Abstract
In the field of tissue reconstruction, the development and improvement of suitable bone grafts is of increasing importance. The implementation of bone banks enables the international distribution of suitable allografts that can be used for defect reconstruction. Currently used procedures have significant drawbacks, especially regarding biomechanical and structural properties. These can be overcome by using the technique of high hydrostatic pressure (HHP) processing. To date, little is known about the impact of HHP protocol alterations including pressure-transmitting medium or temperature regarding bone graft integrity. Data of the present study show that a low-temperature and medium-pressure treatment using isotonic sodium chloride solution as the pressure-transmitting medium generated devitalized bone tissue with preserved extracellular matrix. Specifically, efficient devitalization of human primary osteoblasts (hOBs) was found starting from 150 MPa with cell death being a complex interaction between different mechanisms. Furthermore, protein denaturation in response to HHP treatment that was predominantly observed at 600 MPa led to non-significant impairment of biomechanical properties. Effects of HHP treatment on the bone tissue did not lead to any noticeable compromise in biocompatibility. Accordingly, the presented protocol may be applied as a medical device to improve the outcome of patients undergoing bone defect reconstruction with allogenic grafts.
Introduction
The population in industrialized countries is becoming increasingly older. It is estimated that by 2050, a proportion of 30% people would be older than 60 years in countries like Germany, Poland, the United Kingdom, France, China, and Canada. A key factor for ensuring healthy aging is an appropriate level of movement and physical activity. 1 Consequently, one of the main focuses of musculoskeletal research involves the reconstruction of damaged hard and connective tissue, aiming to sustain mobility, and overall quality of life.
Despite the remarkable regenerative abilities of bone tissue, defects exceeding its self-healing threshold often occur as a result of fractures, tumor resections and joint replacements.2–4 When such defects surpass a critical size surgical reconstruction becomes necessary to ensure the healing process. 3 An ideal bone reconstruction material has to meet several requirements, such as osteoconductivity, osteoinductivity, biocompatibility, degradability, porosity, and mechanical stability while minimizing risks of infection and disease transmission.4,5 A wide range of materials is available, from artificial bone substitute materials to native bone tissue of various origins.2,4,6
Autologous bone, typically harvested from the patient’s iliac crest, is the gold standard for the reconstruction of critical-size defects due to its superior biocompatibility, osteoinductivity, osteogenic potential, and low rate of graft rejection. 3 Additionally, autologous bone requires minimal processing, so its beneficial properties are preserved. However, the extractable amount is limited, and the process entails serious risks of donor-site morbidity caused by complications occurring in up to approximately 20% of cases.2,4,6,7 Allogenic bone is obtained from deceased persons or during procedures like total hip or knee arthroplasty. 7 Its greater availability enables bone banks to store and distribute compatible donor tissue, while offering native human material without the risks of harvesting from the recipient. 4 However, the use of allogenic bone increases the risk of disease transmission or infection due to contamination during extraction or storage.2,4,7
The clinical relevance for decontamination of bone tissue is shown in a recent meta-analysis by Baseri et al. 8 who found >10% of banked bone allografts to be contaminated with bacteria in the past two decades. Deceased donor tissue (approximately 20%) was more prone to contamination than tissue taken from living donors (7.5%). Processing has to inactivate both donor cells as well as bacteria, fungi, and viruses to prevent infections and excessive host immune reactions leading to graft rejection. 7 In addition to ensuring sterility and preventing infections, addressing immune compatibility of the graft is crucial for successful integration and long-term function. Although allografts typically exhibit good histocompatibility, the presence of donor-specific bone cells poses an inflammatory risk for the recipient. 9 Devitalization of bone grafts is linked to the death of related cells such as osteoblasts and osteocytes. To avoid graft rejection, apoptotic clearance of cells is preferred over the inflammatory death or necrosis of cells. Apoptosis initiates specific signaling cascades that lead to controlled cell death which is then recognized and cleared by macrophages in a non-inflammatory process. 10 It is assumed that pro-inflammatory types of cell death can negatively influence the recipient’s immune response due to cell membrane impairment and leakage of biomolecules. Those so-called damage-associated molecular patterns (DAMPs) can be recognized by immune cells and trigger an immune response. 11
Some European countries have strict rules regarding allograft safety involving devitalization and sterilization. Processing of bone tissue typically involves treatment with: (1) peracetic acid/ethanol, (2) gamma irradiation, (3) humid heat, or (4) combinations of (1)–(4). Due to insufficient penetration of bone by chemical detergents, processing mainly relies on physical methods like gamma irradiation and thermal treatment. 12 Notably, these methods are effective in both devitalization and decontamination. On the other hand, they especially interfere with the structural integrity resulting in loss of biomechanical properties and diminished load-bearing capabilities. 13 Ultimately, a reduced load-bearing capacity can lead to fatigue bone fractures, significantly impairing clinical outcome for the patients affected. 14
Considering this significant disadvantage of conventional methods, less destructive methods of bone graft processing are needed. The technique of high hydrostatic pressure (HHP) treatment has been increasingly studied in the field of tissue processing. While this method has been widely explored in various applications including processing of foods such as juices and yogurt since the 1990s, its potential in the medical field remains of great interest.15–19 The application of pressure levels up to 600 MPa demonstrated preservation of biomechanical properties while devitalizing bone and other musculoskeletal tissues.20–24 In addition, an adapted HHP treatment protocol successfully inactivated model bacteria and achieved decontamination in a bone model. 19 However, microbial decontamination requires pressure levels of 400–600 MPa – a pressure level that far exceeds the pressure thought to induce the gentle, non-inflammatory inactivation of eukaryotic cells – and works better at low temperatures.19,25–27
Previous studies on HHP influences focused on the ability of HHP to modulate bone graft devitalization, its impact on structure and biomechanical behavior and on the immunogenicity under selected conditions.16,21,28 Such findings need to be reassessed in the context of improved decontamination protocols to gain deeper insights into the underlying processes. 19 Our present study aims to comprehensively analyze HHP effects regarding cell devitalization and tissue structure as a function of treatment temperature and pressure level and to verify the applicability of a pre-defined protocol successful in bone tissue decontamination.
Methods
We analyzed the effects of HHP on human osteoblasts and human trabecular bone at different treatment temperatures using 0.9% saline as the pressure-transmitting medium. A methodological overview is provided in Figure 1. Follow-up analyses for cells included verification of morphological changes, viability, and regenerative capacity. Bone specimens were analyzed for cell death mechanisms, collagen integrity and structural alterations.

Methodological overview about samples, treatment parameters, and analyses. This image was created with BioRender (Agreement number: PX288QCARQ).
