cells.
Osteoclasts
Bone maintains homeostasis and adapts to factors like activity, age, and disease through continuous remodeling involving bone regeneration and resorption. While the skeletal lineage cells are primarily responsible for bone synthesis, osteoclasts predominantly involved in bone resorption, are derived from HSCs and categorized as myeloid lineage cells. 33
The differentiation of HSCs into osteoblasts requires multiple stages, precisely controlled by a range of cytokines (Figure 2). HSCs give rise to common myeloid progenitor cells (CMPs) under the stimulation of factors such as stem cell factor (SCF), iIL-3, and IL-6. Then granulocyte/macrophage colony stimulating factor (GM-CSF) then promotes the development of CMPs into granulocyte/macrophage progenitor cells (GMPs). Later, GMPs differentiate into monocyte/macrophage lineage cells, which are considered as osteoclast precursors.57–59 The precursors are eventually recruited to a specialized vascular structure called the bone remodeling compartment, characterized by a canopy formed by bone lining cells, where they differentiate into osteoclasts. 60 Monocyte/macrophage colony stimulating factor (M-CSF) and RANKL play a key role during the differentiation from monocyte/macrophage lineage cells to osteoclasts. 61 M-CSF primarily supports the survival and proliferation of osteoclast precursors. while RANKL drives their differentiation into mature osteoclasts. 62 In the final step, osteoclasts bind to the bone matrix through a special structure known as the sealing zone, forming the resorption compartment filled with protons and proteolytic enzymes that dissolve minerals and degrade bone matrix proteins.
While the direct utilization of osteoclasts for the construction of bone organoids remains unreported, their crucial role in bone remodeling has garnered significant attention. Research efforts have focused on incorporating osteoclast precursor cells or stem cells to reinstate osteoclast functionality within bone organoids. Osteoblast precursors (human amniotic fluid MSCs) and osteoclast precursors (human monocytes) were employed to create a bone organoid in a rotational cell culture system by Mandatori et al. 63 Over time, the stem cells in the spheroids differentiated into osteoblast- and osteoclast-like cells with corresponding functions. Similarly, Clarke et al. 64 generated large (>4 mm diameter) 3D mineralized tissue constructs (regarded as bone organoids) from a mixture of human primary osteoblast and osteoclast precursor cells. The histological analysis of those spheroid showed the formation of osteocytes embedded in a mineralized matrix in the core region, surrounded by osteoblasts and osteoclasts. In summary, current reports involving osteoclasts in bone organoids have primarily focused on constructing bone remodeling units in vitro, emphasizing the role of osteoclasts in bone resorption and remodeling. 65
Myeloid and lymphoid cell lines
Myeloid cells residing in the bone marrow play a role in preserving the integrity of the bone and hematopoietic systems. They initiate inflammatory responses, defend against infections, and assist in tissue recovery. The differentiation of myeloid cell lines is a complex process orchestrated by various cytokines, ensuring an effective immune respond to a wide range of diseases and injuries (Figure 2). 66
HSCs differentiate into CMPs, under the influence of cytokines such as granulocyte colony-stimulating factor (G-CSF), GM-CSF, and IL-3. 67 CMPs then diverge into two major lineages: megakaryocyte-erythroid progenitor cells (MEPs) and GMPs. MEPs, driven by erythropoietin, undergo erythropoiesis to produce red blood cells. They are also responsive to IL-3, SCF, and thrombopoietin (Tpo), resulting in the generation of megakaryocytes, which fragment to form platelets upon stimulation by IL-11 and Tpo.25,68 GMPs, on the other hand, differentiate into primitive granulocytes, encompassing neutrophils, eosinophils, and basophils, and can also transform into monocyte/macrophage lineages in response to growth factors, including GM-CSF and M-CSF. 69 Monocytes, in turn, can differentiate into monocyte-derived dendritic cells (DCs) upon stimulation by factors like Flt-3 Ligand, GM-CSF, IFN-α, and IL-4. Notably, DCs are crucial for antigen presentation and initiating immune responses. 70
In addition to myeloid cell differentiation, HSCs also differentiate into common lymphoid progenitor cells (CLPs) under IL-7 stimulation, marking the initiation of lymphoid cell differentiation (Figure 2). 71 CLPs differentiate into B lymphocytes in response to stimulation by IL-3, IL-4, and IL-7 cytokines. 72 Mature B cells, bearing antigen receptors on their cell surface, differentiate into plasma cells and memory B cells upon exposure to microbial antigens. 73 CLPs can also differentiate into T lymphocytes in response to IL-2, IL-7, and Notch. IL-7 functions promotes the survival and proliferation of lymphatic progenitor cells, while IL-2 supports their expansion and commitment to the T cell lineage. Notch signaling is essential for T cell fate specification and guidance. 74 Notably, unlike B lymphocytes, T cell precursors primarily mature in the thymus rather than in the bone marrow.
Programed differentiation and maturation of myeloid and lymphoid cell lines, as well as the self-renewal of HSCs, comprise hematopoiesis. 75 This essential biological process predominantly occurs within the bone marrow, which resides in the marrow cavity and is essential both structurally and physiologically. Therefore, reconstructing functional bone marrow is imperative in the construction of bone organoids.
Currently, studies on bone marrow organoids or organoids encompassing bone marrow tissue are limited. Khan et al. 76 designed a step-wise, directed-differentiation protocol to generate bone marrow organoids from iPSCs committed to mesenchymal, endothelial, and hematopoietic lineages. While these organoids did not recapitulate the anatomical relationship of bone tissue enveloping bone marrow, this limitation can be addressed through organoid assembly methodology in the future. This approach involves separately fabricating bone organoids and bone marrow organoids, followed by their systematic integration to form composite bone organoids containing internalized bone marrow tissue. Another approach was proposed by Dai et al., 77 who established osteo-organoids by implanting scaffolds loaded with bone morphogenetic protein-2 (BMP-2) into the internal muscle pocket near the femur in mice. This implantation recruited stem cells and immune cells, leading to the formation of bone marrow tissue as early as 2 weeks post-implantation. Hence, the construction of ex vivo bone organoids followed by in vivo transplantation to enhance maturation may represent a viable strategy.
