Abstract
Early in vitro oral mucosal infection models (OMMs) failed to consider the suitability of the model environment to represent the host immune response. Denture stomatitis (DS) is mediated by Candida albicans, but the role of Staphylococcus aureus remains uncertain. A collagen hydrogel-based OMM containing HaCaT and HGF cell types was developed, characterised and employed to study of tissue invasion and pro-inflammatory cytokine production in response to pathogens. Models formed a robust epithelium. Despite their inflammatory baseline, 24-h infection with C. albicans, and/or S. aureus led to tissue invasion, and significantly upregulated IL-6 and IL-8 production by OMMs when compared to the unstimulated control. No significant difference in IL-6 or IL-8 production by OMMs was observed between single and dual infections. These attributes indicate that this newly developed OMM is suitable for the study of DS and could be implemented for the wider study of oral infection.
Introduction
The oral immune system is compromised by denture wear. Reduced salivary flow between the denture and the hard palate inhibits the sloughing of epithelial cells and delivery of salivary antimicrobial factors.1 –4 Denture wear can lead to tissue trauma, especially if improperly fitted, compromising the integrity of the epithelial barrier. Additionally, acrylic dentures may support biofilm formation, particularly when inadequately or infrequently cleaned. 5
The fungus Candida albicans is a frequent oral coloniser and a common cause of infection. 4 C.albicans is the main causative agent of Denture Stomatitis (DS), which presents as erythema and oedema in oral tissues that underlie dentures, most notably the hard palate. 3 Locally, the recognition of pathogens, mostly due to damage, by epithelial cells and myeloid cells including tissue resident macrophages and dendritic cells, leads to localised inflammation of the hard palate, which in more severe cases can lead to more generalised inflammation of the oral cavity. 6 DS associated biofilms are thought to be potential reservoirs for disseminated infection, therefore all DS cases should be considered of clinical importance.7 –9
Concurrent bacterial infections are common and may contribute to disease severity. Staphylococcus aureus is a commensal bacterium that can act as an opportunistic pathogen. S.aureus is now an accepted member of the normal oral flora and has been detected in conjunction with C.albicans in the oral cavity, and is thought to be associated with DS .10 –12 More recently, small colony variants of S.aureus were found to associate with C.albicans amongst denture wearers. 13 Elsewhere, S.aureus infections range from superficial, localised wound infections to systemic sepsis, all of which are dependent on a portal of entry through the epithelial barrier. 14 C.albicans and S.aureus are considered to have a synergistic relationship in terms of virulence and disease severity.15 –17 It has also been proposed that S.aureus may be able to hitch a lift across the mucosal barrier by adhering to C.albicans, risking S.aureus gaining access to deeper tissues and subsequently the blood stream.18 –22
C. albicans contains multiple cell wall components that possess conserved structural motifs including chitin, glucans and mannans, which are exposed to the extracellular environment, as well as internal nucleic acid sequences, all of which may act as PAMPs and are subsequently detected by pattern recognition receptors (PRRs) .23 –31 Gram positive bacteria such as S.aureus possess multiple cell-wall components that act as PAMPs including lipoteichoic acid and peptidoglycan, which are recognised by TLR-2.32,33 TLR-2 is expressed at the cell surface of oral keratinocytes, gingival fibroblasts and in vivo on innate immune cells, and can recognise both C.albicans and S.aureus, in conjunction with other PRRs.25,32 –35 Upon pathogenic recognition, epithelial cells and immune cells secrete cytokines, which instigate an immune response, including further immune cell recruitment and, in the case of a functional immune system, ultimately pathogenic clearance and tissue repair.
At the oral mucosal barrier, keratinocytes and local resident immune cells such as macrophages and Langerhans cells possess pattern recognition receptors such as Toll-Like-Receptors (TLRs), which recognise conserved pathogen associated molecular patterns (PAMPs), and damage associated molecular patterns (DAMPs).36,37 Upon TLR activation, cytokines are produced. Depending on the local cytokine milieu, an immune response is orchestrated. This may lead to the recruitment of further innate immune cells for pathogenic clearance, differentiating naïve immune cells into their specific and activated counterparts and presenting pathogenic antigens to the adaptive immune system to instigate a memory response.
Pro-inflammatory cytokines play a key role in instigating an immune response at the oral mucosal barrier. Interleukin 6 (IL-6) is a multifunctional cytokine that is paramount to a functional immune response. It undertakes both inflammatory and regulatory roles and is required to maintain tissue health. IL-6 is secreted by many cell types, including immune cells, oral epithelial cells and fibroblasts, and the IL-6 receptor (IL-6R) is expressed on a multitude of cell types. 38 IL-6 is also important for regulating wound healing; and dysregulation of IL-6 can lead to either fibrotic or chronic wounds. 39 Interleukin 8 (IL-8) is a pro-inflammatory cytokine secreted by both keratinocytes and fibroblasts. IL-8 has a direct effect on immune cells, leading to neutrophil migration, an integral part of the cellular innate immune defence. In addition to this, IL-8 may enhance both keratinocyte and fibroblast migration and has been demonstrated to influence wound healing.40,41 The ability of both IL-6 and IL-8 to drive both inflammation and promote wound healing makes them very interesting indicators for both the induction and the resolution of infection-induced damage at the oral mucosal barrier.