Cultivation of osteoblasts, fibroblasts, and human trabecular bone specimens
Femoral heads from patients (>60 years) undergoing total hip replacement were used for the isolation of primary osteoblasts (hOBs) following a protocol by Jonitz-Heincke et al. 29 Preparation of trabecular bone cylinders were done after patients’ informed consent (local Ethics Committee of the University of Rostock; registration number A 2010-0010). HOBs were cultivated in Dulbecco’s modified Eagle medium (DMEM, Biochrom AG, Berlin, Germany) with 10% fetal calf serum (FCS, Gibco® Invitrogen, Paisly, UK), 1% penicillin/streptomycin, 1% amphotericin B, 1% HEPES buffer, 50 µg/ml L-ascorbate-2-phosphate, 10 mM β-glycerophosphate, and 100 nM dexamethasone (all: Sigma-Aldrich, Munich, Germany). Human dermal fibroblasts (Merck KGaA, Darmstadt, Germany) were cultivated in DMEM GlutaMAX™ (Thermo Fisher Scientific, Waltham, MA, USA) supplemented with 10% FCS, 1% penicillin/streptomycin and 1% amphotericin B. Cells were cultivated in 75 cm2 culture flasks. For experiments, passage four hOBs and passage five fibroblasts were used. Adherent cells were detached with 0.05% trypsin and incubation at 37°C and 5% CO2 for 5 min. Afterward, the cell suspension was pelleted at 118g for 8 min, and cells were counted. Cultivation of cells and tissues was conducted at 37°C and 5% CO2.
The femoral heads for the preparation of trabecular bone cylinders were stored at −20°C and slowly thawed prior to sawing into disks with a height of approximately 10 mm using a diamond-coated bone saw under continuous cooling with water (EXAKT Advanced Technologies GmbH, Norderstedt, Germany). Bone cylinders of approximately 10 mm in height and 6 mm in diameter were generated using a trepan drill (Ustomed Instrumente, Tuttlingen, Germany). The prepared samples were stored at −20°C until HHP treatment and biomechanical testing.
High hydrostatic pressure (HHP) treatment
For HHP treatment, cell suspensions were transferred to 2 ml CryoTubes (Thermo Fisher Scientific, Waltham, MA, USA) and centrifuged at 118g for 8 min. After discarding the supernatant, the pellet was covered with 0.9% saline, and tubes were closed air bubble-free. Bone cylinders were transferred to CryoTubes that were filled with 0.9% saline air bubble-free. CryoTubes were transferred to 50 ml falcon tubes filled with water and finally sealed with Parafilm® (Amcor Limited, Zurich, Switzerland). Samples were pressurized using the HHP device (205797-Druckprüfanlage 6000 bar, Dustec Hochdrucktechnologie GmbH, Wismar, Germany) at set temperature and pressure levels. An overview about the HHP treatment protocols of this study is provided in Figure 2. In general, HHP was applied for 20 min using 0.9% saline as the pressure-transmitting medium.

Overview about experiments, samples and applied treatment parameters (0.9% saline was used as the pressure-transmitting medium). This image was created with BioRender (Agreement number: UP288QDW8I).
Osteoblast morphology regarding size and internal complexity
Size and internal complexity (granularity) of hOBs (105 cells per treatment protocol, n ⩾ 10) were determined using different HHP protocols (Figure 2) using 0.9% saline as the pressure-transmitting medium. After HHP treatment, hOBs were incubated at 4°C for 1 h prior to fixation with 4% paraformaldehyde for 1 h. To perform measurements, the cells were pelleted at 1000g for 10 min and resuspended in 200 µl of autoMACS® running buffer (Miltenyi Biotech, Bergisch Gladbach, Germany). About 5000 events were recorded using the Cytek™ Aurora (Cytek® Biosciences Inc., Fremont, CA, USA) using the SpectroFlo ® software version 2.2.0.3 (Cytek® Biosciences Inc., Fremont, CA, USA). Mean fluorescence intensities for side scatter (SSC, granularity) and forward scatter (FSC, diameter) in a previously defined cell gate (see Supplemental Figure S1 for the gating strategy with two samples) were determined using the FlowJo software version 10.7.1 (FlowJo LLC, Ashland, OR, USA). 30
Viability and regenerative capacity of hOBs
Live/dead fluorescence staining
Viability of HHP-treated hOBs was tested via calcein AM (live)- and ethidium homodimer-1-staining (EthD-1; dead; LIVE/DEAD® Viability/Cytotoxicity Kit for mammalian cells, Molecular Probes, Inc.; Eugene, OR, USA). Per protocol, 1 × 105 hOBs were treated as indicated in Figure 2. After treatment, the cell suspension was centrifuged at 118g for 8 min, and the cells were stained according to the manufacturer’s recommendations. After 30 min of incubation at room temperature in the dark, cells were imaged via fluorescence microscopy using a Keyence BZ-X800 fluorescence microscope (Keyence Corporation, Osaka, Japan). Representative images are shown in Figure 4.
Regenerative capacity
About 5 × 104 hOBs were treated with HHP at pressure levels of up to 600 MPa, as displayed in Figure 2. After treatment, cells were centrifuged at 118g for 8 min, the pellet was resuspended in 1 ml of DMEM with supplements, and the suspensions were transferred to 24-well plates. Cells were incubated for 7 days at 37°C and 5% CO2 and the regenerative capacity was evaluated by phase contrast microscopy using a Keyence BZ-X800 microscope.
Cell death mechanisms via gene expression analysis
Bone cylinders of 6 mm in diameter (n = 4) were prepared (Figure 2) to analyze HHP influences on the induction of different cell death mechanisms. After HHP application, bone cylinders were incubated at 4°C for 1 h, prior to freezing at −80°C.
RNA isolation
For isolation of RNA, 1 ml of TRIzol™ reagent (Thermo Fisher Scientific, Waltham, MA, USA) was added to the tissue. The following steps were performed according to the manufacturer’s manual. The RNA pellet was dried under laminar flow and resuspended in 50 µl nuclease free water. Concentrations were measured using a Tecan Reader Infinite® 200 Pro microplate reader and NanoQuant™ plate (Tecan Trading AG, Maennedorf, Switzerland). RNA was stored at −80°C.
Reverse transcription polymerase chain reaction
Reverse transcription polymerase chain reaction (RT-PCR) was performed with the High Capacity cDNA Reverse Transcription Kit (Applied Bioscience Corp., Beverly Hills, CA, USA) using 1000 ng RNA per sample. According to the manufacturer’s recommendations, the following program was carried out: 25°C for 10 min, 37°C for 120 min, 85°C for 5 min (Personal Thermocycler Biometra Analytik Jena AG, Jena, Germany).
PCR
For gene expression analyses, a master mix for each gene and sample was prepared, containing 1 µl of forward and 1 µl of reverse primer, 7 µl H2O and 10 µl of DreamTaq Green PCR Master Mix (all: Thermo Fisher Scientific, Waltham, MA, USA). The primer sequences used in the experiment are listed in Table 1. For each sample, 1 µl of cDNA and 19 µl of master mix were added. The PCR was performed using the My Cycler™ (BioRad Laboratories, Hercules, CA, USA) with the following protocol: 95°C for 5 min, 40 cycles of 95°C for 30 s each, 58°C for 30 s, and 72°C for 5 min.
Primers and primer sequences used for PCR.

fwd: forward primer; rev: reverse primer.