Immune microenvironment
The bone marrow cavity contains a dense and intricate vascular network that serves as a pivotal hub for immune cell production, underscoring the vital role of the immune response in bone health. The immune system plays a crucial role in regulating bone physiology, including growth, development, osteogenesis, and osteoblastogenesis, and significantly influences the onset and progression of various bone diseases.
For instance, in the context of postmenopausal osteoporosis, apart from the direct negative effects of estrogen deficiency on bone, indirect effects of altered immune status contribute to ongoing bone destruction. Postmenopausal women often display a chronic low-grade inflammatory phenotype with altered cytokine expression and immune cell profile. 78 Under these conditions, specific subtypes of T lymphocytes upregulate TNF-α expression, while Th17 cells increase IL-17 production. These cytokines have been demonstrated to elevate osteoblast apoptosis and indirectly promote osteoclast differentiation.79,80 Moreover, hyperactivated neutrophils induce osteoblast apoptosis by releasing ROS and promoting osteoclastogenesis through the RANKL signaling pathway. 81 In addition, numerous immune cells and complex immune responses are involved in bone regeneration and repair. 82 Neutrophil aggregation and infiltration occur in bone injuries and play a key role in the early stages of bone repair. During the ossification phase of bone regeneration, macrophages support osteoblast differentiation and proliferation by releasing cytokines including BMP-2, BMP-4, and TGF-β1. 83 Other immune cells, such as DCs, innate lymphoid cells, Th17 cells, and T regulatory cells (Treg), are also involved in the regulating bone regeneration and repair.
Although previous reports on bone organoids did not involve immune cells, the immune microenvironment plays a crucial role within bones, making its the reconstruction in bone organoids significantly important. Bar-Ephraim et al. recently conducted a comprehensive review of research on organoids in tumor immunology, providing valuable insights into the establishment of immune-related tumor organoids. This pioneering work offers an essential reference for potential advancements in the field of bone organoids. 84
Direct culturing of patient-derived cancer organoids while retaining endogenous immune cells has demonstrated viability as a holistic method for in vitro modeling of the tumor immune microenvironment. For instance, Neal et al. 85 cultured a range of cancer organoids, encompassing colorectal and lung cancers, using the air-liquid interface method. They discovered that patient-derived cellular immune components, such as tumor-associated macrophages, TH cells, B cells, natural killer (NK) cells, and natural killer T (NKT) cells, were effectively sustained in the organoid cultures for up to 30 days. Conversely, a reductionist approach involves co-culturing cancer organoids with immune cell subsets that have been isolated and separately expanded. For example, a study successfully confirmed the cytotoxic effect of immune cells on tumor cells by establishing triple co-cultures of mouse gastric cancer organoids with DCs and cytotoxic T lymphocytes (CTLs) derived from the bone marrow or spleens of tumor-bearing mice. 86
Both the direct culturing of organoids retaining endogenous immune cells and the co-culturing of organoids with immune cells present viable approaches for reconstructing the immune microenvironment within bone organoids. In addition, based on the structural and functional foundation of bone marrow tissue within the bone, reconstructing bone marrow tissue within the bone organoids and subsequently directing the differentiation of HSCs toward immune cell lineages present another solution. Nevertheless, the feasibility of this solution requires further studies.
Furthermore, it is imperative to investigate the distinctive alterations in immune cells and cytokine profiles that manifest during the construction of bone organoids. This understanding enhances our comprehension of the growth and developmental patterns of bone organoid and facilitates the more efficient design of organoid construction strategies. To clarify the cytokine secretion dynamics in bone organoid development, Dai et al. 77 screened the proteins in the bone organoid, constructed with BMP-2-loaded gelatin scaffold at different developmental stages via a high-throughput protein array (Figure 3). During the early stages of bone organoid development, an immunosuppressive microenvironment was observed before bone marrow tissue maturation. This was driven by a high CD4+:CD8+ T cell ratio and negative feedback regulatory mechanisms: elevated expression of inflammatory cytokines, including TNF-α and IL-17F, was accompanied by a continuously high expression of anti-inflammatory cytokines, including IL-1 receptor antagonist (IL-1ra) and soluble TNF receptor 1 (TNF R1). They also observed the elevated expression of osteochondral differentiation-associated osteogenic and angiogenic cytokines—such as insulin-like growth factors (IGF-1), osteopontin (OPN), osteoprotegerin (OPG), matrix metalloproteinase-3 (MMP-3), MMP-2, basic fibroblast growth factor (bFGF), and vascular endothelial growth factor (VEGF)—during the maturation of bone organoids. 77

Cytokines with varied expression levels during bone organoid development. (a) Heatmap representing the results of the protein array analysis performed on the supernatant of the native bone marrow and the bone organoid at days 3, 7, 10, 14, and 21. (b) Principal component analysis was performed among all six groups. (c to e) Enriched GO terms of associated biological processes (c), molecular functions (d), and cellular components (e) from all differentially expressed proteins (DEPs) are shown. (f) KEGG pathway analysis for all DEPs. (g–j) Representative protein expression of associated inflammatory and chemotactic effect (g), bone generation (h), hematopoiesis and marrow generation (i), and vascular activities (j) in native bone marrow and the bone organoid. This image is from Ref. 77 and is reproduced with permission.
In summary, the complex interaction between the immune system and bone physiology plays a crucial role in comprehending bone health, diseases, and regeneration. As we venture into the domain of bone organoids, a deeper investigation into their immune microenvironment promises to reveal new horizons in bone organoid research.
