Abstract
The repair and regeneration of tissues and organs using engineered biomaterials has attracted great interest in tissue engineering and regenerative medicine. Recent advances in organoids and engineered organs technologies have enabled scientists to generate 3D tissue that recapitulate the structural and functional characteristics of native organs, opening up new avenues in regenerative medicine. The matrix is one of the most important aspects for improving organoids and engineered organs construction. However, the clinical application of these techniques remained a big challenge because current commercial matrix does not represent the complexity of native microenvironment, thereby limiting the optimal regenerative capacity. Decellularized extracellular matrix (dECM) is expected to maintain key native matrix biomolecules and is believed to hold enormous potential for regenerative medicine applications. Thus, it is worth investigating whether the dECM can be used as matrix for improving organoid and engineered organs construction. In this review, the characteristics of dECM and its preparation method were summarized. In addition, the present review highlights the applications of dECM in the fabrication of organoids and engineered organs.

Introduction
The repair and replacement of damaged tissue or organs are driving an expanding need worldwide. However, there is a critical shortage of organs available for transplantation and the post-transplant immunological rejection still remains a significant concern. In the United States alone, the annual expenditure for diseases related to tissue defects and organ failure exceeds $400 billion. 1 Recently, extracellular matrix (ECM)-derived bio-scaffolds due to their excellent biocompatibility have drawed increasing interest in tissue engineering and regenerative medicine due to their excellent biocompatibility, biological activity, and mechanical properties. As a biological three-dimensional (3D) network, ECM plays a critical role in cell signaling, function, phenotype, and morphology, providing a beneficial microenvironment for cellular homeostasis, growth, tissue formation, and repair.2,3 Each tissue or organ has its unique ECM, and the interactions between cells and ECM are crucial in regulating cell behavior, function, and fate.4,5 Due to the complex biological properties and 3D ultrastructure of native ECM, replicating them using traditional manufacturing methods and biomaterials is often challenging. 6
Decellularized extracellular matrix (dECM) materials refer to biomaterials formed by removing immunogenic cellular components from human or animal organs/tissues using decellularization techniques. dECM minimizes changes to ultrastructure and composition and provides an excellent 3D microenvironment for subsequent cell seeding, thereby opening new avenues for tissue engineering. 7 In clinical practice, the use of dECM is progressively being adopted to support tissue repair and transplantation therapies.8 –10 A variety of commercially available dECM products have been developed, including Alloderm® and GraftJacket®, which are derived from human dermis and used for repairing skin wounds as well as tendon and ligament injuries. Additionally, OASIS®, sourced from porcine small intestinal submucosa, is utilized for skin wound healing, while Prima™ Plus and CardioCel®, derived from porcine and bovine cardiac tissues, are employed in cardiac valve repairs.11 –16 The emergence of these products underscores the versatility and potential of dECM-based biomaterials in clinical applications. Currently, tissue engineering materials derived from dECM have been widely applied in the fabrication of engineered tissue and organ-like constructs, including the gastrointestinal tract, kidneys, liver, lungs, pancreas, uterus, bone,17 –19 cartilage,20 –24 heart,25 –29 skin,30,31 brain,32,33 esophagus, 34 and oral cavity, 35 among others. Compared to traditional two-dimensional culture models, organoids represent an innovative technology that can recapitulate the entire physiological processes of an organism. They possess advantages such as a closer resemblance to physiological cell composition and behavior, a more stable genome, and suitability for biological transfection and high-throughput screening. In contrast to animal models, organoid models offer simpler operation and can be utilized to study mechanisms underlying disease onset and progression. Therefore, they hold broad application prospects in various fields including organ development, precision medicine, regenerative medicine, drug screening, gene editing, and disease modeling. Organoids refer to tissue-like structures formed through the in vitro 3D culture of adult stem cells (ASCs) or pluripotent stem cells (PSCs) with a certain spatial organization. While not exact replicas of human organs, organoids can mimic real organs in structure and function, enabling the closest possible simulation of in vivo tissue structure and function as well as long-term stable passaging. Currently, the cultivation of most organoids relies on Matrigel, a commercial matrix derived from the Engelbreth-Holm-Swarm mouse sarcoma, comprising a variety of elements including laminin, type IV collagen, and growth factors. However, in organoid culture, there can be instances where morphogenetic control during the self-assembly process is suboptimal. Possible reasons for this include the lack of tissue-specific cell types, typically the absence of matrix, vasculature, and immune components, as well as potential limitations in nutrient supply leading to cell death and accumulation within the central and luminal cavities of organoids. Matrigel also has several other drawbacks in orgnoid culture. 36 Compared to porcine and human-derived dECM, Matrigel exhibits greater compositional differences from natural human ECM.37,38 Additionally, Matrigel poses potential antigenicity and potential risks of harboring animal pathogens that could infect macrophages and impact the immune system (e.g. lactate dehydrogenase-elevating virus).39,40 It is well established that ECM and dECM provide essential environments for cell growth, proliferation, and differentiation, possessing tissue specificity and containing rich regulatory factors. The mechanical properties of both ECM and dECM, including stiffness and viscoelasticity, are also crucial for maintaining the structural integrity and functionality of organoids. Numerous studies have utilized tissue-specific dECM for culturing organoids. For example, human intestinal dECM has been employed to construct intestinal organoids,41,42 and porcine or human liver dECM has been used to develop hepatic and cholangiocyte organoids.43 –45 Further specific applications are discussed in Section “dECM-based materials for organoids engineering and organ” of this review. Consequently, there is an increasing emphasis on elucidating the interactions among ECM, dECM, organogenesis, stem cells, and the surrounding cellular microenvironment to optimize organoid culture and enhance their potential applications in regenerative medicine and disease modeling. This optimization encompasses not only the selection of appropriate ECM and dECM components and the modulation of their mechanical properties but also the regulation of cellular behavior through biochemical signals and physical environmental factors. Currently, bioengineering of whole organ culture based on dECM scaffolds is getting popular, and dECM-based organoids also have the potential to develop into engineered organs.
This review emphasizes the preparation of dECM-derived biomaterials from tissues/organs and the necessity of post-decellularization processes such as sterilization, as well as their applications in organoid and organ engineering dECM scaffolds are considered promising biomaterials as bioinks for 3D bioprinting due to their capability to transition from pre-gel solutions to hydrogels, which is also discussed herein.
Organoids
Single cells or small clusters of cells proliferate and reorganize to form complex and structured cellular assemblies that mimic the functional attributes of specific organs, termed organoids. The study of organoids traces back to the early 20th century (Figure 1). In 1907, Wilson demonstrated that dissociated sponge cells possess the inherent capability to self-organize and regenerate entire organisms. 46 The foundational work for organoid technology was laid in 1981 when Evans and Martin independently isolated pluripotent stem cell lines from mouse embryos, with Martin naming them “embryonic stem cells”.47,48 By 1998, Jones et al. had successfully isolated and cultured embryonic stem cell lines from human blastocysts, expanding the possibilities for using stem cells to culture organoids. 49 Significant advancements were made in the 2000s. In 2009, Professor Hans Clevers coined the term “Organoid” and he cultivated the first intestinal organoid from Lgr5(+) stem cells derived from mouse intestines. 50 Recent years have seen rapid developments in organoid research, including Mendjan’s generation of self-organized cardiac organoids from human pluripotent stem cells (hPSCs) in 2021 51 and Clevers et al.’s establishment of a human fetal hepatocyte organoid model. 52 Significant advancements in reproductive system organoids were also made from 2018 to 2022.53–56

Organoids can be classified into ASC-derived organoids and PSC-derived organoids, according to the cellular origin. ASCs can be directly harvested from human tissues, simplifying the cultivation protocols for organoids. However, for adult organs like the heart, where tissue-specific cells with stem cell properties may not be obtainable, the generation of organoids using ASCs might not be feasible. Since human ASCs are sourced from adult tissues rather than embryonic sources, their use is ethically uncontroversial. Compared to PSC-derived organoids, ASC-derived organoids more closely resemble mature adult tissues, making them better suited for modeling adult tissue repair. They can also be expanded ex vivo over extended periods while maintaining genetic stability.77,78 PSC-derived organoids can be cultivated using embryonic stem cells (ESCs) or induced pluripotent stem cells (iPSCs). ESCs, originating from the inner cell mass, are capable of differentiating into all three germ layers, exhibiting robust self-renewal and pluripotent differentiation capabilities. 79 iPSCs can also differentiate into all three germline cell types, 80 and can be reprogrammed from patient-derived somatic cells using small molecules or transcription factors to generate organoids. iPSC-derived organoids, with their complex cellular compositions, are often used to study early organogenesis, although they typically do not expand further once they reach their differentiation endpoint.77,81,82 Organoids from various sources possess the capacity to include high-functioning cell populations, encapsulating both the function and structure of the original organs. The cells within organoids follow the differentiation trajectories of the organs, and can also develop vascular-like structures similar to those of the host, potentially enabling long-term survival post-transplantation. 83 Utilizing organoids for model construction offers advantages of reduced model development time, personalization, and genetic editing capabilities.
