Abstract
Vascularized composite allotransplantation (VCA), which can effectively improve quality of life, is a promising therapy for repair and reconstruction after face or body trauma. However, intractable issues are associated with VCA, such as the inevitable multiple immunogenicities of different tissues that cause severe rejection, the limited protocols available for clinical application, and the shortage of donor sources. The existing regimens used to extend the survival of patients receiving VCAs and suppress rejection are generally the lifelong application of immunosuppressive drugs, which have side effects. Consequently, studies aiming at tissue engineering methods for VCA have become a topic. In this review, we summarize the emerging therapeutic strategies for tissue engineering aimed to prolong the survival time of VCA grafts, delay the rejection and promote prevascularization and tissue regeneration to provide new ideas for future research on VCA treatment.
Keywords
Introduction
Vascularized composite allotransplantation (VCA) is generally considered to comprise a variety of tissue components, namely, nerve, muscle, bone, skin, and blood vessels, that can be used to rebuild the form and function of damaged tissue. 1 VCA has been widely used in face transplantation, upper limb transplantation, uterus transplantation, penis transplantation, abdominal wall transplantation, and others. 2 Since the first successful hand transplant in the world in 1998 and the first completed face transplantin 20053,4 the number of patients who are benefitting from VCA is growing. A total of 86 VCAs were performed in the United States from 2014 to 2021.2,5 In contrast to solid organ transplantation (SOT), VCA has not been identified as a life-saving treatment but rather a way to improve the quality of life, thus, VCA is referred to as a “life-enhancing” treatment 6 and has become an important repair method for patients suffering from severe trauma or burn. 7 By way of illustration, it is difficult for patients with severe facial burns are difficult to achieve esthetic and functional restoration through traditional surgical methods, but VCA represents a promising approach.8,9 Correspondingly, according to statistics, in most of the patients treated with hand transplantation, tactile sensation and functional movement can be recovered. 10 Nevertheless, as the number of VCA cases has increases, the treatment challenges have gradually been exposed. For instance, since VCA involves a variety of tissue components, 11 it is difficult to ignore the high immunogenicity of these components, which might cause different kinds of rejection (Figure 1).12,13

Several therapeutic strategies for tissue engineering methods in vascularized composite allotransplantation (VCA).
Allograft rejection frequently occurs in VCA patients and can lead to graft injury and multiple complications that adversely affect the prognosis of patients.14–16 Acute immune rejection (AR) occurs most often in the early posttransplant period, 17 usually manifests as papules, erythema, edema, and other skin lesions.18,19 This process is mediated by T cells and antigen presenting cells (APCs) from both donor and recipient responses to mismatched MHCs, 20 which depend on the recognition of damage-associated molecular patterns by APCs and lead to the activation of CD4+ and CD8+ T cells. In turn, inflammatory cytokines secreted by effector T cells and recruited macrophages contribute to the subsequent inflammatory response and also promote graft apoptosis. 21 AR can be reversed with adequate prevention and treatment. Chronic rejection (CR) due to a prolonged treatment time, increased recurrence of AR, and irregular anti-immune rejection therapy are also associated with VCA. 22 CR mainly damages the skin and deep blood vessels, and histopathologically manifests as skin atrophy, fibrosis, and vascular disease, which is jointly mediated by cell-mediated immunity and humoral immunity. 23 Unfortunately, the reversal probability of CR is not clear, and the problem is often solved by ultimately removing the graft. 24 In recent years, antibody-mediated rejection (AMR) mediated by the donor-specific antibody (DSA) has also been reported in human VCA, 25 confirming that AMR plays a significant role in the long-term survival of grafts with preoperative cross-matched positive VCA. There is an urgent need for further research and improved treatment (Figure 2).

Illustration of patterns of rejection in vascularized composite allotransplantation.
At present, the main treatment for preventing the loss of VCA grafts is the lifelong application of immunosuppressive drugs. 26 The usage of immunosuppressive drugs is almost similar to that of SOT, 27 so their application is limited due to a variety of unavoidable side reactions,28,29 such as opportunistic infections, malignant tumors, leukopenia, abnormal renal or liver function and others. Although some patients receive durable immunosuppressive therapy, acute rejection cannot be completely avoided.30,31 Fortunately, researchers are committed to developing new types of therapeutic strategies, especially the therapies related to tissue engineering, to avoid the risks of the lifelong application of immunosuppressive drugs. Therefore, this review elucidates the emerging strategies for tissue engineering in vascularized composite allotransplantation to provide inspiration for the researchers (Table 1).
Emerging strategies for tissue engineering in vascularized composite allotransplantation.

Cell therapy
Immune cell therapy
VCA cell therapy refers to a therapeutic method that uses cells as functional components, namely, as mediators or carriers in vivo or in vitro that guide immune tolerance after transplantation in VCA. At present, this approach mainly includes immune cell therapy and stem cell therapy. 32 Immune cell therapy is not uncommon in the treatment of graft-versus-host disease (GVHD). Regulatory immune cells, represented by regulatory T cells (Tregs), regulatory dendritic cells (DCregs), regulatory B cells (Bregs), and regulatory macrophages (Mregs), play a significant role in maintaining immune homeostasis and preventing overactivation of the immune system.33–35
Tregs account for 5%–10% of the total number of T cells and include a variety of subtypes, of which CD4+CD25+Foxp3+pTregs have received the most attention. 36 Tregs can act on a variety of immune cells (such as macrophages, CD4+ T cells, CD8+ T cells, and dendritic cells) to realize negative immune regulation by expressing CD25 molecules and releasing immunosuppressive cytokines (such as IL-10) and so on. 37 This property makes the use of engineered Tregs for the treatment of rejection after VCA transplantation a promising research direction. In 2019, Ignacio Anegon and Carole Guillonneau designed A2-CAR CD8+Tregs via lentiviral transfection. The introduction of a chimeric antigen receptor (CAR) derived from an HLA-A*02 antigen-specific antibody (A2-CAR) to human CD81 Tregs, it has a targeted immunosuppressive effect. Compared to nonengineered CAR CD8+Tregs, A2-CAR CD8+Tregs were able to efficaciously and chronically maintain intact skin grafts intact without showing significant cytotoxicity. 38 In particular, when the ratio of peripheral blood mononuclear cells (PBMCs) to CAR-Tregs is 1:1, rejection was inhibited in 100% of grafts. This study introduced CAR-T cell therapy from the field of tumor therapy to the field of post-VCA therapy and adopted the measures of engineered cell editing measures to achieve the precise treatment of anti-immune rejection after VCA transplantation (Figure 3).