Innervation of bone
The mature skeleton is innervated by a complex neural system that infiltrates all types of bone, with a rich nerve network at the periosteum and many nerves accompanying nutritive vessels in the bone marrow and throughout the Haversian system of bone87,88 Furthermore, autonomic and sensory nerve fibers have been recently identified within different regions of bone, such as blood vessels in the periosteum, Volkmann’s canal, bone marrow, and synovial attachments. Dense sympathetic nerve fibers are primarily found in the trabeculae, with a lesser presence in cortical bone and epiphyseal growth plates. 89
Well-established innervation can maintain the stability of the systemic skeletal system by controlling blood flow, regulating bone metabolism, secreting neurotransmitters, and regulating stem cell behavior. The regulatory effects of the innervations upon the skeletal system depend not only on the direct transmission of neural signals but also on the presence of neurogenic factors, released by peptidergic nerves, that subsequently bind to receptors on skeletal cells.90,91 For example, osteogenic differentiation and bone matrix mineralization during bone formation are promoted by substance P (SP) at high concentrations (>10−8 mol/L) but inhibited at low concentrations (<10−8 mol/L).92,93 Neuropeptide Y (NPY) proved to be able to regulate the osteogenic activity of rat MSCs by stimulating Wnt signaling to promote fracture healing. 94 Additionally, paracrine signaling from Schwann cells (SCs), a non-neurogenic factor, has been hypothesized to have consistent effects on bone through a variety of mechanisms. 95
Moreover, the regulation of bone homeostasis by the nervous system is not static; it undergoes adaptive changes in pathological conditions, encompassing osteoporosis, osteoarthritis, heterotopic ossification, psychological stress-related bone abnormalities and bone tumors. Furthermore, in specific instances, the malfunction of the nervous system constitutes one of the key etiological factors underlying the onset of these conditions. On the other hand, the skeleton, a multifunctional complex, also exerts regulatory control over the peripheral nerves within the bone. A broad range of chemical, mechanical, and electrical stimuli provided by the bone orchestrate the physiological activities of intra-bony nerves. Not to mention that bone marrow stromal cells within the skeletal microenvironment may act as an additional cell source for nerve repair. 96
Clearly, in light of the paramount significance of innervation for the skeletal system, it is imperative to reconstruct the neural network within the bone organoid during its initial development to faithfully emulate the regulatory influence of the nervous system on bone homeostasis. Despite the fact that no successful neuralization of bone organoids has been reported so far, the experience of neuralization in other bone tissue engineering research fields is valuable at this point. Implantation of sensory or motor nerve bundles into bone tissue using microsurgical techniques is one of the feasible options to achieve neuralization of tissue-engineered bone. 97 Incorporation of neurotrophic factors such as nerve growth factor (NGF) and glial cell line-derived neurotrophic factor (GDNF) represents another promising way to achieve neuralization of the bone organoid. Multiple investigations have demonstrated that the combination of scaffolds with NGF can effectively promote osteogenesis and enhance the innervation.98–100 It is also considered feasible to construct neuralized bone organoids by co-culturing seed cells possessing neural differentiation capabilities, such as Schwann cells (SCs), alongside cells with confirmed osteogenic potential. This concept is supported by Cai et al.’s research, which demonstrated the feasibility of generating organoids through the co-cultivation of SCs with osteoblasts. 101 Certainly, in the context of in vivo integration, the amalgamation of bone organoids with the host’s neural system to emulate nerve-bone interactions and establish a neurogenic microenvironment is anticipated to effectively address this challenge, much akin to the vascularization of bone organoids in a similar manner. In addition, there have been numerous studies showing that proper electrical stimulation can promote nerve regeneration and repair,102–104 which suggests that those stimuli to the bone organoid may be able to encourage nerve cell growth and maturation. The study of neuralization of bone organoids is still in its early stages, and the above schemes are only reasonable inferences based on the existing results. These inferences have not yet been supported by experimental results, and their feasibility needs to be further verified.
Vascular network of bone
Bone is highly vascularized tissue with a complex and large vascular network system that receives 10%–15% of the total cardiac output. 105 For example, in long bones, the central nutrient artery, metaphyseal-epiphyseal arteries, and periosteal artery comprise the blood vessel network that supplies the bone. 106 At a microlevel, blood vessels localize within the Haversian and Volkmann’s canals in compact bone and extend across the medullary cavity by passing through the trabeculae of spongy bone. 107
Skeletal vasculature plays a significant role in bone development (endochondral and intramembranous ossification), regeneration and remodeling.108,109 Alterations in the vascular network system occur in various orthopedic diseases, including osteoarthritis, 110 osteoporosis, 111 and ischemic osteonecrosis, 112 significantly contributing to their pathogenesis. Vascularized bone organoids serve as valuable in vitro models for studying those diseases, aiding in understanding pathological mechanisms and bone supporting advancements in clinical drug screening. In addition, due to the lack of mature vasculature, the cores of long-term cultured and oversized organoids tend to undergo apoptosis or necrosis because of insufficient oxygen and nutrients supply. This can lead to the formation of dead space or collapse of the organoids, which limits their application. 113 In a word, simulating the vascular network of bone tissue during the construction of bone organoids is necessary.
Addressing vascularization in bone organoids is an imminent research challenge. However, compared with brain organoids and intestinal organoids, vascularization of bone organoids is still in its initial stages. Currently, only a few studies have reported the successful construction of vascularized bone organoids. As early as 2004, Wenger et al. 39 initiated the development of coculture spheroids involving hOB and HUVECs, representing an early effort toward the vascularization of bone organoids. Subsequently, Heo et al. achieved vascularization of bone organoids by embedding mixed 3D cell spheroids of human MSCs and HUVECs in hydrogels. 114 In a separate study, Li et al. 115 substituted HUVECs with dental pulp stem cells (DPSCs) to construct scaffold-free vascularized bone organoids following a similar protocol. Although only a few studies have reported the vascularization of bone organoids, great efforts have been made to achieve the vascularization in brain organoids, which holds significant implications for bone organoids. Organoid co-culture techniques enhance the potential for organoid vascularization. In separate experiments, Ahn et al. 116 and Sun et al. 117 constructed vascular organoids and human brain organoids separately and fused them together to build vascular networks in brain organoids. In the initial stages of organoid construction by Shi et al., 118 brain organoid precursors were created by co-culturing 3 × 106 hESCs/hiPSCs and 3 × 105 HUVECs, ultimately leading to the development of vascularized human brain organoids. Additionally, another study investigated a specific xenotransplantation approach, in which human brain organoids were transplanted into highly vascularized regions of an animal host to vascularize the growing brain organoid. 47 Other technologies such as genetic engineering,119–121 multidirectional differentiation,122–124 microfluidics, and 3D bioprinting are also being applied in this field.125,126
It is worth noting that while the methodologies outlined above have successfully established vascular structures within organoids, these structures exhibit irregularity and disorganization, and lack essential components beyond endothelial cells, such as vascular smooth muscle cells, pericytes, and other constituents. Consequently, the current approaches remain inadequate for replicating the holistic physiological functionality of blood vessels within organoids. In conclusion, the vascularization of bone organoids carries substantial significance, even though it promises to be an extended and challenging undertaking. Valuable experiences from other organoids models should be learned to achieve the vascularization in bone organoids.
