Abstract
Cell sheet engineering provides a scaffold-free strategy for fabricating cohesive tissue constructs, but challenges remain in maintaining structural integrity and mimicking complex tissue architectures. This study demonstrated perfluorodecalin-based liquid-liquid interfaces, known for their inertness and stability, as a simple, and efficient platform for fabricating cell sheets. Using single cells, spheroids, and their combination, we evaluated methods to enhance sheet formation. Single cells formed cohesive sheets at high densities (4 × 106 cells/well) but exhibited limited long-term stability due to nutrient constraints. Spheroids formed robust sheets at lower densities (2 × 106 cells/well), whereas higher densities impaired fusion. The mixed approach combined the advantages of both, improving uniformity, mechanical stability, and spheroid fusion, while mimicking muscle-like structures with vascular networks. Additionally, the cell sheets retained adipogenic and chondrogenic differentiation potential, highlighting their functional viability. These findings establish liquid interfaces as a practical and versatile platform for tissue engineering, regenerative medicine, and in vitro modeling.
Keywords
Introduction
Cell sheet engineering has emerged as a transformative strategy in regenerative medicine, offering a scaffold-free platform to fabricate cohesive, functional tissues.1,2 Unlike traditional three-dimensional (3D) constructs that often rely on biomaterial scaffolds, cell sheets preserve native extracellular matrix (ECM) components and cell-cell junctions, which are crucial for maintaining tissue functionality, and integration upon transplantation.3,4 Clinically, this approach has shown promise in repairing cardiac,5,6 corneal, 7 hepatic, 8 and skin tissues 9 by enabling the transplantation of viable, structurally intact cell layers without eliciting significant immune or inflammatory responses. 10
Despite its advantages, conventional methods for fabricating cell sheets—such as temperature-responsive culture surfaces11,12 or electrical stimulation
13
—are mostly dependent on a
Perfluorocarbon (PFC) interfaces have emerged as a novel cell culture platform due to their inertness, high gas solubility, and ability to provide a non-adhesive liquid-liquid interface.19–25 However, unlike solid substrates, the absence of a physical anchoring surface necessitates rapid and sufficient cell-cell adhesion to prevent cell death by anoikis or diffusion away from the interface.16,26 This limitation necessitates the exploration of alternative strategies to enhance cell-cell contact and promote ECM deposition.
To address these limitations, we hypothesized that
In this study, we systematically investigated these three strategies—(i) single-cell-based, (ii) spheroid-based, and (iii) combined-cell sheet fabrication-on PFC-based liquid-liquid interfaces. We compared their efficiency in terms of morphology, cohesion, viability, and differentiation potential. Furthermore, we explore the application of this platform in fabricating
Materials and methods
Cell culture methods
In this study, human immortalized adipose-derived stem cells (hADSCs; ASC52telo, SCRC-4000, ATCC, USA), human skeletal muscle cells (hSKMCs; Promocell, Germany), and human umbilical vein endothelial cells (HUVEC)-TERT2 endothelial cells (ATCC, CRL-4053) were used to investigate methods for cell sheet fabrication. hADSCs were cultured in Mesenchymal Stem Cell Basal Medium (ATCC, PCS-500-030) supplemented with MSC Growth Kit (ATCC, PCS-999-038), L-Alanyl-L-Glutamine (ATCC, PCS-999-034), and 1% Antibiotic/Antimycotic solution (100 U/mL penicillin, 100 μg/mL streptomycin, 0.25 μg/mL amphotericin B; Hyclone, USA). HUVEC-TERT2 endothelial cells were maintained in Vascular Cell Basal Medium (ATCC, PCS-100-030) supplemented with a Vascular Endothelial Cell Growth Kit (ATCC, PCS-100-040), which includes growth factors essential for endothelial cell proliferation and maintenance. hSKMCs were cultured according to the manufacturer’s protocol using skeletal muscle cell growth medium (Promocell, C-23060). All cell lines were maintained in a humidified incubator at 37°C with 5% CO₂. Culture media were replaced every 48–72 h to ensure optimal growth conditions. Cells were passaged at 80% confluence using Trypsin-EDTA (ATCC, PCS-999-003). Prior to experiments, cell counts and viability were confirmed using a hemocytometer and the trypan blue exclusion method.