(a) Schematic illustrating the procedure for generating CAR CD8+ Tregs. (b) Assessment of in vivo cytotoxicity through monitoring weight loss in mice (mean ± SEM). (c) Survival of skin graft over time based on macroscopic features of rejection in the model of human skin graft in NSG mice. (d) Representative photos of HPS coloration of human skin tissue grafted 100 days after cell transfer. (e) Representative photos of immunofluorescence stains of human skin tissue grafted 100 days after cell transfer. Reproduced by permission of Guillonneau et al. 38
Among the many types of Treg cells, αβ-TCR+CD3+CD4−CD8− double-negative (DN) Treg cells have also been shown to attenuate allogeneic and xenogeneic immune responses, which can mitigate the anti-donor specificity of CD4+ and CD8+ T cells and have strong therapeutic potential for VCA.39,40 Previous studies have confirmed that DN Tregs can be derived from peripheral CD4+ T cells in vitro, and can express a stable phenotype. 41 A study by the team of X.X. Zheng has developed a therapeutic approach based on in vitro-induced DN Tregs combined with ALS, Rapa, and an IL-2/Fc fusion protein applied to a fully MHC-mismatched mouse VCA model. This treatment can induce allograft receptor macrochimerism, increase the percentage of CD4+ Foxp3+ Tregs to approximately 15%–20% of peripheral CD4+ T cells (normally 5%–10%) and ultimately achieve donor-specific graft immune tolerance after VCA. 42 These findings further indicate that Treg cells are important for VCA treatment.
In addition to Tregs, T cell inhibitory coreceptors, which are also known as “immune checkpoints”, play key roles in maintaining the balance of the immune response. When activated, they can mediate the inhibition of the T cell proliferation and control inflammation. The common T cell inhibitory coreceptors include CTLA4 and PD1. 43 T cell immunoglobulin and mucin domain-containing-3 (Tim-3) is an inhibitor coreceptor located on CD4+ and CD8+ T cells, NK cells, and Foxp3+ Tregs that produces IFN-γ, which control the stability of T cells function. 44 Yaojun Wang and Zhao Zheng used Tim-3-modified dendritic cells to explore the protective effect of Tim-3 on composite tissue and the immunomodulatory functions of allotransplantation. MDCs are generated from BM progenitor cells and splenocytes and can be subsequently transfected with recombinant vectors to express Tim-3. Researchers have established an orthotopic hindlimb allotransplantation mode to study the effects of mDCs that have been modified by intraperitoneal injection of Tim-3. After detection by laser speckle blood flow (LSBF) imaging, ELISA, MTT assays, ELISPOT assay and flow cytometry analysis, researchers found that Tim-3-modified mDCs could effectively prolong graft survival and achieve lower lymphocyte reactivity. Further studies showed that Tim-3-modified mDCs induced an increase in CD4+ CD25+ Foxp3+ Treg cells(8.54% in PBMCs). These results suggest that Tim-3 is a potential T cell inhibitory coreceptor for the treatment of VCA rejection. 45 As one of the key celltypes involved in the regulation of VCA rejection, Tregs have great therapeutic potential, and additional Treg-related therapeutic strategies should be developed.
Stem cell therapy
Stem cells, including totipotent stem cells, pluripotent stem cells, and specialized stem cells, have good potential for renewal, differentiation, and regulation. Stem cell therapy has been applied in SOT, and stem cells can regulate immune function and induce tolerance through intercellular interactions and cytokine release, which are tolerogenic cell therapies. 46 Hematopoietic stem cells and mesenchymal stem cells, such as HSCs, AD-MSCs, and BM-MSCs, have been widely used in the field of immune tolerance in transplantation. 47
As early as 1953, researchers discovered that the presence of donor hematopoietic cells can promote the development of immune tolerance after transplantation in the recipient. 48 Subsequently, Howard D. Wang and Samuel A.J. Fidder tested hematopoietic stem cells through syngeneic hematopoietic stem cell transplantation (HSCT), which can prevent AMR in VCA and achieve desensitization. The researchers created a sensitized model by transplanting back flaps from Dark Agouti (DA) rats onto the backs of Lewis rats. Syngeneic Lewis rats were treated with HSCT and tacrolimus after TBI and fludarabine pretreatment in the experimental group and tacrolimus alone in the control group. They verified that the survival of the grafts significantly improved (100% graft survival for >30 days) after the combination of HSCT with conventional immunosuppression. 49
Compared with other types of MSCs, AD-MCSs, which are important therapeutic materials in the field of plastic surgery and reconstruction, have more sources for extraction and better immunomodulatory functions.50,51 In 2017, Jan A Plock MD laid the foundation for the application of AD-MSCs in VCA. They transplanted the hind limbs of Brown Norway rats into Lewis rats and used repetitive AD-MSCs treatments in combination with ALS and FK-506 after surgery. The results showed that repetitive AD-MSCs treatment could prolong allograft survival and induce a significant upregulation of Tregs, which further suppressed T cell function. Surprisingly, AD-MSCs were also able to inhibit the development of vascular lesions in long-term survivors with grafts. 52 This study provides insights into the development of more efficient AD-MSCs therapies in the future. In addition, Riccardo Schweizer’s team published a treatment regimen using AD-MSCs in combination with ALS, tacrolimus, and CTLA4-Ig to promote the long-term survival of VCA grafts. The researchers divided the completely mismatched rat hind limbs into four groups: CTRL, ASC, ASC-CYP, and ASC-ALS. The experimental results indicated that the survival of VCA grafts without rejection was longer in the ASC-ALS group. This finding further emphasized that the ASC-ALS group was also associated with continuous mixed chimerism of donor cells and elevated Treg numbers in the recipient, which further explained the mechanism of immune tolerance induced by this treatment approach. 53 In addition, to deliver AD-MSCs more precisely and improve treatment efficacy, the Beijing Key Laboratory of Neuropsychopharmacology developed an AD-MSCs delivery system targeting secondary lymphoid organs (SLOs). Previous studies have shown that CC chemokine receptor 7 (CCR7) can guide cells to immune organs, which contributes to immune tolerance. 54 As a consequence, the team used a lentivirus to introduce the CCR7-GFP gene into hAD-MSCs to construct hASCs/CCR7, which could target SLOs. A significant prolongation of VCA graft survival was achieved after intravenous infusion of hASCs/CCR7 (MST = 14.38 ± 1.51 days), and alterations in the Th1/Th2 and Treg/Th17 axes in SLOs may underlie the downregulation of rejection. 55
