Abstract
Exosomes are nano-sized extracellular vesicles (EVs) released by diverse types of cells, which affect the functions of targeted cells by transporting bioactive substances. As the main component of exosomes, non-coding RNA (ncRNA) is demonstrated to impact multiple pathways participating in bone healing. Herein, this review first introduces the biogenesis and secretion of exosomes, and elucidates the role of the main cargo in exosomes, ncRNAs, in mediating intercellular communication. Subsequently, the potential molecular mechanism of exosomes accelerating bone healing is elucidated from the following four aspects: macrophage polarization, vascularization, osteogenesis and osteoclastogenesis. Then, we systematically introduce construction strategies based on modified exosomes in bone regeneration field. Finally, the clinical trials of exosomes for bone healing and the challenges of exosome-based therapies in the biomedical field are briefly introduced, providing solid theoretical frameworks and optimization methods for the clinical application of exosomes in orthopedics.

Introduction
Bone healing is a complex physiological process regulated by a precise spatiotemporal network of synergistic effects of different cells, tissues and bioactive molecules.1,2 In the initial stages of bone healing, immune cells release pro-inflammatory cytokines and start an inflammatory response. With the activation of immune cells, tissue debris and bacteria at injured site are removed. 3 Subsequently, during the repair phase of bone healing, different cells are recruited to initiate the tissue repair process. The polarization of macrophages toward M2 anti-inflammatory phenotype promotes the formation of bone immune microenvironment. 4 With the help of M2 macrophages and different mediators, osteogenic-related cells initiate the repairing phase. Angiogenic factors expressed by endothelial cells (ECs) induce the inward growth of blood vessels and facilitate the regeneration of vascularized bone. 5 Mesenchymal stem cells (MSCs) undergo initial proliferation and osteogenic differentiation, and form callus structures via ossification.6,7 Finally, under the synergistic influence of osteoclasts and osteoblasts, bone tissue replacement and remodeling are realized. 8 Thus, it can be seen that cells of different lineages participate in the orderly regulation of bone healing, including but not limited to immune cells, ECs and osteogenic-related cells. However, it can be disrupted with advancing age or under certain pathological conditions, which may lead to abnormal bone remodeling and induce the occurrence of bone diseases including bone defects, osteoporosis, osteoarthritis, etc.9–11
Stem cell therapy is considered to be the most promising approach to promote bone repair. Stem cells with high self-renewal and rapid proliferation capacity are utilized to maintain the orderly regulation of bone tissues. 12 However, considerable limitations hinder the further clinical application of stem cell therapy, including biosafety concerns due to abnormal differentiation and immune rejection, as well as dealing with issues of stable cell storage and targeted transport. 13 In addition, how to maintain optimal cell efficacy and vitality during cell delivery to patients is also an unsolved challenge. 14 Interestingly, an increasing number of studies have shown that the biological function of stem cells is largely due to their paracrine action.13,14 The exosomes they secrete induce cell differentiation and tissue healing by regulating microenvironment surrounding the damaged tissues. 15 Exosome is a kind of extracellular vesicle (EV), which affects various functions of target cells by transporting different bioactive substances (nucleic acids, proteins, etc.). 2 Compared with traditional stem cell therapy, exosomes hold several advantages, such as excellent biocompatibility, high stability, non-immunogenicity, and strong targetability. 16 Motivated by the above considerations, exosome-based cell-free therapy has emerged as an excellent alternative strategy to conventional stem cell therapy in the field of orthopedic research.
Currently, a growing number of preclinical evidence has shown that exosomes show promising efficacy on bone defect repair, 17 femoral head necrosis, 18 osteoporosis9,10 and other aspects. In addition, exosomes and their contents derived from different parental cells exert different specific therapeutic effects on the process of bone healing in diverse tissue microenvironments, including regulation of the immune microenvironment, vascularization, bone regeneration and bone remodeling at the injured sites.19–22 However, to our knowledge, there have been no reviews dedicated to address the role and mechanisms of exosomes in facilitating osteogenesis. In this review article, we first introduce the biogenesis and secretion of exosomes, and elucidate the role of the main cargo in exosomes, ncRNAs, in mediating intercellular communication. Subsequently, we elucidate the potential molecular mechanism of exosomes promoting bone healing from the following aspects: macrophage polarization, vascularization, osteogenesis and osteoclastogenesis (Figure 1). The aim is to provide theoretical blocks for further exploring the mechanism of exosomes promoting bone formation and remodeling. Then, we systematically introduce construction strategies based on modified exosomes in bone regeneration field. Finally, the clinical trials of exosomes for bone healing and the challenges of exosome-based therapies in the biomedical field are briefly introduced, providing more building blocks and optimization strategies for the clinical application of exosomes in orthopedics.
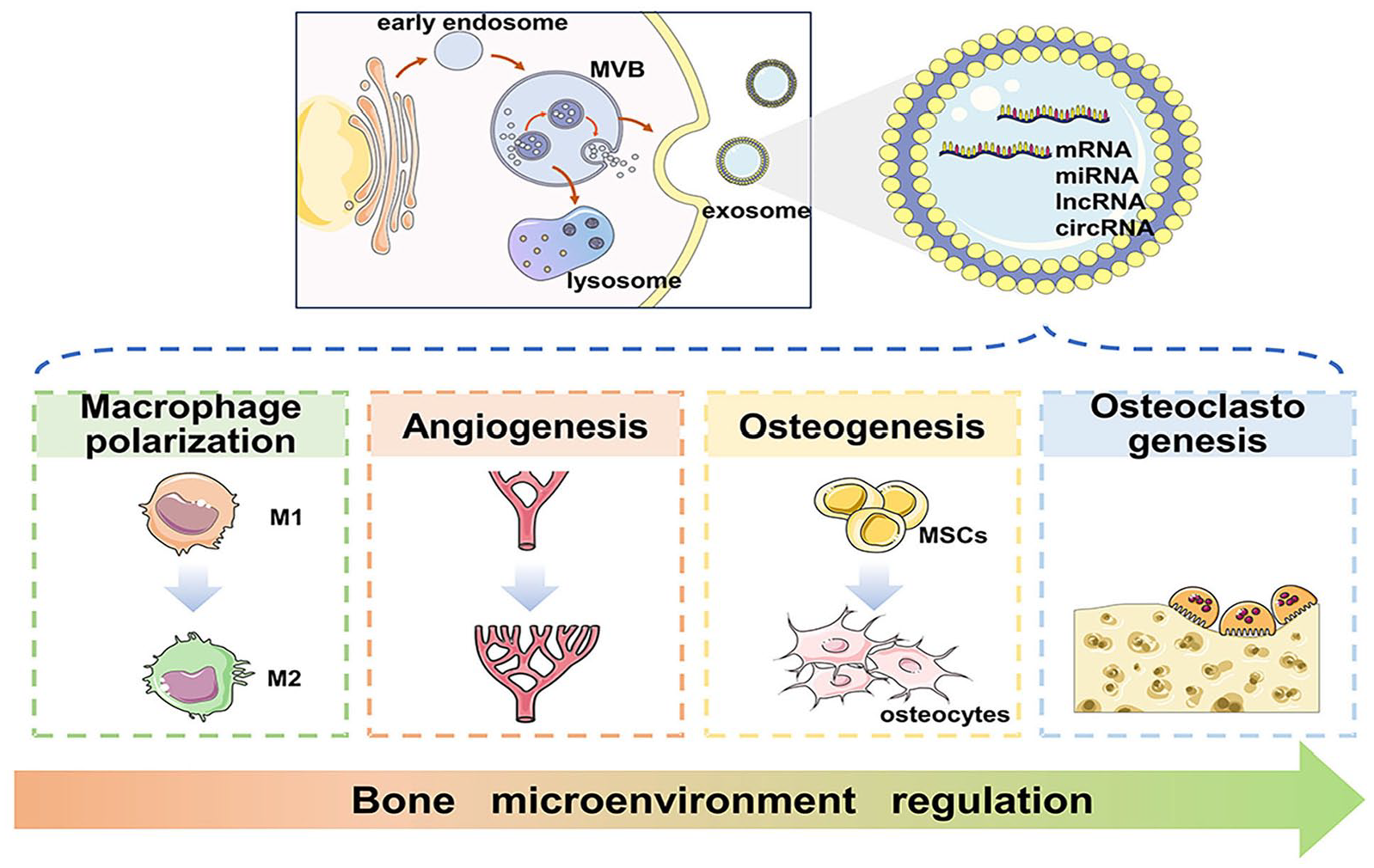
Schematic illustration of exosomes-mediated bone microenvironment regulation.