To gain deeper insight into the synergistic relationship between C.albicans and S.aureus, and further elucidate the role of this synergy in DS, it is desirable to study the interaction of these in an in vitro oral barrier model. A three-dimensional structure incorporating multiple cell types into a matrix material encourages cellular crosstalk and spatial interactions, and allows for cellular migration, stratification and matrix remodeling.42 –48 Depending on the desired application, a different onus is placed on the necessary attributes of a successful oral mucosal model. Selection of incorporated cell types and matrix materials are often dependent on the desired application of the model, each with their own merits and shortfalls. 49 OMMs have been frequently validated for the study of tissue invasion and damage upon infection and have proven to be valuable tools when considering the pathogenesis, virulence and treatment of C.albicans, S.aureus and other oral pathogens.48,50 –61 Additionally, OMMs containing keratinocytes and fibroblasts have been demonstrated to produce pro-inflammatory cytokines upon stimulation with live pathogens such as C.albicans.48,62,63
Few oral mucosal models have gone beyond this, to incorporate immune cells. Those that have been developed included Macrophages and Langerhans cells, however to the best of our knowledge, none have been further implemented for the study of pathogen-host interactions.62,64 –67 Models with the ability to recognise and effectively respond to infection would be informative tools to determine pathogen-host interactions. It is therefore of particular interest to identify any barriers in the current field that may hinder the effective development and implementation of an immunocompetent oral mucosal infection model.
Aims
This paper aims to consider the suitability of a newly devised OMM to model Denture Stomatitis by ensuring that the model effectively represents pathogen-host interactions upon single and dual infections of C.albicans and / or S.aureus. The immunogenicity of the model environment and its incorporated cell types will be studied, to determine whether the model can produce pro-inflammatory cytokines (IL-6 and IL-8) in response to microbial insult. These data will be used to comment on whether the model environment is a) suitable for the study of pathogen-host interactions at mucosal surface and b) identify the barriers to the further development and application of oral mucosal infection models.
Methodology
Cell selection
HaCaT cells, a spontaneously immortalised keratinocyte cell line, derived from histologically normal adult skin, were employed form the epithelial barrier. HaCaT were selected due to their longevity, reproducibility, reported ability to form stratified, keratinised epithelia and previous employment to represent the inflammatory response of the oral epithelium.68 –71 Human gingival fibroblasts (HGF) were selected to form the lamina propria layer of the tissue when embedded in a type-1 collagen matrix. HGFs were selected due to their anatomical location, as fibroblasts from different locations, even within the oral cavity, are physiologically diverse, and their activity is dependent on their origin.53,72
Cell culture
Immortalised keratinocytes (HaCaT), ATCC PCS-200-011 and Primary Human Gingival Fibroblasts (HGF), ATCC PCS-201-018, were maintained in DMEM containing 1 g/L glucose, 584 mg/L L-glutamine, 110 mg/L sodium pyruvate and 15 mg/L phenol red, Life technologies Ltd, Gibco, Paisley UK, and 10% FBS, Life technologies Ltd, Gibco, 10500 batch #08Q1379K. HaCaT were utilised between passages 39 and 48 and HGF, passages 8–14.
OMM production
Three-dimensional oral mucosal models (OMM) were formed in 0.4 μm polycarbonate mesh transwell inserts, Sigma Aldrich, Corning, Gillingham, UK, using rat-tail type-1 collagen, First link (UK) Ltd., Birmingham, UK, with a protein concentration of 2 mg/ml. HGF were embedded into a collagen gel mix, containing: 25.6% collagen, 63.5% DMEM, 7% FBS and 3.8% 1 M NaOH. Once polymerised, a keratinocyte (HaCaT) overlay was added to the gel matrix. Each model was formed of 800 µl total hydrogel mix containing 3.75 × 104 HGF and overlayed with 2.1 × 105 HaCaT. Once the models had contracted (~48 h), remaining media was aspirated and replaced with 500 μl of KGM: Keratinocyte gold basal media, Lonza, Slough, UK, supplemented with KGM gold bullet kit, Lonza (omitting Gentamicin/Amphotericin) and 2.4 μM CaCl2, below the transwell insert. Models were kept at the air-liquid interface (ALI) for the remainder of culture, media was changed every 48–72 h thereafter.
Histology
Formalin fixed paraffin embedded sections (5 μm thickness) were prepared and stained with Haematoxylin and Eosin prior to being mounted using DPX, and imaged.
Microbial preparation
The commonly used strain C.albicans SC5314 was used alongside a S.aureus strain (UOPOSA001), which was isolated from the oral cavity of an individual that wore dentures. Ethical approval and written informed consent were sought to isolate clinically relevant strains of S.aureus (Granted by the Health Research Authority London – Camden & Kings Cross Research Ethics Committee, IRAS ref. 208291). A swab was taken from the hard palate and a streak plate performed on mannitol salt agar (MSA), OXOID, Basingstoke, UK. After 24 h of incubation at 37°C a single colony was isolated and transferred onto tryptic-soy agar (TSA) Sigma Aldrich. The pure colony was identified as S.aureus through its golden appearance on MSA, positive gram stain and both coagulase positive and catalase positive biochemical tests as per the manufacturer’s instructions. Fisher Scientific, Loughborough, UK.