Mechanical microenvironment
The mechanical properties of bones are important for the ability of skeletons to support movement and protect vital organs. Bone tissue is continuously subjected to mechanical loads from human activities, including compression, tension, and shear stress at the macroscopic level. 127 At the microscopic level, the porous structure of bone tissue, along with the fluid it contains, generates fluid flow under mechanical loading, result in fluid shear stress (FSS).128,129
Mechanical stimuli are integral to bone homeostasis and regeneration, not only coordinating site-specific activation of osteoblasts and osteoclasts but also influencing stem cell differentiation. 130 For example, high-frequency, extremely low-magnitude mechanical signals bias MSC differentiation toward osteogenesis and away from adipogenesis. 131 Mechanical stimulation, either high strain or low strain with high-frequency has been shown to provide a significant anabolic stimulus to bone. 132 More than that, by utilizing dynamic compression loadings that match developmental stages McDermott et al. 133 demonstrated that mechanical stress promoted the deposition of matrix around cells, contributing to endochondral bone formation. Dzamukova et al.’s team demonstrated that physiological mechanical loading related to body weight transformed highly angiogenic type H vessels into quiescent type L vasculature, resulting in the maturation of bone and vasculature. 134 Mechanical stress is also a promising technique for regulating osteocyte physiological activities, facilitating faster and more efficient bone organoid construction. 135 For instance, applying mechanical loads can accelerate extracellular matrix deposition by stem cells, enhancing organoid stability in the early stages and promoting osteogenic differentiation.131,133 Therefore, mechanical factors are important in bone development, repair, and bone homeostasis, and should be considered in the construction of bone organoids. However, incorporating mechanical factors into bone organoids have not been widely reported yet. Only Zhang et al. 136 verified the role of cyclic compressive loading in bone organoid construction by using a custom-made mechanical stimulation unit (MSU) to apply mechanical loading to 3D bioprinted functional bone organoids. They found that organoid mineral density, organoid stiffness, and osteoblast differentiation significantly increased compared to non-loaded samples (Figure 4(a)).
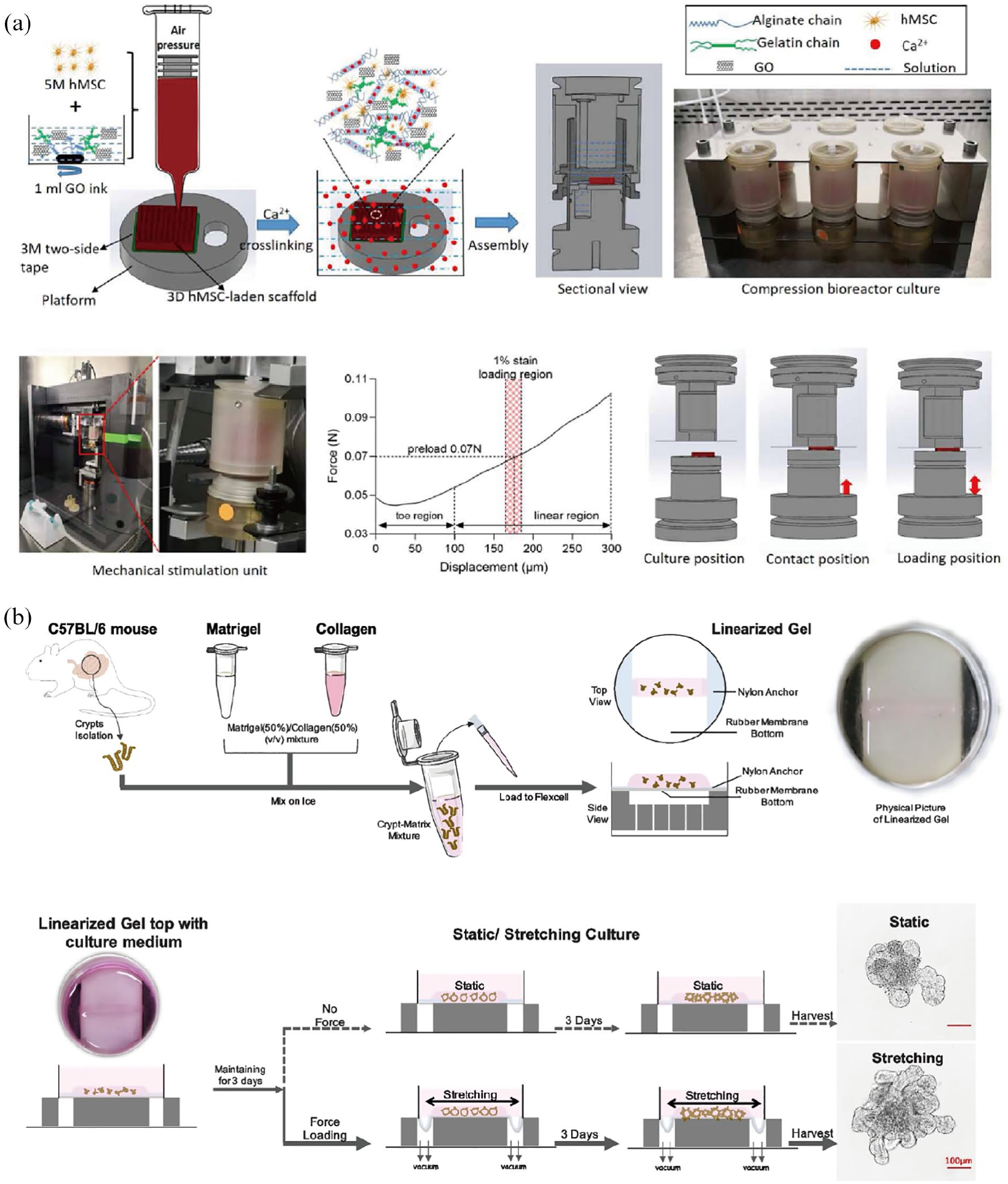
Verification of the influence of mechanical signals on organoid construction using mechanical loading systems. (a) The 3D MSCs-laden scaffold (precursor of bone organoid) was bioprinted with MSCs and graphene oxide (GO)/alginate/gelatin composite bioink, and alginate chains were crosslinked with Ca2+ after bioprinting. Then the MSCs-laden scaffold was cultured with osteogenic medium and assembled in the compression bioreactor. A mechanical stimulation unit was used to apply mechanical loading to 3D MSCs-laden scaffolds during osteogenic differentiation. (b) Schematics of mechanical stretching induced intestinal organoid culture system. Crypts were isolated and mixed with matrix to create gel strips wrapped with intestinal organoids. The linearized gel was sustained for 3-day static incubation, then subjected to stretching for another 3 days. Intestinal organoids in the control group were incubated under static conditions during the whole process. Stretching-induced organoids exhibited an increase in both size and crypt number compared to those in static culture. These images are from Refs.136,137 and are reproduced with permission. Table 1 summarized the recent advances and their significance in organoid research of different lineages and microenvironments.