Exosomes and exo-ncRNAs
Exosomes were first discovered in reticulocytes in 1983, which participated in the release of transferrin receptors during the maturation of reticulocytes. However, when exosomes were just discovered, they were merely regarded as products of cellular metabolism, without affecting other neighboring cells. 23 Exosomes did not attract much attention until Ratajczak et al. first discovered that exosomes transferred RNA they contained to other cells and tissues. 24 With in-depth study, researchers have come to understand that exosomes could mediate intercellular signaling as a natural carrier system (mainly translocating various bioactive materials like proteins and nucleic acids). Thus, the function and activity of target cells are regulated in various physiological and pathological processes, including immune surveillance, inflammatory response, tumor development, etc.25,26
Biogenesis and secretion of exosomes
Exosomes vary in size, density, and shape, mainly depending on the state of their contents. Generally speaking, exosomes are EVs whose diameter range from 30 to 150 nm. These nanoparticles are naturally released by diverse cells through cytoplasmic exocytosis. 27 Exosome biogenesis is a dynamic process. At the beginning, the cell membrane participates in the invagination process to form early endosomes, and gradually late endosomes. Subsequently, the late endosomal membrane buds inward to form multivesicular body (MVB).28–30 Eventually, only a few MVBs fuse with the plasma membrane and release intraluminal vesicles (ILVs) carrying various information into extracellular space to form exosomes. In contrast, most MVBs ultimately fuse with lysosomes and degrade in cells (Figure 1).28,31 During MVBs fusing with the plasma membrane, several intracellular components play an important role, the most import being Rab GTPase. It interacts with cytoskeleton to stimulate vesicle budding and transport. The fusion of MVBs with membrane is then completed mediated by the SNARE complex.28,31
Classification and function of ncRNA
The biological characteristics and effects of exosomes are determined by their composition. 32 NcRNA is the main component of exosomes, whose type and content determine the specificity and function of exosomes. NcRNAs encompass three types of RNA molecules, responsible for controlling cellular functions, namely long ncRNAs (lncRNAs), small ncRNAs (sncRNAs) and circular RNAs (circRNAs). 33 Length is the basic criterion for defining ncRNAs. NcRNAs with more than 200 nucleotides (nt) are called lncRNAs, emerging as a regulator of numerous cell processes. MiRNA with less than 22 nucleotides long are well-recognized sncRNA groups, considered as regulators of post-transcriptional gene expression. CircRNAs are stable and endogenous RNAs produced via reverse splicing events. 34 These ncRNAs not only serve as intercellular signals mediating exosome transport in intercellular communication, but also modulate gene expression at the transcriptional, post-transcriptional, and translational levels of target cells, thereby modulating signal transduction networks. 32 Their characteristics and functions are summarized in Table 1. It is worth noting that the expression levels of exosomal ncRNAs vary significantly in various cells, suggesting their potential role in the treatment of diseases.
The characteristics and functions of ncRNAs in exosomes.

Mechanisms of exo-ncRNAs on bone healing
Recent studies have reported the potential therapeutic benefits of exosomes-mediated osteogenesis. Exosomes containing ncRNAs are significantly involved in various stages of bone healing, including macrophage polarization, angiogenesis, osteogenesis, and osteoclastogenesis. Tables 2–5 and Figures 2–5 summarize the main molecular mechanisms and therapeutic effects of exo-ncRNAs on bone healing, respectively.
The underlying mechanism of macrophage polarization mediated by exosomal ncRNAs in bone healing.

The underlying mechanism of angiogenesis mediated by exosomal ncRNAs in bone healing.

The underlying mechanism of osteogenesis mediated by exosomal ncRNAs in bone healing.

The underlying mechanism of osteoclastogenesis mediated by exosomal ncRNAs in bone healing.

The underlying mechanism of macrophage polarization mediated by exosomal ncRNAs in bone healing.

The underlying mechanism of angiogenesis mediated by exosomal ncRNAs in bone healing.

The underlying mechanism of osteogenesis mediated by exosomal ncRNAs in bone healing.

The underlying mechanism of osteoclastogenesis mediated by exosomal ncRNAs in bone healing.
Regulation of macrophage polarization
Bone healing is a highly dynamic and intricate process, covering three sequential phases, namely inflammation, repair, remodeling. 98 The local microenvironment of bone changes in the initial phase of bone healing, various immune cells accumulate at the defect site, especially macrophages. 19 Activated macrophages are divided into two phenotypes, M1 and M2. M1 macrophages exert proinflammatory effects in bone healing, amplifying inflammatory cascade and inducing osteoclastogenesis by secreting proinflammatory cytokines. This ultimately leads to bone resorption and the removal of necrotic tissues at the fracture site. 3 M2 anti-inflammatory macrophages facilitate tissue regeneration and repair by releasing anti-inflammatory cytokines. Thus, they are recognized as critical regulators of bone formation. 42 Key to the success of bone healing relies on polarizing macrophages toward a reparative M2 phenotype at appropriate time. Conversely, persistent hyperinflammatory response may delay fracture healing. 4 Therefore, rational regulation of macrophage polarization has implications for bone healing.
Previous studies have revealed that exosomes derived from various cells promote forming an M2-like phenotype, thereby reducing inflammatory response and accelerating tissue repair, including bone marrow mesenchymal stem cells (BMSCs), 98 adipose-derived stem cells (ADSCs), 42 human umbilical vein endothelial cells (HUVECs), 4 dental pulp stem cells (DPSCs), 40 macrophages, 99 Schwann cells, 100 etc (Table 2). For instance, Hao Z et al. first reported a neural tissue engineering hydrogel-encapsulated Schwann cell-derived exosomes (SC Exo). This system significantly promoted M2 macrophage polarization to enhance bone regeneration. 100 Interestingly, altering exosome contents through physicochemical stimuli is also verified to be an emerging technology promoting osteo-immunomodulation, such as constructing three-dimensional-printed scaffolds with macrophage-derived exosomes and β-tricalcium phosphate (β-TCP) bioceramics, 101 pretreating exosomes with LPS, 41 TNF-α, 3 or under hypoxic environment. 40 Nakao Y et al. discovered for the first time that TNF-α upregulated CD73 expressed on exosomes, which produced adenosine from ATP to mediate immune suppression and to induce M2 macrophages polarization. 3 All these prior studies hint that exosome contents are the key to influencing the polarization status and attenuating early inflammatory response via delivering bioactive materials and modulating signaling pathway.
Recently, a mounting number of studies suggest that exosomes accelerate the transition from M1 to M2 polarization state via suppressing nuclear factor kappa B (NF-κΒ) pathway, thus leading to attenuation of the inflammatory cascade. 19 Liu S et al. developed tannic acid (TA)-modified polyetheretherketone (TPEEK) to achieve sustained release of miR-21a-5p. It was further confirmed to promote polarization of M2 phenotype macrophages and osteogenic differentiation of BMSCs via NF-κB pathway. 45 Similarly, combining TA modified sulfonated polyetheretherketone (SPEEK) with exosomes exerted strong immunomodulatory effects. The exosomes that released contained inflammation-associated miRNA, promoting M2 macrophages polarization through downregulating NF-κB pathway. 43 It was further demonstrated that hypo-sEV downregulated NF-κB1/p105. As a precursor of p50, p105 can be processed into mature p50 to form dimers with p65. The p65/p50 heterodimer, as the most common form of the NF-κB dimer, binds to the DNA consensus sequences after translocation to the nucleus. 40 Actually, it has been demonstrated that suppressing NF-κΒ pathway deactivates the priming signal of the pyrin domain-containing 3 (NLRP3) inflammasome. NLRP3 inflammasome is confirmed to exacerbate bone inflammation through enhancing osteoclast differentiation. Zhang P et al. first confirmed that LPS pre-Exo loaded with mi-222-3p reduced inflammation by targeting NF-κB pathway and thus downregulating the expressions of NLRP3/caspase-1/IL-1β. 41 Except for NF-κB pathway, the inhibition on NLRP3 inflammasome can be realized by downregulating DDX3X via HUVECs derived exosomal NEAT1 as well. 4
Other signaling pathways have also been demonstrated to convert macrophages from an M1-like to an M2-like phenotype, modulating osteoimmune tissue microenvironment and thus accelerating bone healing. For instance, suppressing IRF1, 19 migration inhibitory factor (MIF), 42 PI3K/AKT pathway in macrophages 99 is also considered as an effective strategy against inflammatory responses. MiR-23a-3p in BMSC-Exos can target the 3′-untranslated region (3′UTR) of IRF1 by bioinformatics analysis, to further modulate macrophages. 19 Exosomes enriched with miR-451a mediate polarization through targeting MIF. 42 Furthermore, analyses and validation of transcriptome sequencing showed that TIM3 in EVs entered macrophages to inhibit p38/MAPK signals and to release bone morphogenetic protein 2 (BMP2) cytokines, thus enhancing osteointegration. 98 As another key protein participating in macrophage polarization, AKT is composed of three serine/threonine protein kinase. It was demonstrated that miR-181b significantly triggered M2 polarization and suppressed inflammation by downregulating PRKCD and activating p-AKT. 44
As discussed above, much effort is currently focused on signaling pathways on how exosomes modulate macrophage phenotypes during bone healing (Figure 2). Given the superior performance of exosomes on metabolic regulation, further exploration in macrophages is warranted from this perspective in the near future. Moreover, the mechanism of exosomes acting on other immune cells has not been revealed. Lin Z et al. proposed that exosomal PD-L1 could act as an immunosuppressant for the treatment of bone fracture. It inhibited T cell activation via specific binding to T cell surface PD-1. 102 While exosome uptake by neutrophils is demonstrated to be involved in the occurrence and development of cancers,103,104 its influence on bone healing is poorly defined. Therefore, deeper investigation is required upon the regulation of exosomes targeting other immune cells.