Preparation of spheroids
Spheroids were generated using ultra-low attachment (ULA) culture dishes with integrated mesh (35 mm; LabToLab Co., Korea), designed to prevent cell adhesion and facilitate uniform spheroid size. Adherent cells were harvested from monolayer cultures and resuspended in fresh culture medium at a density of 1 × 106 cells/3 mL. The single-cell suspension was seeded into the ULA dishes at a total volume of 3 mL per dish. Cultures were maintained in growth medium specific to each cell type. Fifty percent of the medium was replaced every 48 h to ensure consistent nutrient supply without disturbing spheroid formation. Spheroid growth was monitored daily using an optical microscope (Leica DMi8, Germany). The diameter and morphology of spheroids were documented using calibrated imaging software.
Dissolved oxygen measurement
To assess the oxygen delivery capacity of PFC, perfluorodecalin was pre-oxygenated by bubbling 100% O₂ for 10 min. The dissolved oxygen concentration in culture medium above the PFC interface was measured over a 48-h period using a NeoFox Phase Fluorometer (Ocean insight®) and a fiber-optic oxygen sensor (Probe: FOSPOR-R) in a humidified incubator at 37°C with 5% CO₂. A two-point calibration was performed prior to measurements. The zero-point calibration was performed using 100% nitrogen gas, and the second point calibration was performed using ambient air with known oxygen concentration. The liquid sample was placed in a clean container and the sensor probe was immersed to a constant depth to minimize interference due to surface oxygen exchange. Results were compared with non-oxygenated PFC well and control wells without PFC.
Fabrication of cell sheets on liquid-liquid interfaces using perfluorodecalin
A PFC interface using perfluorodecalin (C10F18, Sigma, P9900, USA) was established to create a liquid-liquid interface for cell sheet formation. For all fabrication methods, 300 μL of perfluorodecalin was carefully dispensed into each well of a 48-well plate to establish a stable liquid-liquid interface.
For the fabrication of single-cell-based sheets, cell suspensions were prepared at various concentrations (1 × 10⁵, 5 × 10⁵, 1 × 106, 2 × 106, 3 × 106, and 4 × 106 cells/well) to determine optimal conditions. One milliliter of the prepared cell suspension was carefully added onto the PFC surface to minimize disruption of the interface. The cells were then incubated under standard culture conditions (37°C, 5% CO₂) for 7 days to allow for spontaneous aggregation and formation of cohesive cell sheets. Cell sheet formation was monitored daily using phase-contrast microscopy.
For spheroid-based sheets, spheroids were generated using mesh-integrated ultra-low attachment (ULA) dishes (35 mm; LabToLab Co., Korea). Single cells were seeded at a density of 1 × 106 cells per dish, resulting in the formation of approximately 9000 uniform spheroids within 24 h. To maintain equivalent cell numbers with the single-cell approach, multiple dishes of spheroids were used for each sheet fabrication condition. Specifically, for sheets containing 2 × 106 cells, spheroids from two dishes (approximately 18,000 spheroids) were collected, and for sheets containing 3 × 106 cells, spheroids from three dishes (approximately 27,000 spheroids) were used. The collected spheroids were suspended in 1 mL of culture medium and carefully seeded onto the PFC interface in each well. The spheroids were cultured for 7 days to allow for spheroid fusion and sheet formation. The progression of spheroid fusion and sheet development was monitored daily using phase-contrast microscopy.
Spheroids were prepared as described above and then mixed with single-cell suspensions at various ratios (1:1 ratio by cell number), maintaining a total cell count of 2 × 106 cells/well. One milliliter of this mixed suspension was carefully added onto the PFC surface. The combined cultures were incubated for 24 h to allow for the formation of integrated cell sheets, with single cells filling the gaps between spheroids. Fluorescent labeling with DiO (single cells, green) and DiI (spheroids, red) allowed visualization using fluorescence microscopy (Leica DMi8, Germany). Labeled single cells and spheroids were seeded onto the PFC surface and incubated under standard culture conditions to facilitate the formation of cohesive cell sheets. Fluorescence microscopy was used to monitor the spatial distribution of single cells and spheroids within the sheets, and high-resolution images were captured for analysis. To evaluate scalability, we fabricated sheets in various well formats (48-well, 24-well, and 6-well plates). The medium and perfluorodecalin ratios were cultured under the conditions presented in Supplemental Figure 1.
Viability assessment of cell sheets
The viability of cell sheets was assessed using the LIVE/DEAD® Viability/Cytotoxicity Kit (Invitrogen, USA). After labeling with Calcein-AM (live cells; green) and ethidium homodimer-1 (dead cells; red) as per the kit’s instructions, sheets were carefully transferred to glass-bottom chamber slides (ibidi, USA). Samples were imaged using fluorescence microscopy (Leica, Germany) equipped with excitation/emission filters for green and red fluorescence. Images were analyzed using LAS X software to determine live/dead cell population.