In the cultivation and application of organoids, a critical challenge lies in developing mature organoids that possess the functional and physiological properties of native organs, essential for tissue repair and biological function reconstruction. Advances in bioengineering have enabled the standardized, programmable, and scalable cultivation of organoids. For instance, the use of photolithography or micropore technology to fabricate 3D porous plates allows cells to form aggregates with specific parameters through micropore arrays, enhancing the consistency of organoid modeling.84,85 Bioreactors increasingly facilitate the large-scale, high-quality, efficient, and homogeneous cultivation of organoids. Bioreactor systems, by employing rotational forces, enhance the uniform diffusion of internal materials and support the growth of 3D structures, 86 offering a cost-effective solution with higher morphological stability of the cultivated organoids and the potential for large-scale and homogeneous organoid cultivation.87,88 The development of hydrogel matrices with high biocompatibility, which can dynamically mimic the physiological ECM due to their unique 3D structures and editable properties, has also become a focal point of research. Organoids cultivated in specific hydrogels outperform those lacking in vivo-like microenvironment conditions. 89 Utilizing cell-containing hydrogels as bio-inks for 3D bioprinting allows for the construction of organoids. This technique holds tremendous potential for accurately and reproducibly creating complex microstructures, crucial for cultivating organoids that closely resemble the structure and function of original organs.90,91
Current organoid models are limited in maturity and functionality, exhibiting only certain functions of the tissues and failing to replicate the complete physiological lineage of cell types, maturity, and function of their respective original organs. As organoids increase in size, nutrient supply to cells located at the center of the organoid becomes restricted, which can lead to severe cell death. This issue is more likely to occur in densely structured organoids (such as the brain) and those forming cavitated structures (such as bile ducts and pancreas). 92 The majority of organoid models are derived from a single type of stem cells and lack tissue-specific cell types, including niche-specific stromal cells, immune cells, vascularization, innervation, or microbiomes. These cell types play a crucial role in establishing a specific microenvironment, enabling organoid models to more effectively mimic the physiological responses of real tissues. Additionally, studies on cell behavior within organoids derived from stem cells remain scarce. Consequently, even under the same experimental conditions, cultivated organoids exhibit high heterogeneity and significant variability in phenotypic characteristics (such as shape, size, and cellular composition), which is disadvantageous for high-throughput screening experiments and research requiring high spatial and temporal resolution imaging.93,94 Optimizing the interactions between tissue-specific cells and the ECM, as well as local physical and biochemical microenvironment characteristics, can significantly reduce batch-to-batch heterogeneity. 95 Synthetic matrices can be used to control the composition and stiffness of the culture medium. Currently, a promising approach is the use of dECM that provides a matrix rich in tissue-specific regulatory factors, offering a better environment for the growth, proliferation, and differentiation of stem cells and organoids.38,96
Decellularization extracellular matrix
dECM is a promising natural biomaterial prepared from human or animal tissues or organs through decellularization. 97 dECM retains the 3D structure and biological properties of ECM, exhibiting bioactivity, biocompatibility, and non-immunogenicity.98,99 It preserves numerous cell growth factors, such as fibroblast growth factor, transforming growth factor, and hepatocyte growth factor, 100 which can enhance the growth, migration, proliferation, differentiation, and angiogenesis of seeded cells. This “real-time interaction” with seeded cells can reshape the structure of tissues and organs, playing a crucial role in tissue and organ regeneration and functional repair. Therefore, various scaffolds and materials based on dECM have garnered increasing attention in recent years.
The origin of ECM
ECM can be derived from three primary sources: human tissues, cell cultures, and animal tissues. ECM from human sources significantly reduces the risk of immune rejection associated with xenogenic ECM, such as the response to α-gal epitopes. 101 Moreover, human-derived ECM best mimics the structural and functional properties of native tissues and organs, offering superior biocompatibility and clinical efficacy. Therefore, human tissues are the preferred source. 102 Ideally, tissues from young, healthy donors are the best source, but human tissue is relatively scarce and its availability is limited, especially for younger, healthier tissues. 103 Cadaveric tissue can be used as donors but availability is again limited. 104 Cadaveric skin, sold as Alloderm and GraftJacket®,105,106 is used clinically for wound treatment. Tissues associated with birth, such as the placenta, umbilical cord, and fetal membranes, are often discarded after delivery, making them more abundant and accessible. Studies have shown that ECM derived from placental tissues possesses favorable immunogenic and regenerative properties, containing various growth factors (e.g. epidermal growth factor, TGF-β, VEGF) that support the growth and differentiation of diverse cell types.107,108 For instance, research by Murchison et al. demonstrated that placental ECM hydrogels can serve as suitable substrates for hiPSCs differentiation into the three embryonic layers. 109 Wang et al. utilized placental dECM hydrogels to construct spinal cord-like organoids that are more stable and mature than those developed using Matrigel. 110
Human ECMs secreted by human cells culture can also be applied in tissue engineering and regenerative medicine (TERM). 111 For example, Gonzalez-Fernandez et al. enhanced the survival and proliferation of mesenchymal stromal cells (MSCs) by incorporating ECM secreted during the spheroid formation of MSCs. When these ECM-loaded spheroids were encapsulated in inductive alginate gels, their osteogenic differentiation capability was enhanced. 112 Wan et al. found that dECM obtained from MSCs differentiated into osteoblasts exhibited stronger osteoinductive effects on reseeded MSCs. 113 Furthermore, Choi et al. demonstrated that ECM secreted by decellularized human iPSC-derived chondrocytes enhanced chondrogenesis of hiPSCs and promoted cartilage repair when specific dECM was implanted into cartilage defects in rat knees. 114 These studies and concepts provide a promising avenue for isolating cell-type-specific ECMs, though yield remains limited. Some researchers are focusing on using stable cell lines to produce these ECMs, minimizing batch-to-batch variations due to different donors, thereby offering new insights into these challenges. 115
ECM from animal tissues is more abundant, but shares many limitations with Matrigel derived from murine sources. Antigenic glycans found in animal tissues, such as α-gal and Neu5Gc, can lead to chronic rejection and calcification issues. Additionally, certain protein components (e.g. protein antigens in bovine bioprosthetic valves) might trigger immune responses, and the presence of porcine endogenous retrovirus (PERVs) genes poses potential immunological and pathogenic risks in clinical applications.40,116 Nonetheless, due to their availability, animal-derived ECMs remain the most commonly used resources. Compared to Matrigel, tissues from cattle and pigs share greater structural similarity with human tissues, particularly pigs, gaining increasing attention and research focus. During decellularization, optimizing methods to reduce immunogenicity, such as the removal of α-gal epitopes, helps lower the risk of immune rejection. Therefore, the careful selection of animal sources is crucial to ensure protein structures are closely related to human tissues, alongside effective antigen removal and decellularization processes to mitigate risks.45,117–119
Decellularization methods
Effective decellularization maximally removes all cellular and nuclear components from tissues or organs, reducing or eliminating immune rejection reactions and providing a foundation for cell development of new tissues and organs (Figure 2). 116 Therefore, various factors such as cell type, tissue density, thickness, and lipid content determine the effectiveness of tissue decellularization methods. 120

Overview of common decellularization techniques and post-treatment processes. Decellularization of target tissues is effectively achieved through chemical, physical, or enzymatic methods, with combinations of these approaches often being employed. The harvested dECM materials are subjected to sterilization procedures to eliminate potential microbial contamination, reduce toxicity, and enhance biocompatibility. Inevitably, these processes can impact the structure and functionality of the dECM, which is frequently addressed by employing solubilization and crosslinking methods for improvement.
Various decellularization methods involve the use of chemical, physical, or enzymatic approaches to remove cells and genetic material residues from the target tissue (Table 1). Different methods are often combined to more effectively eliminate cells and genetic material residues from the scaffold. Physical methods include temperature, pressure, supercritical fluids, polyethylene glycol, and electroporation. The basic principle is the mechanical disruption of cell membrane structures in the tissue, leading to cell death, followed by decellularization through methods such as solution washing, nucleic acid, and lipid removal. Chemical methods are the most commonly used and include acids, alkalis, detergents, low and high osmotic solutions, and various chemical solvents, primarily to dissolve cell membranes and degrade DNA. Biological methods mainly involve various enzymes and chelating agents, which can selectively hydrolyze various proteins and genetic material. However, residual enzymes may cause adverse reactions,121,122, so they are often used in combination with other chemical and biological methods.
Commonly decellularization methods and the effects.

The ultimate goal of decellularization is to produce two primary end products: a complete organ scaffold or specific dECM materials. Whole organ decellularization aims to preserve not only the three-dimensional architecture of the organ but also its vascular network, facilitating subsequent recellularization and the development of transplantable human organs. The most commonly employed method is perfusion decellularization, which utilizes custom bioreactors to perfuse reagents through the organ’s native vasculature. These reagents include ionic and non-ionic surfactants (such as SDS and Triton X-100) and enzymes (such as DNase). The perfusion duration and reagent concentration are adjusted according to the type and size of the organ.150–152 Whole organ decellularization techniques are predominantly developed in research institutions and have not yet seen widespread commercialization. The future direction involves improving decellularization efficiency and enhancing scalability, automation, and standardization to better serve regenerative medicine and organ transplantation. Commonly used dECM materials include dECM scaffolds and hydrogels. dECM scaffolds are frequently employed in applications that requires to maintain the tissue/organ’s original shapes and structures. By designing the diameter and distribution of microfibers, the architecture of the ECM network can be systematically manipulated to induce specific cellular functions. This is particularly useful in studies involving tissues/organs with multilayered or multi-regional characteristics, diverse cell types, and functional features such as glands and ducts. On the other hand, dECM hydrogels, which require processes such as solubilization and cross-linking, possess high water content and offer greater design flexibility and tunability. Cross-linking can enhance their gelation kinetics and mechanical properties. These hydrogels are well-suited for minimally invasive applications, such as injections, and are often used in scenarios requiring more irregular shapes or where precise shape conformity is not critical. In summary, dECM scaffolds and hydrogels each have their own advantages and the choice depends on specific requirements and application objectives. For preparing dECM materials, the decellularization process must not only remove cellular content and nuclear material while preserving the native ECM structure, but also eliminate potential contaminants or residual detergents. However, different decellularization protocols can still impact the immunogenicity and functionality of the final product. For instance, damage-associated molecular patterns (DAMPs) are danger signals released upon cell and ECM damage during graft processing or implantation. Incomplete or harsh decellularization protocols may leave residual nuclear and mitochondrial DNA, as well as fragmented components like hyaluronic acid and fibronectin, which are renowned DAMPs. These remnants can interact with the innate immune system post-transplantation, potentially leading to graft damage or even transplant failure. Therefore, selecting appropriate decellularization and postprocessing protocols and further developing new methods is crucial. Additionally, the dECM may be digested, degraded, or extracted to create coatings or thermoreversible gels as required. This process can be divided into three main stages: the washing phase, sterilization phase, and processing phase. The washing phase typically employs detergents, enzymes, or denaturing agents to lyse cells and degrade animal tissue. This is followed by rinsing with agents such as PBS to remove residual detergents from the dECM, as these contaminants are often cytotoxic and can inhibit cell proliferation during recellularization. The methods summarized above are commonly combined and can be optimized by adjusting concentration, time, and other parameters to enhance decellularization outcomes. 125 We consider the sterilization and processing phases as the “postprocesses of dECM,” which will be introduced in the following discussion.