Agarose gel electrophoresis and analysis
The full volume of the PCR product was loaded onto a previously prepared 1% agarose gel with 0.01% SYBR™ Safe DNA gel stain (Invitrogen AG, Waltham, MA, USA). A 100 bp marker (Thermo Fisher Scientific, Waltham, MA, USA) was used as a reference for band size. An initial voltage of 80 V was increased to 120 V after the samples had left the gel pockets. The gel was imaged using the ChemiDoc™ Molecular Imager with the Image Lab Software (both: BioRad Laboratories, Hercules, CA, USA) and the application “Nucleic Acid Gels.” Gel bands were quantified using the Image Lab Software for Mac Version 6.1 (BioRad Laboratories, Hercules, CA, USA). Respective bands were marked, and pixel intensities were used for the calculation of percentage changes toward control samples.
Matrix integrity of HHP treated trabecular bone
Bone cylinders of 6 mm in diameter were prepared as described in Section 2.1 and treated (Figure 2) to analyze HHP influences on the integrity of the extracellular matrix. Analyses included multi-photon laser scanning microscopy and Western blotting for collagen I.
Multi-photon laser scanning microscopy
Samples for multi-photon laser scanning microscopy were stored at 4°C overnight. In addition to the HHP treatment, controls included native (untreated) bone and autoclaving at 121°C for 20 min. The multiphoton images were taken utilizing an ultra-fast laser-scanning multiphoton microscope (TriMScope II, LaVision BioTec, Bielefeld, Germany) in combination with a mode-locked femtosecond-pulsed Ti:Sa laser (Chameleon Vision II, Coherent, Santa Clara, CA, USA). The laser was focused onto the surface of the sample through a 25× water immersion objective with a numerical aperture of 0.95 (HC FLUOTAR L 25×/0.95 W VISIR, Leica Microsystems GmbH, Wetzlar, Germany) and tuned to 810 nm with a pulse frequency of 80 MHz and average output power of 148 mW. Since the second harmonic generation (SHG) signal intensity strongly correlates with the polarization of the incident light, 31 an adjustable quarter-wave (λ/4) plate was installed at the back aperture of the excitation objective to ensure elliptical light polarization and to eliminate polarization-dependent intensity variations. The backwards-scattered emission signal was collected by the excitation objective and detected using two ultrasensitive transmission photomultiplier tubes (H 7422-40 LV 5M, Hamamatsu Photonics, Herrsching, Germany), equipped with a 405/20 nm bandpass filter (CHROMA ET 405/20×, Chroma, Olching, Germany) for the SHG-signal and another 525/50 nm bandpass filter (CHROMA ET 525/50×, Chroma, Olching, Germany) for the autofluorescence signal, respectively. The imaging parameters were set to 1024 × 1024 pixels per image with a corresponding image size of 400 μm and line scanning frequency of 1000 Hz, which results in a lateral physical pixel size of 0.7 μm and a pixel dwell time of 1.41 μs. Representative images are shown in Figure 6.
Isolation of protein from HHP treated bone cylinders
Bone cylinders were immersed in liquid nitrogen and frozen at −80°C. Analogous to RNA isolation, total protein was purified via TRizol™ isolation performed according to the manufacturer’s manual. After the last washing step, the protein pellet was dried under laminar flow and resuspended in 200 µl of 1 × MOPS buffer. Protein concentrations were measured using the Qubit® 2.0 fluorometer and the Qubit® protein assay kit (both: Invitrogen AG, Waltham, MA, USA) according to the manufacturer’s protocol. Protein was stored at −20°C.
Native PAGE and Western blot
Native PAGE with 1000 ng of protein per well was performed on a 4%–20% Mini-PROTEAN TGX Stain-Free Precast gel (Bio-Rad Laboratories, Inc., Hercules, CA, USA). Electrophoresis was run at a voltage of 80 V until samples left the wells, then at 110 V for 1 h. Imaging was done using the ChemiDoc™ Molecular Imager and Image Lab Software (both: BioRad Laboratories, Inc., Hercules, CA, USA). The gel was equilibrated in transfer buffer (1× Towbin buffer without SDS; ChemCruz®, Santa Cruz Biotechnology, Dallas, TX, USA) for 15 min. A nitrocellulose membrane (Merck Millipore Ltd., Cork, Ireland) was equilibrated in methanol and transfer buffer for 1 min each. The membrane transfer was carried out using a Mini Trans-Blot ® Cell (BioRad Laboratories, Inc., Hercules, CA, USA) at a current of 350 mA under cooling for 1 h. The membrane was blocked with 5% skim milk (Carl Roth GmbH + Co. KG, Karlsruhe, Germany) in Tris-buffered saline with Tween 20 (TBS-T; 20 mM Tris, pH 7.5, 150 mM NaCl, 0.1% Tween 20) at room temperature for 1 h. Collagen I primary antibody (1:5000 in blocking solution; Abcam plc., Cambridge, UK) was applied and the membrane was incubated at 4°C under agitation for 16 h followed by four TBS-T washes for 5 min each. The secondary antibody (1:5000 in blocking solution; Anti-rabbit Antibody #7074S, Cell Signaling Technology, Inc., Danvers, MA, USA) was applied at room temperature for 1 h, followed by four washes. Bands were visualized with 1 ml of chemoluminescent substrate (SuperSignal™ West Pico PLUS Chemoluminescent Substrate, Thermo Fisher Scientific, Waltham, MA, USA) and quantified using Image Lab Software for Mac Version 6.1 (BioRad Laboratories, Hercules, CA, USA) to calculate percentage proportions of total protein.
Biomechanical characterization
Bone cylinders of 6.08 ± 0.05 mm in diameter and 10.8 ± 1 mm in length were prepared as described in Section 2.1. The bone density was measured as the ratio of total volume to bone volume by µCT scanning (Skyscan1076, Bruker, Billerica, MA, USA; resolution: 18 µm, voltage: 10 V, current: 10 mA, filter: 0.5 mm aluminum) and analyzed in Materialize Mimics 25 (Materialize NV, Leuven, Belgium). Since bone density significantly affects the mechanical properties of trabecular bone, 32 four groups (n = 9) were assembled as follows: control (bone density: 0.448 ± 0.096), 250 MPa (bone density: 0.449 ± 0.099), 350 MPa (bone density: 0.447 ± 0.095), and 600 MPa (bone density: 0.448 ± 0.097). Before the mechanical testing, the specimens were stored in 0.9% saline overnight. In the mechanical testing with a uniaxial test machine (Zwick Z050, ZwickRoell GmbH & Co. KG, Ulm, Germany), a preload of 0.1 N was applied, followed by a compressive load-to-failure test at a crosshead speed of 0.05 mm/s, 21 which is within a physiological range.33,34 The stress-strain curves were used to determine the compressive modulus and maximum strength (first significant drop in the stress-strain curve) to evaluate the influence of the treatment on the mechanical properties. The theoretical cross-section of a fully dense cylinder was used to calculate the stress values.