Adipogenic differentiation of cell sheets
Adipogenic differentiation was induced by culturing cell sheets in adipogenesis differentiation medium (Gibco, USA) supplemented with StemPro Adipogenesis Supplement (Gibco). Sheets were cultured for 14 days, with medium changes performed daily. After differentiation, sheets were fixed with 4% PFA, washed with DPBS, and stained with Oil Red O. Staining involved immersing the sheets in a working solution (prepared by diluting Oil Red O stock solution in isopropanol) for 15 min at room temperature. Excess stain was removed by washing with distilled water. Lipid droplet formation was visualized using bright-field microscopy, and images were captured for documentation.
Chondrogenic differentiation of cell sheets
To induce chondrogenesis, cell sheets were cultured for 14 days in StemPro Chondrogenesis Medium (Gibco) containing Chondrogenesis Supplement (Gibco). Following differentiation, the sheets were fixed with 4% PFA, embedded in paraffin, and sectioned at 4 μm using a microtome. Sections were stained with Alcian Blue to detect proteoglycans and glycosaminoglycans. Stained sections were imaged using a slide scanner (Aperio GT450, Leica, USA) for quantitative and qualitative analysis.
Histological and immunofluorescence analysis
Cell sheets were fixed with 4% paraformaldehyde (PFA) for 1 h at room temperature and washed twice with Dulbecco’s phosphate-buffered saline (DPBS; Hyclone, USA). For histological evaluation, fixed sheets were embedded in paraffin and sectioned at a thickness of 4 µm. Sections were then processed for both Hematoxylin and Eosin (H&E) staining and immunofluorescence analysis. For H&E staining, sections were deparaffinized and rehydrated through a graded ethanol series, followed by staining to assess cellular morphology and structural integrity. Sections were permeabilized with 0.3% Triton X-100 in TBST and blocked with 10% normal goat serum for 1 h at room temperature. Primary antibodies targeting β-tubulin, VE-Cadherin, SA-αactinin, and CD31 (Abcam, USA) were diluted in blocking buffer and incubated with the sections overnight at 4°C. Following primary antibody incubation, sections were washed and treated with secondary antibodies. Counterstaining was performed using DAPI to visualize nuclei. Stained sections from both analyses were imaged using a confocal microscope (Olympus, Japan) for immunofluorescence and a light microscope for H&E staining to ensure comprehensive evaluation of cellular organization, morphology, and specific marker expression.
Apoptosis detection by TUNEL assay
Cell sheets were assessed for apoptosis using the TUNEL (Terminal deoxynucleotidyl transferase dUTP Nick End Labeling) assay, which detects DNA fragmentation characteristic of programed cell death. The procedure was performed using a commercially available TUNEL detection kit (In Situ Cell Death Detection Kit, Roche, Germany) according to the manufacturer’s recommended protocol. Stained sections were visualized using a confocal microscope (Olympus, Japan).
Statistical analysis
Quantitative data were collected from at least three independent experiments and are presented as mean ± standard deviation (SD). Statistical significance was assessed using Student’s t-test, as appropriate. All analyses were conducted using Origin with p-values < 0.05 considered statistically significant.
Results
Cell sheets using single cells on Liquid Interface
To form a liquid-liquid interface for cell sheet fabrication, perfluorodecalin and cell culture medium were sequentially added to cell culture plates. The two solutions remained unmixed and formed a stable interface (Supplemental Figure 2). To validate the hypothesis that perfluorodecalin enhances oxygen availability at the liquid-liquid interface, we measured the dissolved oxygen (DO) concentration in culture medium over time. As shown in Supplemental Figure 3, DO levels remained significantly higher in the PFC-pretreated interface group compared to the control group without PFC. The oxygen-saturated PFC maintained elevated oxygen levels in the overlying medium for up to 48 h.
To evaluate whether this interface could support cell sheet formation, adipose-derived stem cells (ADSCs) were used due to their multipotent differentiation capabilities (Figure 1(a)). 30 Perfluorodecalin (300 µL) was added to each well of a 48-well plate, followed by the careful addition of a 1 mL cell suspension. To determine the optimal cell density for sheet formation, we tested concentrations of 2 × 106, 3 × 106, and 4 × 106 cells per well. After 24 h, cohesive cell sheets were successfully formed only at the highest density of 4 × 106 cells per well (Figure 1(b)). At lower cell densities, the sheets were incomplete, with visible gaps, and lacked mechanical integrity, making them difficult to handle with forceps.