Postprocesses of dECM
Postprocesses of dECM to improve biocompatibility before application is necessary. Although methods such as acid-base or certain solvents can effectively remove cellular components and have sterilization effects, they may not achieve ideal sterilization due to inadequate penetration, and over-penetration may damage dECM components. Therefore, appropriate disinfection and sterilization processes can be selected based on different sources, decellularization methods, and application scenarios.
Common physical methods include UV radiation and irradiation. UV radiation is simple and easy to perform, 153 and can be used for surface and environmental disinfection. However, it has weak penetration and is usually used in combination with other disinfection methods.27,154 Irradiation sterilization can directly damage and induce the production of peroxides and free radicals from water molecules, disrupting the genetic material, enzymes, and proteins of microbes, thereby impairing their metabolic functions.155,156 Ethylene oxide (EtO) sterilization inactivates microbial macromolecules, 157 representing a well-established method. However, EtO is toxic and soluble in water, leading to potential residual toxicity post-sterilization, necessitating weeks for complete sterilization. It is noteworthy that the use of EtO sterilization is not recommended for loose and porous freeze-dried dECM due to these concerns.156,158 Peroxyacetic acid (PAA) concentrations of 0.001%, 0.003%, and 0.3% exhibit bactericidal, fungicidal, and sporicidal activities, respectively, with their degradation products being non-toxic and thus safer, while minimally impacting the dECM proteins and its physicochemical properties.159,160 Antibiotics can help reduce contamination during the decellularization process, 161 yet their efficacy is limited and may leave microbial residues, necessitating subsequent sterility and antibiotic residue testing for dECM.162–164 Supercritical carbon dioxide (ScCO2) presents a promising approach with its superior penetration and solubility features, offering an eco-friendly method for disinfection and sterilization. Utilizing ScCO2 can reduce the time required to produce the final dECM and is beneficial for its long-term preservation.22,128,165 It is well-known that disinfection can kill or inhibit the activity of pathogenic microorganisms, but it may not completely eliminate all microorganisms, such as bacterial spores. Sterilization, on the other hand, eradicates all forms of microorganisms, including viruses, fungi, and spores, achieving a sterile state. Disinfection is more convenient and cost-effective, making it suitable for some laboratory-based tests, but it is not appropriate for clinical applications. Ethanol and UV are common disinfection methods; 70%–90% ethanol or isopropanol is frequently used for surface disinfection of materials, and thinner dECM materials may be treated with UV irradiation or radiation. However, for clinical application, sterilization of dECM materials is required. EtO and autoclaving are the routine use of sterilization methods in clinical applications. For dECM materials with high structural integrity, peracetic acid and ScCO2 are recommended. For heat-sensitive dECM materials, EtO and PAA are considered appropriate options. 166 Both disinfection and sterilization may alter the physical and chemical properties of dECM to some extent (e.g. certain collagen cross-linking and macromolecular chain breakage) or lead to the presence of residual sterilizing agents. After all, successful application of dECM materials in clinics require both safety and efficiency. Further studies are needed to explore the clinically acceptable sterilization methods for various dECM materials.
After decellularization and sterilization processes, organs and tissues inevitably undergo changes in ECM shape, biomechanical properties, and collagen fiber exposure. These alterations not only hinder the preservation of the native ECM structure but also adversely affect cell growth and penetration into internal regions, thereby limiting the infiltration and diffusion of oxygen and nutrients. Solubilization and crosslinking are therefore recommended to tune the mechanical properties and physical characteristics of ECM scaffolds. The primary purpose of dissolution is to break down proteins, glycoproteins, and other macromolecules within the dECM and enhance their solubility in water. Additionally, this process can partially dissolve residual cellular components, further reducing immunogenicity. In contrast, the main purpose of crosslinking is to enhance the mechanical strength and stability of the dECM, retard the degradation rate, and mask exposed antigenic sites to reduce immunogenicity. Decellularized tissues are typically digested using chemical and biological agents, such as proteases, and then subjected to physical agitation to achieve the desired concentration of a gel-like solution. Upon termination of the digestion process, the dissolved dECM proteins spontaneously reassemble through intramolecular cross-linking.167,168 This approach maximally preserves the corresponding bioactivity of dECM materials, reduces purification steps, and retains essential growth factors and small molecule peptides present in natural ECM.
Crosslinking enhances the material’s mechanical strength and maintains a stable 3D network structure. Glutaraldehyde is a commonly used chemical crosslinking agent for collagen-based materials, which can reduce calcification and preserving tissue mechanical properties.169,170 Additionally, ethylene glycol and genipin can serve as crosslinking agents with reduced cytotoxicity.171,172 Studies have also found that weak crosslinking with genipin and modification with fibronectin and fibrinogen coatings yield excellent cell viability. 173 Furthermore, a poly (2-hydroxyethyl methacrylate) (pHEMA) coating for materials may offer better biocompatibility, anti-calcification, and anti-thrombotic properties than glutaraldehyde. 174 Photooxidation crosslinking offers the advantages of being non-toxic, causing minimal calcification and inflammation, preventing ECM degradation, promoting regeneration of collagen and glycosaminoglycans, and cell infiltration. 175
The application of solubilization and crosslinking techniques in the processing of dECM has distinct focuses. Solubilization is primarily employed to reduce scaffold stiffness, whereas crosslinking is used to enhance the mechanical properties and stability of the scaffold. Consequently, these techniques are often combined as complementary steps to optimize the final dECM material. However, some challenges remain. Solubilization may lead to the loss of certain ECM components, such as elastin and GAGs, potentially affecting the scaffold’s mechanical properties and biocompatibility. Although crosslinking can slow the degradation rate of dECM, providing sufficient time for angiogenesis and new ECM synthesis, extensive crosslinking—especially with chemical crosslinkers—may increase the M1/M2 macrophage polarization ratio, leading to chronic inflammation and foreign body reactions. Therefore, the degradation rate of dECM scaffolds must be finely tuned to balance stability and functional remodeling, minimizing fibrosis and chronic immune responses. In general, while the decellularization process aims to remove cellular and nuclear components to reduce immunogenicity, incomplete removal may lead to immune reactions post-implantation. Residual DNA and cellular proteins, such as vimentin, could trigger immune responses. Selective antigen removal and solubilization are two key methods for reducing the immunogenicity of dECM. However, excessively harsh methods should be avoided as they may damage the ultrastructure of dECM or even increase its immunogenicity.
dECM-based materials for organoids engineering and organ systems
The construction of organoids and engineered organs necessitates three fundamental components: seeded cells, culture medium, and matrix. Seeding cells primarily include ESCs, iPSCs, ASCs, and tumor-derived cells. The culture medium is tailored to the specific characteristics, tissue features, and genotype of different types of organoids, requiring specific additives such as growth factors and signaling pathway inhibitors to optimize culturing conditions. The matrix provides structural support and certain nutritional components for the cultivation of organoids. Currently, the most common matrices are animal-derived commercial hydrogels, such as Matrigel, which have significantly advanced the development of organoids in recent years. However, their complex composition and performance instability remain limitations that restrict the clinical application of cultured organoids. An ideal organoid matrix should possess excellent mechanical properties, high biocompatibility, non-toxicity, and non-immunogenicity. Moreover, it should support and enhance cellular activities, mimic the specific microenvironments of various cells, and interact favorably with cells to facilitate component renewal. 176 Utilizing dECM derived from tissue-specific ECM is a promising approach in matrix design, displaying tissue and organ specificity, and can be enhanced with relevant proteins and cellular molecules to foster the development and construction of organoid models that closely mimic natural conditions. Figure 3 provides a summary of the advancements and applications in the field of organoids and engineered organs utilizing dECM materials.