Biocompatibility
Indirect biocompatibility of HHP-treated human trabecular bone cylinders (n = 4 per protocol) was determined based on DIN EN ISO 10993-5. 35 Trabecular bone cylinders were treated with HHP (Figure 2) and afterward immersed in 2 ml of fresh cell culture medium (DMEM GlutaMAX™, Thermo Fisher Scientific, Waltham, MA, USA) at 37°C and 5% CO2 for 24 h.
Fibroblasts were seeded in 96 well plates in duplicates and incubated at 37°C and 5% CO2 for 24 h prior to stimulation with 200 µl of HHP-treated bone supernatants. Cells were incubated at 37°C and 5% CO2 for 48 h. WST-I (TaKaRa Bio Inc., Saint-Germain-en-Laye, France) for metabolic activity and CyQuant cell proliferation assay (Thermo Fisher Scientific, Waltham, MA, USA) were performed according to the manufacturer’s recommendations.
Graphical illustration and statistical analyses
Methodological overviews were created with BioRender.com (Science Suite Inc., Toronto, Canada). GraphPad Prism 9.0 for MacOS (GraphPad Software, Boston, MA, USA) was used for graphical illustration and statistical analyses. The image analysis of the multiphoton microscopy data was evaluated, using ImageJ (Fiji). 36 Data were tested for statistical significances by two-way ANOVA with Šidák’s and/or Dunnett’s multiple comparisons tests. Biomechanical data were tested for normal distribution via the Shapiro-Wilk test and for statistical significances using Kruskal-Wallis and Dunn’s multiple comparisons test. A p-value of <0.05 was considered statistically significant.
Results
Cellular morphology, viability, and regenerative capacity of hOBs in response to HHP treatment
First, responses of osteoblasts to HHP treatment were analyzed regarding morphological changes (Figure 3), viability and regenerative capacity (both: Figure 4). The morphology of osteoblasts treated with HHP at 10°C or 30°C was investigated using flow cytometry. Mean fluorescence intensities of forward and side scatter were determined and used to infer to cell size and internal complexity (granularity).
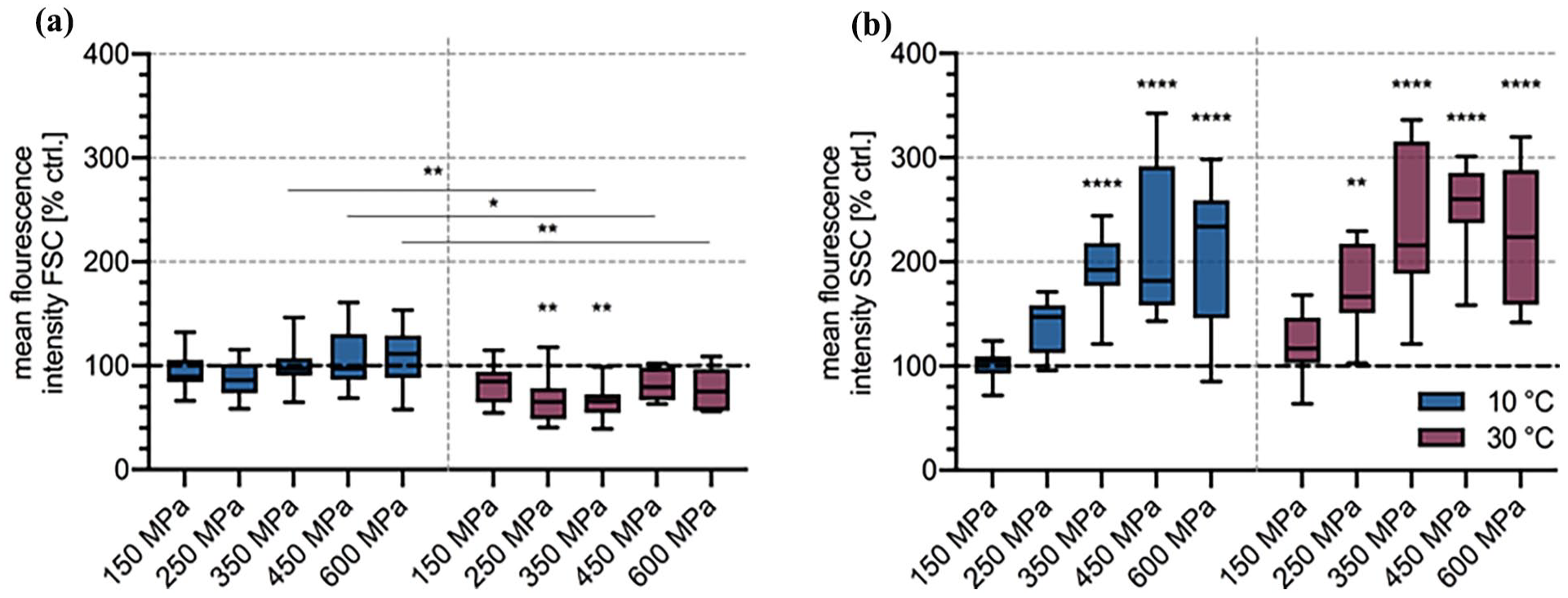
Size (a) and granularity (b) of hOBs treated with different HHP protocols regarding pressure level and pressurization temperature as determined by flow cytometry. About 105 cells were treated with HHP at 150, 250, 350, 450, and 600 MPa at 10°C (blue) or 30°C (red) for 20 min using 0.9% saline as the pressure-transmitting medium. Post-treatment incubation was carried out at 4°C for 1 h. Tukey box plots based on percentage change in fluorescence intensity of SSC and FSC toward untreated control cells (dotted line) for n = 11 (10°C) and n = 10 (30°C) different donors. Statistical analysis: two-way ANOVA with Šidák’s multiple comparisons test for significance between treatment at 10°C versus 30°C and Dunnett’s multiple comparisons test for significance relative to respective controls.

Viability and regenerative capacity of hOBs in response to HHP treatment at different treatment temperatures and pressure levels. Cells were treated with HHP at 10°C (a, c, e, g, i, k, m, o, q, s, u, w) or 30°C (b, d, f, h, j, l, n, p, r, t, v, x) at pressure levels ranging from 150 to 600 MPa for 20 min. Pressure levels shown: 0.1 MPa (controls; (a–d)); 150 MPa (e–h); 250 MPa (i–l); 350 MPa (m–p); 450 MPa (q–t); 600 MPa (u–x). Post-treatment incubation of cells was carried out at 4°C for 1 h prior to the immediate performance of the live/dead assay or cell seeding with incubation for 7 days and imaging at 20×. Scale bars: 100 µm for both phase contrast microscopy and fluorescence microscopy.