Formation of cell sheets using single hADSCs on a liquid interface between medium and PFC. (a) Schematic representation of the fabrication method for single hADSC-derived cell sheets using perfluorocarbon (PFC). (b) Morphological changes in cell sheets fabricated at varying cell densities (2 × 106, 3 × 106, and 4 × 106 cells/well) observed at different time points (0, 6, and 24 h). Images are presented from both top and side views to illustrate the structural characteristics of the cell sheets. Scale bars: 1 mm.
To further investigate whether longer culture durations could compensate for insufficient cell density, we conducted additional experiments using lower cell concentrations (0.1 × 106, 0.5 × 106, and 1 × 106 cells/well) and monitored sheet formation for up to 7 days. Partial cell aggregation was observed during the first 3 days, forming small multicellular clumps, but no continuous sheet structures were observed during the observation period. Instead, the aggregates became increasingly compact over time without fusing into a sheet, indicating that low-density cultures lack sufficient cell-cell interaction and ECM deposition required for sheet formation at the liquid-liquid interface (Supplemental Figure 4).
Cell sheets formed at 4 × 106 cells per well were further cultured to evaluate their long-term stability. Despite daily medium changes, the sheets gradually lost their structural integrity over time (Figure 2(a)). By day 3, the sheets maintained their form, but by day 5, they began to fragment, and by day 7, they were significantly disintegrated. TUNEL assay results showed a sharp increase in dead cells starting on day 5 (Figure 2(b)). H&E staining corroborated these findings: on day 1, cells retained their morphology and nuclei appeared intact, but by day 7, most cells in the center of the sheet exhibited fragmented nuclei or lacked nuclei, indicating cell death (Figure 2(c)). A key limitation was observed during these experiments. The 48-well plates, which could hold a maximum of 1 mL of medium per well, appeared insufficient to support the metabolic needs of 4 × 106 cells. This inadequate supply of fresh nutrients likely contributed to the cell death and sheet disintegration observed during extended culture periods.

Characterization of high-density single-cell-derived cell sheets cultured for 7 days. (a) Morphological changes in single hADSC-derived cell sheets (4 × 106 cells/well) observed from top and side views over a 7-day culture period. Scale bars: 1 mm. (b) TUNEL assay results indicating apoptotic cells within the sheets over time, with nuclear staining (DAPI, blue) and apoptotic cells marked by TUNEL-positive signals (green). Scale bars: 100 µm. (c) H&E staining of cell sheets seeded at 4 × 106 cells/well, comparing structural integrity between day 1 and day 7 of culture. Scale bars: 500 µm (upper image), 50 µm (lower image).
Cell sheets using spheroids on liquid interface
To address these challenges, we explored an alternative approach to forming sheets more efficiently at lower cell densities. Instead of plating single cells onto the interface, we pre-formed spheroids and plated them onto the interface to investigate whether this method could improve sheet formation. Spheroids were generated using mesh-integrated ultra-low attachment dishes. Within 24 h of plating single cells into these dishes, spheroids were uniformly formed (Figure 3(a)). By day 3, the spheroids exhibited greater uniformity in shape and increased in size to 100–120 µm (Figure 3(c)). Most cells within the spheroids remained viable (Figure 3(b)), and H&E staining confirmed that the spheroids were uniformly structured without necrotic cores (Figure 3(d)). A density of 1 × 106 cells yielded approximately 9000 spheroids.