Overview of the application and research progress in organoid and engineered organs based on dECM materials. dECM materials exhibit tissue and organ specificity and can be enhanced with relevant proteins and cellular molecules to promote the development and construction of organoid models that closely mimic natural conditions. The utilization of stem cells and organoids to populate whole-organ decellularized scaffolds, followed by cultivation in bioreactors, has significantly advanced the field of whole-organ bioengineering. With the rapid progress of these technologies, they demonstrate immense potential for applications in regenerative medicine and organ transplantation, offering promising avenues for addressing current challenges in these fields. (Created with bioRender.com).
dECM materials for gastrointestinal organoids and engineered organs
For gastrointestinal diseases, particularly those prone to cancer cell dissemination, the current standard treatment involves surgical intervention, known as “en bloc” resection. This encompasses the removal of not only the affected organs but also the concomitant resection of the mesentery containing lymphatic vessels and lymph nodes. Following partial or total organ resection, defects in the gastrointestinal tract are typically compensated by adjacent organs, such as the reconstruction of the esophagus and stomach using the stomach and jejunum. However, adverse sequelae such as loss of reservoir function, impaired anti-reflux barrier, dumping syndrome, and drastic changes in food intake and nutritional status often impact the postoperative course of the disease. The use of tissue-engineered materials obtained from the patient’s own body or other healthy donors for the construction of gastrointestinal-like organs represents an ideal approach for repairing or regenerating tissue or organ defects resulting from disease, surgical resection, and trauma. 177 Three-dimensional microscale organoids of the gastrointestinal tract, referred to as gastrointestinal-like organs, have been established through the in vitro culture system by embedding stem cells into ECM materials. 178
Currently, the cultivation of most gastrointestinal organoids relies on Matrigel. Due to the aforementioned limitations associated with Matrigel, there is growing interest in utilizing dECM derived from porcine gastric and intestinal tissues for the cultivation of gastrointestinal organoids. The dECM hydrogels extracted from these tissues have been found to possess physiological characteristics and mechanical properties similar to commercially available gels, capable of supporting the development and function of gastrointestinal organoids, thereby enabling long-term passaging and transplantation (Figure 4). The gel maintains appropriate hardness, which is foundational for the formation, survival, and differentiation of gastrointestinal organoids. In comparison to Matrigel, the dECM hydrogel exhibits higher abundance and diversity of collagen subtypes and proteoglycans, upregulation of ECM and cell proliferation-related genes, specific enrichment of key ECM proteins associated with organoid formation, as well as the preservation of numerous exosomal proteins involved in cell adhesion. Therefore, dECM hydrogels derived from porcine gastric and intestinal mucosa/submucosa can support the survival and growth of human gastrointestinal organoids, and even hepatic and pancreatic organoids, both in vitro and in vivo, maintaining structural and characteristic protein expression at the molecular level. They can also serve as a tool for the growth of cells derived from human organoids with stable transcriptomic features and for the delivery of organoids into the body.38,179 Functional loss of the small intestine due to disease or injury hinders adequate nutrient absorption and may lead to malnutrition and dehydration, a condition known as intestinal failure (IF). Short bowel syndrome (SBS) describes a state of malabsorption that typically occurs following extensive small bowel resection and is the most common cause of chronic irreversible IF (SBS-IF).180,181 The applicability of small-scale organoid units in extensive SBS-IF is limited. A proposed approach involves the engineering of a human intestinal mucosal transplant by seeding patient-derived organoids and intestinal fibroblasts onto a scaffold derived from decellularized human and patient-derived intestines, forming a functional human intestinal organoid in vitro, which can be utilized for functional analysis and in vivo transplantation.41,42 This approach represents the first use of pure primary human-sourced cells and biomaterials for bioengineered transplants, closely resembling true organ nanoarchitecture and physiology. The variability of fluid dynamics and its impact on epithelial morphology, as well as the ability to capture neuro-muscular peristaltic activity, are future directions for improving and developing this approach.43,182
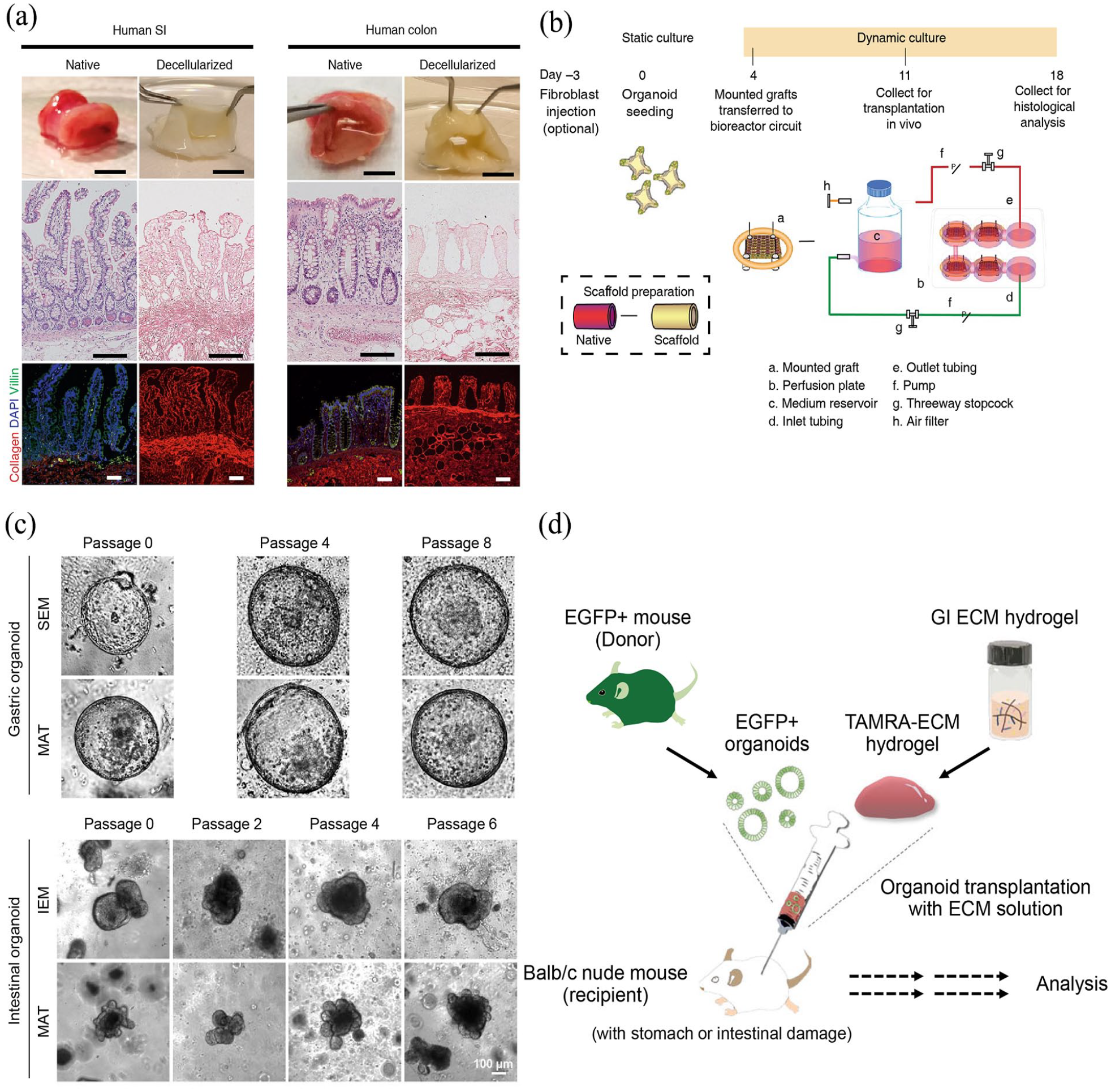
Application of dECM materials in gastrointestinal organoids and engineered organs culture: (a) decellularization characterization of human SI and colon scaffolds, (b) a schematic representation of a strategy for the vitro preparation of functional human jejunal mucosal grafts using a bioreactor, (c) brightfield images of gastrointestinal organoids cultured in decellularized stomach-derived ECM (SEM) or decellularized intestine-derived ECM (IEM) hydrogels and Matrigel, (d) a schematic diagram illustrating the application of dECM hydrogels carrying GI organoids as grafts for treating acute epithelial injuries in mouse models of the stomach and intestine.
dECM materials for renal organoids and engineered organs
The kidneys are a pair of bean-shaped organs in vertebrates, responsible for the elimination of waste products and uremic toxins from the bloodstream, contributing significantly to maintaining internal balance. It has been established that a significant portion of renal diseases are caused by or involve alterations in ECM, for example, fibrosis; however, current therapeutic approaches of treating ECM changes in kidneys are still limited. Notably, recurrent acute kidney injury can lead to irreversible kidney damage, progressing to chronic kidney disease (CKD) and potentially culminating in end-stage renal failure. In advanced stages of chronic kidney failure, the kidneys are unable to function properly, and current mainstay clinical treatments rely on lifelong dialysis or kidney transplantation. However, dialysis is costly, adds to the psychological burden of patients, and is associated with complications such as cardiovascular diseases and infections. Kidney transplantation is an effective treatment for end-stage kidney disease but faces a chronic shortage of kidney donors. Kidney organoids represent a potential tool for producing transplantable kidneys. The decellularization and recellularization of kidneys into kidney organoids provide a solid foundation (Figure 5). Kidney dECM preserves the complex structure, composition, and microenvironment of natural kidney tissue, supporting the self-organization and differentiation of stem cells or progenitor cells into specific kidney cell types, such as nephrons, collecting ducts, and stromal cells, closely resembling the cellular diversity found in natural kidneys.183–185 Different decellularization protocols can yield materials with varying histological properties, 186 human cell re-endothelialization degrees, 150 varying degrees of preserved microvascular integrity and functionality, thereby influencing the differentiation of various embryonic stem cells (ESCs) into renal lineages.187–189 This offers new insights into the importance of optimizing decellularization protocols.