Recent advances in cell lineages and microenvironments for bone organoid engineering.

The research on mechanical stress in other organoid systems is also still in its early stages. In the skin organoids, Wang et al. have demonstrated the crucial role of mechanical tensile forces originating from dermal cells in the attachment of dermal cells to the epidermal aggregate and the initiation of mesenchymal-epithelial interaction (MEI) during skin organoid development. 139 The mechanical forces of the mammary tumor organoids such as Luminal Pressure and Tangential Stretching Stress have been analyzed by Fang et al., with the conclusion that the mechanical pressure within the organoids increases as the lumen grows and can reach 2 kPa after 2 weeks of culture, which in turn impacts the organoids’ response to doxorubicin and latrunculin A. 140 Meng et al. 140 developed a mechanically dynamic technique involving cyclic stretching for cultivating intestinal organoids (Figure 4(b)). They found that mechanical stretching with refined parameter settings remarkably promoted intestinal organoid stemness, including up-regulation of stem cell marker genes and proliferation of stem cells.
The studies hold significant importance in the study of mechanical stress in bone organoids. Compared with other tissues and organs, the mechanical properties of bone are complex. Factors such as cortical thickness, the spatial distribution of trabecular bone, cross-sectional area, bone size, and bone shape all influence bone mechanics. In addition, the mechanical characteristics of bone are not static; they change with age, activity, disease, and other factors. Hence, simulating the mechanical bone microenvironment within organoids remains highly challenging. In summary, the critical role of the mechanical microenvironment in bone organoid construction is emphasized. While mechanical stress regulates bone metabolism, its application in bone organoids remains limited. Cross-disciplinary insights from other organoid fields (e.g. intestinal models in Figure 4(b)) offer transferable methodologies for stress application systems in bone research.
Construction of bone organoid
Construction process of bone organoid
The construction of bone organoids typically involves several key steps141,142 : (1) Cell Source Selection, where mesenchymal stem cells (MSCs) or induced pluripotent stem cells (iPSCs) are commonly used due to their osteogenic potential(Figure 5(a))76,143; (2) Scaffold Preparation, utilizing biomaterials such as hydrogels (e.g. collagen, alginate, or Matrigel) or synthetic polymers to provide a 3D microenvironment mimicking the extracellular matrix(Figure 5(b))144,145; (3) Osteogenic Differentiation, achieved through biochemical cues like growth factors (e.g. BMP-2, TGF-β) and mechanical stimulation (e.g. cyclic loading or perfusion bioreactors)146,147; (4) Maturation and Vascularization, where co-culture with endothelial cells or angiogenic factors enhances vascular network formation55,148; and (5) Characterization, involving histological, molecular, and functional assays to validate bone-like tissue properties (e.g. mineralization via Alizarin Red staining, osteogenic marker expression like RUNX2 and OCN). Recent advances also incorporate bioprinting or microfluidic systems to improve spatial organization and scalability (Figure 5(c)).25,149

The process of construction strategies of bone organoids. (a) Multiple cell source for fabrication of bone organoids. (b) Extracellular matrix and biochemical cues for bone organoids preparation. (c) Fabrication techniques for constructing bone organoids.
Seed cells for bone organoid
The appropriate selection of stem cells becomes a crucial step in bone organoid construction. In vivo, the regeneration and repair are initiated in response to factors compromising bone tissue integrity, with osteogenic progenitors, not mature osteoblasts, being the primary source of osteoblasts involved in bone regeneration. BMSCs, as a highly dynamic and stress-responsive heterogeneous stem/progenitor cell population, have proven to be the osteogenic progenitors that meet the high cell replacement demands of bone maintenance and regeneration. 150 The significant role of BMSCs in bone repair, along with their easy accessibility and low cultivation costs compared to other stem cells, makes BMSCs the preferred choice for the construction of bone organoids. The feasibility of this approach has been substantiated by numerous independent studies.151–153 Certainly, other autologous stem cells with osteogenic differentiation potential, such as SSCs, 154 adipose-derived stem cells (ADSCs), 155 periosteum-derived cells (PDCs), 156 and the like, hold promise for bone organoid development.
As organoid technology advances and bone organoid applications expand, the construction of bone organoids has become more intricate, like osteoblasts, osteoclasts, immune cells, vascular endothelial cells, and more.40,64,101,115 To meet these requirements, seed cells must exhibit pluripotent differentiation potential or require the combined application of multiple types of stem cells. The limited differentiation potential of BMSCs alone is insufficient for this purpose. Although ESCs have pluripotent differentiation potential, their extensive use in the field of organoid generation is restricted due to ethical issues.
In contrast, iPSCs are a promising source of patient-specific stem cells with high efficacy and fewer immune rejection and ethical concerns. 157 Furthermore, owing to the self-renewal activity, iPSC-derived organoids can be generated in an unlimited manner, solving problems such as scarcity of donors, unity of organoid quality, risk of disease transmission. Currently, iPSCs are extensively employed in organoid construction,158–161 with ongoing new attempts in the field of osteochondral research. For example, both O’Connor et al. 162 and Limraksasin et al. 163 successfully prepared osteochondral organoids with mouse iPSCs using a stepwise induction method. Yamashita et al. 164 pioneered the utilization of BMP-2, TGF-β1, and GDF5 to induce cartilage spheroid construction from human iPSCs in scaffold-free suspension culture conditions. Likewise, Abe et al. 157 achieved cartilage defect repair in primate knee joints by constructing cartilage organoids with iPSCs. The widespread use of iPSCs undoubtedly provides more possibilities for bone organoids, making them an excellent choice for stem cells alongside MSCs.