As a kind of MSCs, bone marrow-derived mesenchymal stem cells (BM-MSCs) also have functions such as pluripotency, immunosuppression, and angiogenesis stimulation.56,57 Accordingly, researchers have attempted to use BM-MSCs as alternative treatment options for VCA. Studies have confirmed that BM-MSCs can increase the ratio of CD4+/CD25+ and CD4+/Foxp3+ T cells in grafts. 58 In 2018, a team from NYU Langone Health attempted to evaluate the therapeutic effect of BM-MSCs activated by inflammatory and hypoxic microenvironments on rejection after VCA in vitro. They used a rat hindlimb allograft model, followed by perioperative infusion of ex vivo-expanded receptor-derived BM-MSCs. Through subsequent observations and experiments, the team found that in vitro delivery of donor-derived BM-MSCs could delay the onset of acute immune rejection of grafts and ameliorate graft perfusion. BM-MSCs could also be activated by inflammatory and hypoxic microenvironments and play a role in preventing alloreactivity, so stimulation before ex vivo perfusion could become an optimized treatment modality. 59
Donor recipient chimeric cells
Experimental research has validated that BMT therapy is useful for the induction of mixed chimerism and reduction of rejection after VCA 60 which is also valuable for identifying donor-recipient chimeric cells (DRCCs). 13 Additionally, Maria Siemionow’s group designed a treatment to induce DRCCs by in vitro cell fusion. PKH26/PKH67 double-labeled BMCs were obtained by mixing BMCs derived from ACI rats (PKH26) and Lewis rats (PHK67) followed by PEG-mediated fusion. After VCA of the inguinal region with the ACI rat as the donor and the Lewis rat as the recipient, the rats were treated with 2-4 × 106 doses of DRCCs in combination with 7 days of the anti-αβTCR monoclonal antibody and CsA immunosuppressive regimen. After 21 days of observation, flow cytometric analysis and other assays revealed that DRCC treatment prolonged the survival of VCA grafts to 79.3 ± 30.9 days and induced long-term mixed chimerism. The authors also highlighted the important role of the DRCCs in the thymus and spleen in the induction of long-term mixed chimerism, which inspired us to investigate this topic further. 61
To sum up, cell therapy as a popular treatment method, also play a part in the treatment for the rejection after VCA transplantation. In summary, cell therapy, a popular treatment method, also plays a part in the treatment of rejection after VCA. Further clinical evaluation and safety evaluations are needed to improve the treatment, and more accurate preparation methods and dosages will be more conducive to the progress of cell therapy in for VCA.
Cell-free therapy
Cell-free therapy refers to a new strategy for treatment using extracellular vesicles (exosomes, apoptotic bodies, and ectosomes) secreted by cells. 62 Compared with cell therapy, cell-free therapy can prevent the risk of an immune response and differentiation of cells and is easier to administer to the treatment area. 63 At the same time, extracellular vesicles are more conducive to transport and preservation, which are important features. 64 According to previous studies, EVs can participate in the process of immune recognition or tolerance in allogeneic tissue and organ transplantation. However, the mechanism and regulatory components of EV-induced tolerance are still unclear. We know that it is affected by many factors, such as the nature of EVs, the type of APC that acquires them, and the process of antigen presentation. 65
Exosomes from donor-derived AD-MSCs (ADMSC-exos) have biological activity similar to that of AD-MSCs. 66 Furthermore, they are also nontumorigenic, do not induce a host immune response, are rich in origin, are stable, and can be preserved for a long time. Accordingly, ADMSC-exos could be a better alternative. Professor Qifa Ye led the team to corroborate that donor-derived ADMSC-exos can prolong the survival of VCA grafts safely and effectively. The experimental team extracted ADMSC-exos from the adipose tissue of the donors by ultracentrifugation, transplanted the hind limbs of the donor Brown Norway rats into the Lewis rats, and intravenously injected ADMSC-exos in combination with short-term immunosuppression. The control group was treated with phosphate buffered saline combined with short-term immunosuppression. Similar to AD-MSCs, ADMSC-exos induced donor cell chimerism, promoted the growth of Tr1 cells and Tregs (14.80±7.39% in PBMCs) in vivo, and the decreased of CD4+ T cells and Th1 cells, thereby lengthening the survival of the VCA grafts (MST >58 days). 67 This study provides inspiration for the application of cell-free therapy in VCA, and the development of engineered extracellular vesicles against VCA immune rejection is a direction that could be explored.
Furthermore, some studies have shown that mesenchymal stem cell-derived exosome (MSC-exo)-derived miRNAs also regulate immune responses. Zheng et al. tested this theory in an acute GvHD mouse model. The authors selected the highly expressed miR-223 in MSC-exos by high-throughput sequencing and bioinformatics analysis. In vivo and in vitro experiments demonstrated that it could inhibit the migration and adhesion of T cells, reduce the expression of the inflammatory cytokines IFN-γ, TNF-α, and IL-17, and reduce the infiltration of T cells, thereby alleviating the symptoms of GvHD in mice. 68 Moreover, the application of MSC-exo-derived miRNAs in the treatment of chronic cutaneous GvHD has also been reported. 69 Therefore, the results of this study can also be applied to the treatment of immune rejection caused by VCA.