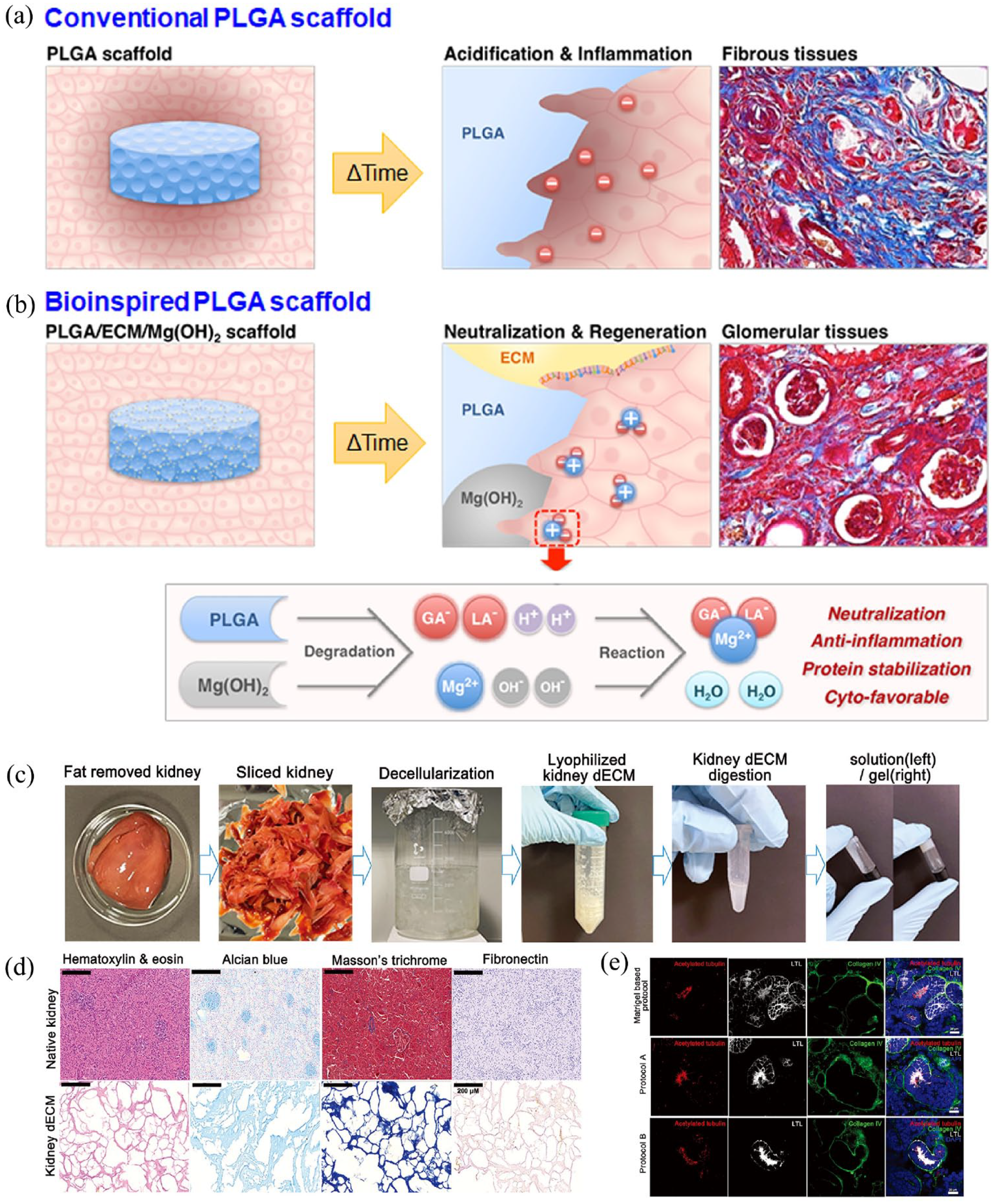
Application of dECM materials in renal organoids and engineered organs culture. Schematic illustration of (a) conventional and (b) dECM-derived bioinspired scaffold for renal tissue regeneration, highlighting their biological and chemical mechanisms, (c) the decellularization and gelation processes of porcine kidney and (d) characterization, (e) renal organoids cultured on kidney dECM exhibit more mature tubular structures
Kidney dECM is currently considered one of the most promising biomaterials for constructing renal organoids and bioartificial kidneys.190,192 However, insufficient recellularization of scaffolds in vivo still challenges the applications of kidney dECM. Kidney is a highly vascularized organ and complete vascular structures is crucial for recellularization of engineered kidney. Kim et al. have utilized kidney dECM hydrogels to culture renal organoids derived from hPSCs, which have a broad vascular network and self-derived ECs, as well as a more mature glomerular development pattern and higher similarity to human kidneys. Transplanting renal organoids into the kidneys of mice accelerated the recruitment of ECs from the host mouse kidneys, maintaining vascular integrity through more organized slit diaphragm-like structures. 190 Researchers have established an in vitro induction protocol for the generation of stromal progenitors (SPs) from mouse PSCs. When these induced SPs are combined with two distinct types of induced parenchymal progenitors (nephron progenitors and ureteric buds), the resultant organoids, entirely derived from PSCs, are capable of recapitulating complex renal structures, with multiple types of stromal cells interspersed among differentiating nephrons and branching ureteric buds. This approach holds promise for better replicating the intricate architecture of organs. However, the proximal tubules (PT), responsible for the majority of renal reabsorption functions, remain immature in renal organoids, with limited expression of key transporters critical for nephron functionality. Vanslambrouck et al. have shown that enhanced specification of metanephric nephron progenitors leads to elongated and radially aligned proximal nephrons, featuring distinct S1–S3 PT cell types. Compared to standard renal organoid protocols, this approach yields organoids with improved PT maturity, exhibiting an increase in the expression of a range of critical transporters, which translates to enhanced functionality as measured by substrate uptake and transport.193,194
Recent years have also seen progress in kidney fabrication via bioprinting. Combining 3D bioprinting with renal organoid technology allows for the rapid, high-throughput generation of renal organoids with reproducible cell numbers and cell type ratios, facilitating easier access to oxygen and nutrients due to the organoid’s structural integrity. 183 However, generating complex organs like the kidney via 3D bioprinting presents several challenges. Firstly, the kidney’s structure is complex, composed of both macrostructures such as renal arteries, veins, and ureters, and microstructures like nephrons and the stroma that occupies the space between them. Secondly, with over 20 different cell types in the kidney, selecting and correctly positioning these cells is challenging. Moreover, choosing suitable biomaterials to preserve the printed kidney’s structure and function warrants careful consideration.
dECM materials for hepatic organoids and engineered organs
The liver is a vital organ for metabolism and homeostasis in the body. The high mortality rate associated with end-stage liver disease (ESLD) poses a significant global public health challenge. According to data from the World Health Organization, liver disease ranks as the 12th most common cause of death worldwide. Currently, orthotopic liver transplantation (OLT) is the most effective therapeutic option to enhance the survival and life quality of ESLD patients. Whole-organ bioengineering and regenerative medicine represent promising new technologies that could alleviate liver shortages by increasing the number of organs available for transplantation (Figure 6). Functionally bioengineered livers (FBLs) are a promising alternative to OLT, with recent studies reporting successful in situ transplantation of FBLs prepared using rat whole decellularized liver scaffolds (DLSs) in rats subjected to total hepatectomy, effectively prolonging survival. 195 Transplantation of human liver organoids co-cultured in decellularized natural liver scaffolds into rabbits subjected to bile duct ligation improved short-term survival and liver function. 196 Decellularized liver scaffolds have been successfully prepared from a variety of species, including rats, pigs, dogs, and humans, with porcine-derived decellularized materials being widely utilized. 197 In recent years, due to the broad availability of porcine organs and their compatibility with human liver size and structure, decellularized porcine livers have attracted increasing interest for constructing engineered liver.118,119,198,199 Ansari et al. have indicated that a combination of saponin, sodium deoxycholate, and deionized water perfused through the hepatic artery and portal vein may yield decellularized scaffolds with an intact vascular system and preserved ECM. 119 Faccioli et al. described a protocol promising complete decellularization of an entire porcine liver within 3 days, resulting in a scaffold that preserves tissue integrity, supports cell adhesion, and produces vital hepatic proteins such as albumin, demonstrating functional viability. 118

Application of dECM materials in hepatic organoids and engineered organs culture: (A) The preparation process of DLSs and (B) the morphology of their intrahepatic bile duct tree. (C) The vascularization process of DLSs. (D) Macroscopic images and (E) SEM images of DLSs (a) and vascularized liver scaffolds (VLSs) (b) after blood perfusion. (F) A photograph of the perfusion culture apparatus placed in a CO2 incubator, utilized for the recellularization of bile ducts in DLSs. (G) Reconstruction of biliary tree-like structures was achieved through the recellularization of bile ducts using liver ductal organoids.
In the field of liver regenerative medicine, ex vivo preservation of the liver on organ perfusion devices is essential for liver and bile duct reconstruction and repair, with the use of stem cells or hepato-biliary cells being indispensable. Functional ductal organoids (FDOs) and bile duct organoids are valuable sources of hepato-biliary cells for application, as these organoids can be generated from relatively small liver biopsies and yield a large number of genetically stable cells, which are crucial for the construction of liver organoids and organ engineering.44,202 Recent research has successfully constructed FDOs with well-developed biliary networks from primary biliary cells isolated from mouse bile ducts in rat DLSs. 200 Other studies have cultured differentiated organoids showing uniform differentiation and even polarization using porcine liver scaffolds and effective recellularization. Additional research has successfully reconstructed a hollow intrahepatic duct-like organ with a single-layered cell membrane within a decellularized 3D scaffold of the liver. Prior to differentiation, this organ exhibited characteristics resembling bile duct cells and retained the potential for proliferation and stem cell differentiation. Introduction of the hepatic duct-like organ into the bile duct network of rat-derived dECM led to successful recellularization and reconstruction of intrahepatic bile ducts, maintaining bile duct cell properties, exhibiting major bile duct functions, and reconstructing a ductal tree-like network with luminal structures. Furthermore, it was observed that when the hepatic duct-like organ underwent recellularization in conjunction with primary liver cells, it maintained its characteristics as bile duct cells, thus providing a more accurate simulation of the natural cellular distribution within the liver. 201
dECM materials for pulmonary organoids and engineered organs
Respiratory diseases rank as the third leading cause of mortality globally, with lung transplantation facing similar challenges of donor shortage and the necessity for lifelong immunosuppression. 203 Decellularized lung scaffolds and hydrogels are increasingly utilized in lung and lung organoid bioengineering through decellularization and recellularization processes (Figure 7); however, a fully functional pulmonary organ has yet to be realized. Numerous studies have employed stem cells from various sources, including induced iPSCs, ASCs, and ESCs, to cultivate lung organoid models.