As previously indicated, the function of bone and the maintenance of its homeostasis rely on the intricate physiological structure and cellular composition inherent to bone tissue. Consequently, the construction of bone organoids necessitates the coordination between various cellular components and the multidirectional differentiation of stem cells. The composition of cell types in bone organoid construction requires tailoring based on the intended application. For example, when constructing bone organoids for screening drugs for osteoporosis, the inclusion of both osteoblasts and osteoclasts is necessary. However, for tissue engineering purposes, the emphasis is typically on enhancing osteogenesis, leading to minimal consideration for osteoclast inclusion in bone organoid construction.
Extracellular matrix
Supplementary components are essential for maintaining organoid stability. The extracellular matrix (ECM), a pivotal non-cellular component, serves as the organoid’s framework, ensuring stability, and facilitating the proliferation and differentiation of seed cells (Figure 5). 165 Stem cells, such as ESCs and iPSCs, as well as organ progenitors, can autonomously organize into organoids via cell sorting and spatially restricted lineage commitment. 12 However, at the early stage of organoid construction, these stem cells may not produce sufficient ECM to ensure structural stability, necessitating the addition of exogenous matrix components. 166
The ECM is in direct contact with cells and serves as the primary pathway for nutritional exchange and waste removal between cells and their culture environment, as well as the main way for drug delivery into cells. Therefore, any additional ECM must meet the criteria: (1) be highly biocompatible, non-toxic, and nonimmunogenic; (2) have excellent mechanical properties; (3) be biodegradable; (4) support and improve cellular life activities including cell differentiation, proliferation, adhesion, and migration; (5) mimic the specific microenvironment of various cells; and (6) interact well with cells to update their components. 167 Currently, the most common used exogenous ECMs are animal-derived commercial hydrogels, such as Matrigel and basement membrane extract (BME). 167 These matrixes are obtained from the basement membrane produced by Engelbreth–Holm–Swarm mouse sarcoma cells, 168 leading to variable compositions and potential impurities. Furthermore, their availability can be influenced by supplier monopolies, leading to high costs. These issues significantly impede organoid research and applications, prompting substantial efforts to identify alternative substitutes for these matrixes.
Therefore, synthetic hydrogels of hydrophilic polymers including polyethylene glycol, polyvinyl alcohol, polylactic acid, and polyacrylamide have become better alternatives. 169 Recently, Gai et al. developed a novel engineered bionic matrix hydrogel containing calcium phosphate oligomers (CPO), decellularized extracellular matrix (ECM), and salmon-derived deoxyribonucleic acid (DNA). Through promoting BMSC proliferation and network formation, BMSC-loaded biomimetic hydrogel matrices facilitate the sequential construction of vascularized and mineralized bone organoids within in vitro dynamic culture systems and in vivo heterotopic ossification models. 147 These hydrogel systems provide a favorable microenvironment that mimics the native extracellular matrix, facilitating spatial-temporal coordination between osteogenesis and angiogenesis. Such findings highlight the potential of bioengineered materials to mimic key physiological cues necessary for bone organoid engineering.
Application of decellularized extracellular matrix
Decellularized extracellular matrix (dECM) refers to biomaterials formed by human or animal organs/tissues after the removal of immunogenic cellular components via decellularized technologies, which is another choice for additional extracellular matrix. 170 Composed of extracellular macromolecules such as collagen, elastin, fibronectin, laminin, and matricellular proteins, dECM maintains its physicochemical signals and biological properties even after decellularization, making it an ideal mechanically supportive substrate and 3D bio-support for subsequent cell seeding.171,172 Capitalizing on these traits, dECM finds extensive application in organoid construction, such as in the development of brain, 173 intestine, 174 and kidney organoids. 175 While direct utilization of dECM in bone organoid construction has not been reported, dECM has found extensive applications in bone tissue engineering. The versatility of dECM allows for diverse uses, ranging from whole tissue scaffolds and microparticles to digested solutions that can be employed as bio-ink for 3D printing. 176 These findings suggest the feasibility of utilizing dECM for more rapid, cost-effective, and efficient bone organoid construction.
Application of fibroblasts
Fibroblasts are primarily responsible for maintaining the ECM and serving as the building blocks for tissue and organ maintenance or remodeling. Therefore, enhancing ECM secretion through the inclusion of fibroblast cells in organoids represents a dependable approach. For example, Tan et al. 177 built airway organoids by combining lung fibroblasts, human adult primary bronchial epithelial cells, and lung microvascular endothelial cells in supportive 3D culture conditions. A heart microtissue organoid composed of human-induced pluripotent stem cell-derived cardiomyocytes (hiPSC-CMs) and human cardiac fibroblasts (hCFs) was constructed by Xu et al. 178 It was found to have higher mechanical strength than organoids without hCFs and could be cultured in vitro for up to 68 days. The bone organoids composed of human umbilical cord blood-derived fibroblasts (CB-BFs) signifies a substantial advancement in the field of regenerative medicine, particularly in contexts necessitating the reconstruction of the hematopoietic stem cell (HSC) niche.179,180 Their inclusion may hold promise for future applications in bone organoid construction.
Macromolecular crowding effect
In recent years, macromolecular crowding has been applied to tissue engineering. 181 Introducing soluble macromolecules such as Dextran Sulfate or Ficoll into the culture medium induces macromolecular crowding, which impacts the behavior of neighboring molecules. This effect reduces the available volume and conformational freedom of neighboring molecules, resulting in a more compact protein conformation. Macromolecular crowding accelerates ECM assembly, promotes a native matrix architecture, enhances matrix remodeling, and reduces cellular phenotypic drift.182–185 This effect has been verified in a variety of cells cultured in 2D, such as chondrocytes and BMSCs, but its applicability in 3D models lacks sufficient evidence. The study by Chen et al. 183 demonstrated enhanced total collagen and glycosaminoglycan production in porcine chondrocytes in monolayer cultures (2D model) due to macromolecular crowding, while matrix production and tissue formation were significantly inhibited in in vitro engineered cartilage (3D model) by these macromolecules. Conversely, Lareu et al. 186 observed a 20–30 fold increase in collagen deposition in fibroblasts in 2D culture conditions and a 3–6 fold increase even in 3D models with the application of macromolecular crowding. Utilizing macromolecular crowding to enhance ECM secretion for the rapid construction of stable organoids is a novel approach, contingent on additional investigation to validate its viability.