Decellularized scaffold therapy
Decellularizatied/recellularizatied engineered tissue
The decellularization/recellularization of engineered tissue is a kind of treatment in which native cells are removed to produce extracellular matrix (ECM) scaffolds through the introduction of sources of cells from healthy patients, combined with proliferation after reconstruction of the tissue microstructure.70,71 According to this scheme, Qixu Zhang designed an engineered vascularized composite soft tissue flap-a decellularized skin/adipose tissue flap (DSAF). DSAF retains the 3D nanostructure of the ECM in the VCA graft, the complete vascular system and neural network, and even well-preserved cytokines. The researchers obtained free flaps from donor rats, after which human adipose-derived stem cells (hASCs) and human umbilical vein endothelial cells (HUVECs) were recellularized in vitro. Finally, the DSAFs were transplanted into the recipient nude mice. This study demonstrated that DSAFs achieved vascularization in the recipient within 7 days and that complete structural remodeling and highly vascularized adipose tissue regeneration were achieved within 3 months, while DSAFs regulated the immune system by activating M2 macrophages. The use of engineered tissue, as an alternative to autologous tissue sources, can avoid the toxic side effects caused by immunization and drug use. 72 The decellularization/recellularization method validated through DSAFs has great potential in the field of engineering composite tissue transplantation.
Perfusion-decellularization of human ear grafts
Due to the esthetic and functional importance of the external ear, auricle reconstruction has been a challenging treatment in the field of plastic surgery. 73 Compared with autogenous grafts, the application of VCA for auricle reconstruction is a promising method in the case of total ear defects. 74 To avoid the adverse effects of the application of immunosuppressive agents, the team of Jérôme Duisit designed a kind of decellularized human ear graft that can be used as an ECM-based scaffold in VCA. 75 The decellularized human ear ECM scaffold was constructed according to the SDS/polar solvent protocol. HE staining and DAPI staining revealed that the antigens and cells were removed, while the collagen and three-dimensional structure of the ECM were preserved. In this way, it not only has the reconstructive function of the scaffold but also reduces immunogenicity. According to the in vivo biocompatibility evaluation, no adverse events occurred after transplantation. This study further broadens the application of decellularized scaffolds in VCA; however, allograft experiments were not performed, and the effective reduction in rejection was not verified. Further improvement in these areas would help to verify the possibility of applying this strategy for VCA (Figure 4).

(a) Schematic illustration, perfusion-decellularization of human ear grafts. (b) DNA content expressed in ng/mg wet weight. (c) Relative preservation of total protein extract, pro-inflammatory cytokines, chemokines, and growth factors. (d) ECM major proteins and 2D structure preservation. (e) ECM 3D-histology reconstruction. (f) Macroscopic assessment of subcutaneous implantation in rat. (g) Live/dead and MTT vital stainings in whole-ear scaffold perfusion-bioreactor seeding. Reproduced by permission of Duisit et al. 75
Biomaterials
Hydrogels
Biomaterials have become common in various medical disciplines in recent years, 76 and VCA is no exception. The common application forms of biomaterials include hydrogels, nanoparticles, etc., which can be loaded with various bioactive components to achieve different functions. For example, Thusitha Gajanayake and Radu Olariu developed an enzyme-reactive hydrogel that carries TAC at a single topical dose. The hydrogel is composed of a biodegradable glycolide-co-clatide-co-caprolactone polymer, which can target the release of tacrolimus by proteolytic enzymes in the inflammatory environment of rejection. Injection of the hydrogel on the first day after transplantation prolonged the survival of VCA grafts to more than 100 days, and only mild cellular infiltration was observed histologically. 77 Moreover, C. Anton Fries designed a graft-implanted, enzyme responsive, TAC-eluting hydrogel. This hydrogel is constructed of amphiphilic triglycerol monostearate (TGMS) and releases TAC into grafts upon the activation of matrix metalloproteinases (MMPs). Notably, this research used mismatched porcine orthotopic forelimb transplantation as the VCA model to assess the safety and effectiveness of the treatment more accurately. Different doses of TAC were used in the experiments, and the effect and tolerance of the hydrogel were evaluated by histopathological evaluation, blood concentration evaluation, and animal behavior changes. Consequently, they confirmed that graft-implanted, enzyme-responsive, TAC hydrogel can delay the immune rejection and prolong the graft survival after VCA with mismatched large animals. 78 We are looking forward to the development of more kinds of immunosuppressant-loaded active hydrogels that can achieve controlled release and targeted delivery in VCA.