Application of dECM materials in pulmonary organoids and engineered organs culture: (a) procedural workflow for the production and transplantation of bioengineered lung into a large animal model, (b) schematic representation, and (c) images of branching morphogenesis over a 20-day culture period, illustrating the promotion of airway-like tubular morphology in lung organoids through epithelial-mesenchymal interactions and the physicochemical properties of the ECM.
Researchers have induced these stem cells to differentiate into lung-specific cell types, such as tracheal, bronchial, and alveolar cells, by employing specific culture conditions and signaling pathways, including the use of particular growth factors (e.g. Fibroblast Growth Factor (FGF)-2, FGF-10, FGF-7) and cytokines.206–208 Additionally, Leiby et al. have proposed an organoid model utilizing decellularized lung slices (preserving alveolar, airway, and vascular substructures for cell interaction within a physiologically relevant 3D environment) for the multilinear recellularization to generate engineered lung tissue, comprising alveolar-like structures with multiple cell lineages. This organoid model facilitates the study of cell-cell and cell-matrix interactions within the alveoli, as well as the biochemical signaling regulating type II alveolar epithelial cells and their microenvironment. 208 Güney et al. introduced a novel scaffold material, Agar-Matrigel scaffold (Agrigel), forming a mechanically tunable ECM with adjustable viscoelasticity and stiffness, capable of cultivating long-lived, contractile lung tubule organoids, termed bronchotubules. This method enables the long-term stable culture of the bronchotubule system, offering possibilities for simulating different bronchial diseases’ microenvironments, such as COPD and IPF. 204 Valdoz et al. discovered that soluble ECM and dECM in the 3D suspension culture of lung organoids, by enhancing carbohydrate metabolism and preventing cell dissociation, improved cell viability. The cultured lung organoids developed luminal structures, showcasing viable gas exchange units, branching formation, perfusable vascular systems, and luminal structures maintained for approximately 70 days. 209
One of the goals of lung organ engineering is to generate functional capillaries with healthy microvascular walls to maintain blood passage in the capillaries and restore the natural barrier properties between the vasculature and airway compartments. 210 However, current recellularization techniques struggle to achieve complete endothelial coverage.151,211 A theoretical model has been constructed to calculate quantitative values for pulmonary capillary recruitment and barrier hydraulic conductivity. It was found that in native tissues, increasing pulmonary artery perfusion to normal cardiac output resulted in elevated average capillary pressure and diameter, rather than an increase in microvascular/capillary recruitment. Conversely, in decellularized tissues, increased perfusion rates led to increased microvascular recruitment without affecting the size and fluid dynamics of a given capillary flow path, at least for the tested perfusion rates. This suggests that decellularization may lead to changes in the microvascular hemodynamics of the lung. 211 Excitingly, Nichols et al. reported the formation of normal microvasculature and systemic support within bioengineered lungs (BEL). Following the transplantation of BEL into porcine subjects, it was observed that the vasculature continued to develop, expressing a range of markers associated with vascular function, including CD31, ERG1, eNOS, and ACE. Additionally, within two weeks, BELs developed a vascular system through collateral circulation, showing no signs of rejection; however, pulmonary artery and vein anastomosis was not achieved. 205 Of course, there are many aspects that require further research, such as achieving engineered barrier functions with ultra-low levels of capillary-alveolar hydraulic conductivity to mimic natural whole-organ barrier functions. Even with high levels of cellularization, natural levels of hydraulic conductivity require endothelial tubulization and organized tight junction formation, design criteria that thus far are challenging to achieve ex vivo. 211 Additionally, the anastomosis of the bronchial circulation and lymphatic system to establish functional vascular circulation has yet to be realized.
dECM materials for pancreatic organoids and engineered organs
Diabetes is a global health condition necessitating long-term management and monitoring. Currently, pancreatic or islet transplantation represents the a reliable treatment modality for achieving glucose homeostasis in patients with Type I diabetes, with intraportal islet transplantation standing as the standard approach for β-cell replacement. Organ decellularization and recellularization are emerging as a promising solution to address the scarcity of viable organs available for transplantation by offering potential alternative sources of donor organs.212–214 (Figure 8). The microenvironment is crucial for the regeneration of islet-like organoids from stem cells, with organoids comprising all major endocrine cell types, including α, β, δ, and pancreatic polypeptide cells, assembled into tissue structures akin to human islets. Studies have utilized a natural material derived from decellularized rat pancreatic extracellular matrix (dpECM) to induce the self-assembly of human islet-like organoids during the pancreatic differentiation of iPSCs. Exposure of iPSCs to dpECM during differentiation resulted in significant upregulation of critical pancreatic transcription factors such as PDX-1, MAFA, and NKX6.1, and prompted the production of all principal hormones, including insulin, glucagon, somatostatin, and pancreatic polypeptide from stem cell-derived organoids. 215 Further research, through bioinformatics analysis of collagen derived from decellularized pancreatic ECM, has for the first time demonstrated that ECM-derived peptides dictate the fate of iPSCs, giving rise to endocrine progenitor cells and subsequent islet-like organoids. 216

Application of dECM materials in pancreatic organoids and engineered organs culture: (a) schematic diagram of a custom bioreactor and the assembly steps for vascularized endocrine pancreas (VEP), (b) long-term in vivo analysis following VEP transplantation, (c) pre-transplant VEP imaging, along with gross pathological assessment, and (d) vascular density analysis at 9 weeks post-transplantation, (e) schematic representation of the protocol for decellularized mouse pancreas perfusion and the recellularization of directed differentiation into PP using hPSCs within a bioreactor.
Recent studies have indicated the potential of dECM from mouse, rat, pig, sheep, and human pancreata to generate usable decellularized pancreatic scaffolds.102,103,217–223 These studies have optimized various decellularization protocols for pancreatic tissue to generate dECM scaffolds with improved 3D structural preservation, GAG retention, and cellular biocompatibility. Goh et al. has investigated the differentiation of hPSC into pancreatic progenitor (PP) lineage cells and their reseeding into decellularized organ scaffolds, showing a more favorable outcome for cell proliferation and reconstitution of pancreatic organ functionality. Interestingly, it was observed that reseeding decellularized liver scaffolds with hPSC-PP cells yielded more promising results compared to pancreatic scaffolds. Through chemical complementation and bioreactor cultivation, these hPSC-PP cells demonstrated robust growth throughout the entire liver scaffold, with the engrafted cells exhibiting endocrine functionality. This enhanced performance in the liver scaffold may be attributed to its higher vascular density, which facilitates more uniform support for cell reseeding. Additionally, the liver’s vascular architecture allows for easier perfusion, thereby promoting nutrient delivery and waste removal within the 3D reconstructed organ. Furthermore, the liver may serve as a potential supportive site for pancreatic islet transplantation. 215 Recellularization and refunctionalization of native dECM scaffolds using mesenchymal stem cells from recipients could potentially restore organ structure and function while reducing adverse immunological reactions during transplantation. 218 Citro et al. have combined decellularized lung scaffolds as a platform for β-cell replacement with neonatal pig islets and EC s derived from healthy subjects to construct endocrine pancreatic organs with immunoprotected and vascularized features. This technique not only promotes the maturation of neonatal pig islets ex vivo but also sustains immediate function in vivo post-transplantation for over 18 weeks. 117 Islet infusion into the portal vein remains the preferred method for islet transplantation, despite almost immediate loss of islet quality post-implantation. Therefore, methods that avoid direct intravascular placement are needed, with the omentum emerging as a promising extrahepatic site. Berkova et al. compared different methods of decellularization of the pancreas, including perfusion through the pancreatic duct, gastric artery, portal vein, and a novel method via the splenic vein, aiming to provide a more suitable microenvironment for islet survival in the omental flap. They found that pancreatic perfusion via the splenic vein may offer a smaller ECM scaffold, which aids in the transplantation of islets into the omentum. 224
dECM materials for uterine organoids and engineered organs
Numerous intractable uterine pathologies, leading to reproductive disorders, as well as conditions such as uterine failure, may be amenable to treatments involving uterine organoids, bioengineered reconstruction of the uterus and its tissues, and uterine transplantation.225–227 Daryabari et al. have provided the first efficacious perfusion-based decellularization protocol for obtaining human uterine whole-organ scaffolds. Employing a peristaltic pump to perfuse SDS through the uterine artery generates a scaffold that contains ECM components similar to the native human uterus. 228 A combined physical and chemical method for uterine decellularization has also been reported to more effectively remove cellular remnants and detergents, while preserving the scaffold’s three-dimensional structure and mechanical properties. 229 Studies have demonstrated that combining decellularized extracellular matrix derived from the uterus with chorionic villus mesenchymal stem cells for uterine restoration can lead to efficient live births and regenerate functional uterine tissue in rats with severe uterine damage. The reconstructable uterine material (RUM) can be tailored into different states (such as liquid RUM and solid RUM) and shapes (e.g. rectangular, triangular prism, and cube), effectively preventing intrauterine adhesions, promoting endometrial regeneration and myometrial collagen reconstruction, and accelerating wound healing through the construction of physical barriers and secretion of cellular factors, thus achieving effective uterine restoration.230,231
Recellularization of dECM with MSCs can create a conducive regenerative microenvironment by increasing anti-inflammatory interleukin-10 levels, enhancing the number of FOXP3+ regulatory T cells and CD163+ M2 macrophages, and reducing the number of CD8+ cytotoxic T cells 232 (Figure 9(a)–(c)). Currently, most human endometrial organoids (hEOs) are cultured in 3D microenvironments that are significantly different from the human endometrium, limiting their applicability in bioengineering. Gómez-Álvarez et al. have introduced a hybrid endometrial-derived hydrogel that combines the rigidity of PuraMatrix (PM) with the bioactive components of natural scaffolds and the interactive properties of decellularized porcine endometrial extracellular matrix (EndoECM) hydrogel. Due to the biochemical similarity of this hydrogel to natural tissues, it has enhanced the differentiation efficiency of hEOs, demonstrated excellent in vivo stability, and exhibited xenobiocompatibility in mice within two weeks. 233 Additionally, hydrogels developed from decellularized human and bovine endometrium have been shown to support the growth of mouse and human endometrial organoids, displaying proteomic profiles more akin to native tissues than organoids cultured in Matrigel, with the solid form of scaffolds being suitable for supporting high-throughput cell culture-based applications 234 (Figure 9(d)–(g)).