Regulation of angiogenesis
Angiogenesis is a key factor in achieving high-quality bone regeneration. Sufficient vascular networks support the growth of osteogenesis-related cells through nutrient and oxygen delivery and metabolic waste removal. 36 Conversely, poor vascularization contributes to the lack of oxygen and growth factors, thus impacting bone formation and bone remodeling. 105 Therefore, bone formation depends highly on neovascularization. This multilayered regulatory network is called osteogenic-angiogenic coupling. 7
Type-H vessels, with high expression of CD31 and Endomucin (CD31hiEMCNhi), play a fundamental role in osteogenic-angiogenic coupling. They regulate the growth of bone vasculature, recruit osteoprogenitors. 7 Currently, a growing number of studies has demonstrated that the formation of type-H vessels is strongly associated to exosomes (Table 3). The combination of USC-EXOs with GelMA-HAMA/nHAP hydrogels upregulated HIF1A, which further promoted the formation of type-H vessels. 106 Serum-Exo was reported to inhibit macrophage inflammation, thus leading to the overexpression of type-H vessel markers CD31 and EMCN via upregulating VCAM1 in HUVECs. 107 Interestingly, mechanical stimulation was confirmed to improve vasculogenic potential of exosomes, especially type-H vessels. This appears to be extremely associated with downregulated exosomal miR-214-3p. 51 The mechanism of type-H vessels was studied in depth by Cui Y et al. They discovered that Shn3 gene silencing increased production of slit guidance ligand 3 (SLIT3) and consequently facilitated forming type H vessels. Based on this finding, they developed the engineered exosomes BT-Exo-siShn3, achieving vascularized bone regeneration via specific delivery siRNA to osteoblasts. 7
VEGF is considered as a key regulator in angiogenesis, including, but not limited to type H vessels. 51 It is involved in the activation, proliferation, migration of ECs, thus enhancing blood supply and accelerating bone healing. 51 Therefore, increasing number of studies focus on exosomes-mediated vascularization through VEGF. Hypoxia preconditioning of exosomes is verified to be an effective strategy for activating VEGF pathways.50,108 Gao Yet al. discovered that hypoxic precondition promoted the secretion of exosomes. The corresponding hypoxic exosomes (H-Exo) induced vascularized bone regeneration via stimulating VEGF signals. 108 A similar conclusion has also been reached by Liu PP et al. H-Exo secreted from hypoxia pretreated upregulated VEGF mRNA transcription and protein synthesis via overexpressing let-7f-5p and argonaute 1 (AGO1). 50 HIF-1α, as a critical regulator in a hypoxic environment, dramatically activates VEGF expression. 53 As is demonstrated, PL and PRP-derived exosomes, 109 BMMSC-Exos, 110 USC-Exos, 106 uMSC-Exos 52 are able to trigger angiogenesis by activating HIF-1α and VEGF. Moreover, initiating Wnt/β-catenin pathway is found to overexpress VEGF as well. LncRNA HOTTIP accelerated both osteogenesis and vascularization by interacting with TAF15 to stabilize DLX2. Subsequently, DLX2 initiated Wnt/β-catenin pathway to promote vascularization. 35 Furthermore, Aday et al. Set al. combined lipid nanoparticles (LNPs) with a proangiogenic miRNA, let-7b-5p, to develop a bioinspired “artificial exosomes” (AEs). It was found that inducing neovascularization might be contributing to targeting CTNNB1 (catenin beta 1, aka β-catenin), a part of the Wnt/β-catenin pathway. 111
Acting as the downstream pathway of VEGF and HIF-1α, PI3K/AKT signals also play a substantial role in proliferation, migration and tube formation of HUVECs.112,113 For instance, miR-126 overexpressed exosomes significantly triggered angiogenesis by suppressing phosphoinositol-3 kinase regulatory subunit 2 (PIK3R2), regarded as a negative regulator of VEGF, thus activating PI3K/Akt signaling pathway. 20 The upregulated miR-210-3p in hypo-sEVs was confirmed to downregulate EFNA3 expression, thereby enhancing the phosphorylation of PI3K/AKT pathway. 47 As a putative protein arresting vascularization, overexpressed THBS1 might lead to the inhibition of PI3K and further VEGF/Akt/PI3K cascade, thus suppressing neovascularization. Liao Fet al. reported that THBS1 suppression in ECFC-Exos activated the aforementioned pathway. 114 As is well established, PTEN functions as a negative regulator in AKT pathways. In the study of Gao WL et al, H-Exo significantly downregulated PTEN in endothelial cells, to initiate PI3K/Akt pathways. 115 Another research showed that miR-23a-3p could bound the 3′UTR of PTEN, leading to the suppression of PTEN. 49 Thus, exosome-delivered miR-23a-3p was corroborated to be another effective strategy for achieving vascularized bone regeneration.
Inhibiting TGF-β/SMAD signals can stimulate angiogenesis as well. Tregs-Exos-mediated transfer of miR-142-3p was demonstrated to accelerate angiogenesis through TGFBR1/SMAD2 suppression. 46 Similarly, under the stimulation of bioactive trace element strontium (Sr), elevated pro-angiogenic miR-146a bound to 3′UTR of Smad4 and NF2 mRNA, inhibiting its translation and thus promoting in vitro angiogenesis. 53 In addition to the above-mentioned signaling pathways, other regulators are also thought to be essential. For example, exosomal miR-21 promoted vascularization by upregulating the NOTCH1/DLL4 pathway. 52 Overexpressed miR-210-3p in H-Exo decreased ephrinA3 in HUVECs and enhanced tube formation. 50 Exosomal miR-21-5p targeted SOX5 and negatively regulated its expression, subsequently suppressing EZH2 transcription and promoting angiogenesis. 48 Behera J et al. first demonstrated the NO-dependent nature of exosomes stimulating vascularization via lncRNAH19-Angpt1-Tie2/NO axis. 36 Wu D et al. combined low doses of Fe3O4 nanoparticles with a static magnetic feld (SM), discovering the expression of COL4A2 was repressed by upregulated miR-1260a, enhancing osteogenesis and angiogenesis. 74
The majority of the above-mentioned studies have focused on the vital signaling pathways or specific regulators (Figure 3), however, few studies interrogate the potential mechanisms on forming and assembling focal adhesions (FAs). As is well-known, FAs maintain the adhesion strength of endothelial cells to the extracellular matrix (ECM). Paxillin and vinculin are required primarily for robust FAs. Conversely, the reduced adhesion is likely to trigger enhanced migration and angiogenesis potential. 105 Cheng P et al. reported for the first time that Nidogen1 (NID1), an ECM protein in exosomes, diminished the activity of myosin-10, which targeted FAs to affect cell adhesion and cytoskeletal remodeling. This significantly suppressed the expression of paxillin and vinculin, the reduced adhesion then contributed to enhanced angiogenesis. 105 The study suggests that mechanical insights into FAs should also become a focus of research attention on exosomal-mediated angiogenesis in the near future.
Regulation of osteogenic differentiation
Compared with other systems, the skeletal system shows significant healing capability. During this complex process of bone reconstruction, the regulatory network between the surrounding cells and osteogenic-related cells exerts essential functions. There has been increasing evidence suggesting that acting as vectors of this cellular crosstalk, exosomes released by various cells (such as skeletal muscle cells, 116 HUVECs, 117 macrophages, 118 osteocytes, 55 osteoclasts, 60 etc.) show a key regulatory role in osteoblast differentiation and mineralization (Table 4). Compared with other cellular sources, macrophages-derived exosomes have attracted extensive attention because of unique inflammatory regulation capacity. The study of Wang D et al. first demonstrated that exosomal lncRNA LOC103691165, expressed by macrophages, entered surrounding microenvironment to promote BMSCs osteogenesis. 118 Liu K et al. further highlighted the significance of crosstalk between M1 macrophage and BMSCs. M1-derived exosomes containing miR-21a-5p mediated an original, transient inflammatory process to enhance osteoblast differentiation. In contrast, M2 macrophages significantly enhanced the mineralization capacity of MSCs due to the soluble factors that released, rather than exosomes. 119
The key for exosomes-mediated osteogenic effects lies in the productive uptake of exosomes cargo by recipient cells. 118 After exosomes are transported to indicated cells during stable cycling, the endocytosed ncRNA induces functional changes in osteogenesis-related cells, facilitating MSCs maturation into bone progenitor cells, osteoblast precursors, and eventually osteoblasts. 117 Therefore, how to improve the uptake efficiency of exosomes has become an emerging focus of investigation. Zhou YK et al. first reported the strong association between moesin protein and exosomes endocytosis. After pretreated with H2S, no observable changes were present in the sizes and number of M2 exosomes. Instead, moesin protein in exosomes were overexpressed, which mainly distributed in the surface structure of actin-rich cells to participate in forming cellular skeleton and cell adhesion. Highly-enriched moesin protein in exosomes promoted its endocytosis into MSCs, to modulate bone regeneration. 120 Similarly, hypoxia can also accelerate the uptake by altering the protein profiles of exosomes. Hypoxia preconditioning significantly increased the amount of Bgn protein, which directly interacted with various osteogenic-related proteins. 121 Moreover, hypoxia preconditioning promoted the secretion of exosomes, by increasing the intracellular expression of vesicular transporters (including SNARE complexes (Snap25), Pkm2, and Rab5a), thereby promoting the secretion of EVs. 121 In the study of Su HR et al, significantly overexpressed SLIT2 in osteoblasts was found to participate in the internalization of miR-382-containing exosomes, boosting the capacity of osteogenesis. 66 Recent evidence has showed that traumatic brain injury (TBI) accelerates bone healing. This is mainly because after TBI, ECM protein FN increased on small EVs, allowing osteogenic miRNA-enriched exosomes to preferentially metastasize to skeleton. 21 Compared with target-mediated internalization, the vast majority of studies on exosomes have placed a focus on the regulation of osteogenic-related signaling pathway mediated by ncRNAs predominated in exosomes. 113 Researcher has recently explored the potential mechanism, involving Wnt/β-Catenin, PI3K/AKT, MAPK, STAT, BMP/Smad signaling pathway, etc (Figure 4).