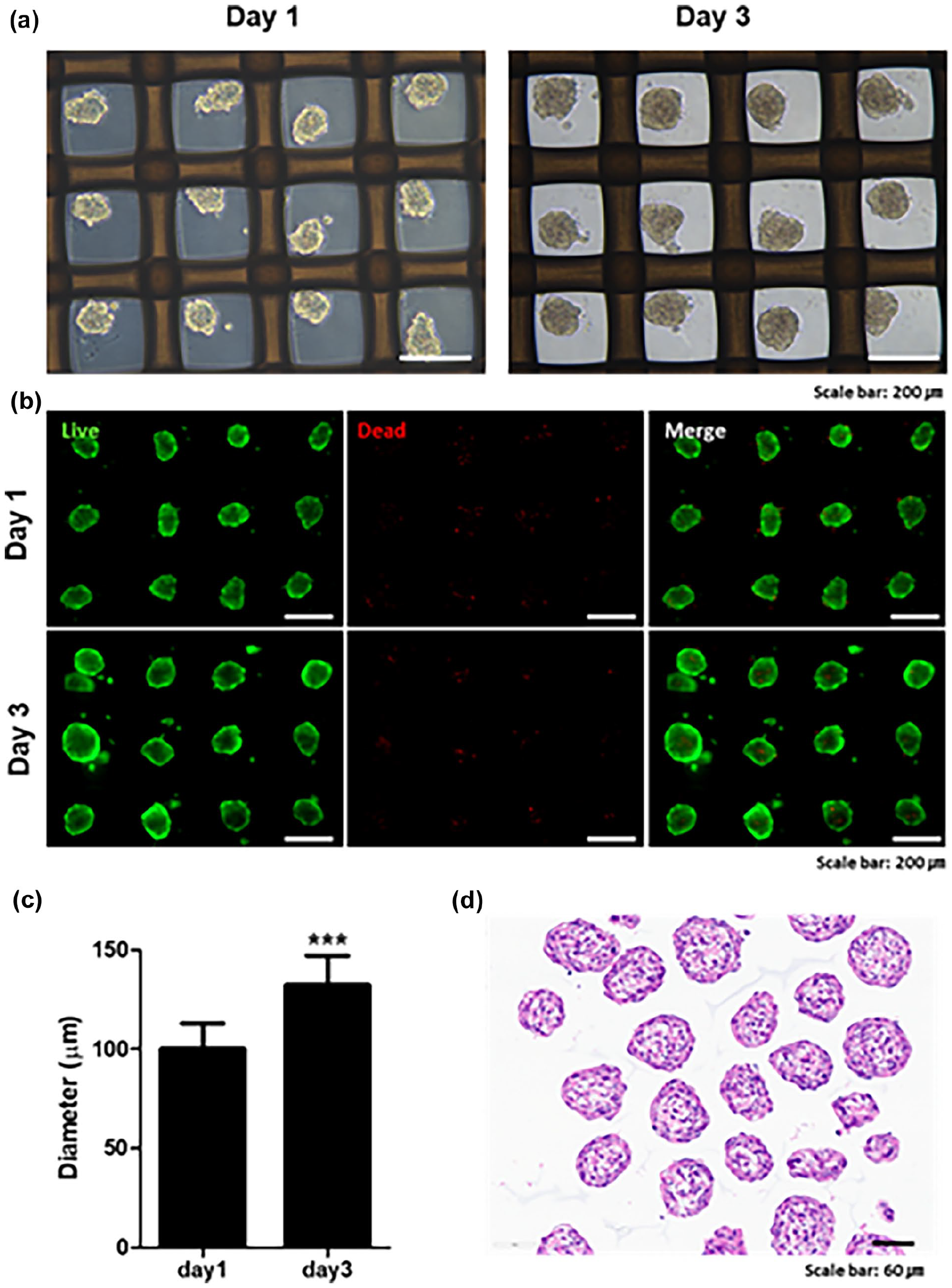
Preparation and characterization of spheroids formed using ULA culture dishes with integrated mesh. (a) Optical microscopy images of spheroids formed in ultra-low attachment (ULA) culture dishes with integrated mesh at day 1 and day 3. Scale bars: 200 µm. (b) Fluorescence microscopy images showing live/dead analysis of spheroids, with live cells stained green (Calcein-AM) and dead cells stained red (Ethidium homodimer-1). Scale bars: 200 µm. (c) Graph showing the changes in spheroid diameter measured on day 1 and day 3. Data are presented as mean ± standard deviation (SD). (*** indicates a significant difference (p < 0.0001)). (d) H&E staining of spheroids, demonstrating uniform structural integrity and absence of necrotic cores. Scale bars: 60 µm.
The spheroids were carefully plated onto the liquid interface as shown in Figure 4(a). Spheroids corresponding to 2 × 106 and 3 × 106 cells were plated in 24-well plates to observe sheet formation (Figure 4(b)). In both groups, the spheroids were evenly distributed across the liquid interface and formed cohesive cell sheets within 24 h. Live/dead analysis revealed that most cells were viable regardless of the cell density (Figure 4(c)). However, H&E staining revealed differences in sheet morphology based on cell density (Figure 4(d)). At 2 × 106 cells per well, the spheroids fused effectively with each other, forming a robust and cohesive sheet. In contrast, at 3 × 106 cells per well, the sheet was thicker, but the spheroids did not fuse properly, resulting in a less organized and weaker structure.