C.albicans was maintained on Sabouraud-dextrose agar, Sigma Aldrich, and S.aureus was maintained on TSA. Prior to use, an overnight culture was created by resuspending a single colony of C.albicans or S.aureus in Sabouraud-dextrose broth or tryptic-soy broth, respectively and grown in a 37°C shaking incubator. Prior to use, C. albicans was washed three times via centrifugation at 3000 × g for 5 min in PBS. Cultures were diluted 1 in 10 and counted using a haemocytometer. Prior to use, S. aureus was washed three times via centrifugation at 10,000 × g in PBS and due to its smaller size, was quantified spectrophotometrically, titrated to an OD of 0.2 at 595 nm (~2 × 107 CFU/ml, as revealed by preliminary experimentation), and subsequently diluted to the desired infectious dose.
LDH assay
A time-course was performed to assess LDH release over the duration of 21 days of OMM culture. A Pierce-LDH cytotoxicity assay kit, Thermo-scientific 88953, was used as per the manufacture’s instruction. OMM LDH release was controlled against a maximum LDH release control (MC) by lysing OMMs at each time point, as per the manufacturer’s instruction. LDH was firstly represented as a percentage of the MC for each time point, and then represented as a percentage of day-1 LDH release.
Quantification of cytokine secretion
Secretion of IL-6 and IL-8 into cell culture supernatants were quantified by sandwich ELISA, using commercially available paired capture and detection antibodies (BD-Biosciences, Wokingham, UK). Capture antibodies were used at 1 µg/ml (BD Biosciences, 554543) and 2 µg/ml (BD Biosciences, 553716) for IL-6 and IL-8, respectively, and detection antibodies at 0.5 µg/ml (BD Biosciences 554546 and 554718). Protocols were followed according to manufacturer’s instructions and compared to standard curves of recombinant human cytokines (using recognised international cytokine standards available from NIBSC, Potter’s Bar, UK) between the range of 7 and 5000 pg/ml. Colorimetric development was determined spectrophotometrically by an OPTIMax tuneable microplate reader at 450 nm and analysed by Softmax Pro version 2.4.1 (Molecular Devices Corp., Sunnyvale, CA, USA).
Monolayer infections
HaCaT cells were seeded at a density of 7.1 × 104 / cm2, HGF cells were seeded at a density of 1.42 × 104 cells/cm2, seeding densities were selected due to cell size and to proportionally represent the number of each cell type included in the three-dimensional models (below). Both were grown until confluence at 37°C in 12-well plates. C.albicans and S.aureus were cultured and quantified as above and then set to a count of 5 × 105 CFU/ml in DMEM + 10% FBS. Control wells contained media only, 1 ml of each organism in DMEM + 10% FBS was added to each single infection well. For dual infections 0.5 ml of each organism was added to the same well ~MOI 0.5 for each.
Three-dimensional infections
OMMs were utilised at day 19, to permit time for sufficient model maturation and infected with C. albicans SC5314 ~MOI 0.1 (1 × 105 CFU/ml), S.aureus P116 ~MOI 1 (1 × 106 CFU/ml), and both C. albicans and S. aureus MOI 0.05 and MOI 0.5, respectively. Models were used for both ELISA and Histological evaluation, proportions of microbes were selected to be more representative of the proportion of bacteria to yeast found in in vitro infections. Organisms were co-cultured with cells/models for 24 h, and supernatant collected for subsequent ELISA analyses. A 24-h culture was selected to balance the ability to detect the initial response of the OMM to both IL-6 and IL-8 in the model supernatant, and to maximise the amount of time C.albicans and S.aureus had to commence tissue colonisation, prior to risking the model elevating cytokine levels due to hypoxia, from both prolonged culture time and competition for nutrients from introduced microbes. OMMs were then fixed in 4% PFA for >24 h at 4°C prior to histological analysis.
Model interactions
HaCaT cells were seeded at a density of 5.4 × 105 cells/ml and HGF 1.2 × 105 cells/ml and allowed to adhere overnight. Each cell type was stimulated with collagen and supernatant of the other cell type contained within the model. Cells were embedded in collagen as per the OMM protocol, cultured for 24 h prior to supernatant collection by aspiration after centrifugation at 200 × g. Supernatant was collected from cultured cell types, at the same seeding density as detailed above introduced to the other cell type in a pulse-chased manner, to ensure that cytokines contained within the conditioned supernatant did not skew the final ELISA readouts. Supernatant was added to the cell line at a 1:1 ratio with culture medium for 4 h, and then removed. Cells were cultured for a further 18 h in refreshed media. Supernatant was then collected and analysed via ELISA.
Results
OMMs are similar in structure to native oral mucosa
Firstly, to ensure that the model would be suitable for the study of tissue invasion, it was important to ensure that the structure of the OMM suitably represented that of the native oral mucosa. Figure 1 demonstrates cross-sections of OMMs at day-19 compared with native oral mucosa. Both tissues exhibit an organised multi-layered epithelium. Epithelial layers are distinct from the lamina propria layer in both models and native tissue. Native oral mucosa appears highly stratified, the OMM displays a level of stratification that is less organised than that of the native oral mucosa, in image D, whereas image C exhibits even lower stratification. Within the lamina propria layer, fibroblasts are identifiable by their haematoxylin-stained nuclei. There are fewer visible nuclei within the lamina propria layer of the OMM compared with the native tissue. At the apical layer of the epithelium, within both the native oral mucosa and the OMM there are fewer nuclei present, and a flattened appearance indicative of squames. Image C indicates the lamina propria and epithelium have separated.