There were significant effects for cell size and granularity in osteoblasts treated with HHP at 10°C and 30°C. A statistically significant influence of both temperature and pressure level was demonstrated via two-way ANOVA for size (pt < 0.0001; pp = 0.0043) and granularity (pt = 0.0026; pp < 0.0001). A tendency for a decrease in the cell size was observed, especially when treatment was performed at 30°C (Figure 3(a)). Here, the decrease in size was significant for 250 MPa (p = 0.0029) and 350 MPa (p = 0.001). For the size, the percentage variation was between ~110% at 600 MPa and 10°C and a decrease to a minimum of ~65% at 350 MPa and 30°C. For the granularity (Figure 3(b)), an increase in the applied pressure level corresponded to an increase in internal complexity. Statistically significant differences compared to the untreated control cells could be detected with pressures of 350 MPa and higher (pall < 0.0001) at 10°C. At 30°C, these effects were even more pronounced with the increase already being significant for the treatment at 250 MPa (p = 0.0025), 350, 450, and 600 MPa (pall < 0.0001). The mean increase in granularity reached its maximum at ~220% and ~255% at 450 MPa for 10°C and 30°C, respectively. Granularity was not significantly influenced by the treatment temperature, however, there were significant differences in the hOBs size when HHP treatment was performed at 350, 450, and 600 MPa (p350 MPa = 0.0013; p450 MPa = 0.0173; p600 MPa = 0.0047).
Cellular viability, as reflected by fluorescence intensities of the “live” stain calcein (esterase activity) and the “dead” stain Ethidium homodimer-1 (EthD-1; DNA binding in permeabilized cells) in response to HHP treatment is shown in Figure 4.
Untreated controls, incubated at 10°C or 30°C (Figure 4(a)–(d)), respectively, showed few EthD-1 positive cells and extensive adherence after 7 days of incubation. No adherent osteoblasts were observed when hOBs were treated from 150 MPa (Figure 4(g), (h), (k), (l), (o), (p), (s), (t), (w), and (x)). Positivity for EthD-1 increased with increasing pressure levels for both treatment at 10°C and 30°C. Simultaneously, calcein fluorescence was gradually reduced with increasing pressure levels. This effect was more intense when treatment was performed at 30°C.
Cell death mechanisms in response to HHP treatment
To evaluate cellular responses to different HHP applications in detail, gene expression analyses were carried out with trabecular bone cylinders. Specific markers for apoptosis, pyroptosis, necroptosis, and ferroptosis were taken into account.
CASP8 tended to be up-regulated by the higher pressure levels of 450 and 600 MPa independent of the pressurization temperature (Figure 5(a)). CASP9 showed a tendency (p = 0.0813) for up-regulation in the treatment at 250 MPa at 10°C. When treated at 600 MPa at 30°C, there was significant up-regulation of CASP1 compared to the untreated control bone cylinder and the treatment at 10°C (both: p = 0.0025). Gene expression of MLKL tended to be higher after treatment with 600 MPa at 10°C as well as at 30°C (Figure 5(d)). The expression of GPX4 was temperature-dependent. Here, the treatment at 30°C led to a decrease, while treatment at 10°C caused an increase in expression of at least 200% for 250 MPa and 320% for 450 MPa (significant vs treatment at 30°C; p = 0.0125, and control bone, p = 0.0377).

Gene expression analyses of trabecular bone cylinders in response toward different HHP protocols regarding pressure level, pressurization temperature and post-treatment incubation temperature. Trabecular bone cylinders (n = 4 per protocol) were treated with HHP at 10°C or 30°C at 250, 350, and 450 MPa, or 600 MPa, respectively, for 20 min before post-treatment incubation of bone cylinders was carried out at 4°C for 1 h. PCR was performed to quantify gene expression of CASP8 (a), CASP9 (b), CASP1 (c), MLKL (d), and GPX4 (e). Band intensities from agarose gel electrophoresis as determined by Image Lab Software are expressed as mean percentage change toward controls as shown in the heat map. Statistical analysis: two-way ANOVA with Šidák’s multiple comparisons test for significance between treatment at 10°C versus 30°C and Dunnett’s multiple comparisons test for significance toward respective controls: *p ⩽ 0.05; **p ⩽ 0.01.
Matrix integrity and biomechanical properties
Bone matrix integrity was determined via different techniques. Microscopically, multiphoton microscopy was utilized to visualize structural details. On a molecular level, Western blot analysis quantified collagen I degradation (Figure 6). Biomechanical evaluation via uniaxial compression tests was performed to analyze the mechanical properties of the matrix.

Bone matrix integrity in response to HHP treatment. Human trabecular bone was treated with HHP at 250, 350, and 600 MPa at 10°C for 20 min using 0.9% saline as the pressure-transmitting medium before post-treatment incubation of bone cylinders was conducted at 4°C for 1 h. Multiphoton microscopy images (a–j), showing tissue autofluorescence at 525/50 nm (green; organic matrix components)) and second harmonic generation (SHG) signal at 405/20 nm (blue; fibrillar collagen), using an excitation wavelength of 810 nm. Here, treated bone cylinders (c–h) are compared to untreated bone (a and b) and bone that was autoclaved (i and j) 121°C for 20 min). Scale bar: 100 µm. The collagen I Western blot (k) is based on a native PAGE (1000 ng of protein per band) with the corresponding quantification (l) that was performed using Image Lab quantification of respective band intensities. Mechanical characterization of human trabecular bone cylinders (n = 9) regarding compressive modulus (m) and strength (n) via a tactile extensometer. Compressive modulus and strength are displayed as means with standard deviation and single values, represented as dots colored according to corresponding bone density values. Statistical analysis of M and N: Kruskal-Wallis test with Dunn’s multiple comparisons test: p > 0.05.
Bone autofluorescence is maintained regardless of the treatment protocol, whether HHP application or autoclaving. In contrast, the second harmonic generation (SHG) signal at 405 nm visualizing fibrillar collagen molecules (mainly collagen I)37,38 was clearly visible in the untreated control bone cylinders (Figure 6(b)) and in the HHP-treated samples (Figure 6(d), (f), and (h)) but strongly decreased when autoclaving was performed (Figure 6(j)). Collagen I denaturation was determined via Western blot followed by quantification of band intensity. Control bone and treatment with 250 and 350 MPa resulted in a collagen integrity of 95.5% (350 MPa) to 97.3% (250 MPa), whereas the treatment at 600 MPa resulted in a decrease in native protein to 80.3%. This effect results from the degradation of α1- and α2- subunits as demonstrated by the presence of the lower two bands in the last lane of Figure 6(k). According to the one-way ANOVA analysis of the biomechanical testing data, no statistically significant influence of the HHP treatment was observed. In general, there was a considerable scattering of the measurement results. Compared to the control samples, treatment at 350 and 600 MPa appeared to reduce the compressive modulus, with a stronger effect observed at the higher pressure level. In contrast, treatment at 250 MPa tended to result in an increase in compressive modulus. Regarding maximum strength, treatments at 250 and 350 MPa were associated with an apparent increase, while treatment at 600 MPa was linked to a reduction.
Biocompatibility
HHP-treated bone cylinders were used to prepare conditioned medium and stimulate fibroblasts to identify potential cytotoxic effects induced in the bone tissue. Here, metabolic activity and cell number were determined using two different photometric/fluorometric assays (Figure 7).