Microparticle-based systems
Microparticle-based systems (MSs) are frequently used as drug delivery vehicles in the design of bioactive materials. 79 Clinically, the commonly used immunosuppressive strategy in VCA treatment, standard triple therapy, which is a regimen of TAC, MMF, and steroids, is usually applied through systemic delivery, 80 so it can cause many inevitable side effects. Therefore, Jun Yang and Binbin Sun designed a microbead system equipped with FK506/MMF/prednisolone (PDNN) based on triple therapy. This FK506/MMF/PDNN-PLGA MS can be injected locally into the grafts of a rat hind limb transplantation model. Compared with oral administration, the application of FK506/MMF/PDNN-PLGA MSs can significantly prolong graft survival while maintaining a higher drug concentration in VCA tissue and preventing systemic side effects. 81 Other kinds of immunosuppressants can also be used as components of microparticle-based systems. For example, a team from Bern University Hospital also attempted to develop a PLGA-based in-situ forming implant (ISFI) loaded with rapamycin. This ISFI was demonstrated to induce an immune microenvironment and increase the proportion of Tregs in the VCA graftas well as to suppress immune rejection and prolong the survival time of the VCA grafts (Figure 5). 82

(a) Schematic representation of the Rapa-ISFI formation and drug release properties. (b) Graft survival represented with Kaplan-Meier survival curves. (c) Representative microphotographs of the histology sections of the skin stained with HE and histopathological grading of rejection based on Banff working classification. (d) Quantitative summaries for the frequency of Treg, HeliosNegTreg and HeliosPosTreg in the peripheral blood at POD21. (e) Correlation analysis between chimerism levels and allograft survival. (f) Survival of hind limb allografts in Rapa-ISFI treated rats with unsuccessful ALS therapy. Reproduced by permission of Plock et al. 82
We have previously shown that Tregs are a type of cell that negatively regulates the immune system, and researchers are working to develop a biological material that can recruit Tregs. Work by teams from the University of Pittsburgh and Wake Forest School of Medicine offers a solution. CCL22 is a chemokine of Tregs that can recruit Tregs in vivo through its corresponding chemokine receptor (CCR4).83,84 In this study, synthetic CCL22 was loaded into a poly(lactic-co-glycolic acid) (PLGA) microparticle system (recruitment-MP), which is biodegradable and controllable and allows the gradual release of CCL22 in vivo. The results demonstrated that recruitment-MP could recruit CD4+CD25+Foxp3+Tregs, extend the survival of VCA grafts, decrease the levels of TNF-α, IFN-γ, IL-17A, and perforin-1, and promote the construction of immune tolerance. 85 As we have shown, more efficient drug delivery systems can alleviate the side effects and dosage of drugs that doctors are concerned about and provide more options for VCA treatment.
3D bioprinting strategy
The successful implementation of VCA requires a complex process. In addition to coping with the immune system, the regeneration of nerves, muscle, bone, skin, and blood vessels is also crucial due to the shortage of donors. However, 3D bioprinting strategies are expected to play a role by creating biomimetic structures through computer technology-assisted simulation of biological structures and functions. 86
In recent years, in terms of skin regeneration, 3D bioprinting methods have been used to create mature bionic skin. This kind of bionic skin can reproduce full-thickness skin structures and skin appendages and can also induce vascularization after skin transplantation. 87 For instance, Luca Pontiggia’s group has developed a new robotic platform, “SkinFactory”, to generate a prevascularized and pigmented dermo-epidermal skin substitute (DESS) composed of a variety of human-derived cells. 88 This DESS contains fibroblasts, blood and lymphatic endothelial cells, keratinocytes, melanocytes, and collagen type I hydrogels, and can maintain pigmentation and reconstruct complete capillary and lymphatic networks in vivo. This finding is expected to solve the two major problems of skin 3D bioprinting, which is a promising achievement for application in VCA.
In VCA, it is also difficult to reconstruct bone and muscle tissue defects by traditional grafting methods, while 3D bioprinting is also effective for preparing bioactive substitutes. 89 Zhiguang Huan led the team to design a biomimetic ink capable of mimicking tendon/bone tissues for 3D bioprinting. 90 This biomimetic ink consists of Mo-containing silicate as the active ingredient to induce the differentiation of tendon stem/progenitor cells (TSPCs) and BMSCs and GelMA as the scaffold. It can promote the regeneration of tendon-to-bone interfaces in a rabbit rotator cuff tear injury model. This invention provides insights into the application of 3D bioprinting in complex composite tissue transplantation.
Similarly, 3D bioprinting has led to new breakthroughs in nerve injury repair. At present, artificial nerve conduits with three-dimensional structures can be manufactured by 3D bioprinting, which is expected to be useful for filling nerve defects during VCA. This method mainly involves using stem cells and neural cells as bioinks, combining them with growth factors and biocompatible biomaterials, and finally printing them with a suitable 3D printer. 91 Therefore, the preparation of artificial nerve conduits with good biological and mechanical properties has become an important issue. To address this problem, Yin et al. developed a bilayer nerve conduit using multinozzle addition-lathe 3D bioprinting technology. This bilayer nerve conduit is composed of an inner layer of GelMA/BMSCs and an outer layer of GelMA/PEGDA. Experimental results have shown that it has good mechanical properties and promotes the differentiation and proliferation of PC12 cells, which has the potential to promote nerve regeneration. 92 In summary, 3D bioprinting is a promising technology to compensate for the shortage of VCA donors, and the generation of safer and more stable 3D bioprinting products that can replace real human tissue is the direction of future research.
Biological components therapy
Antibody-based therapy
IL-1β plays an important role in the rejection of composite tissue allograft transplantation. 93 In rejection, inflammation, and ischemia-reperfusion injury after transplantation, pathogen-associated molecular patterns (PAMPs) and damage-associated molecular patterns (DAMPs) are usually released. IL-1β is activated through a series of effects, starting with the recognition of PAMPs and DAMPs by TLRs and inflammasomes-NLRP3 and subsequently triggering the activation of T cells and the recruitment of monocyte-macrophages to the graft. 94 Therefore, subcutaneous administration of a neutralizing IL-1β antibody has been attempted to prolong the posttransplant survival of VCA patients. The Daniel Swarovski Research Laboratory used an orthotopic allogenic rat hind-limb transplantation model with Brown Norway rats as donors and Lewis rats as recipients. The graft survival time (partially reached POD 100) and rejection level were also evaluated. Taken together, the results demonstrated that intragraft injection of an IL-1β antibody (1 mg/kg weekly) effectively improved graft survival and reduced rejection, while there was no significant difference in cell infiltration. 95 Therefore, the mechanism of IL-1β antibody treatment needs to be further explored.