Preparation and characterization of spheroid-based hADSC sheets on PFC. (a) Schematic illustration of the fabrication process for spheroid-based hADSC sheets. Two conditions were tested: 2 × 106 cells/well (18,000 spheroids) and 3 × 106 cells/well (27,000 spheroids). The spheroids were plated onto a PFC interface in 48-well plate to assess sheet formation over time. (b) Morphological changes in spheroid-based sheets at different time points (0, 3, 6, and 24 h), presented from top and side views to demonstrate the structural evolution of the sheets. Scale bars: 1 mm. (c) Live/dead assay results showing cell viability in spheroid-based sheets fabricated with 2 × 106 and 3 × 106 cells. Scale bars: 100 µm. (d) H&E staining results of spheroid-based sheets, demonstrating the structural organization and fusion of spheroids within the sheet at different densities. Scale bars: 500 µm (upper image), 60 µm (lower image).
To further evaluate the scalability of this approach, we attempted cell sheet formation in larger well formats (24-well and 6-well plates), following the same seeding protocol and PFC-based interface. Cell sheets were successfully formed in both formats, indicating that the method can be extended beyond the 48-well scale. Live/dead staining confirmed that the majority of cells in the sheets remained viable across all well sizes (Supplemental Figure 5). However, histological analysis by H&E staining revealed that the sheet formed in the six-well plate exhibited irregular cell distribution, with visibly uneven cell density compared to those formed in smaller wells. These findings suggest that while the technique is scalable, larger formats may require further optimization to ensure uniformity and reproducibility.
We further evaluated whether the cell sheets retained their differentiation potential after formation. Sheets were induced to differentiate into adipocytes and chondrocytes. Oil Red O staining confirmed successful adipogenic differentiation, with lipid droplets visible under the microscope and strong red staining throughout the sheets (Figure 5(a)). Similarly, Alcian Blue staining indicated successful chondrogenic differentiation, as the extracellular matrix within the sheets was stained blue, showing proteoglycan and glycosaminoglycan production (Figure 5(b)). These results demonstrate that the sheets retained both structural integrity and functional differentiation potential.

Adipogenic and chondrogenic differentiation of spheroid-based hADSC sheets cultured on PFC. (a) Oil Red O staining of spheroid-based hADSC sheets after 2 weeks of adipogenic differentiation. Lipid droplet formation is indicated by red staining, confirming successful adipogenic differentiation. Scale bars: 500 µm (left image), 50 µm (right image). (b) Alcian Blue staining of spheroid-based hADSC sheets after 2 weeks of chondrogenic differentiation. The presence of proteoglycans and glycosaminoglycans in the extracellular matrix is highlighted by blue staining, demonstrating successful chondrogenic differentiation. Scale bars: 500 µm (left image), 50 µm (right image).
Cell sheets using a mixture of single cells and spheroids on liquid interface
To achieve an ideal cell sheet for broader applications, uniform initial cell distribution, enhanced structural stability, and the ability to mimic in vivo environments are essential. 31 To meet these criteria, we developed a mixed approach, combining single cells and spheroids to fabricate cell sheets (Figure 6(a)). The single cells and spheroids were fluorescently labeled to track their arrangement. Single cells filled the gaps between spheroids, increasing the uniformity of sheet formation and facilitating robust sheet development within 24 h (Figure 6(b)). Additionally, the single cells acted as bridges between spheroids, promoting their fusion and improving overall sheet stability (Figure 6(c)).

Formation and characterization of single and spheroid combined cell sheets on PFC. (a) Schematic illustration of the fabrication process for combined cell sheets. Single hADSCs (1 × 106 cells/well) and spheroids (1 × 106 cells/well) were mixed and plated onto a PFC-coated 48-well plate to evaluate sheet formation. (b) Morphological observations of cell sheets formed using single cells, spheroids, and a combination of single cells and spheroids (single + spheroids), presented from top-view and side-view perspectives, showing the structural differences among the groups. Scale bars: 1 mm (c) Fluorescence imaging of combined cell sheets at days 1, 3, and 5, with single cells labeled with DiO (green) and spheroids labeled with DiI (red). Images demonstrate the spatial distribution, integration, and fusion of cells and spheroids within the sheets over time. Scale bars: 100 µm.
To mimic a prevascularized muscle-like environment, we selected hSKMCs to generate spheroids and co-cultured them with HUVECs in single-cell form.32,33 To determine the optimal culture medium for co-culturing these two cell types, we initially seeded hSKMCs and HUVECs at a 1:1 ratio and cultured them separately in SKMC growth medium, HUVEC growth medium, and a 1:1 mixture of both media. Among the tested conditions, the mixed medium supported the highest growth rate of the co-cultured cells (Supplemental Figure 6), and thus was selected for subsequent cell sheet formation.