Cytotoxicity of human trabecular bone previously treated with HHP at different pressure levels and pressurization temperatures as represented by metabolic activity (a) and cell number (b) of fibroblasts. Human trabecular bone cylinders (n = 4 per protocol) were treated with HHP at 250, 350, and 600 MPa at 10°C and 30°C for 20 min and afterward, post-treatment incubation was carried out at 4°C for 1 h. Cytotoxicity was determined according to DIN EN ISO 10993-5. After 10,000 fibroblasts/well were stimulated for 48 h, metabolic activity was determined via WST-I assay, and cell number was determined via CyQuant™ cell proliferation assay. Tukey box plots based on n = 4 human trabecular bone cylinder supernatants each related to fibroblasts stimulated with control bone cylinder supernatants (dotted line). Statistical analysis: two-way ANOVA with Dunnett’s multiple comparisons for significances toward control bone cylinders and Šidák’s multiple comparisons test for significances between treatment temperatures.
In general, HHP-treatment of human trabecular bone cylinders did not significantly alter bone biocompatibility. A significant increase in metabolic activity compared to the untreated control bone blocks was obtained for the treatment at 10°C and 350 MPa (p = 0.0291) and 250 MPa at 30°C (p = 0.0493). For the treatment at 600 MPa, a significant decrease in metabolic activity was observed (p = 0.0063). Furthermore, significant differences were detected for the different treatment temperatures 10°C versus 30°C (p250 MPa = 0.0031 and p350 MPa = 0.0004). No significant differences were detected for the relative cell count and thus, proliferation of fibroblasts stimulated with supernatants of HHP-treated versus untreated bone. Both metabolic activity and relative cell count of fibroblasts were slightly decreased to ~75% for the stimulation with the supernatants of the treatment at 600 MPa at 10°C.
Discussion
With bone being the most frequently transplanted human tissue after blood transfusions, there is the need to investigate eligible processing methods in detail. 39 HHP showed significant potential to enhance allogenic bone transplant quality while preserving biological properties and biomechanical behavior.20–24 Effective devitalization was described for porcine skin, 40 human nevus tissue, 41 cholesteatoma on human ossicles, 42 human head and neck squamous cell cancer (HNSCC) tissue, 17 and, finally, human trabecular bone. 43 Previous studies by Waletzko et al. 28 showed positive effects of HHP treatment on human trabecular bone tissue. These included effective devitalization of osteoblasts, matrix integrity via protein structure analyses as well as electron microscopy, 43 biomechanical integrity, 21 immunological safety, 16 and successful osteointegration in a pilot animal study. 44 Accordingly, fundamental studies showed that gentle, devitalizing HHP treatment in the range of 250–300 MPa enables reproducible processing of tissue.43,45 While the preference for immunologically safe mechanisms of cell death (mainly apoptosis) is postulated at intermediate pressure levels, immunogenic cell death is suggested for higher pressure levels.28,46,47 This study considered the broader implications of tissue processing, including sterilization, which targets prokaryotic cells that usually require pressure ranges of 400–600 MPa for effective inactivation.18,19 The discrepancy of effective pressure levels between eukaryotic and prokaryotic cells might be overcome by investigating HHP efficiency as a function of treatment temperature (10°C and 30°C) and pressure-transmitting medium (isotonic saline). 19
Nutrient media, like DMEM or bacterial growth media, are commonly used in studies for the high-pressure treatment of cells and tissue.17,21,28,43,48–51 Some other pressure-transmitting media used are PBS,52,53 isotonic saline,40,41 or distilled water. 40 By utilizing isotonic saline, a clinically approved and cost-effective medium, fundamental research on HHP processing is approaching clinical practice. 54 Furthermore, we demonstrated that treating Escherichia coli at 245 MPa with isotonic saline instead of a nutrient medium led to an increased microbial reduction, improving from a 2.2 log10 to a 5.63 log10 reduction, achieving successful sterilization. 19 Consequently, changing the nutrient medium to isotonic saline also has a mechanistic effect that needs to be examined in the context of tissue treatment. While the cultivation and, formerly, HHP treatment of hOBs were carried out in DMEM medium (~330 28 mosm/l; water activity (aw) ~0.99), the HHP treatment was performed in 0.9% saline, (~306 28 mosm/l; aw ~0.99). Media with a low osmolarity and thus, a high water activity are assumed to have an increased efficiency. 55 Furthermore, the efficiency of HHP treatment depends on the solute concentration and composition, with moderate salinity levels enhancing stress responses, while high concentrations of salts and sugars exert protective effects.55,56 Here, the influence of the increased saline concentration of 154 mM compared to the culture medium (110 mM) may be considered regarding cellular stress reactions during HHP treatment. Concerning clinical applicability, storage temperature is another impact factor. Low temperatures support tissue sterility and integrity while maintaining cellular viability in stored bone grafts. 57 Here, the effects of the treatment temperature on osteoblasts were investigated with a focus on protein denaturation. It is assumed that protein denaturation occurs at approximately 300–600 MPa depending on the specific conditions under which the pressure is applied.58–60 Pressure-temperature phase diagrams of proteins suggest cooler conditions to enhance protein denaturation and thus, sterilization during HHP treatment.18,59 For bacteria, studies showed increased HHP efficiency for at temperatures below the moderate range of 20°C–30°C.19,26,27 Accordingly, treatments at 10°C and 30°C were tested on osteoblasts.