Modified mRNA-based therapy
Messenger ribonucleic acid (mRNA)-based therapy is an emerging gene therapy strategy that can replace DNA gene therapy to achieve translation in the cytoplasm to play a specific role. 96 Compared with DNA gene therapy, mRNA therapy avoids the risk of genome integration, but it still has the problems of immunogenicity and instability that have challenged researchers, and it is difficult to use as an effective component of therapy. 97 Therefore, modified mRNAs that can improve stability and reduce immunogenicity through structural modifications have gained increasing interest. Modified mRNA has higher transfection efficiency and a longer half-life period, which is more conducive to its use as a therapeutic method. 98 In fact, COVID-19 mRNA vaccines that have received regulatory approval are also based on modified mRNA technology. 99 This promising technology has also been applied to VCA. Aline Yen Ling Wang’s team designed a therapeutic approach applying IL-10 modRNA based on the immunomodulatory function of IL-10. 100 Previous studies have shown that IL-10 can promote the differentiation of naive T cells to Tregs, further supporting its role in negative immune regulation. 101 The experiments showed that the IL-10 modRNA developed by the team could express functional IL-10 in vitro. Moreover, repeated low-dose local injections of IL-10 modRNA around the graft significantly prolonged the survival of the grafts in a face transplantation mouse model. The researchers also found that local lymphocyte infiltration was reduced after the injection of IL-10 modRNA, while the proportion of CD4+CD25+Foxp3+Tregs in CD4+T cells was significantly increased. Moreover, an assessment of the expression of proinflammatory factors in grafts revealed that the levels of IL-12, TNF-α, and IL-2 were significantly reduced, confirming the suppression of the inflammatory response in the face transplant group. Further studies showed that IL-10 modRNA could also induce elevated levels of mixed chimerism, thereby protecting the graft from rejection (Figure 6). 100 This study demonstrated the possibility of using modRNA technology in VCA, and modRNA based on additional mechanisms to inhibit immune rejection need to be further developed.

(a) Schematic illustration of IL-10 modified mRNA monotherapy for composite facial allografting. (b) Procedure of semi-face transplantation surgery in mouse. (c) Survival of facial allografts in IL-10 modRNA-treated mice. (d) Histological changes in facial OMC allografts of IL-10 modRNA and buffer groups on PODs 10–14. Histological Banff classification is as shown in the right. (e) Immunohistochemistry in facial OMC allografts of IL-10 modRNA and buffer groups on PODs 10–14. (f) Representation of gating strategy for donor Tregs (CD4+CD25+Foxp3+H2Dd+). Reproduced by permission of Wang et al. 100
Other treatments
Donor-derived lymph nodes
In addition to the above categories, lymphatic organs are also important targets for immune tolerance therapy for VCA, thus, lymphatic targeted therapy has become a new treatment method. 102 Recent studies have confirmed that the implantation of donor-derived lymph nodes (LNs) at the same time as VCA transplantation can delay immune rejection after transplantation. 103 The models were divided into three groups: VCA receiving donor LNs, VCA eliminating donor LNs, and VCA using the lymph angiogenesis inhibitor VEGFR3. The results showed that VCA of donor LNs could effectively delay the occurrence of rejection (median rejection onset time 5.0) and accelerate lymph angiogenesis, which was related to the enrichment of VEGF-C in the skin. However, the travel of tertiary lymphoid organs (TLOs) was not affected by LN transplantation, and there was no significant delay in graft survival. 103 This study suggested that LNs and VEGF-C have promising applications in VCA therapy, but maximizing their functions is an urgent issue. Moreover, the specific underlying mechanism is not clear, which indicates the need for further research.
Donor splenocytes
As early as 2008, researchers proposed that ECDI-fixed allogeneic splenocytes (ECDI-SPs) can prolong allograft survival after islet transplantation by inducing antigen-specific tolerance. 104 EDCI is a cross-linking agent that can couple peptides to the cell membrane and thus can achieve specific tolerance of antigens by linking them to splenocytes. In subsequent studies, it has also been confirmed that this method has an effective protective effect on allogeneic heart transplantation in mice, 105 and the mechanism is related to the induction of anergy of T cells and the promotion of Treg production. Whether ECDI-SPs affect the induction of immune tolerance in VCA has attracted the interest of researchers; accordingly, Shuzhong Guo’s team applied ECDI-SPs combined with rapamycin to allogeneic vascularized skin transplants in a mouse model. The model used C57BL/6 mice as the recipients and BALB/c mice as the donors, which were then tested in vivo and in vitro. ECDI-SPs were extracted and prepared from donor splenic tissue and applied to the recipient by injection of a single-cell suspension. The results of in vitro experiments showed that ECDI-SPs stimulated increases in IL-10 and TGF-β levels and induced the decreases in IL-1β and TNF-α levels. More importantly, ECDI-SPs also increased the proportion of CD4+Foxp3+ Tregs, which was further confirmed by in vivo experiments. In vivo, infusion of ECDI-SPs effectively prolonged the MST of vascularized skin grafts to 28 days, confirming that this regimen is not only feasible for islet and heart transplantation but also has the same efficacy for VCA. 106 In summary, the use of ECDI-SPSs, a relatively safe treatment method, has many advantages over previous immunosuppressive protocols used in the clinic, such as no requirement for overall depletion of T-cells depletion or comprehensive costimulation blockade; consequently, ECDI-SPSs have good potential for clinical application. 107
Vascularized bone marrow
Conventional bone marrow transplantation (CBMT) involves continuous supply of donor-derived hematopoietic stem cells. 108 It can induce mixed chimerism in costimulation blockade-based therapy, which is currently a widely accepted method at present. The vascularized bone marrow component (VBM) is a tissue engineering treatment based on CBMT. In contrast to CBMT, VBM preserves the bone marrow microenvironment, provides nutritional support for the proliferation of bone marrow cells, and has potential for immune regulation. A team of researchers based their treatment on costimulation blockade. They compared the efficacy of VBM transplantation with that of low-dose CBMT in the treatment of rejection after VCA in mice and reported that VBM transplantation was associated with a longer survival of up to 20 days, a higher chimerism level of 3.8% in circulating donor leukocytes, and a greater proportion of Treg/Teff cells and a lower proportion of CD4+ and CD8+ T cells. 108 The proposal of VBM provides more options for the therapy involving the combination of VCA with BMT, which can be further engineered with vascularized tissue. However, the bioactive components of VBM and the mechanism by which it controls rejection need to be further studied (Figure 7).