Using the optimized co-culture medium, we formed hybrid sheets composed of muscle spheroids and single HUVECs. The resulting cell sheets formed within 24 h, with endothelial cells aligning between spheroids to mimic vascular structures (Figure 7A(a) and (b)). H&E staining confirmed strong fusion between spheroids (Figure 7(c)), and beta-tubulin staining revealed higher expression in the mixed group compared to the spheroid-only group, indicating enhanced muscle cell activity (Figure 7(d)). Furthermore, immunofluorescence staining was performed to assess the functional preservation of each cell type within the sheets after 5 days of differentiation. Sarcomeric alpha-actinin (SA-α-actinin), a skeletal muscle-specific marker, was prominently expressed in hSKMCs, while VE-cadherin and CD31 were strongly expressed in HUVECs (Supplemental Figure 7), confirming that both cell types retained their functional characteristics within the engineered tissue.

Prevascularized skeletal muscle mimicry using hSKMC spheroids and single HUVECs on PFC. (a) Morphological assessment of cell sheets formed using single hSKMCs combined with hSKMC spheroids, and hSKMC spheroids combined with single HUVECs (spheroids + single cells). Images captured on Day 1 from both top-view and side-view perspectives, highlighting structural differences among the groups. Scale bars: 1 mm (b) Fluorescence imaging of hSKMC spheroid + HUVEC sheets at Days 1, 3, and 5, with hSKMCs labeled with DiI (red) and HUVECs labeled with DiO (green). Images depict the spatial distribution, integration, and interaction between the two cell types within the sheets over time. Scale bars: 100 µm. (c) H&E staining of cell sheets formed from single hSKMCs + hSKMC spheroids and hSKMC spheroids + single HUVECs groups, demonstrating structural integrity and organization. Upper images scale bars: 1 mm, lower images scale bars: 70 µm. (d) Immunofluorescence staining for VE-cadherin (endothelial marker) and beta-Tubulin (skeletal muscle marker), illustrating the formation of endothelial networks and skeletal muscle architecture in the cell sheets. Scale bars: 50 µm.
The combination of single cells and spheroids not only enhanced mechanical stability but also mimicked more complex cell-cell and cell-ECM interactions, closely resembling in vivo environments. These findings suggest that our hybrid sheet platform can effectively replicate the structural and cellular complexity of vascularized skeletal muscle tissues. This approach provides a promising basis for developing 3D biomimetic models in regenerative medicine, tissue engineering, and preclinical drug screening platforms.
Discussion
This study presents a novel and practical method for fabricating cell sheets using a perfluorodecalin (PFC)-based liquid-liquid interface. Compared to conventional cell sheet fabrication techniques that rely on solid surfaces and detachment mechanisms (e.g. temperature-responsive polymers or electrical stimulation),12–14,34 our platform eliminates the need for specialized substrates or mechanical processing, enabling direct and non-invasive retrieval of the sheets using simple tools like forceps or spatulas. The sheet can be gently detached without damaging cell-cell junctions or the extracellular matrix (ECM), which are critical for functional tissue assembly and transplantation. 35 This ease of retrieval provides a distinct advantage over traditional solid-liquid interface systems, where harvesting the sheets typically involves complex procedures, including surface stimulation, or enzymatic treatment, which may compromise cell viability or sheet integrity. In contrast, the liquid-liquid interface allows for seamless sheet handling, which may be highly advantageous for clinical applications requiring rapid, intact tissue delivery. To support this, we have included a Supplemental video demonstrating how easily the sheet can be lifted and transferred using only spatulas, highlighting the simplicity and reproducibility of the process (Supplemental Movie 1).
Unlike traditional approaches that utilize a solid-liquid interface where cells can immediately adhere and proliferate, the liquid-liquid interface lacks solid adhesion sites.36,37 This feature offers both advantages and limitations. On one hand, it promotes enhanced cell-cell interaction, leading to compact sheet formation. On the other hand, single cells that fail to participate in cohesive aggregation remain suspended in the medium and undergo cell death. This was particularly apparent at low seeding densities, where extended culture did not improve sheet formation, as confirmed by our time-course experiments (Supplemental Figure 4). Thus, successful single-cell sheet formation on the PFC interface required high initial seeding densities (4 × 106 cells/well) to ensure rapid aggregation and survival.