The Wnt signals, especially the canonical signaling pathway, Wnt/ β-Catenin, is one of the most essential pathways during bone healing mediated by exosomal ncRNAs. 85 Exosomes first target glycogen synthase kinase-3β (GSK-3β) to promote its phosphorylation. This activation subsequently induces β-catenin accumulation and its translocation into the nucleus, thus activating Wnt signaling pathway and accelerating bone regeneration. 122 After exosomes being pre-treated, Wnt signaling together with osteogenic effect are differentially impacted. For example, in the study of H2S-pretreated exosomes, the osteogenic effect induced by Wnt signaling pathway is amplified. 120 HU Y et al. discovered that, under the inflammatory condition, GMSC-Exo exhibited effective cross-regulation capacity between NF-κB and Wnt/βcatenin pathway, through partly increasing the phosphorylation of GSK-3β. To be more specific, excessive GSK-3β activity not only upregulated Wnt/β-catenin pathway to enhance osteogenesis, but also adversely affected the NF-κB functionality through inhibiting its nuclear accumulation of NF-κB and binding to promoters. 123 Surprisingly, different conclusion was reached by Lei Fet al. They discovered that PDLSCs-exosomes under the inflammatory microenvironment overactivated the Wnt signals in PDLSCs to suppress bone regeneration. Conversely, exosomes under physiological microenvironment suppressed the over-activation of canonical Wnt signaling to recover osteogenic differentiation capacity. 124 This suggests that microenvironment where exosomes are placed greatly affects the initiation of Wnt signals. And exosomes derived from various cells under the inflammatory microenvironment play divergent roles in Wnt signals and bone regeneration. Furthermore, many ncRNAs in exosomes are verified to upregulate Wnt/β-catenin signaling pathway. For instance, EVs carry miR-335 promoted bone fracture recovery through Wnt/β-catenin pathway. MiR-335 could also target VapB to amplify osteogenic effects. 76 Hao LZ et al. manufactured miR-26a loaded exosomes, modified with bone-targeting peptide Asp8. This novel engineered exosomes showed enhanced osteogenic potential via upregulating Wnt/β-catenin signaling pathway. 57 Exosomes with overexpressed miR-130a-3p led to the downregulation of SIRT7, interacted with Wnt signaling pathway-associated protein through its deacetylation activity to participate in the regulation of osteogenesis. 54 Exosomes are also confirmed to modulate Wnt/β-catenin pathway by targeting receptor gene or protein. For example, miR-136-5p delivered via exosomes inhibited lipoprotein related protein 4 (LRP4), which exerted suppressive effects on osteogenesis. Wnt/β-catenin pathway was then activated to facilitate fracture healing. 85 Exosomes with high-expressed miR-424-5 attenuated osteogenesis via WIF1/Wnt/β-catenin pathway. 84 MiR-328a-3p enriched in the sEVs promoted osteogenesis by directly targeting the 3′UTR of an anti-osteogenic target gene FOXO4. The suppression of Wnt/β-catenin transcriptional activity was subsequently removed. 21 Another research showed that miR-19b repressed the expression of WWP1 or SMAD ubiquitination regulatory factor 2 (Smurf2) to elevate the stability of target protein Kruppel like factor 5 (KLF5). Overexpressed KLF5 subsequently initiated Wnt/β-catenin pathway and facilitated osteogenesis. 73
BMP is regarded as one of the three subfamilies of the transforming growth factor-β (TGF-β) family. 125 BMP receptor complex drives the process of bone repair and regeneration by regulating Smad protein. 89 Exosomes derived from various cell types can regulate the BMP/Smad/TGF-β pathway. 67 Hao Z et al. encapsulated Schwann cell-derived exosomes (SC Exo) in the neural tissue engineering hydrogel. This controllable sustained release system activated TGF-β to stimulate BMSCs osteogenesis. 100 It was further confirmed that SC-exos regulated the TGF-β pathway to accelerate the osteogenic differentiation by transferring let-7c-5p. 126 Macrophages-derived exosomes can also drive osteogenesis via targeting BMP/TGF-β, 127 such as M2D-exos carrying miR-142-3p 64 and miR-486-5p. 63 The Tregs-Exos-mediated transfer of miR-142-3p was likely to accelerate osteogenesis through TGFBR1/SMAD2 suppression. 46 It has been recently found that exosomes in which noggin were suppressed elicited BMPR1A expression to increase BMP/Smad signaling by inhibiting miR-29a. 16 SMAD1/5 is a main driver of osteogenesis, can be mediated by exosomal cargo, such as miR-30a, 72 miR-23a, 72 miR-128-3p, 89 miR-144-5p, 75 miR-100-5p, 5 etc. It was confirmed that miR-30a 72 and miR-144-5p 75 were more effective in reducing SMAD1, while miR-23a 72 and miR-128-3p 89 in reducing SMAD5. Moreover, the induction of mechanical forces is an important initiator of exosome-mediated BMP-Smad1/5 signaling pathway. 15 Wang R et al. discovered that PDLSCs under stretch secreted functional EVs with high levels of miR-200b/c, leading to the promotion of alveolar bone regeneration. After miR-200b/c transferring into cells, it targeted and silenced Smurf1, which then promoted the phosphorylation of Smad1/5, and activated the BMP-Smad signaling pathway, together with a negative regulator of osteogenesis. 58 Li ZC et al. found that miR-17 was enriched in BMSC-Exos under healthy conditions and promoted bone regeneration by targeting Smad7. In contrast, in diabetic conditions, down-regulated miR-17 increased the expression of SMAD7 and hindered the bone regeneration process. 62 Nevertheless, not all kinds of miRNAs directly regulate BMP/Smad signaling. Liu AQ et al. first reported that, Bmpr2 and Acvr2b might have a competitive relationship in Smad signaling. 1 Multicomponent exosomal miRNAs (let-7a-5p, let-7c-5p, miR-328a-5p and miR-31a-5p) led to elicited Smad1/5/9 phosphorylation and enhanced osteo-inductivity of exosomes by regulating the competitive balance of Bmpr2/Acvr2b. 1
PI3K/AKT is another critical signaling pathway to drive osteogenesis, mainly responsible for cell growth, cell proliferation and osteogenic differentiation. 128 Zhao B et al. found that mediated by exosomes, BMSCs cultured in vitro accelerated cell migration and proliferation, which was partially attributed to the adenosine receptor-mediated initiation of AKT and ERK1/2 pathway. 128 The PI3K/Akt signals can be activated by differentially expressed miRNAs within the exosomes, such as overexpressed osteogenic miRNAs (miR-3182, miR-1468) and downregulated anti-osteogenic miRNAs (miR-182-5p, miR-335-3p, miR-382-5p). 59 Compared with normal condition, the osteogenic effect in a hypoxic environment based on PI3K pathway appeared to be more apparent, including osteoblast proliferation, migration, differentiation, and mineralization. 121 Furthermore, Tang YF et al. declared that exosomal miR-140-5p could target IGF1R to suppress IRS1/PI3K/Akt phosphorylation and mTOR pathway, which further hindered osteogenic differentiation of MSCs. 69 PTEN can negatively regulate PI3K/AKT signaling pathway. 61 Exosomes derived from mechanical stress are demonstrated to effectively suppress PTEN activity and to initiate PI3K/AKT signals by upregulating miR-92a-3p. 61 In the study of Xun J et al, serum exosomes from young rats highly expressing miR-19b-3p were found to reverse the decreased osteogenic capability in aged rats. This correlated closely with miR-19b-3p downregulating the expression of PTEN. 129 Similarly, reduced expression of miR-494-3p senescent osteocyte-derived exosomes accelerated age-related bone loss via PTEN/PI3K/AKT pathway. 55
MAPK signaling pathway comprises ERK, p38 and Jun amino-terminal kinases (JNK) signaling. P38/MAPK has currently been corroborated to play a major role in exosome-mediated regulation of osteogenesis. It was found that exosomes activated p38 MAPK pathway to facilitate PDLSCs proliferation, migration and osteogenic differentiation. 130 Furthermore, other upstream signals are also likely to initiate MAPK signals. For instance, PLC/PKC/MAPK pathways contribute to MVs-mediated bone healing. 131 As one of the HOX family, down-regulation of Hoxa7 can activate MAPK pathway as well. Qi L et al. implanted 3Dprinted porous Mg-containing Akermanite biocreamics (Akt) scaffolds into cranial defects of aged rats. Effectively increased bone regeneration was observed. Mechanically, this is because Mg-containing Akt-mediated exosomal miR-196a-5p cargo targeted Hoxa7 to downregulate its expression, which further leading to the activation of MAPK signaling pathway. 132
STAT, functioning as a negative regulator of osteogenic differentiation, plays an important role in exosomal-mediated bone remodeling. STAT directly interacts with Runx2 in cytoplasm to inhibit Runx2 nuclear localization, thereby hindering osteogenesis. 79 Exosome carrying miR-5p-72106_14, 56 miR-935, 79 are both confirmed to negatively target STAT1 and subsequently initiate osteogenic differentiation. It was reported for the first time that osteoclast-derived exosomal miR-5134-5p inhibited bone formation and accelerated the resorption of alveolar bone via the JAK2/STAT3 pathway. 60 They confirmed that miR-5134-5p combined with receptors of tyrosine kinase to form ligand-receptor complexes, which triggered the phosphorylation of JAK via tyrosine sites. Stat3 subsequently get phosphorylated and translocated into the nucleus to regulate transcription. 60
Hippo signaling is another significant pathway in modulating bone regeneration. The activated Hippo signaling sequentially phosphorylates large tongue suppressor kinase 1/2 (LATS1/2) kinases and YAP/TAZ. 17 The effector of Hippo signaling, YAP1, was found to be the direct target of miR-375. 2 Exosomal miR-335 17 and miR-365a-5p 78 were reported to target LATS1 to inhibit Hippo signaling, thus promoting the proliferation and osteogenic differentiation.
RhoA/ROCK pathway-mediated cytoskeletal recombination is critical for osteogenic commitment. 10 MiR-324 released from active osteoclast-derived exosomes promoted osteogenesis by regulating the ARHGAP1/RhoA/ROCK axis. 77 Similarly, exosomal miR-140-3p could promote osteogenic commitment by suppressing plexinB1/RhoA/ROCK signaling pathway. 6
Besides the above-mentioned pathways, ncRNAs in exosomes can also regulate osteogenesis by binding to other targeted proteins. For instance, exosomal lncTUG1 suppressed the expression of miR-22-5p, leading to an increase in the expression of Anxa8 in osteoblast cells, leading to enhanced osteogenesis. 70 It was demonstrated that exosomes from miR-378-modified ADSCs enhanced osteogenesis and angiogenesis by suppressing Sufu expression and activating Shh signaling pathway. 18 Elevated miR-23a in exosomes inhibited osteogenic differentiation of BMSCs via targeting CXCL12. 90 By directly targeting GATA2, miR-21a-5p enhanced MC3T3-E1 activity. 11 Exosomal miR-92b-3p secreted from MVECs under mechanical unloading partially attenuated the function of osteoblasts by downregulating ETS-like transcription factor 4 (ELK4). 71 Exosomal lncRNA-H19 functioned as a molecular sponge for the endogenous function of miR-106a, to negatively modulate Angpt1. Angpt1 administration upregulated NO production, angiogenesis and bone regeneration via Angpt1-Tie2/NO signaling. 36 BMSC-derived exosomal miR-206 promoted cell proliferation and differentiation by reducing E74-like factor 3 (Elf3). 82 M2 macrophage-derived exosomal miR-5106 induced BMSCs toward osteoblastic fate by targeting salt-inducible kinase 2 and 3 (SIK2 and SIK3), which both involved in different processes such as cell cycle regulation, growth and differentiation. 88
Autophagy is a process of cell self-degradation and recycling of intracellular components, which also plays a key role in the process of osteogenesis. 39 Exosomal cargo (circHIPK3, 39 mir-27a-5p, 81 miR-381 87 ) has been reported to promote bone formation through autophagy. This aspect of study will be worth pursuing in the near future.