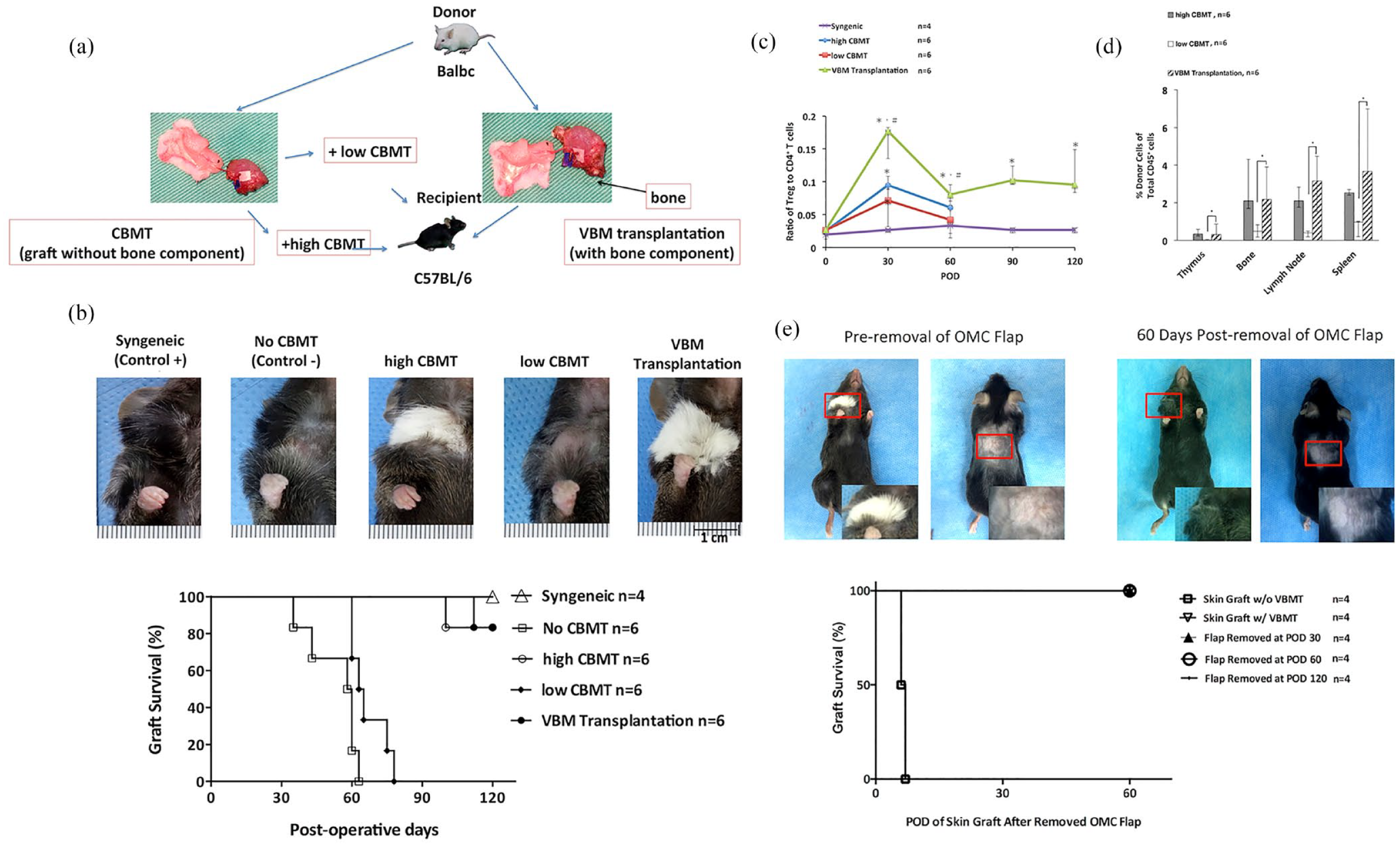
(a) Illustration of Experimental design. (b) VBM transplantation with CoB and RPM resulted in prolonged VCA survival. (c) Ratio of Tregs to CD4+ and CD8+ T cells. (d) Flow cytometric analysis of donor cells (H2d) residing in the thymus, bone, lymph nodes, and spleen of VCA recipients. (e) Survival of the skin graft is maintained after removal of the OMC flap. Reproduced by permission of Brandacher et al. 108
Strategies for prevascularization
In addition to therapies targeting immune rejection, tissue engineering strategies that facilitate prevascularization will also contribute to the success of VCA and should be considered by researchers. The prevascularization strategy usually refers to the formation of a vascular network in grafts before transplantation. 109 Microvessels are an important part of blood circulation and play a role in material exchange via anastomosis with host vessels after transplantation. 110 Here, we present some tissue engineering strategies for prevascularization that have the potential to be used in VCA.
Finding a suitable source of microvessels has always been an important challenge in prevascularization applications. Recently, the role of human induced pluripotent stem cell (hiPSC)-derived microvessels in angiogenesis, which is achieved by their differentiation into pericytes and vascular smooth muscle cells, has attracted increasing attention. Li Liao’s group recently successfully constructed an hiPSC-derived microvascular system (iMV). Through the induction of small molecules and growth factors, hiPSCs can differentiate into blood vessels and eventually generate iMVs. Experiments on various models, such as muscle injury models and skin wound models, have shown that iMVs have potential for angiogenesis and tissue repair. The results of this study provide new ideas for prevascularization strategies in the VCA. 111
In addition, there are prevascularization strategies that combine microvascular networks with porous scaffolds. Geraldine M Mitchell’s group achieved this by seeding primary human lymphatic (hLEC) and blood endothelial cell (hBEC)-induced lymphatic capillary networks and capillary networks in a 3D porous polyurethane scaffold, which has unique functions for lymphangiogenesis and prevascularization in vivo. The use of porous scaffolds provides mechanical support for the generation of vascular networks and is also a strategy worthy of reference. 112 Ying-Chieh Chen’s team also developed a prevascularization strategy by encapsulating angiogenic cells (HUVECs, MSCs) in methacrylate collagen (CMA) hydrogels, which promoted the prevascularization of the implant by hydrogels with different vascular densities. The results were validated in the volumetric muscle loss (VML) model. 113 In addition, there are other options for prevascularization. For instance, one of the in situ engineering strategies used in angiogenesis and lymphangiogenesis is called the arteriovenous loop, which induces the formation of vascularized structures by forming a circular arteriovenous fistula.114,115
The application of the above prevascularization strategies has made significant progress. Of course, some new mechanisms have yet to be proposed. Recently, the role of mechanobiology in vascularization, which has the potential to be developed into innovative therapeutic strategies, has also begun to be recognized. Mechanobiology has received increasing attention in related research on fracture healing. It is generally believed that mechanobiology can stimulate the repair of bone tissue to a certain extent. It has been proposed that mechanical loading further regulates the biological functions of bone cells by stimulating mechanoreceptors. 116 After being stimulated by mechanical load, osteoblasts and osteocytes can promote the proliferation and migration of endothelial cells through the secretion of extracellular vesicles (EVs), thereby regulating angiogenesis. 117 The discovery of new mechanisms provides new ideas for the development of prevascularization strategies. The effects of mechanically activated osteocyte-derived EVs on prevascularization in VCA need to be further verified (Table 2).