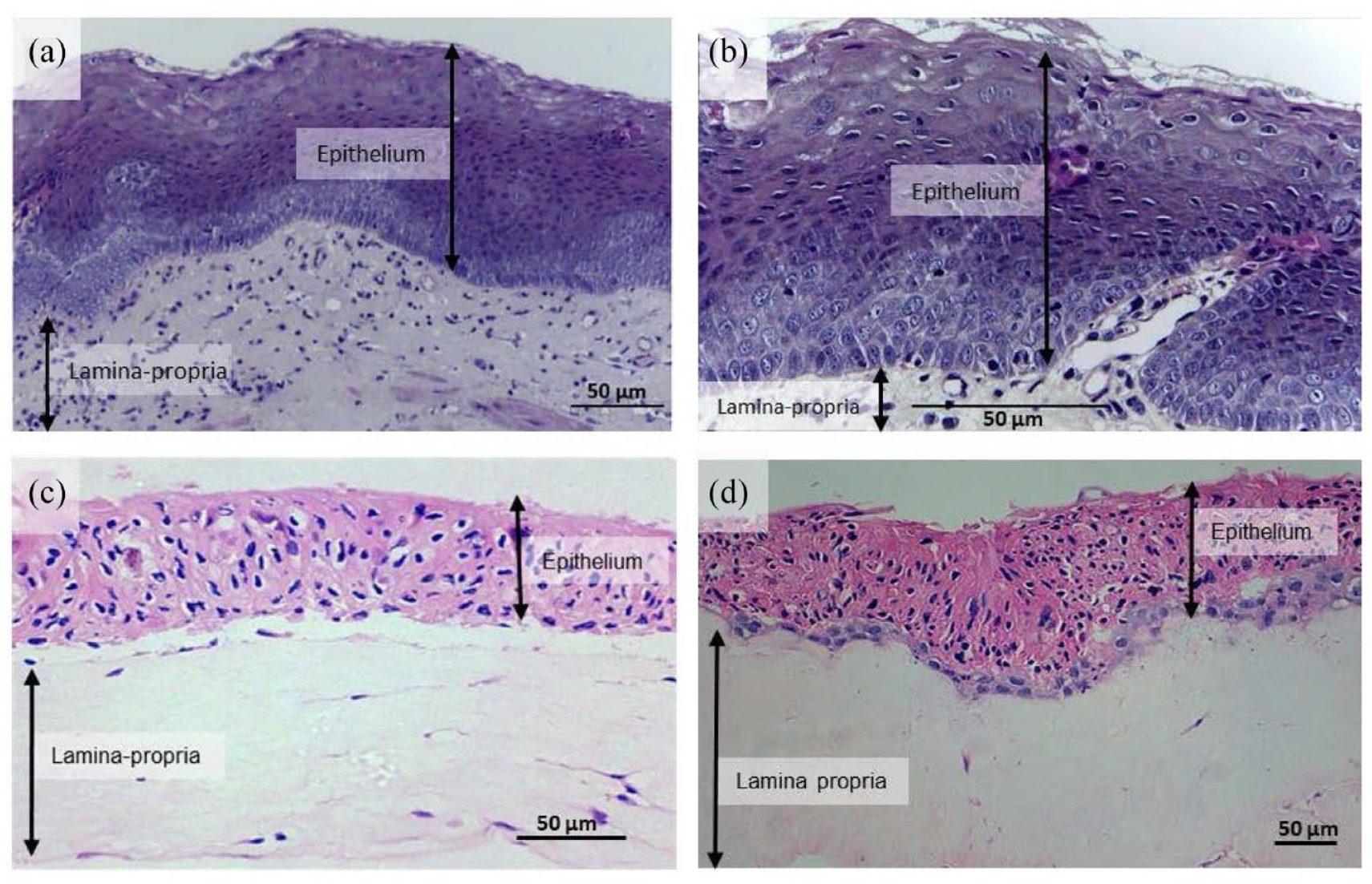
OMMs display histological similarities to native oral mucosal tissue. Images of normal human oral mucosa (a and b) were obtained from: A histopathological image repository of normal epithelium of Oral Cavity and Oral Squamous Cell Carcinoma by Rahman et al. 73 , under CC BY 4.0, images were re-oriented, cropped, labelled and post-fitted with a scale bar The two images were selected as they represented a similar field of view to the OMMs formed in this study. A representative sample of paraffin embedded, 4 μm sectioned, haematoxylin and eosin-stained oral mucosal tissue (images c and d) at day 19 of growth (100× magnification) were evaluated. Two images are selected to demonstrate the range of histological presentation of the OMMs and post-fitted with scale bars. Image scale was calculated using pixel size, number of pixels, width of the output image and objective magnification. Labels indicate the epithelial (keratinocyte-containing) and the lamina propria (fibroblast-containing) layers of the tissue.
Cell-death and baseline pro-inflammatory production are interlinked
Preliminary experimentation revealed unexpectedly high levels of the pro-inflammatory cytokines IL-6 and IL-8 during model development. It was necessary to further explore this observation to determine (a) what was the cause of this high inflammatory baseline, (b) whether this was likely to inhibit the use of the OMM to model host response to infection and (c), if not, what is the best timepoint in which to utilise this model.
Figure 2 demonstrates a close relationship between IL-6 and IL-8 pro-inflammatory cytokine production, and LDH release. Spearman’s rank correlation analysis indicates a strong positive correlation between LDH release and IL-6 production (r = 0.936), LDH release and IL-8 production (r = 0.945) and IL-6 and IL-8 production (r = 0.891; reporting p values of p = 0.00008, 0.00005 and 0.00052, respectively). A strong correlation is observed between IL-6, IL-8 and LDH release. Higher levels of all factors are observed at day 1, which declines over the first 7 days. After this point the IL-6, IL-8 and LDH activity does not return to its original high level over the 21-day period, however there is gradual increase in all from day 7 onwards. Across all experimental replicates mean IL-6 secretion was 7542.42 ± 1322.65 pg/ml and mean IL-8 secretion was 2481.72 ± 351.4 pg/ml at day 1.