Supplementary components of the extracellular matrix
The interaction between cells and the ECM in organoids regulates cellular behaviors, including adhesion, migration, proliferation, differentiation, and apoptosis. These effects are contingent upon the ECM’s structure and composition (Figure 6). Given the distinct cell composition and differentiation characteristics among different organoids, their requirements for the ECM vary from one another. Therefore, supplementary components, particularly bioactive factors like TGF-β1 and FGF, are essential for the ECM. In addition, the physical characteristics of the ECM should be adjusted to meet the specific needs of organoids. For instance, bone organoids necessitate an ECM with higher stiffness, less elasticity and ductility, which can be achieved by incorporating elements such as laminin, fibronectin, and synthetic polymers like PEG and its derivatives. 167

Stem cells interact with the surrounding ECM.
The extracellular matrix is a complex structure located outside the stem cell. It is composed of various molecules, including proteoglycan, collagen, and other biological molecules. The ECM forms a supportive framework around stem cells and plays a crucial role in shaping stem cell morphology, proliferation, differentiation, migration, and signal transduction.
By incorporating engineered microenvironmental strategies, organoids are capable of establishing gradients of nutrients, oxygen, and signaling molecules, thereby facilitating the coordinated integration of immune modulation, angiogenesis, and osteogenesis. Recent studies have shown that bone marrow-like organoids can be generated on microporous hydrogel platforms with the assistance of natural bone niche components. 187 Structurally, these organoids develop self-organized, vasculature-like networks, providing strong support for the advancement of vascularized bone organoid construction. 188 Modulation of the surrounding physical microenvironments can induce either reversible or irreversible alterations in the extracellular matrix’s physical properties or chemical composition, thereby directing cell fate decisions and enhancing bone tissue repair and regeneration. Notably, recent investigations have further demonstrated that re-establishing the physical microenvironment through electrical stimulation significantly promotes bone regenerative processes. 189
Structure
Most organoids reported to date are spherical because, in the absence of scaffolds, cells naturally aggregate together to form spheroids due to surface tension. However, the spherical shape has limitations; for example, the excessive volume of spherical organoids often leads to apoptosis or necrosis in the core due to insufficient nutrient and oxygen supply, and the spherical shape not adequately simulate physiological characteristics of certain organs or tissues. Clearly, shape and structure, as one of the key features of organoids, significantly impact their physiological and morphological characteristics. Unfortunately, the majority of research on organoids overlooks the importance of the structure and fails to consider the potential for generating heteromorphic organoids. Cell self-assembly in non-adherent agarose hydrogel molds is an important method for organoid fabrication.190–192 This method enables the fabrication of heteromorphic or even polymorphic organoids, including loop-ended,153,166,193–196 and honeycomb-shaped197,198 expanding beyond the common spherical shape (Figure 7(a)–(c)). Comparing with honeycomb-shaped design, loop-ended design exhibited advantages such as simplified biofabrication process, enhanced molecular binding via curved surfaces, better simulation of capillary curvature, and potentially improved nutrient diffusion kinetics.

Design and Applications of heteromorphic Organoids. (a) Spheroids, rods, ring-shaped, and honeycomb-shaped self-assembled cellular structures that meet the definition of organoids were fabricated using micromolded, nonadhesive agarose hydrogels. (b) Schematic overview of the 2-phase screening approach to construct pancreatic duct-like organoids from hPSCs, and the bright-field images of organoids during differentiation. (c) Schematic overview for constructing cartilage tubes by stacking ring-shaped cartilage organoids longitudinally to meet the structural requirements for tracheal repair. Macroscopic and histological images of cartilage tubes of different inner diameters are shown in (d); GAGs were stained with Safranin O (pink/red); immunohistochemistry was performed for human type II collagen (red). (e) Marsha’s team developed MSCs tubes containing microparticles loaded with TGF-β1 and BMP-2 to repair bone defects, following a similar approach as shown in (c). Representative histological staining of those MSCs tubes, including Hematoxylin & Eosin (H&E), Safranin-O/Fast-green (Saf-O), and Alizarin Red S (ARS), is presented in (f). These images are from Refs.153,194,197,199 and are reproduced with permission.
In the following section, we consider loop-ended organoids, also named ring-shaped organoids by ourselves, as an illustrative case to clarify the influence of distinctive structure on physiological and morphological traits, and investigate their unique applications in contrast to conventional spherical organoids. Marsha’s team pioneered the creation of self-assembled tissue rings by seeding cells into non-adhesive agarose wells.190,191 A polydimethylsiloxane (PDMS) negative was first cast in 3D-printed plastic, and then agarose was gelled in the PDMS negative to create ring-shaped cell seeding wells. 190 Various cell types, including human MSCs and smooth muscle cells, can be introduced into the cell seeding wells. Within 24 h, they self-assemble into tissue rings, which serve as precursors to ring-shaped organoids (Figure 7(c)).153,166,193–196 Notably, Marsha’s team referred to the structures created by this method as “tissue rings” rather than ring-shaped organoids. However, the preparation process and properties of these “tissue rings” closely align with the organoid definition, and therefore, this review incorporates them within our scope.
Distinct from spherical organoids, ring-shaped organoids offer a key advantage by preventing core necrosis. In organoids that are not adequately vascularized, the core often undergoes apoptosis or necrosis due to inadequate nutrient supply and hypoxia, resulting in a dead space during prolonged in vitro culture with excessive volume. 113 The ring-shaped structure significantly increases the organoid’s surface area comparing with spherical organoids, facilitating efficient nutrient exchange and waste removal, making the organoid more stable in long-term in vitro culture.178,200 Meanwhile, it also greatly increases the limit size of organoids theoretically. Importantly, the ring-shaped structure can be scaled up through self-assembling and automatically fusing into a larger size, forming a highly regular and reproducible system. 178 This allows the application of ring-shaped organoids to be further expanded. For example, by stacking ring-shaped organoids longitudinally, tubular organoids can be formed to meet the structural requirements of organs or tissues like the trachea (Figure 7(d)),194,196 blood vessels, 200 glands, 199 and long bones (Figure 7(e) and (f)).153,166 Furthermore, ring-shaped organoids with different diameters can be forge into one complex organoid that has multiple layers. For example, combining vessel organoids within ring-shaped bone organoids can simulate long bones with medullary cavity structures, while overlapping ring-shaped smooth muscle organoids with endothelial cell rings can replicate more intricate vascular structures.