To overcome limitations in long-term survival of single-cell-based sheets, we introduced spheroid-based cell sheets and a hybrid method that combined single cells with spheroids. Spheroids pre-cultured in ultra-low attachment (ULA) mesh-dish maintained cell-cell interactions and ECM prior to seeding, resulting in improved survival and fusion when transferred to the PFC interface. At equivalent cell densities, spheroid-based sheets maintained structural integrity longer than single-cell-based sheets. The compact, pre-formed architecture of the spheroids not only protected cells from anoikis but also accelerated sheet fusion. Importantly, all comparisons between single-cell and spheroid methods were conducted under standardized conditions, including equivalent cell densities and identical media volumes (1 mL per 48-well), ensuring experimental consistency.
Furthermore, we demonstrated that spheroid-based sheets retained their multipotent differentiation capacity. After induction, adipogenic and chondrogenic differentiation was confirmed via Oil Red O and Alcian Blue staining, respectively. However, extended culture required for differentiation (2–3 weeks) was only feasible with spheroid-based sheets due to their greater structural and metabolic resilience. Sheets formed solely from single cells were unable to sustain long-term viability under these conditions.
To mimic vascularized tissue, we fabricated prevascularized skeletal muscle-mimetic sheets by combining hSKMC spheroids with HUVEC single cells. While we did not directly compare our vascular network with microfluidic or co-culture models, our system allowed spontaneous spatial arrangement of HUVECs among muscle spheroids, resulting in a rudimentary yet functional endothelial-muscle interface. CD31 and α-sarcomeric actinin immunofluorescence confirmed endothelial and myogenic identity, respectively. Although we did not achieve long-term culture beyond 7 days, these findings show the feasibility of forming heterogeneous tissue-like constructs without complex scaffolds or microengineering.
Scalability remains an important consideration for translational applications. 38 Although we successfully fabricated sheets from 48-well to 6-well plates, reproducibility decreased with scale. Larger interfaces led to uneven sheet morphology and occasional cell clumping, likely due to instability in initial cell distribution. Nevertheless, the simplicity and accessibility of our fabrication method—requiring only basic lab equipment—make it well suited for further optimization and integration into automated systems or bioreactors.
Perfluorodecalin, as a component of clinically approved artificial blood substitutes, offers a safety profile advantageous for clinical translation. 25 The use of perfluorodecalin offers further benefits beyond physical support. PFCs are chemically inert, biocompatible, and known for their high gas-dissolving capacity, particularly for oxygen.23,25,39 To evaluate this, we measured DO levels in culture media over 48 h, with and without pre-oxygenated PFC. The results confirmed that DO levels remained consistently higher in PFC-conditioned media, suggesting enhanced oxygen delivery to cells cultured at the interface. This is particularly relevant in thick or dense tissue constructs, where hypoxia can be detrimental to cell viability and function.40,41 However, future studies should focus on improving long-term stability, incorporating oxygen-diffusing microparticles (e.g. HA, gelatin, or collagen microspheres), and developing multilayer or compartmentalized systems tailored to specific tissue types.
Conclusion
In this study, we demonstrated the potential of perfluorodecalin-based liquid interfaces for fabricating cell sheets using single cells, spheroids, and their combination. While single cells formed cohesive sheets at higher densities, nutrient limitations hindered long-term stability. Spheroids offered structural integrity at lower densities but required optimization for proper fusion. The mixed approach combined the strengths of both, enhancing uniformity, mechanical stability, and mimicking complex tissue structures like muscle with integrated vascular networks. Additionally, the cell sheets retained adipogenic and chondrogenic differentiation potential, underscoring their applicability in regenerative medicine, tissue engineering, and in vitro disease modeling. These findings provide a versatile platform for developing advanced 3D biomimetic models with broad biomedical applications.
Supplemental Material
sj-docx-1-tej-10.1177_20417314251350316 – Supplemental material for Enhanced cell sheet engineering through combination of single cells and spheroids on liquid interface using perfluorocarbon
Supplemental material, sj-docx-1-tej-10.1177_20417314251350316 for Enhanced cell sheet engineering through combination of single cells and spheroids on liquid interface using perfluorocarbon by Ji Hye Park, Ji-Seok Han, Eun-Jung Ann, Cho Yeon Kim, Byoung-seok Lee, Ji Su Kang, Sun-Sook Song, Junhee Lee and Sun-Woong Kang in Journal of Tissue Engineering
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the challengeable future defense technology research and development program through the agency for defense development (ADD) funded by the defense acquisition program administration (DAPA) in 2024 (No. 915060201).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