Regulation of osteoclast differentiation
Bone healing is a highly coordinated process of bone remodeling, balanced by the coupling of bone formation and bone resorption. 133 To be more specific, osteoclasts adhere to decalcified bone, degrading the bone matrix and forming resorption cavities by secreting matrix degradation enzymes. Osteoblasts subsequently fill in the cavities with bone matrix proteins to trigger mineralization. 134 Exosomes are confirmed to optimize the balance between osteoblasts and osteoclasts, which is vital for bone remodeling. 8 Currently, numerous researches investigated the regulation of osteoblast-mediated bone resorption via exosomes. Exosomes derived from various cells are demonstrated to influence the balance of bone remodeling, including MSCs,3,40,91,134 osteogenic-related cells, 93 macrophages,92,133 skeletal muscle cells, 116 pericytes, 135 etc (Table 5).
As is well-known, osteoclasts originate from the monocyte/macrophage hematopoietic lineage, 92 they can be differentiated from M1 macrophages, instead of M2 phenotype. 136 Hakki SS et al. elucidated the impact of macrophages polarization on osteoclastogenesis. They treated macrophages with preosteoblastic MC3T3-E1 clone 4 (MC4)-derived exosomes, discovering the overexpressed mRNA of Cd86, Rankl, etc. This indicated the macrophage polarization toward the M1 phenotype. TRAP staining analysis further suggested a significant increase in RANKL-induced osteoclastogenesis under the treatment of M1 macrophages. 136 Actually, this interaction and complex regulatory network between macrophages and osteoclasts is inextricably linked to macrophages-derived exosomes. Exosomes from M2 macrophages were demonstrated to suppressed RANKL-induced osteoclast differentiation by inactivating proinflammatory-associated CSF2/TNF-α signaling. 133 Similarly, macrophage-derived exosomes induced osteoclast differentiation via miR-3470b targeting TAB3/NF-κB. 92
Mechanistically, increased osteoclastogenesis mediated by M1 macrophages is closely related to pro-inflammatory cytokines that secreted promoting the overexpression of NF κB and receptor activator of NF κB ligand (RANKL). This contributes to the excessive activation of osteoclasts, and the imbalance of bone resorption and bone formation. Therefore, the inhibition of NF-κB and RANKL is considered to be the critical signals to suppress bone pro-inflammatory cytokines in macrophages and osteoclast formation. 2 For instance, silencing of the osteoblastic Shn3 gene suppressed RANKL expression and further osteoclast formation. 7 MiR-210-3p enriched in hypo-exosomes targeted NF-κB1 p105, to induce M2 macrophage generation as well as to inhibit osteoclastogenesis. 40 Exosomes derived from cyclic mechanical stretch (CMS)-treated BMSCs caused diminished osteoclast activity by attenuating NF-κB pathway activity. 137 Given the importance of NF-κB and RANKL in osteoclastogenesis, majority of researchers have recently devoted to excavating their critical regulators. Tumor necrosis factor receptor-associated factor (Traf) is regarded as the upstream of NF-κB pathway. As was reported, exosomal circ_0000722 promoted osteoclastogenesis by upregulating TRAF6 expression and activating downstream NF-κB and AKT signaling pathways. This promoted the translation of NFATc1 and c-Fos, thus enhancing osteoclast formation. 91 Conversely, Traf3 in PC-EVs negatively regulated osteoclastogenesis by inhibiting the non-classical NF-κB pathway. 135 Thus, in-depth and effective research studies on TRAF are required in the near future. Moreover, Wnt5a could modulate the expression of RANKL. To be more specific, miR-1260b in TNF-α preconditioned-GMSC-derived exosomes specifically targeted Wnt5a and JNK to suppress RANKL signaling pathway as well as bone-resorbing activity. 3 Acting as the specific markers of osteoclast differentiation, NFATc1 is the most powerful transcription factor gene stimulated by RANKL23. Zhang C et al. confirmed that lnc-MALAT1 enriched in osteoblasts-exo promoted osteoclasts differentiation by downregulating miR-124/NFATc1 axis. 93 Similarly, Guo S et al. found that driven by GATA4, miR-206-3p suppressed osteoclast differentiation through targeting the 3′UTR of NFATc1 via exosomes. 22 Exosomes enriched with OPG are regarded as another strategy for dampening osteoclastogenesis, because they are nature inhibitors of RANKL, effectively blocking the interaction between RANKL and rank. 95
Researchers have recently revealed several new pathways of osteoblast-osteoclast crosstalk mediated by exosomes. For instance, exosomal circ_0000722 activated downstream AKT signaling pathways. 91 Exosomal miR-501-3p directly targeted the 3′UTR of PTEN gene to inhibit its expression, leading to the subsequent activation of PI3K/AKT signaling pathway. 97 Mechanical force upregulated the expression of ANXA3 protein in exosomes, facilitating exosome internalization to activate the phosphorylation of ERK. 138 The activation of the above pathways, AKT and ERK, both triggers osteoclast differentiation ultimately. Conversely, upregulating Wnt/β-catenin is likely to negatively regulate osteoclastogenesis. Wang Yet al. observed that exosomal miR-27a activated Wnt/β-catenin pathway by inhibiting the expression of dickkopf2 (DKK2) in osteoclasts and osteoblasts. This reduced bone resorption and enhanced bone formation simultaneously. 94 Moreover, Wang X et al. confirmed the interaction between upregulated ubiquitin specific peptidase 7 (USP7) in exosomes and YAP1. The overexpressed YAP1 stimulated the nuclear translocation of β-catenin, thus activating Wnt/β-catenin and inhibiting osteoclast activity. 134 Hu CH et al. shed light on a novel mechanism underlying sympathetic regulation of osteoclasts via exosomes independent of RANKL. Sympathetic neuro-stress through the β1/2-adrenergic receptor (β1/2-AR) signaling was found to trigger the transfer of exosomal miR-21 from osteoblasts to osteoclast progenitors for dictating osteoclastogenesis. 139 Another study demonstrated that miR-6924-5p directly suppressed osteoclastogenesis via exosomes by binding to 3′UTR of OCSTAMP and CXCL12, which are known as well-established regulators governing osteoclast function. 96 These studies mentioned above suggest the multiple possibilities for exosomes-mediated osteoclastogenesis.
In addition to the above signaling pathways (Figure 5), m6A methylation is confirmed to be an effective strategy for osteoclastogenesis as well. As an epigenetic modification, it modulates a variety of essential functions of RNA. Yang JG et al. found a strong correlation between m6A methylation level of NFATc1 gene and osteoclast-induced bone resorption. Specifically, METTL14 released by exosomes could increase the m6A methylation level of NFATc1, which inhibited osteoclast bone absorption via binding to functional site 4249 A. 140 Similarly, this team discovered in previous studies that circ_0008542 upregulated target gene RANK in osteoclast through m6A methylation, to recruit osteoclast-mediated bone resorption. 141
Constructive strategies of exo-ncRNAs to bone healing
Most of purified exosomes lack strong bone-targeting capacity, leading to the ease of their removal from the body in a relatively short period.142,143 Thus, exosomes require proper constructive strategies to realize effective targeted delivery and sustained release of bioactive substances. 144 There are mainly two types of exosomes-mediated bone-targeting constructive strategies, including exosomes combined with biomaterials, and engineered modification of exosomes (intracellular or extracellular strategies; Figure 6).
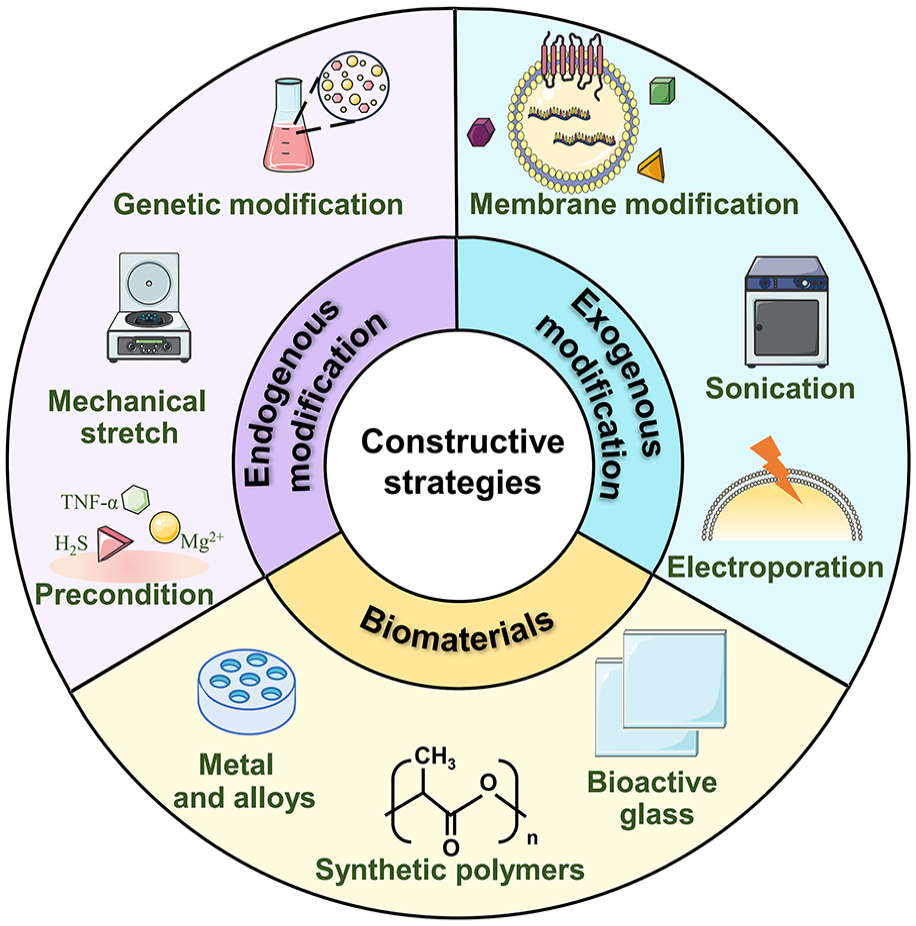
Engineering modification of exosomes for bone healing.