In fluorescence staining, the calcein signal in cells treated at 10°C was maintained over a broader range of applied pressure levels compared to the 30°C treatment. Here, “live” fluorescence was sporadically observed even in cells treated at pressure levels of up to 600 MPa. This effect, also described for human umbilical vein endothelial cells (HUVECs) treated with HHP, is not necessarily correlated with functionally intact cells. 45 Primarily, this observation indicates the relative retention of esterase activity responsible for the conversion of the dye. In this experiment, a retention of proteins was observed, which, contrary to expectations, occurred rather at 10°C than at 30°C. In addition to the detected loss of non-specific esterase activity, a pressure-dependent increase in the internal complexity, also expressed as granularity, was observed by flow cytometry. Intracellular disorganization, also observed in bacteria treated with HHP, is discussed as an indicator of protein denaturation.19,61 Interestingly, the increase in granularity observed in some samples might not solely be due to intracellular disorganization and protein structure damage, but also, or primarily, due to a decrease in cell size, which could increase the intracellular density. 62 For example, at 250 and 350 MPa at 30°C, a reduction in cell size to approximately 65% was observed alongside a significant increase in granularity (up to 255%). However, such a correlation was not found in all samples, suggesting that more complex mechanisms like cell death mechanisms, discussed later, may contribute to both cell shrinkage and increased granularity. As both, protein denaturation and preservation of specific functional proteins was observed, questions regarding the integrity of structural proteins are raised. Constituting 90% of the organic matrix, collagen I is the main structural protein of the bone.63,64 Its integrity is crucial for maintenance of the maximum strength ensuring load-bearing capabilities if used in surgical reconstruction.13,65 Here, native collagen I structure was investigated using multi-photon laser scanning microscopy and Western blotting of HHP-treated bone samples. A structural alteration in collagen I at the molecular level is evident, as shown by the separation of α1- and α2-chains in the Western blot when 600 MPa were applied.66,67 Interestingly, theoretical calculations even assume a stabilization of the collagen helix up to pressures of 324 MPa, since their application leads to a reduction in volume and thus, to a thermodynamically favorable reaction in response to HHP according to Le Chatelier principle.25,68 However, the observed separation could point toward modified interactions between the α-chains, potentially due to partial denaturation or conformational changes within the triple helix. The separation of subunits in the collagen triple helix suggests alterations in the molecular structure or stability of individual collagen molecules. 69 Despite these molecular-level alterations, the higher-order fibrillary structure appears unaffected. Since second harmonic generation (SHG) primarily reflects the supramolecular, highly ordered structure of collagen fibrils, it provides insights into the intact fibrillar organization rather than molecular-level changes.37,38 SHG emissions of multi-photon laser scanning microscopy seemed to remain unchanged in response to HHP treatment. This result suggests that the fibrillar shell, responsible for SHG signals, retains its highly organized structure, even if minor molecular changes occur internally.38,69,70 This observation corresponds to studies by Diehl et al. 52 who demonstrated biological functionality in the adhesion of osteoblast-like cells to collagen I even after treatment at 600 MPa. In contrast, after autoclaving, we observed a decrease in the 405 nm SHG signal, indicating a loss of fibrillar organization, 69 while the autofluorescence at 520 nm showed a slight reduction. Since autofluorescence arises from intrinsic matrix components, including collagen cross-links and non-collagenous proteins, its persistence suggests that certain molecular structures remain despite fibrillar degradation. While SHG is highly sensitive to disruptions in collagen organization, autofluorescence provides complementary insights into the integrity of the organic matrix, which is also essential for biomechanical properties.37,38,65
In addition to biological functions in the tissue, collagen I has a structural function which accounts for the maintenance of biomechanical properties. It is known that the integrity of mineral components of the bone influences the stiffness and thus, the Young’s modulus while the collagen I integrity affects the maximum strength.33,34 Denaturation of structural proteins leading to impairment of structural integrity and thus, decreased biomechanical properties of bone is described for established methods in tissue processing. For example, the application of gamma irradiation with a standard dose of 25–29.5 kGy reduces bone strength by about 21%–35%. 13 In previous studies Waletzko-Hellwig et al. have reported no negative effects of HHP treatment of up to 300 MPa 21 and demonstrated osteointegration and structural functionality of allogenic bone samples in a pilot animal study that investigated bone defects in the lateral condyles of rabbits. 44 In contrast, a tendency reduction in strength after HHP treatment at 600 MPa might be due to the partial denaturation of collagen I at this pressure level. A tendency for an increase rather than a decrease in maximum strength as a result of the treatment at 250 and 350 MPa might be due to the above-mentioned phenomenon of collagen-stabilization up to approximately 324 MPa. 68 The stiffness (measured by the compressive modulus) of the bone specimens also showed no significant differences between the groups. This is explained by the fact that that the stiffness is majorly influenced by the calcium content of the bone33,34 and HHP has no known effect on these anorganic structures. 71 However, it has to be mentioned that the evaluation of the effects of HHP on the biomechanical properties is superimposed by highly deviating properties of the biological tissue. Despite equalized bone density and a relatively high sample size (n = 9), high standard deviations arose from tissue heterogeneity. In addition, the bone samples were predominantly from older individuals with a reduced bone tissue quality and density, as they were taken during total hip arthroplasty. Interestingly, Zhang et al. 72 have shown a negative effect of 0.9% saline solution storage of up to 60 days on cortical bone regarding biomechanical integrity. Other authors concluded that short-term storage in normal saline did not influence the compressive modulus of trabecular bone samples. 73 Hence, we conclude that 0.9% saline might have an influence on bone properties in long-term storage but, did not negatively influence bone structure when used as a pressure-transmitting medium during the HHP for 20 min.
Furthermore, immunological compatibility of the processed bone is of great importance. Seeding assays (Figure 4, phase contrast microscopy) showed that cellular adherence and thus, proliferation were not possible after treatment at 150 MPa and above, indicating effective devitalization of osteoblasts. 45 Beneath the loss of regenerative capacity, cell death in response to HHP treatment has been previously associated with a decrease in cell size (Figure 3(a)), as observed for the treatment at 250 and 350 MPa. 62 This effect was apparent in the treatment at 30°C but not at 10°C. The difference between the two temperatures may be attributed to the faster progression of underlying cell death mechanisms at 30°C, where the cells are closer to their physiological temperature, accelerating membrane permeabilization and subsequent dye diffusion. On the other hand, at 10°C, cellular processes are slower, resulting in less intensive permeabilization and EthD-1 binding. An increase in EthD-1 signal was shown for the HHP treatment, indicating intensive permeabilization of cellular membranes that allows dye diffusion and binding to the DNA. Previous studies demonstrated membrane alteration due to HHP treatment in osteoblasts via electron microscopy.28,42 An increase in the pressure level typically induces a phase change in the membrane lipids from a fluid-like liquid crystal toward a gel phase resulting in modified membrane properties most likely affecting cellular survival.59,74 Building upon the observed effects of HHP on membrane integrity and cellular permeability, it is crucial to further investigate the underlying cell death mechanisms and their dependence on treatment parameters.
In this context, a more in-depth analysis of cell death mechanisms is necessary. In extrinsic apoptosis (type 1-cells), which is mediated by the binding of so-called death ligands to death receptors at the surface of the cell, Caspase 8 operates as an initiator caspase. Ultimately, cell death occurs as a result of the activation of effector Caspases 3, 6, and 7, which cleave target proteins. Intrinsic apoptosis (type 2-cells) is induced by a mitochondrial pathway. A specific trigger activates p53, and again a signaling cascade that leads to the activation of the above-mentioned effector caspases via Caspase 9. This pathway is also activated if an extrinsic stimulus is not sufficient in the activation of extrinsic apoptosis. Here, the cellular stress finally leads to the activation of intrinsic mechanisms.10,11 Differing patterns of CASP8 and CASP9 expression were observed in the present study. Treatment of human trabecular bone cylinders at 450 and 600 MPa appeared to up-regulate CASP8 independent of the treatment temperature, potentially indicating a type-I reaction of the associated cells. An elevated CASP9 expression, however, was observed in the samples treated at 250 MPa and 10°C, but also 600 MPa and 30°C. This observation suggests that the HHP treatment temperature may influence the activation of apoptosis pathways. It is possible that higher pressure levels (e.g. 600 MPa) promote immediate activation of the extrinsic pathway, whereas lower pressure levels (250 MPa, 10°C) might induce cell stress, leading to delayed intrinsic pathway activation. Concurrent upregulation of CASP8 and MLKL was observed mainly at higher pressure levels. Caspase 8 typically counteracts the pathway leading to the activation of MLKL and, therefore, the realization of necroptotic processes. 11 This highlights the increased risk of necroptosis at elevated pressures and the potential cellular response to regulate this process. Further protein expression analyses could provide deeper insights into the feasibility and regulation of these pathways. Significant upregulation of CASP1 after treatment at 600 MPa at 30°C suggests the occurrence of pyroptotic and thus, pro-inflammatory cell death mechanisms also associated with the expression of Interleukin-1 and Interleukin-17.11,75 This form of cell death is involved in inflammation in the context of bone loss in vivo. 75 While necrosis is a lytic type of non-programed cell death that is caused by excessive chemical or physical stimuli, necroptosis and pyroptosis are regulatory types of necrosis. In necroptosis, cell death is initiated via binding of specific death ligands to death receptors, whereas pyroptosis is mediated by Caspase 1.11,75,76 We observed a contrasting pattern for the expression of GPX4 depending on the HHP treatment temperature applied. While downregulation was determined for the treatment at 30°C, GPX4 was significantly upregulated when treatment was performed at 10°C. GPX4 is a marker of ferroptosis which is an iron-induced, pro-inflammatory form of cell death. This enzyme is responsible for reversing radical-induced lipid peroxidation and is increasingly expressed to promote damage repair. Cell death occurs if the damage to the membrane becomes excessive.77,78 Apparently, the devitalization of bone tissue by HHP is not specifically apoptotic or necrotic, as described in the literature, but might include other variants on the spectrum of cell death mechanisms, either differentially or simultaneously. The remaining knowledge gaps on the interplay of cell death mechanisms in HHP treatment may be addressed by more detailed investigation of various cell death markers in gene and protein expression and studies on the time progression after treatment.