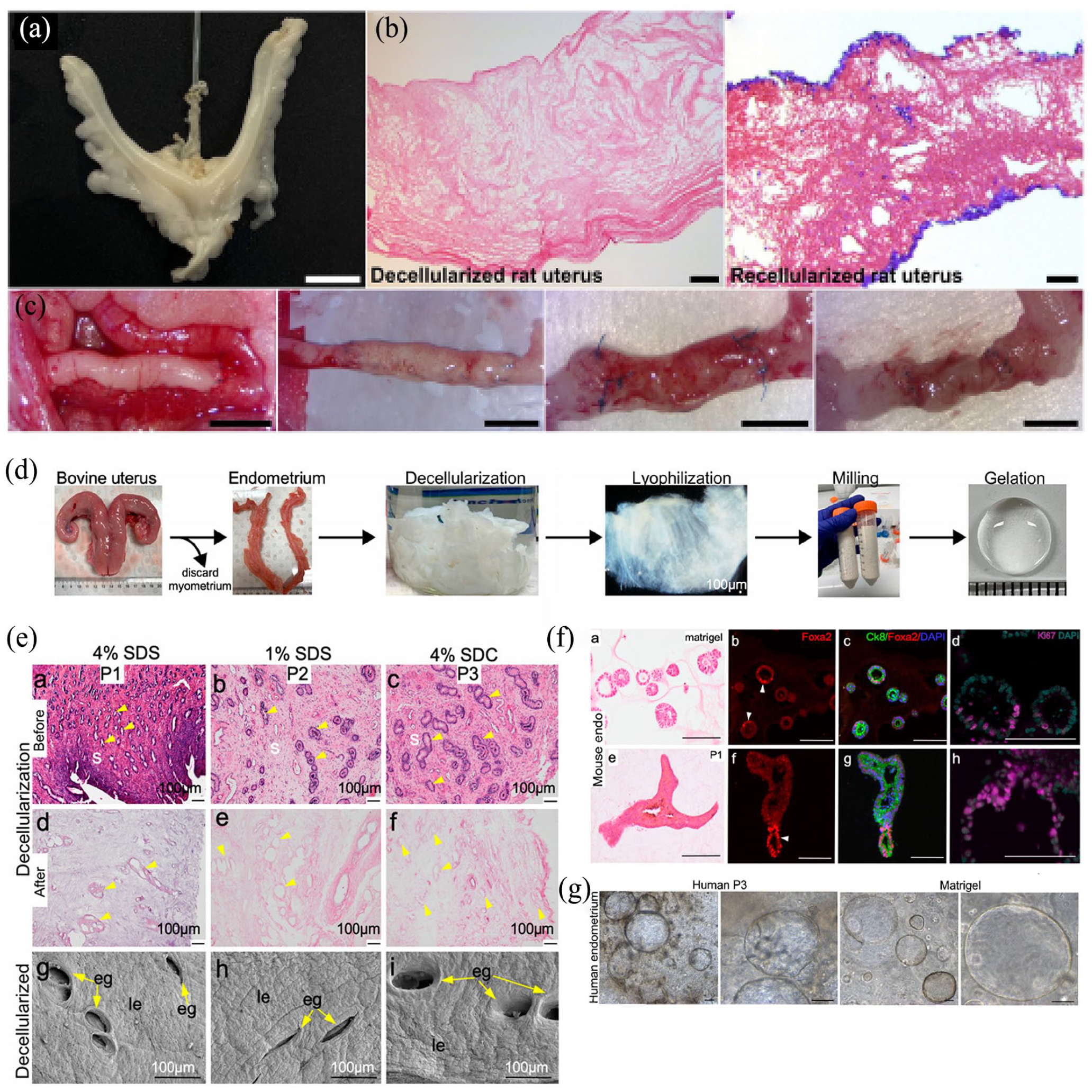
Application of dECM materials in uterine organoids and engineered organs culture: (a and b) decellularization and recellularization of rat uterus via vascular perfusion, followed by (c) graft evaluation at 14, 30, and 120 days post-transplantation, (d) overview of the key steps in the preparation of dECM hydrogel from bovine endometrium, (e) histological images of natural and decellularized endometrial tissue sections, (f and g) Endometrial dECM hydrogel supports the culture of human and mouse uterine organoids.
dECM materials for neural and other fast developing organoids and engineered organs
Diseases in the field of neuroscience remain difficult to cure. A major difficulty is the lack of ideal model systems that can help to the discovery of effective therapies for complex central nervous system (CNS) disorders. Human CNS organoids, including brain and spinal cord organoids, have garnered significant attention due to their closer anatomical and physiological resemblance to the human brain and spinal cord 235 (Figure 10). Spinal cord injury often results in severe loss of motor and sensory functions, and due to the extremely limited regenerative capacity of spinal cord neurons, the local pathological microenvironment is unfavorable for repair. The surrounding tissues have difficulty compensating effectively, leading to challenges in functional recovery, and current treatment options show limited efficacy. Organoid technology can be combined with stem cell therapy to provide a supportive microenvironment that promotes neural regeneration and functional restoration. Recent work by Wu et al. utilized decellularized brain ECM hydrogel (DBECMH) derived from rat brain tissue to culture spinal cord organoids from hiPSCs. Compared to Matrigel, organoids cultured in DBECMH exhibited higher expression levels of natural spinal cord segment-specific markers. 236 Wang et al. developed a temperature-sensitive hydrogel using dECM from human placenta (DPECMH), constructing a stable and quantifiable spinal cord organoid system. Spinal cord organoids cultured in DPECMH displayed a richer expression of regional spinal cord markers, such as FOXA2 and PAX7, significantly accelerating differentiation and maturation. 110 The development of brain organoids has also opened new avenues for research into neurodevelopmental disorders and diseases such as microcephaly, schizophrenia, and Alzheimer’s disease. Organizing brain-like structures can be achieved by culturing neurons derived from hiPSCs on brain dECM prepared from adult pig brains. 32 Seo et al. introduced a microfibril-aligned 3D culture platform utilizing pig brain dECM, which resulted in hydrogels rich in neurotrophic factors capable of better supporting neuronal growth and maturation, thereby promoting the formation of neural circuits. 237 Additionally, Cho developed a 3D organoid culture platform that simulates brain tissue by integrating dECM from human brain tissues with a bioreactor (microfluidic device). The finely regulated dynamic flow provided by this system significantly reduced neuronal apoptosis and the formation of necrotic areas, enhancing the maturity and quality of the brain organoids. 238 Pancreatic cancer (PC) are characterized by tumor cells that frequently invade neural tissues. Chenyun Song et al. constructed a coculture system of human brain organoids (hBrO) and mouse pancreatic cancer organoids (mPCO) using Matrigel, partially recapitulating the pathophysiological processes of neural invasion in PC. However, a limitation of this system is the lack of an immune cell population and the maturity of the myelination of the neurons within the brain organoids. Utilizing dECM may offer a pathway to create more mature organoid systems. 239

Application of dECM materials in neural organoids and engineered organs culture: (a) light-sheet microscopic bright-field images of brain organoids cultured for 60 days using a microfluidic decellularized human brain tissue-derived brain ECM (BEM) system, (b) Flowchart illustrating the preparation process of DPECMH, (c–d) DPECMH induces the formation of spinal cord organoids exhibiting complex layered structures similar to those of natural spinal cord tissue.