Exosomes combined with biomaterials as therapeutics
Although exosomes are verified to have a remarkable capacity for tissue repair, exosomes are likely to be rapidly scavenged in the circulatory system without vectors. 143 Various biomaterial scaffolds have become a feasible option for carrying exosomes, because they allow the controlled release of exosomes in a dose- and time-dependent manner. 144 Furthermore, as delivery vehicles of cells and cytokines, biomaterials provide proper structural support as well as biochemical clues, to enhance the biomechanical properties of bones.144,145
Metallic orthopedic implants show superior biocompatibility and mechanical properties, playing an important role in the long-term therapeutics for bone healing. 144 In order to improve the osteo-inductivity, researchers attempt to develop novel therapeutic strategies for bone remodeling via altering the surface morphological topography of biomaterials and developing biochemical coatings.142,146 For example, alkali- and heat-treated nano-topography is able to stimulate osteogenic differentiation of MSCs by inducing the secretion of pro-osteogenesis exosomes. 142 In another study, strontium (Sr) and highly bioactive serum exosomes (sEXOs) were both integrated inside a 3D-printed Ti scaffold to enhance osteointegration. 112 Nevertheless, a range of issues lead to unsatisfactory osteogenic effects, including complex fabrication procedure and failure in achieving functional diversification. In comparison, exosomes are enriched in proteins, cytokines, which enable to induce complex and durable biological reactions in vivo in a more safe, high-efficient and cost-effective manner. Thus, the application of exosomes for metallic osteo-implant surface modification is preferred for osseointegration strategy. To achieve mild and stable tethering of exosomes on Ti implants, Ge GR et al. combined mussel-like molecular adhesion with bio-orthogonal click conjugation. This novel surface decoration strategy was confirmed to enhance the osteo-immunomodulatory effect in diabetic patients. 145 The exosome-fibrin combination on the tantalum (Ta) coated surface can also substantially activate osteogenesis, through modulating cell adherence, proliferation, and osteogenic differentiation. As is well-known, fibrin, a kind of reticular protein, provide initial substrate for fundamental cellular processes including adhesion, proliferation and differentiation, during tissue repair. In this research, fibrin functioned as the binding site of exosomes, exerting excellent biological effects by effectively slowing down release of exosomes. Moreover, compared with Ti-based surface, the negative potential of Ta surface favors carrying exosomes as well. 143 Except for fibrin, positively charged polyethyleneimine can also immobilize exosomes to the implant surface. The immobilized exosomes are then slowly released and phagocytosed by BMSCs and macrophages. 125
Currently, acting as delivery vectors, hydrogels are efficiently used for bone defect healing. Hydrogels are a kind of polymer networks, sharing similar properties to ECM (such as excellent biocompatibility and superior biodegradability 108 ). Hydrogels can also carry and sustained release cytokines, such as vascular endothelial growth factor A (VEGF-A) and BMP-2 mRNAs,147–149 stromal cell derived factor-1α (SDF-1α), 150 etc. Compared with other biomaterials, hydrogels, especially injectable in situ forming hydrogel systems, can help address the challenge of exosomes-induced poor stability. 121 They offer a favorable microenvironment for the storage and gradual adsorption of exosomes, and achieve targeted transportation. For example, injectable porous poly(lactide-co-glycolide) (PLGA) microspheres with bioinspired polydopamine (PDA) coating exhibited an effective adsorb and sustained release of exosomes, which induce vascularized bone regeneration. 108 In order to achieve continuously deliver of hypo-EVs in vivo, Deng JJ et al. manufactured an injectable bioactive hydrogel composed of poly(ethylene glycol)/polypeptide copolymers. The injectable polypeptide-based hydrogel exhibited a sol-gel transition with rising temperature. To be more specific, under the trigger of external conditions, free-flowing sols are transformed into solid hydrogels, serving as the depots for sustained release of EVs after being injected into the sites of injury. 121 Different from the aforementioned traditional hydrogels, hydrogel microparticles (HMP) exhibited more outstanding injectability, allowing accurate injection at particular pathological sites to achieve minimally invasive treatment. 151 Moreover, HMP showed greater surface area, facilitating extensive interactions with cells in a local environment. 152 To enhance the adhesion of cells or exosomes, the surface modification of hydrogels is a feasible strategy, mainly modifying the surface with RGD (Arg-Gly-Asp) peptides.152,153 As an efficient adhesion coating, PDA coating is another proper approach for achieving efficient loading and sustained release. 154 This is because after adsorbing on a material surface, PDA is likely to form covalent and noncovalent bonds. 155 Additionally, PDA can be rapidly crosslinked to form a polymer, which stably adsorb bioactive factors. 155
A wide variety of synthetic materials has been explored for the fabrication of hydrogels. Gelatin methacrylate (GelMA) hydrogels have become a broadly useful tool in the field of orthopedic surgery.100,156 This is because GelMA undergoes rapid photo-crosslinking due to the methacrylation on the gelatin side chain.106,126 GelMA can also significantly improve cell adhesion, regulate cell viability and promote cell proliferation. 100 Nevertheless, pure GelMA hydrogels exhibit poor mechanical performance and rapid degradation rate. 157 Lu W et al. incorporated hyaluronic acid methacrylate (HAMA) and nano-hydroxyapatite (nHAP) into GelMA hydrogel to reinforce the degree of densification in fibril network. 106 LAP can also improve the hydrogel mechanical properties by accelerating the gelation of polysaccharide matrices through hydrogen bonds. And because of the rheology modifying capabilities offered by LAP, the shape fidelity of 3D printed GelMA/nanoclay composite hydrogel was significantly enhanced. 157 3D printed bi-layer GelMA composite scaffold was recently developed by Sun T et al. The upper scaffold added black phosphorus (BP) in GelMA to obtain a relatively lower elastic modulus, which was conducive to the differentiation of MSCs into cartilage. While in the lower scaffold, the addition of β-TCP significantly enhanced the elastic modulus, conducive to the osteogenic differentiation of MSCs. 156 The addition of 3-(3,4-dihydroxyphenyl) propionic acid (CA) is another approach for improving mechanical properties of hydrogels. This is likely because catechol groups of CA could form a cross-linking network between collagen molecules. 158 As an essential component of ECM, hyaluronic acid (HA) is regarded as tissue adhesive. 159 HA hydrogels present excellent tissue adhesion and exocrine sustained-release effects. 122 Zhang Y et al. encapsulated exosomes in HA hydrogel. Pore structures of nanohydroxyapatite/poly-ε-caprolactone (nHP) scaffolds were then filled with hydrogels. Excellent osteogenic effects were observed in the cranial defects in rats. 52 Zhao X et al. combined high-biocompatible F127 with o-nitrobenzyl alcohol-modified hyaluronic acid (HA-NB). The novel F127/HA-NB hydrogel was verified with dense network structure, tissue adhesiveness, and dual sensitivity to temperature and light. 122 PEGylated poly (glycerol sebacate) acrylate (PEGS-A) hydrogels are another type of widely used hydrogels, for ensuring the integrity of exosomes and sensitive mRNAs, to maximize the translational efficacy of the genetic materials inside. 147 Furthermore, exosomes combined with other kinds of hydrogels show prior bone regeneration ability, such as poly(D, L-lactide-co-glycolide)-b-poly(ethylene glycol)-b-poly(D,L-lactide-co-glycolide) (PLGA-PEG-PLGA) hydrogel, 160 polyethylene glycol (PEG)/DNA hybrid hydrogel, 113 injectable chitosan hydrogel16,161, chitosan/β-glycerophosphate hydrogel, 162 alginate hydrogels, 153 etc.
The structural innovation on hydrogels has attracted much attention gradually. Hierarchical hydrogel containing dual EVs has been fabricated recently. 163 EVs-200b and EVs-130b were loaded into a hierarchically injectable hydrogel composed of PF-127 (outer layer) and SA (inner layer), respectively. Effective amelioration on bone loss was then verified. 163 Mi Bet al. constructed a kind of cocktail therapy, regulating the balance between osteogenic-related cells and macrophages. This injectable HA hydrogel system allowed for specific delivery of engineered endothelial cell-derived exosomes (EC-ExosmiR-26a-5p) and APY29 (an IRE-1α inhibitor) for osteoblast/osteoclast and macrophage regulation, respectively. 164
Except for metal implants and hydrogels, bioceramics,53,101,165 bioactive glass 1 can also be used for exosomes vectors to promote osteogenesis, which represents a novel and effective treatment option for the application of exosomes. Sun YH et al. successfully constructed a 3D printed bioceramic-induced macrophage exosomes (BC-Exos) scaffolds, with persistent release of exosomes and distinct osteo-immunomodulatory effects. This is because bioceramics alters the paracrine effects of BC-Exos via regulating exosomal miRNA cargos. 101 The mesoporous bioactive glass (MBG) scaffold also achieved sustained release of exosomes, because of their entrapment in the surface microporosity of the scaffold. 1 Nonetheless, given the complexity of the microenvironment in vivo, the intrinsic instability of exosomes has not been improved in this mode of binding, compared with hydrogels. Further exploration is warranted for the combination of exosomes with biomaterials, so as to better understand the implications of exosomes-based treatment.