Although bone tissue typically exhibits good histocompatibility and no cytotoxic effects, the triggering of pro-inflammatory signaling cascades may have negative effects on biocompatibility. 9 Indirect biocompatibility tests revealed no cytotoxic effects of HHP-treated bone independent on the HHP treatment temperature, despite the treatment at 600 MPa and 10°C with 73.4% of proliferation and 76.6% of metabolic activity being classified as slightly toxic regarding to DIN EN ISO 10993-5:2009. 35 Interestingly, we observed a significant increase in metabolic activity of fibroblasts treated with supernatants of HHP-treated bone (350 MPa at 10°C and 250 MPa at 30°C). We suspect that HHP treatment at distinct pressure levels may be able to stimulate the release of bioactive factors like peptides, proteins and matrix components from the bone that might temporarily enhance metabolic activity in the fibroblasts. The analysis of the supernatants via protein multiplex panels or the targeted stimulation of other cell types, such as osteoblasts, may help to identify specific components released from the treated bone matrix. Also, direct in vivo investigation of the immune response and tissue reaction following HHP treatment may provide additional insights to fully assess clinical safety. In general, the treatment at 600 MPa and 10°C resulted in minor drawbacks, not only regarding biocompatibility but also structural integrity and thus, biomechanical properties of the bone. Interestingly, different treatment temperatures appeared to have a complex influence on the effectiveness and specific effects of HHP treatment. The observed temperature-dependent effects in our present study suggest that HHP treatment at lower temperatures, such as 10°C, appears to provide a more controlled response, with cells retaining some structural integrity even at higher pressure levels (up to 600 MPa).19,59 The lower temperature may slow down cellular processes, leading to a slower progression of membrane permeabilization and a more gradual loss of cellular function.59,74 In contrast, treatments at 30°C resulted in a faster onset of cell death mechanisms, likely due to the increased kinetic activity of the cells, which could facilitate more extensive damage. 11 These findings indicate that treatment at 10°C may be preferable when a gentler devitalization of bone tissue is desired, as it preserves more cellular integrity while still enabling effective sterilization. Based on these results, it is recommended to use 10°C for HHP treatment at moderate pressure levels (250–350 MPa) for optimal balance between sterilization efficiency and tissue preservation. Higher pressure levels, such as 600 MPa, should be applied cautiously, particularly at 30°C, as they may induce more rapid and extensive tissue damage, potentially compromising the mechanical properties and biological compatibility of the treated bone. Therefore, for clinical application involving bone graft processing, a temperature of 10°C combined with moderate pressures offers a promising approach to maximize both safety and functional preservation.
The present study demonstrates that HHP treatment holds promise as a viable method for processing bone grafts. In clinical practice, HHP treatment could be integrated into existing workflows during the harvesting, processing, and storage of bone grafts, with incubation carried out at the lowest possible temperatures and using clinically approved media like isotonic saline. This approach could support both the safety and quality of allografts, positioning HHP treatment as a promising method for medical device applications. Specifically, its ability to preserve bone structure while enhancing sterilization could make it an attractive alternative to current methods such as gamma irradiation compromising the bone’s biomechanical properties.13,21 While this study demonstrates promising results, future research is needed to refine the HHP treatment parameters and explore potential challenges in its application, such as scalability, uniformity in treatment, and long-term effects on bone graft performance. Addressing these aspects could provide valuable insights into the feasibility of integrating HHP into routine clinical practices for bone tissue transplantation, potentially offering a safer and more effective alternative to conventional processing methods.
In the present study, the combination of qualitative and quantitative findings offers a comprehensive picture, which could be further validated by histological analyses and in vivo studies, such as in a rabbit model. This type of HHP treatment will be addressed in subsequent studies to explore complex relationships in greater detail, optimize treatment and, ultimately, enhance the outcome for patients receiving bone tissue transplantation.
Supplemental Material
sj-docx-1-tej-10.1177_20417314251337193 – Supplemental material for Comprehensive characterization of cell and tissue responses toward high hydrostatic pressure treatment: Molecular feedback and structural integrity in bone graft processing
Supplemental material, sj-docx-1-tej-10.1177_20417314251337193 for Comprehensive characterization of cell and tissue responses toward high hydrostatic pressure treatment: Molecular feedback and structural integrity in bone graft processing by Henrike Loeffler, Jan-Oliver Sass, Lorena Muelders, Julian Bauer, Oliver Friedrich, Rainer Bader, Annett Klinder and Janine Waletzko-Hellwig in Journal of Tissue Engineering
Footnotes
Acknowledgements
We would like to thank Doris Hansmann and Katja Simon (Biomechanics and Implant Technology Research Laboratory, Department of Orthopedics, Rostock University Medical Center), the Core Facility for Cell Sorting and Cell Analysis, Rostock University Medical Center as well as Daniel Wolter (Department of Oral, Maxillofacial and Plastic Surgery, Rostock University Medical Center) for their excellent technical support.
Ethical considerations
The use of human cells and tissues within this study is based on the informed consent of respective patients. Ethical approval was obtained from the Local Ethics Committee (ethics approval number: A 2010-0010).
Author contributions
Conceptualization: HL; JWH; Methodology: HL; JOS; LM; JB; JWH; Validation: HL; JWH; Formal Analysis: HL; Investigation: HL; JOS; LM; JB; JWH; Resources: OF; RB; Writing – Original Draft: HL; Writing – Review & Editing: HL; JOS; LM; JB; OF; RB; AK; JWH; Supervision: OF; RB; AK; Funding Acquisition: RB.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data of this study will be made available upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