Cell death and IL-6 and IL-8 pro-inflammatory cytokine production correlate across the duration of OMM culture. Three experimental repeats, each with four biological replicates (performed in parallel). LDH activity was controlled using a maximum LDH control at each time point, by lysing models at the same day of growth. IL-6 and IL-8 production were measured via sandwich ELISA. LDH release was normalised to the maximum LDH control for the corresponding time point. LDH, IL-6 and IL-8 values for each time point are reported on this graph as a percentage of mean day 1 release. Data normality was assessed using a Shapiro-Wilk test. Correlation analysis was performed using the non-parametric Spearman’s rank correlation test, to determine the relationship between IL-6, IL-8 and LDH release. Error bars represent standard deviation.
Considering the initial high levels of pro-inflammatory cytokines released upon the formation of the OMMs, it was hypothesised that the introduction of the cell types to the model environment may lead to this marked inflammatory environment. It was therefore of interest to elucidate, if, and if so which components of the model were stimulating which cells to produce such high levels of IL-6 and IL-8.
Collagen and secretory factors affect pro-inflammatory cytokine production
Figure 3 demonstrates that the HaCaT and HGF cell types used to form the OMM react to the matrix environment and soluble factors secreted by the other cell type. Collagen and HGF supernatant upregulated IL-6 production by HaCaT cells, when compared to the unstimulated control (p = 0.0019 and <0.0001, respectively). Conversely, both collagen and HGF supernatant downregulated IL-8 production by HaCaT (p < 0.0001 for both). For HGF, IL-6 production was significantly greater when stimulated with collagen, compared with the unstimulated control (p < 0.0001), whereas pulse-chasing with HaCaT supernatant demonstrated no significant difference. Both collagen and HaCaT supernatant upregulated IL-8 production by HGF when compared to the unstimulated control (p < 0.0001 for both). Across the three experimental replicates, at baseline, the HaCaT unstimulated control produced on average 44.40 ± 26.48 pg/ml of IL-6 and 93.91 ± 41.85 pg/ml of IL-8, and the HGF unstimulated control produced 130.26 ± 72.23 pg/ml of IL-6 and 12.45 ± 6.90 pg/ml of IL-8.

Collagen and conditioned media differentially induce fibroblast and keratinocyte pro-inflammatory cytokines. IL-6 production by HaCaT (a), IL-6 production by HGF (b), IL-8 production by HaCaT (c) and IL-8 production by HGF (d), in response to collagen and supernatant (SN). Supernatant was pulse-chased to ensure that any IL-6 or IL-8 observed by ELISA was because of stimulation by the other incorporated cell-type. Data are from three independent experiments performed in triplicate. Significant differences in cytokine production were compared between the unstimulated control (media only) and stimulated groups. Data normality was assessed using the Shapiro-Wilk test for normality. Statistical significance was determined using a Kruskal-Wallis test followed by a Dunn’s multiple comparisons test. Statistical significance is indicated on the graph (*p < 0.05. **p < 0.01. ***p < 0.001. ****p < 0.0001, ns = not significant). Error bars represent standard deviation.
Next, Histological evaluation was used to demonstrate tissue invasion upon infection, and ELISAs demonstrated that models were able to respond to single and dual infections of these microbes. Additionally, following the identification that a variety of unavoidable factors contribute to the already present IL-6 and IL-8 at the model’s baseline, it was next important to determine whether the OMMs were capable of exhibiting upregulation of these cytokines in response to infection with C.albicans and S.aureus. In addition, it was of interest to determine which, if any, of the two-incorporated cell types were able to recognise and respond to C.albicans and S.aureus, to gain a more thorough insight of this system and how each cell type may contribute to the overall picture.
OMMs demonstrate tissue-invasion during C.albicans and S.aureus infection
Within Figure 4, the uninfected control model (a) demonstrates a multi-layered intact epithelium anchored to a lamina-propria that is abundant with fibroblasts. Single C. albicans infection (b) can be observed as a layer of hyphae atop the epithelial layer of an intact model. Single infection with S.aureus (c) indicates bacteria lining cavities in the lamina-propria of the OMM. Dual infections with both C. albicans and S. aureus indicate both organisms present on top the epithelium, within the epithelium and within the lamina propria layer (d and e). C. albicans formed a layer at the upper most of the section, above the epithelium. S. aureus appeared throughout the Candida layer. C. albicans hyphae span the entire epithelium, with varying biofilm thickness and S. aureus can be observed in clusters below this layer. During dual infections, C.albicans appears to infrequently penetrate the epithelium (d and e).

OMMs are suitable to demonstrate biofilm formation and microbial invasion. A representative sample of paraffin embedded, 4 μm sectioned, haematoxylin and eosin stained OMMs infected with C. albicans (~MOI 0.1), S. aureus (~MOI 1) and both (~MOI 0.05 and 0.5, respectively). Image a displays a control model (100× magnification), in absence of any infection. Image b displays a model infected with C. albicans (200× magnification). Image c displays a model infected with S. aureus (200× magnification). Image d displays a model infected with both C. albicans and S. aureus (400× magnification). Image e displays a full-length cross section of an OMM infected with both organisms (100× magnification). Images were post-fitted with scale bars which were calculated using pixel size, number of pixels, width of the output image and objective magnification.