Researches in application of dECM materials in regenerative medicine are increasing in the past decade. Beyond the aforementioned research advances, the applications of dECM in cardiovascular, bone and cartilage, and male reproductive systems are drawing more and more attention recently. In studies focused on the repair and reconstruction of bone and cartilage, bioactive scaffolds derived from dECM materials not only provide suitable resistance to mechanical stress and physicochemical properties but also exhibit excellent capabilities to promote the regeneration and differentiation of bone and cartilage cells. 17 For example, Yuan et al. developed an osteo-microenvironment stage-regulative scaffold by combining decellularized bone matrix microparticles and multifunctional magnesium hydroxide nanoparticles with polylactic acid-glycolic acid at an optimized ratio. This scaffold demonstrates enhanced stimulatory effects on bone immunomodulation, angiogenesis, and osteogenesis, allowing better adaptation to different post-transplant stages, including early inflammation, intermediate angiogenesis, and late bone formation. 19 In addressing the increasing prevalence of infertility, especially among young men unable to cryopreserve sperm due to cancer treatments, in vitro sperm culture and maintaining the integrity of immature testicular tissue in vivo are promising solutions. 240 For instance, Gholami et al. optimized decellularization protocols to more effectively remove cells from rat testicular tissue, potentially providing a suitable scaffold for in vitro reconstruction of seminiferous tubule-like structures and functions, aimed at sperm generation. 241
Heart transplantation is the ultimate treatment for end-stage heart failure, and developing cardiac organoids and functional engineered hearts presents potential alternatives. In a structurally complex organ like the heart, using cardiac dECM scaffolds retains the major protein component and structure of heart ECM and shows superior performance in mechanical testing compared to synthetic scaffolds. Goldfracht et al. combined heart dECM hydrogels, enhanced with chitosan and derived from porcine hearts, with hiPSC-derived cardiomyocytes from patients with genetic heart diseases to construct novel engineered heart tissues. These tissues displayed anisotropic muscular structures, and the embedded cardiomyocytes exhibited more mature characteristics than those in 2D culture, demonstrating various phenotypes of arrhythmias under drug treatment, with enhanced sensitivity to a range of pharmacological agents and programmed electrical stimulation. 242 In treating valvular heart disease, traditional mechanical and bioprosthetic heart valves have limitations. Decellularized pulmonary valve allografts are gaining attention. Vafaee et al. utilized low-concentration SDS to decellularize porcine pulmonary roots, which were then implanted into juvenile sheep’s right ventricular outflow tract. After 12 months, these grafts functioned well, resembling their pre-implantation state, with leaflets remaining soft, thin, and pliable. The study also showed effective recellularization of porcine pulmonary root dECM by sheep cells, predominantly mediated by M2 macrophages. 243 Aortic aneurysm and other large vessel diseases often require repair surgery, yet current implants poorly match native tissue, increasing blood flow pulsatility and negatively impacting perfusion supporting related organs. Decellularized natural aorta is an ideal scaffold for aortic implants. Giovanniello et al. employed an optimized decellularization regimen using SDS and DNase to minimize mechanical and structural alterations in tissue, yielding porcine aorta dECM with static and dynamic layered scaffold characteristics closely resembling native human aorta, offering potential as the next generation of aortic implants. 27
dECM materials for cancer organoids
Despite significant advances in cancer research, cancer continues to pose a severe global health threat. Tissue-engineered tumor models, such as organoids, are getting attention in disease modeling and drug development in cancer research. The ECM in tumors differs considerably from that in normal tissues in terms of deposition, composition, stiffness, and post-translational modifications, indicating a close interplay between ECM and tumors. Therefore, constructing decellularized tumor ECM (dT-ECM) for tissue-engineered tumor models can better simulate the actual tumor microenvironment (TME). 244 Tienderen et al. discovered that compared to tumor-free liver matrices (TFL-M), patient-derived decellularized cholangiocarcinoma matrices (CCA-M) better preserved the CCA-related TME. Organoids cultured with CCA-M exhibited transcriptomic features very similar to patients’ tumor tissues in vivo and showed resistance to clinical chemotherapeutic drugs. Furthermore, the growth and proliferation dynamics of organoids differed under the environmental conditions of CCA-M and TFL-M, with CCA-M enabling CCA organoids (CCAO) to better replicate in vivo tumor invasion patterns and ECM-epithelial-mesenchymal transition (EMT), further proving the interaction between tumor cells and their ECM. This finding suggests that ECM may serve as a new therapeutic target for CCA, providing new insights into CCA therapy 245 (Figure 11). Choi et al. developed a lung tissue-derived dECM (LudECM) hydrogel using porcine lung dECM as a 3D-printing bioink, incorporating endothelial cells, lung cancer-associated fibroblasts, and immune cells for recellularization to create a vascularized lung cancer model. Compared to Matrigel and non-vascularized models, the LudECM hydrogel provided signaling stimuli that sustained continuous interactions between cancer cells and stromal cells or cancer cells and stroma within the TME, making lung cancer organoids more representative of real tumor tissues. The organoids in this model better recapitulated pulmonary fibrosis and exhibited significantly increased resistance to targeted anticancer drugs. 246 Digestive system tumors, such as colorectal cancer, are prone to peritoneal metastasis, which significantly lowers survival rates and worsens prognoses. dECM scaffolds derived from patients’ tumor peritoneum exhibited higher stiffness than normal peritoneum, indicating that increased stiffness of ECM facilitate colorectal cancer cell invasion. Normal dECM and Matrigel lack signals associated with cancer metastasis. Experimental results showed that colorectal cancer organoids grew earlier on tumor-derived peritoneum dECM than normal peritoneum dECM, indicating that tumor-derived dECM contribute to cancer metastasis. 247

Application of dECM materials in cancer organoids culture: (a) schematic representation of the steps for generating a comprehensive tumor model by culturing organoids using dECM from excised CCA specimens and TFL, (b) representative 3D live cell confocal images demonstrate that CCA-M enhances the growth and migration of CCAO.
Current limitations and future directions
dECM is considered as a promising material for constructing organoids and engineered organs, as it helps to make organ models more closely resemble their natural counterparts. However, there are several limitations remains to be addressed. For instance, dECM derived from different tissues and animal sources exhibits variations in composition and structure. To better mimic the complex environment of human native tissues and organs, human-derived dECM materials are ideal; however, the relatively limited availability of human sources restricts their application. In addition to the issues previously mentioned regarding decellularization and postprocessing, the consistency between batches may be affected due to differences in the source and preparation processes of dECM. Moreover, different dECM materials has a unique component and require a specific sterilization method, so that the product can meet the safety requirement of clinical application. Further research is still needed to improve our understanding and application of dECM, especially in selecting ECM sources that more closely mimic human tissues, improving decellularization methods, preserving incorporating more ECM components, reducing immune rejection responses, and promoting in vivo angiogenesis.
dECM materials are considered to undergo a dynamic remodeling process during recellularization process both in vivo and in vitro. 248 In the early stages, they experience matrix remodeling that leads to a fetal-like state, which facilitates the release of bioactive fragments and factors within the dECM. This release enhances cellular activation and chemotaxis, supporting tissue regeneration. 249 As the process progresses, matrix deposition facilitates a transition to a mature state. This transition involves the regulation of various immune and inflammatory cell factors, where moderate deposition supports tissue regeneration and functional recovery, whereas excessive deposition results in fibrotic tissue formation, leading to dysfunction and regenerative failure. 250 Therefore, an ideal dECM material must adapt to this dynamic process, meeting the growth and developmental needs of seeded cells or organoids to effectively promote tissue regeneration and repair.
Different organ models necessitate distinct substrate components. For instance, laminin-111 is a pivotal component of the intestinal basement membrane, while laminin-11, type IV collagen, and fibronectin are critical for the biliary tract.251,252 The stiffness and composition of the ECM undergo significant changes in pathological tissue. dECM not only provides adhesion properties but also regulates organ development and organoid formation by offering an appropriate mechanical and physical environment. Diseases such as inflammatory disorders and cancer are often associated with organ and tissue fibrosis. Hyaluronic acid, collagen, and laminin degradation products have been shown to induce inflammation, while pathological changes, such as the marked increase in collagen and laminin content, lead to the replacement of low-density ECM with high-density ECM.253–255 To better replicate the authentic environment of various organs, it is crucial to optimize the culture conditions of organ models. For example, culturing intestinal organoids in hypoxic conditions and co-culturing with anaerobic bacteria can more closely mimic in vivo conditions. 256 Therefore, when constructing disease models, it is vital to consider the differences in dECM stiffness and composition and to identify methods that better simulate the in vivo environment for cultivating organ models.
The risk of inflammation and immune rejection can be mitigated by utilizing natural matrices, optimizing implantation pathways, or employing various novel decellularization techniques, particularly for xenogeneic or allogeneic donor organs. However, the potential of dECM as an ideal transplantable organ material is challenged by the difficulty of achieving uniform distribution of endothelial cells throughout the organ. This issue leads to thrombosis, loss of vascular patency, and subsequent cell and organ death due to the inability to deliver oxygen and nutrients throughout the organ. 257 Therefore, an intact and continuous endothelial layer is crucial for maintaining normal vascular system functionality. Additionally, to ensure the functional characteristics of organs produced during the recellularization process, it is essential to maximize the diversity, initial density, and specific distribution of various cell types present in the parenchymal space of natural organs.
Conclusions and outlooks
dECM is designed to remove immunogenic cells while preserving the original tissue architecture and composition. Owing to its inherent structure, enhanced bioactivity, reduced immunogenicity, and favorable biodegradability, dECM has garnered widespread attention in the fields of tissue engineering and biomedical applications. The prevalent decellularization techniques include physical, chemical, and enzymatic methods, which can be employed individually or in combination. Currently, dECM-derived materials have been increasingly applied in the preparation of organoids engineering and other engineered organs. Further research into the specific impact of ECM from different tissues and disease sources on dECM and subsequent bioengineering is needed to enhance the physiological complexity and clinical relevance of organ models.
Despite the challenges in the exploration of dECM for the fabrication of organoids and engineered organs, ongoing research and technological advancements are bringing us closer to the goal of creating viable, transplantable tissue and organs. These developments include the optimization of the decellularization process, refinement of organoid construction methods, scale-up production, and the enhancement of the matrix’s biological cues to guide cell behaviors. The continuous improvement of these technologies holds great promise for the future of organoids engineering and engineered organs fabrication.
Footnotes
Acknowledgements
The authors would like to express their sincere gratitude to all copyright holders and organizations who have generously granted permission to reproduce and cite figures and images in this review. We acknowledge the contributions of numerous individuals and institutions whose work has been pivotal in illustrating and supporting the content of our article. We have made every effort to respect and adhere to the copyright policies and permissions granted by each copyright holder.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was supported by the National Natural Science Foundation of China (8217081586), and Shenzhen Science and Technology Major Project (KJZD20230923114302006).