In addition to fabricating heteromorphic organoids within molds of different shapes through cell self-assembly and automatic fusion, gene editing presents a viable approach. For example, Breunig et al. 199 developed a protocol that gave rise to a population of homogeneous pancreatic duct-like organoids from hiPSCs by upregulating the KRT19 gene, recapitulating the pancreatic lineage in vitro (Figure 7(b)). Furthermore, organoid reassembly techniques offer more possibilities for creating organoids with intricate structures. Liu et al. 201 obtained upscaled, scaffold-free airway tubes with predefined shapes by assembling airway organoids of varying sizes, employing a technique called “Multi-Organoid Patterning and Fusion.”
In the aspects of constructing bone organoid structures, Wang et al. constructed a cylindrical large-scale self-mineralizing bone organoid. Their study showed that this organoid has good self-mineralizing capacity and multicellular differentiation potential. 145 The integration of a hierarchical Haversian bone structure within a Haversian bone–mimicking scaffold revealed its considerable potential for facilitating multicellular delivery, effectively inducing differentiation along osteogenic, angiogenic, and neurogenic pathways. 202 Similarly, researchers successfully combined various bone cell types to create an organoid capable of generating new woven bone autonomously. These woven bone organoids exhibit spatiotemporal architectures supporting dynamic mineralization and tissue remodeling.203,204
In summary, organoids, as scaffold-free 3D cell culture technology, stand out due to their diverse structures. This structural adaptability caters to the demands of simulating the physiological characteristics of different tissues and organs, establishing a robust foundation for the extensive utilization of organoids in bone research.
Challenges and prospective
Bone organoids, created from stem cells and additional cellular components, often exhibit astonishing resemblances to actual bone, both in terms of microscale tissue architecture and the cellular diversity and key functions.162,204–206 However, challenges remain to maximize their translational relevance and applicability (Table 2).
Current challenges and solutions in bone organoid research.

Currently, organoid construction relies on extracellular matrixes or alternative biological materials, facilitating the transition from 2D to 3D cell cultures. Consequently, the application of bone organoids in clinical settings remains hindered by issues related to the potential toxicity and degradability of these biomaterials. Furthermore, additional extracellular matrixes, such as Matrigel and BME, rely on supply from manufacturers, leading to higher costs compared to traditional two-dimensional cell culture systems and impacting the consistency of the organoids due to the quality control of additional biological materials. To address these challenges, exploring new alternative materials or utilizing cell self-assembling and matrix-free organoid construction protocols may represent novel solutions.
Moreover, due to the lack of a mature vascular system, long-term cultivation and excessive volume of organoids often lead to apoptosis or necrosis in the core. This limitation restricts the potential applications of bone organoids, particularly in the field of tissue engineering, which aims to produce large-scale in vitro tissues and organs for transplantation to replace damaged or diseased organs. While limited studies have addressed vascularization in bone organoids, lessons from successful vascularization in other organoids offer valuable insights. These strategies include co-cultivation with vascular organoids, 116 the introduction of vascular stem cells and growth factors, 118 xenotransplantation, 47 and optimization of organoid structure. 207
One remaining challenge is the comprehensive characterization of all established bone organoids to determine their fidelity in recapitulating in vivo development. Decellularised bone matrix (DBM) has been used to create a bone perivascular niche. The channel in these devices contains bovine DBM and is seeded with hBMSCs and ECs. This supported ECs forming capillary networks throughout the bone matrix and hBMSCs adopting a perivascular role, mimicking one of their roles in vivo. 208 Gai et al. recently developed a novel engineered biomimetic hydrogel derived from decellularized extracellular matrix of bone tissue, which enables the sequential vascularization and mineralization of the constructed bone organoids, thereby facilitating efficient bone regeneration in critical-sized defects. 147 The mechanical load-bearing capacity of bone organoids has emerged as a prominent focus in current research. Recent studies have demonstrated that assembling pure silk fibroin (SF) into a layered three-dimensional scaffold enables the construction of functional bone organoids suitable for repairing large segmental bone defects (LSBD) in load-bearing regions. 209
Bone organoid seed cells often exhibit limited cell phenotypes and lack actual functionality following in vitro differentiation. Contributing factors to this issue may include the ill-defined growth environments within the organoids, leading to uncontrolled differentiation of stem cells, along with a deficiency in synergistic interactions among different cells, tissues, and even organs.
Despite these challenges, there remain fascinating possibilities for the advancement of this growing biological technique. Multiple studies have demonstrated the potential of organoids in various areas, including disease modeling, personalized medicine, drug screening and development, regenerative medicine, and gene therapy. Meanwhile, novel applications for bone organoids continue to emerge. For example, in a recent study by Dai et al., 77 osteo-organoids were utilized as a “cell factory” to consistently produce ample high-quality autologous cells for cell therapies. Dai et al. had proposed an in vivo osteo-organoid approach for obtaining high-quality and large amounts of therapeutic hematopoietic stem/progenitor cells for Hematopoietic stem cell transplantation using bioactive molecules-loaded scaffolds. 210 In the study by Qiao et al., the construction of a neuro-vascularized bone organoid was achieved using a double small interfering RNA-loaded cell membrane-coated scaffold, which attenuates inflammation and promotes osseointegration by regulating the coupling of vascularization and innervation in vivo. 211 In summary, as an emerging biomedical technology, organoids hold great promise in the field of bone-related research, despite the obstacles that lie ahead.
Footnotes
Author contributions
Y.X. and L.S. wrote the manuscript. M.Z. contributed to the graphic generation, Z.H. contributed to the literature collection. H.W. conceived the topic and revised the manuscript. X.Y. revised the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Hunan Province Natural Science Foundation (NO. 2023JJ60467) and Zhejiang Province Basic Public Welfare Research Program (TGD24H160005). The drawing materials were supported by BioRender.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to this research, authorship, and/or publication of this article.