C.albicans and S.aureus stimulate pro-inflammatory cytokine production in 2D and 3D culture
Single and dual infections of C.albicans and S.aureus stimulated IL-6 and/or IL-8 pro-inflammatory cytokine production by HaCaT and HGF monolayers and OMMs, when compared to the unstimulated control for each group (Figure 5). C.albicans infection significantly increased IL-6 and IL-8 production by HaCaT (p < 0.0001 for both). S.aureus led to an increase in IL-6, but not IL-8 production within HaCaT cells (p = 0.0397 and 0.8171, respectively). Dual infection with both organisms led to a significant increase in both IL-6 and IL-8 production by HaCaT cells (p = 0.0002 and 0.0020, respectively). HGF infection with C.albicans led to a significant increase in both IL-6 and IL-8 production (p < 0.0001 for both). S.aureus also significantly increased IL-6 and IL-8 production within HGF (p = 0.0035 and <0.0001, respectively). For HGF, dual infections of both organisms demonstrated a significant increase in both IL-6 and IL-8 (p = 0.0175 and 0.0010, respectively). OMMs infected with C. albicans, or S. aureus, or dual infections of both microbes led to a significant increase in IL-6 and IL-8 production (p < 0.0012, 0.0070 and 0.0015 for IL-6; and p < 0.0001, 0.0001 and 0.0312 for IL-8, respectively).

C.albicans and S.aureus stimulate pro-inflammatory cytokine production by OMMs and individual cell types. IL-6 and IL-8 pro-inflammatory cytokine production by HaCaT (a and d), HGF (b and e) and OMMs (c and f) infected with single infections of C. albicans SC5314 and S. aureus UOPOSA001, each at a density of 5 × 105 CFU/ml, ~MOI 1 (HaCaT), ~MOI 5 (HGF) and dual infections of both. OMMs were cultured until day 19 and then infected with 1 × 105 CFU/ml C.albicans ~MOI 0.1 and 1 × 106 CFU/ml and S.aureus ~MOI 1. Dual infections were seeded at half of the amount of each organism. Graphs indicate three experimental repeats performed in triplicate. Models/cells were co-cultured with the infective organisms for 24 h, and cytokine production was assessed via Sandwich ELISA. Data were standardised to the percentage of the unstimulated control group and statistical significance determined between all groups of the same graph. Data normality was assessed using the Shapiro-Wilk test for normality. Statistical significance was determined using a One-Way ANOVA followed by a Dunnett’s multiple comparisons test for parametric data (d) or a Kruskal-Wallis followed by a Dunn’s multiple comparisons test for (all other) non-parametric data. Statistical significance is indicated on each graph (*p < 0.05. **p < 0.01. ***p < 0.001. ****p < 0.0001, NS = Not significant). Error bars represent standard deviation.
Within HGF cells and OMMs there was no significant difference in either IL-6 or IL-8 production between single and dual infections of C.albicans and/or S.aureus. However, in HaCaT cells when compared with S.aureus, C.albicans was found to stimulate significantly more IL-6 and IL-8 p = 0.0108 and <0.0001, respectively. In addition, for IL-8, dual infections lay somewhere between, stimulating significantly more cytokine production than S.aureus alone, p = 0.0227, and significantly less than C.albicans alone, p = 0.0121. This was not observed for IL-6.
Across the three experimental replicates, at baseline, the HaCaT unstimulated control produced an average of 949.82 ± 578.99 pg/ml of IL-6 and 6718.393 ± 4104.485 pg/ml of IL-8, which was highly variable. The HGF unstimulated control produced 1589.99 ± 590.92 pg/ml of IL-6 and 479.202 ± 346.41 pg/ml of IL-8. For OMMs, the baseline was also variable between experimental replicates, on average producing 1511.41 ± 1075.728 pg/ml of IL-6 at baseline and 1258.61 ± 978.279 pg/ml of IL-8.
Discussion
This paper characterises an oral mucosal infection model (OMM) consisting of collagen 1-embedded human gingival fibroblasts (HGF) overlaid with a HaCaT epithelia and explores the potential of this model to accurately represent host-pathogen interactions for the study of oral infections such as denture stomatitis (DS). The OMM characterised formed a simple, effective organotypic full-thickness model that is structurally similar to the native oral mucosa. This model was used to effectively demonstrate tissue-invasion and pro-inflammatory cytokine production in response to the infective agents Candida albicans and Staphylococcus aureus. Importantly, this work highlighted common challenges and limitations of such models, including their inflammatory baseline, and explores the potential to further develop oral barrier models to further represent the immune system upon infection.