Strategies for prevascularization of vascularized composite allotransplantation.

Limitations and prospects
Even though VCA is an important part of transplantation, research on VCA has more limitations than that on SOT. There are many unavoidable problems in the study of VCA, such as complex immunogenicity, rapid and severe rejection, inefficient drug delivery, few clinical cases, long experimental periods, and the immature legislation.
Systemic administration of immunosuppressive agents has been indispensable in VCA treatment even though the shortcomings of these drugs are well known. Certainly, in the field of tissue engineering for VCA, researchers have also developed a variety of treatment modalities, such as stem cell therapy and some kinds of bioactive materials. However, compared with those for bone tissue repair or wound healing,118–120 tissue engineering strategies for VCA are still limited. For instance, genetic engineering and bioactive materials have developed particularly rapidly in recent years, resulting in the development of a variety of efficient, targeted, and precise treatments. In contrast, research on VCA is still limited by the more common strategies that have been used before. In mechanistic research, the role of Tregs in the immune regulation of VCA has been frequently mentioned, but the underlying molecular mechanism has not yet been fully elucidated, and further research on the application of Tregs needs to be conducted. On the other hand, it is more difficult to conduct clinical research on VCA than on SOT. Due to the limited number of clinical cases, the causes of trauma vary, and the operation mode varies greatly because of differences between individuals, making it difficult to unify. In addition, the donor source for VCA is still a tricky matter. Currently, solid organ donation has been accepted by many people, and there is a stable source of donors. Nevertheless, the donor source for VCA is relatively limited, and the acceptance of VCA by the public is lower, 121 which limits the development of VCA.
Moreover, ethical and legal issues are inevitable topics for both SOT and VCA. In particular, hand transplantation and face transplantation have special clinical characteristics that can change the identification factors of recipients, which has led people to pay more attention to regulations. Therefore, it is important to improve the legislation of VCA and clarify the supervision content for scientific and technological progress. Since 2014, the United States has implemented the supervision of VCA through the Organ Procurement and Transplant Network (OPTN), including the transplant process, donor authorization, recipient allocation, and other content.10,102 Nevertheless, in the face of emerging clinical problems, such as treatment procedures in the posttransplantation period and the application of tissue engineering methods, updated regulations are still lacking, which is conducive to both basic research and clinical trials.
At the same time, whether the emerging tissue engineering methods for VCA can achieve a balance between the efficacy and cost is also a significant question. Even if some of the current research protocols are novel, but they are expensive, complicated to operate, and have limited sources of materials, which may lead to unaffordable expenses after clinical transformation. Therefore, how to achieve the feasibility of transformation is also a topic that needs to be considered.
Notably, these limitations are not irremediable. The development of tissue engineering for VCA still has broad prospects and great clinical significance for overcoming the drawbacks of immune rejection and vascularization. Although public perceptions and policy support for VCA will not change quickly, biomaterials, 3D printing technology and engineered autologous tissues have the potential to promote innovation. Moreover, immunity and rejection are hot research fields, and new mechanistic findings could emerge rapidly because of the development of bioinformatics technology. In addition to solid organ transplantation therapy, the introduction of gene engineering technology, such as CRISPR-Cas9, for the treatment of VCA rejection is also a new strategy to explore.
Finally, the translation of the aforementioned studies into clinical application is difficult, and several problems must be solved. First, experimental animal models of VCA need to be standardized, reproducible, and representative to ensure that clinical trials are conducted. Second, technologies should be developed with the goal of achieving commercial production so that the balance between development costs and therapeutic efficacy can be maintained. The clinical translation of emerging technologies may be difficult due to the high cost and uncertainty of effectiveness, so the integration of new technologies with established treatments is also an option. At the same time, the number of VCA cases in different countries is not comparable, so high-quality multicenter studies should be carried out to promote the clinical transformation of tissue engineering treatment strategies for VCA.
In summary, if more researchers in the fields of transplantation, plastic surgery, and tissue engineering focus on the related aspects of VCA, it will be of great help to the development of VCA. Moreover, allograft rejection and related immune regulation are very important topics. Researchers can conduct more in-depth discussions on this issue, which is conducive to more effectively solving these problems.
Footnotes
Acknowledgements
The authors greatly appreciate Lihong Chen, Yanshan Liu, and Chunchun Li from Center for Basic and Translational Research, The Second Affiliated Hospital, Zhejiang University School of Medicine.
Author contributions
Danyang Ren: Participated in research design and writing of the paper. Jun Chen: Participated in the writing of the paper. Meirong Yu: Participated in the editing of the paper. Chenggang Yi, Xueqing Hu, and Junjie Deng: Provided formulation or evolution of overarching research goals and aims. Songxue Guo: Participated in the management and coordination responsibility for the research activity planning and execution.
Availability of data and materials
Data availability is not applicable to this article as no new data were created or analyzed in this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Grant No. 82272259), National Key Research Project of China (Grant No.2022YFC2403100), and Zhejiang Provincial Natural Science Foundation of China (Grant No.Q23H150010).