Engineered modification of exosomes for accelerated bone healing
Compared with natural exosomes, the modified ones exhibit greater treatment efficiency and targeting functions. This is mainly because modified exosomes are rich in numerous exogeneous molecules, including nucleic acids, proteins, etc. 148 In general, engineered modification of exosomes broadly fall in two main categories, namely intracellular modification (endogenous engineering strategy) and extracellular modification (exogenous engineering strategy). Intracellular modification refers to modifying parent cells before exosomes isolation, mainly including genetic and physical manipulations. While extracellular modification means functionalization of the nanoscale-EVs after isolation, such as sonication,166,167 electroporation, 147 co-incubation,164,168 etc. Table 6 summarizes engineering modification of exosomes for bone healing, including specific fabrication method, advantages, disadvantages and main applicable exosomal cargo.
Engineering modification of exosomes for bone healing.

Intracellular modification of exosomes
Intracellular modification of exosomes is a kind of engineered strategy based on parent cells, that is, modifying parent cells before the secretion of exosomes. Genetic engineering strategy is one of the most common modification approaches. With the assistance of transgenic vectors (i.e. plasmid, liposome, lentiviral vectors, etc.), target genes are transferred into cells. The additional functionalization of exosomes is then realized through genetically manipulation on protein biosynthesis of targeted cells.152,169,170 In the research of Huang CC et al and Li Fet al, MSCs were transfected with BMP2-overexpressed lentiviral particles and liposome, respectively. Engineered exosomes were then successfully manufactured with enhanced osteo-inductive properties.169,170 Similar prior osteo-inductive capacity was discovered in engineered exosomes derived from BMP2-overexpressed NIH-3T3 cell lines. 148 The main reason is that BMP2 complementary DNA (cDNA) was locally delivered to the defective bone region, according to the cellular transcription mechanism, which achieved the continuous production of BMP2 in situ. 149 However, a number of issues remain according to this gene-based regulatory strategy, such as high cost, low loading rate of exosomes. 171 In contrast, altering cells’ epigenetics through post-translational modifications has become a more effective and safer alternative methods. This is mainly due to epigenetic regulation only modulating the transcriptional activity of the genome, instead of altering nucleotide sequence. 157 As is well-known, histone plays an essential role in modulating the structure of the chromatin and modifying cell transcriptional activity. The histone deacetylase (HDAC) inhibitor Trichostatin A (TSA) was used to induce hyperacetylation, so as to augment osteoblast epigenetic functionality as well as the mineralization capacity of exosomes, 171 because of the enrichment in pro-osteogenic miRNAs and transcriptional regulating proteins. 157 Methylation is another essential epigenetic mechanism, which activates transcription by augmenting chromatin remodeling. 172 Epigenetic reprograming via synergistic hypomethylation and hypoxia was demonstrated to improve the exosomal efficiency on bone healing. 172
Physical engineering strategy can also directly achieve exosomal functionalization in a safe, low-cost and convenient manner. 173 Specifically, after altering physical forces and culture environment surrounding donor cells, the types and content of exosomal cargo has significantly changed. Cytokines or therapeutic factors that carried increase, thus optimizing treatment efficiency.137,173 Common physical approaches include mechanical stimulation,15,58,71,137,174,175 electroporation, 147 low-intensity pulsed ultrasound (LIPUS), 173 magnetic actuation,74,176 etc. Wang R et al. compared the elevated miRNAs in PDLSCs-derived exosomes after tension stimulation with the untreated ones. MiR-200 family were found to be mechanoresponsive in exosomes. 58 Cells stimulated by tensile stress secreted exosomes enriched with vital molecules in YAP/TAZ-Notch circuit, which was regarded as a positive pro-osteogenic circuit. 174 The opposite phenomenon was found in another research. Exosomes produced by MVECs under clinorotation-unloading conditions prevented MC3T3-E1 cells from differentiating into mature osteoblasts. 71 Nevertheless, the production of stable mechanical stimulation generally requires high-cost large equipment. And difficult issues seem to be intractable, such as how to enable cells to receive consistent and large-scale stimulation. 175 Microfluidic-based mechanical squeezing technique improves exosome secretion and realizes more precise physical stimulation by permeating cell membrane. 175 Employing cellular nano-electroporation with track-etched membranes (TM-nanoEP) is also verified to be effective for allowing high loading of multiple functional mRNAs (BMP2 and VEGFA) in abundant exosomes. 175 What is more, exosomes secreted by hypoxia-induced MSCs appear to be effective cell-free therapies that facilitate bone regeneration. Compared with conventional in vitro culture, (oxygen concentration remains 21%), when cells are exposed to oxygen concentration of 2%–8%, enhanced bone healing capacity are discovered, for exosomes are enriched with more cytokines, pro-osteogenic proteins,177–179 such as VEGF signals, 108 biglycan (Bgn), 121 high mobility group box 1 protein (HMGB1), 180 etc. Some other types of stimulation can also significantly alter exosome cargo. 181 For example, hydrogen sulfide (H2S) stimulated the high expression of moesin protein in exosome, facilitating its endocytosis into MSCs to promote osteogenic differentiation. 120 Metallic ions, such as Zn2+ and Mg2+, can change the exosomal miRNAs to drive osteogenesis.8,182
Extracellular modification of exosomes
Extracellular modification refers to direct loading cargo into isolated exosomes through exogenous methods. The strategies include sonication, 13 electroporation,7,166 co-incubation,164,168 mechanical extrusion, 167 direct EV membrane modification,7,145,183–186 etc. Compared with the intracellular modification, the extracellular ones significantly enhance the generation efficiency of modified exosomes.
Among the widely-available exogenous engineering strategies, sonication shows the highest loading efficiency. 13 According to this strategy, mechanical shear force induced by ultrasonic probe damages the membrane integrity of exosomes. And during the process of membrane deformation, exogenous cargo are promoted to disperse into exosomes, such as fusing CXCR4+ exosomes with liposomes carrying antagomir-188, 167 combining BMP2 with exosomes to develop eBMP2-EVs. 166 The therapeutic activity of modified exosomes, however, may be impacted for the mechanical forces generated by ultrasound can influence the integrity of exosomal membrane. 13 Electroporation is also regarded as one of the most promising strategies, which aims to carrying nucleotides into the isolated exosomes. With the high-intensity current in a short period of time, the phospholipid layer of exosomes is destroyed. After the diffusion of nucleotides into the interior of exosomes, membrane integrity is restored. In the research of Cui YZ et al., the exosomes were loaded with siRNA of Shn3 through electroporation. 7 BMP2 was also successfully loaded into exosomes with the assistance of electroporation. 166 Co-incubating exosomes with genetic modifier is a simple and inexpensive engineered strategy, without destroying the integrity of exosomal membrane. Gui LY et al. conjugated a bone-targeting peptide, (Asp-Ser-Ser)6 ((DSS)6), onto the surface of ApoEVs by 2 h-incubation with rotation at 37°C, to enhance the bone-targeting ability of natural ApoEVs, 168 miR-26a-5p was transferred into exosomes via coculturing with CD9-HuR fusion protein. 164 Mechanical extrusion endows modified exosomes with extra therapeutic effects, by encapsulating various cargo (especially synthetic nanoparticles) into exosomes. The mixture of exosomes and cargo can be achieved by membrane deformation of exosomes. 167 Nevertheless, continuous mechanical extrusion makes it difficult to ensure the stability of exosomal membrane, which is the major limitation of mechanical extrusion. 13 Some novel strategies have been gradually performed. For example, exosomes for protein loading via optically reversible protein-protein interactions (EXPLOR) is a powerful system for loading proteins into exosomes based on optogenetics, which realizes the high-efficient loading of proteins in exosomes. However, long-term safety and stability remains to be clarified. 186
Direct EV membrane modification is considered as another essential exogenous approach, realizing functionalization of exosomes by taking different approaches (such as clicking chemistry and hydrophobic interaction). Compared with natural exosomes with poor retention, engineered sEVs by direct EV membrane modification endow them with tissue targeting ability. 183 Clicking chemistry connects the molecules on exosomal surface with chemical bonds. To be more specific, it is a synthetic process during which molecules are rapidly synthesized via the splicing of small units. 145 Ge GR et al. combined biomimetic peptides containing lysine and noncoding levodopa (DOPA) with clicking reaction, to develop surface modification strategy on Ti-based material. This guaranteed the stable adhesion on implant surfaces and high efficiency of clicking reaction. 145 Nonetheless, such bioconjugation is likely to impair functions of exosomes by obscuring active sites of surface proteins. In this regard, hydrophobic insertion appears to be a proper alternative. 183 Hydrophobic interaction allows membrane modification in a non-covalent fashion, without interfering with proteins on the membrane and their biological functions. Cui YZ et al. modified the bone-targeting peptide with a diacyl-lipid tail, then it was successfully anchored onto the exosome membrane through hydrophobic interaction. 7 Similarly, “DMPE-PEG-CREKA” was fabricated firstly and inserted into the membrane of sEVs with the assistance of hydrophobic insertion. CREKA-sEVs were then constructed to target fibrin to accumulate and retain in situ. 183 Moreover, 3-way junction (3WJ) RNA nanoparticles can also be used for direct EV membrane modification, for its size, shape, and stoichiometry can be precisely regulated. 185 And their properties (negatively charged and hydrophilicity) avoided them from accumulating in vivo, thereby reducing both immunogenicity and toxicity. 185
Clinical application and challenges of exosomes for treating bone healing
Exosomes-related pre-clinical experiments has gradually garnered the attention of researchers in recent decade. On the basis of positive outcomes in previous mechanical researches and animal studies, further exploration on clinical experiments of exosomes is warranted. Actually, to date, there have been few clinical trials evaluating the application of exosomes during bone regeneration.