OMMs are robust structures that consistently formed a multi-layered epithelial layer atop a fibroblast populated lamina propria layer, comparable to that of the native oral mucosa. The OMM epithelial layer exhibited less organisation than the highly stratified native oral mucosa. Cells were often denser towards the basal layers of the epithelium, which may indicate a partial stratification. The upper epithelial layers of the 3DOMM indicated a low degree of keratinisation, exhibiting one to two layers of enucleated flattened squames. This lower degree of stratification is due to the use of the immortalised HaCaT cell type. 69 Alternative models utilising primary cells, or h-TERT immortalised cells have achieved stratification that is more comparable to their in vivo counterpart.66,74
Due to the reduced keratinisation, these models better represent the buccal mucosa as opposed to the hard palate as intended. 75 Considering that the common site for DS infection is that underlying the denture plate, in some cases, such as to model initial DS onset, it may be necessary to consider alternative keratinocyte cell types to better represent the keratinised barrier of a healthy oral mucosa, prior to the introduction of colonised denture-mimics. 59 In future, these models should be stained with laminin, to draw conclusions regarding the ability of HaCaT-HGF OMMs to permit the development of a physiologically relevant basement membrane. 76
Within an increasing body of literature implementing sophisticated technologies to produce OMMs, this model features towards the more simplistic end of the spectrum. However, with simplicity comes ease of production, affordability and most importantly as demonstrated from our findings, the potential to indicate tissue invasion and cytokine response upon infection. To this model’s merit, the epithelial barrier was robust, remaining intact and exhibiting no significant disruption upon pathogenic invasion. This desirable attribute indicates the OMM as a suitable candidate to further elucidate the cellular and molecular mechanisms underpinning DS pathogenesis.
As indicated by the time-course, cell death was at its highest during the first few days of culture, it then declined until day 7, and then steadily rose until day 21. Increased LDH release correlated with high levels of IL-6 and IL-8 and IL-8 pro-inflammatory cytokine production. These elevated levels may be due to uncontrolled cytokine release during cell lysis and damage receptor activation. This particular OMM was used at day-19 to allow enough time for epithelial development and reduce the likelihood of substantial death due to hypoxia if cultured for too long. If models are required for longer-term culture perfusion systems are available to mitigate hypoxia. 77 Considering IL-6 and IL-8 are also known to have roles in cellular migration and matrix remodelling, the observed highly pro-inflammatory baseline may also result from such expected activities during model formation. Considering the above, the length of time a model is left in culture, period of microbial co-culture and timing of supernatant collection from infected models should be carefully balanced.
The incorporated HaCaT and HGF cell types were responsive to both the collagen and secreted factors (supernatant) present in the model environment, mostly inducing a stimulatory effect, increasing IL-6 and/or IL-8 in response to model components, with the exception of collagen and HaCaT cells, which actually led to a reduction in IL-8 production, even though it still stimulated IL-6 production in this cell type. This already high baseline caused by the model environment did not hinder the model’s ability to further respond to pathogens. The differential HaCaT pro-inflammatory response to collagen could be due to the incorporation of cells into the matrix, as opposed to their seeding on top of the matrix when producing the models. This study is not the first to identify that cytokines are differentially expressed between cells cultured in 2D and their collagen-embedded 3D counterparts. In fact, multiple hydrogels including agarose, alginate, collagen, Matrigel and RGD-functionalized polyethylene glycol (PEG) constructs, exhibited differential cytokine expression, this alteration was observed across multiple cell types. Biomaterial properties and cell-type inclusion therefore require thorough consideration when developing a disease model; particularly one where cytokine expression is an intended outcome measure. 78 When selecting model matrices, media and cell-types, it is proposed that attention is paid to the individual responses and crosstalk occurring due to cells being introduced to their new environment.
Standardisation of data to a percentage of the unstimulated baseline without reporting the absolute control values may hinder the appropriate selection of an infection model from the literature. In this study, infected OMMs exhibited varying IL-6 and IL-8 baseline values between the three experimental replicates. Higher baselines demonstrated a less marked upregulation in cytokine production upon infection, compared to replicates with lower baselines. Difficulties may be faced when introducing immune cells to OMMs, due to the ability of cytokines to activate and tailor immune cells such as macrophages towards either a regulatory or inflammatory response. 37 It is also possible that immune-cell crosstalk may help to modulate the model’s environment; this remains to be explored. It is therefore proposed that baseline cytokine production should be considered, and that close attention should be paid to cell-matrix interactions if these OMMs, or similar, are taken forward for to further incorporate immune cells.
Pro-inflammatory cytokine production (IL-6 and IL-8) was upregulated in response to both pathogens. These OMMs are therefore likely to be a useful tool for studying a variety of gram-positive bacterial and fungal pathogen-host interactions, due to their ability to produce pro-inflammatory cytokines in response to C.albicans and S.aureus. However, as the incorporated HaCaT-epithelial cell type are typically unresponsive to LPS and lack expression of the MD-2 adaptor protein, which is required for TLR-4 activation, it should be noted that this model is unlikely to demonstrate a pro-inflammatory cytokine response to gram-negative bacteria. 79 This lack of response is characteristic of barrier cells, due to the necessity of tolerance to maintain homeostasis. In fact, high levels of TLR-4 expression at the skin and mucosal barriers are linked with inflammatory diseases such as systemic sclerosis and periodontitis.80,81
It was of particular interest to determine whether this OMM was suitable to model DS with specific reference to the pathogens C.albicans and S.aureus. This early look demonstrated that C.albicans hyphae were able to penetrate the epithelial barrier, and S.aureus were subsequently found amongst the lamina propria layer. C.albicans can exist as both yeast and hyphal forms. Strain SC5314 is dimorphic but exhibits virulence that can in part be attributed to gene expression that promotes its more invasive, filamentous hyphal form. Yeast to hyphal transition can be promoted by the presence of serum, such as the FBS present in the model environment.82,83 The OMMs epithelial barrier remained visually intact irrespective of infection with C.albicans and / or S.aureus. Previous reports have identified significant disruption to the epithelial layer upon C. albicans infection of similar full-thickness models with frequent penetration of Candida hyphae.54,56,59 Models were also treated with a higher dose of C.albicans SC5315 (MOI 1), and still demonstrated no visible damage to the epithelium (data not shown). Due to this, considering that 24-h incubation with C. albicans SC5314 caused obvious epithelial damage in alternative models, it is plausible to consider the epithelia of the newly developed OMM more robust than current alternative infection models.
Models infected with C. albicans formed Candida biofilms at the surface of the epithelium, regardless of whether S. aureus was present. S.aureus appeared to line cavities within the lamina propria layer of the OMM regardless of C.albicans presence, indicating that S.aureus could gain access to deeper tissue layers irrespective of hyphal invasion. However, due to model contraction during culture, S.aureus would not have necessarily had to penetrate the epithelial barrier to gain such access and may have entered the construct directly through the lamina propria layer. C.albicans hyphal invasion was infrequent and appeared to occur more when S.aureus was present. It has been previously reported that the extent and depth of infection with dual species of C. albicans and S. aureus, were greater than when alone; this was not observed in the present study. 54
The OMMs and the constituent HaCaT and HGF cell types upregulated pro-inflammatory cytokine production upon infection. However, for HGF and OMMs, no differences in either IL-6 or IL-8 production were observed between single and dual infections of C. albicans and S. aureus. Within HaCaT cells there was found to be a significant difference in IL-8 production between single and dual infections of C.albicans and S.aureus, however, this value lay somewhere between that of a single infection with either organism. This could indicate multiple things. It may be the case that these particular strains of synergistic C.albicans and S.aureus do not exhibit a synergistic response. It may also be the case that regardless of pathogen synergy, host pro-inflammatory cytokine production is not indicative of such. Or, most interestingly, this reduced inflammatory milieu could constitute a synergistic response. If the presence of C.albicans reduces inflammation upon S.aureus infection, this could prolong the period of time in which S.aureus has to evade detection and therefore cross epithelial barriers. Interestingly, the host immune system, namely phagocytosis, appears to play an important role in the synergy between C.albicans and S.aureus and the subsequent potential for S.aureus dissemination. 22 Small colony variants of S.aureus have been associated with denture wear. In vitro, S.aureus isolates from denture-wearers exhibit enhanced monocyte activation, but reduced phagocytosis when compared to S.aureus isolated from non-denture wearers.13,84 This reduced phagocytosis could promote microbial evasion of the host immune system. Considering that the strain from this study UOPOSA001, is an oral isolate from a denture wearing participant, it is possible that such, or similar suppressive mechanisms may be at play.
The effect of C. albicans and S. aureus single and dual infections on pro-inflammatory cytokine production has not been frequently reported in vitro. In a murine intra-abdominal infection model, and a murine peritonitis model, it was identified that pro-inflammatory cytokine levels (IL-6, GCSF, KC MCP-1 and MIP1α) were elevated in co-infection compared with single infections of C. albicans and S. aureus. 85 The current findings do not indicate the same, however in a live murine model other cytokine producing cells will be at play, such as macrophages and neutrophils. Within the present study, microbes were only exposed to epithelial and fibroblast cells. It may be the case that upon the further development of the 3DOMM into an immunocompetent model, the pro-inflammatory cytokine balance is shifted upon response to these different combinations of infective agents, due to the ability of multiple cell types to collectively respond. It may also be possible that this synergistic action may be strain dependent. The previously used, well characterised C.albicans type strain, SC5314 was used in the present study, alongside a clinical isolate of S.aureus (UOPOSA001, IRAS ref. 208291). 54 Due to the relatively recent acceptance of S.aureus as an oral coloniser, at the time of experimentation it was difficult to obtain well characterised oral type strains of S.aureus. In future, when considering the interactions of these organisms in the context of DS, clinically relevant oral isolates should be used for both strains, to study strains that possess virulence factors that permit oral colonisation. This would allow the study of C.albicans and S.aureus in a more relevant context and may better represent their proposed synergistic relationship.
Conclusions
Oral mucosal models formed of HaCaT epithelial cells and primary human gingival fibroblasts are suitable for the study of Denture Stomatitis. Models formed reproducible multi-layered epithelia, which were suitable for the histological evaluation of cross-kingdom S.aureus and C.albicans infection. Culturing models for >7 days but <21 days prior to use indicates a suitably low pro-inflammatory baseline to study IL-6 and IL-8 induction upon infection, selecting day 19 to use the models and a 24-h microbial infection maximises the time in which the epithelium can mature. S.aureus and C.albicans stimulated pro-inflammatory cytokine production by OMMs upon infection but did not exhibit an additive nor deleterious effect when co-cultured. C.albicans formed biofilms consisting of hyphae at the epithelial barrier, and infrequently penetrated deeper tissue. S.aureus was found in the lamina propria layer of the OMM irrespective of C.albicans presence. OMMs produced pro-inflammatory cytokines in response to single and dual infections study of C.albicans and S.aureus. As such, this OMM proves suitable for the study of gram-positive bacterial and fungal infections at the oral mucosal barrier.
Footnotes
Acknowledgements
The authors wish to thank Mr Matthew Emery, Dr Barbara Durante and Ms Hélène Stern for their technical support and advice, Dr Nicole Thomas for the collection of oral swabs, Ms Pam Baxter for advice regarding the IRAS application and Professor Carlos Vergani and Dr Paula Barbugli for their discussions and collaboration in projects prior to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This PhD research was funded by a PhD studentship awarded the Peninsula Dental School, University of Plymouth. This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