Exosome-based clinical trials for treating bone healing
We accessed the ClinicalTrials.gov database on September 6, 2024 and searched for the term “bone” and “exosome or extracellular vesicle.” Only two trials were found so as to assess the impact of exosomes on bone healing (Table 7). Both clinical trials are in the state of “Not yet recruiting.” Of those, one aims to use MSCs enriched by EVs to treat patients with bone tissue defects. The treatment efficacy and safety are used as the final evaluation index. The other one uses autogenous MSCs culture-derived signaling molecules as enhancers to evaluate bone density and quantity in bone grafting.
Clinical trials evaluating the effects of exosomes for treating bone healing.

Given the low number of clinical trials mentioned above, we then searched for the term “disease” and “exosome or extracellular vesicle.” More than 300 trials are listed on the website. However, a closer inspection reveals that in most cases, exosomes are regarded as potential biomarkers for the diagnosis of disease, including lung cancer, pancreatic cancer, liver cancer, Parkinson’s disease, myocardial infarction, Crohn’s disease, etc. Only very few of them applying exosomes as clinical therapeutic approaches. In comparison, more clinical trials based on stem cells, rather than exosomes, have been conducted, such as transplantation of autologous stem cells for the treatment of type 1 diabetes, ischemic cardiomyopathy, inflammatory bowel disease, incurable neurological disorders, etc.
Cell-based versus exosome-based therapies in the field of regenerative medicine
Both cell-based and exosome-based therapies have become an emerging strategy for personalize treatment in the field of regenerative medicine. Since Terpos E et al. first reported the therapeutic efficacy of autologous stem cell for normalizing abnormal bone remodeling in patients with multiple myeloma in 2004, 187 cell-based therapies have been quickly initiated. The major reason for the rapid rise of therapeutic strategy based on stem cell is that the clinical application was both safe and well tolerated without any adverse effects. Furthermore, high efficiency of tissue repair and regeneration has been observed. 188
Some challenges, however, hinder its further clinical application. First, the standardized large-scale production of stem cells is hardly an achievable goal due to their highly heterogenous, including donor age, donor gender, tissue location, etc. 189 Second, in vitro culture of stem cells shows some degree of risk, such as chromosomal aberrations and attenuated cell proliferation rate. 190 Third, long-term in vivo safety still needs to be investigated through further clinical trials.
Recently, exosome-based therapies have emerged as a potential alternative strategy, characterized with high stability, strong targetability and non-immunogenicity. 7 They have some salient advantages over cell-based therapies, such as high bioactivity and safety of exosomes. Moreover, the successful fabrication of engineered exosomes enables tissue-specific delivery and controllable release of drugs.13,183 However, utilizing exosomes as bioactive therapeutics is still in the stage of preliminary development. Potential technical limitations and challenges must be addressed, prior to clinical implementation.
Potential technical limitations and challenges of exosome-based therapies in clinical applications
During the development of clinical-grade exosomes, the major obstacle appears to be the immature production procedure, especially how to effectively purify clinical-grade exosomes, for subtle molecular differences in individual exosomes may ultimately contribute to significant differences in biological functions.7,183,185 Therefore, there is an urgent need for developing good manufacturing practice (GMP) guidance for producing exosomes. What is more, prior to formal clinical use, further investigations are clearly warranted to address these following issues. For instance, how to improve the therapeutic efficiency mediated by the combination of biomaterial scaffolds and exosomes, how to ensure the stability and safety of engineered exosomes, how to adjust the administration plan at any time in the whole treatment process, etc. Overall, more-rigorous statistical methods and larger patient population are required in future studies to determine the long-term clinical benefit of exosomes-based treatment.
Another challenging issue is the optimization of EV isolation approach, so as to obtain a sufficient quantity of exosomes for clinical utility. The main isolation technique currently, ultracentrifugation, presents limitation in maintaining the integrity and improving the yield of exosomes. 148 How to improve the yield of exosomes with high regenerative ability is increasingly becoming an urgent issue that needs to be properly addressed. It was found that the yield of exosomes with plasmid DNA were significantly increased by suppressing lysosome, which can be applied in the mass production of plasmid DNA/exosome complexes. 170 Fan J et al. reported a kind of strategy for achieving exosomes with high yields, namely extrusion approach. Unlike the conventional isolation approaches, this method is not only convenient but also scalable. 16 What is more, exosomes, collected from MSCs with extrusion approach, exerted an improved purity and increased concentration. Compared with those complex and expensive technologies, Ma Y et al. presented a clinically viable and scalable method to improve the yield and efficiency of exosomes via cellular nano-electroporation. In this approach, substantial quantities of small EVs are generated containing therapeutic mRNAs and associated miRNAs. 147 Artificial exosomes are regarded as another strategy for extending production, which ensures in vivo delivery of nucleic acids without immunogenic responses. 111 Nevertheless, before incorporation into routine clinical use, it is necessary to conduct clinical trials, in order to fully evaluate the safety and efficiency of the strategies mentioned above. Additionally, according to previous studies, exosomes show great deal of disparity in their functions and cargos. Therefore, there is a growing need for developing more reliable and accurate assays, to help researchers define the molecular composition of exosomes and the genetic information they contain. This will facilitate the development and fabrication of artificial exosomes, and further elucidate the molecular mechanism by which exosomes promote bone healing.
Conclusion and future perspectives
Exosomes have become an emerging cell-free therapy for accelerating bone healing because of their high stability, strong targeting and non-immunogenicity. As the mediators of intercellular communication, exosomes deliver bioactive substances (mainly ncRNAs) between cells to trigger biological responses in recipient cells. Recently, it has been extensively studied and well-established that exo-ncRNAs play a significant role in bone healing during the different stages, including macrophage polarization, angiogenesis, osteogenesis and osteoclastogenesis. Furthermore, exosomes are beneficial for bone maintenance and healing when combining with biomaterials or constructing engineered exosomes based on endogenous or exogenous pathway. These findings provide new research directions for treating bone-related diseases.
Although our review shed light on a great potential for exosome-based therapies toward bone defect repair, there are still some critical issues that must be addressed: (i) Technology optimization on the isolation and purification of exosomes must be resolved, so as to realize reproducible large-scale production. Despite the commercialized kits for isolating and purifying exosomes have been developed currently, high expense and low efficiency restricts their clinical applications. (ii) Future studies should focus on the comparison on exosomes from various cells of origin in vivo, in order to provide the optimal option for bone healing. Furthermore, it is necessary to elucidate the whole genetic information contained in exosomes, other than just monofunctional molecules carried by exosomes. The next step is to ascertain the specific substances in exosomes that accelerating bone healing, and to develop engineered exosomes with superior targeting properties. (iii) Novel technologies are required to be developed for tracking in vivo distribution of exosomes. To be more specific, further in vivo studies are needed to experimentally identify whether exosomes accumulate in nontarget organs and result in adverse side effects.
While much work remains to be done before exosomes being employed in formal clinical applications, their positive role in bone healing provides us with new perspectives on the treatment of bone related diseases. Taken together, it is reasonable to believe that with the rapid and intensive technology development across biology, medicine and materials science, clinical application of exosome driving bone healing will become of broader use, after resolving these issues.
Footnotes
Abbreviations
ADSCs: adipose-derived stem cells; AEs: artificial exosomes; Akt: Akermanite biocreamics; AGO1: argonaute 1; BP: black phosphorus; BMP: bone morphogenetic protein; BMSCs: bone marrow mesenchymal stem cells; cDNA: complementary DNA; CMS: cyclic mechanical stretch; circRNAs: circular RNAs; DKK2: dickkopf2; Elf3: E74-like factor 3; DPSCs: dental pulp stem cells; ELK4: ETS-like transcription factor 4; ECM: extracellular matrix; EVs: extracellular vesicles; FAs: focal adhesions; GelMA: gelatin methacrylate; GSK-3β: glycogen synthase kinase-3β; GMP: good manufacturing practice; HA: hyaluronic acid; HAMA: hyaluronic acid methacrylate; HDAC: histone deacetylase; H-Exo: hypoxic exosomes; HMGB1: high mobility group box 1 protein; HMP: hydrogel microparticles; HUVECs: human umbilical vein endothelial cells; H2S: hydrogen sulfide; ILVs: intraluminal vesicles; JNK: Jun amino-terminal kinases; KLF5: Kruppel like factor 5; LATS1/2: large tongue suppressor kinase 1/2; LNPs: lipid nanoparticles; LRP4: lipoprotein related protein 4; lncRNAs: long ncRNAs; LIPUS: low-intensity pulsed ultrasound; MSCs: mesenchymal stem cells; MIF: migration inhibitory factor; MVB: multivesicular body; ncRNA: non-coding RNA; nHAP: nano-hydroxyapatite; NID1: nidogen1; NF-κΒ: nuclear factor kappa B; PDA: polydopamine; PEG: polyethylene glycol; PIK3R2: phosphoinositol-3 kinase regulatory subunit 2; PLGA: poly(lactide-co-glycolide); SIK2 and SIK3: salt-inducible kinase 2 and 3; SC Exo: schwann cell-derived exosomes; SDF-1α: stromal cell derived factor-1α; sEXOs: serum exosomes; SLIT3: slit guidance ligand 3; sncRNAs: small ncRNAs; SM: static magnetic feld; Sr: strontium; SPEEK: sulfonated polyetheretherketone; Ta: tantalum; TA: tannic acid; TBI: traumatic brain injury; TPEEK: tannic acid-modified polyetheretherketone; TGF-β: transforming growth factor-β; TSA: Trichostatin A; Traf: tumor necrosis factor receptor-associated factor; USP7: ubiquitin specific peptidase 7; VEGF-A: vascular endothelial growth factor A; β1/2-AR: β1/2-adrenergic receptor; β-TCP: β-tricalcium phosphate; 3WJ: 3-way junction
CRediT authorship contribution statement
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Project of Liaoning Xingliao Talents Plan (XLYC2002103). Basic applied research program of Living Province of China (No.202220347-JH2/1013). All authors reviewed and approved the final manuscript before submission.
Data availability
Not applicable.
